Abstract
Introduction:
Little is known about the implications of multivessel occlusions (MVO) in large vessel occlusion stroke patients who undergo endovascular treatment (EVT).
Patients and methods:
We report data from the MR CLEAN Registry: a prospective, observational study on all stroke patients who underwent EVT in the Netherlands (March 2014–November 2017). We included patients with an intracranial target occlusion in the anterior circulation. An MVO was defined as an MCA occlusion (M1/M2) or intracranial ICA/ICA-T occlusion, with a concurrent second occlusion in the ACA or PCA territory confirmed on baseline CTA. To compare outcomes, we performed a 10:1 propensity score matching analysis with a logistic regression model including potential confounders. Outcome measures included 90-day functional outcome (modified Rankin Scale, mRS) and mortality.
Results:
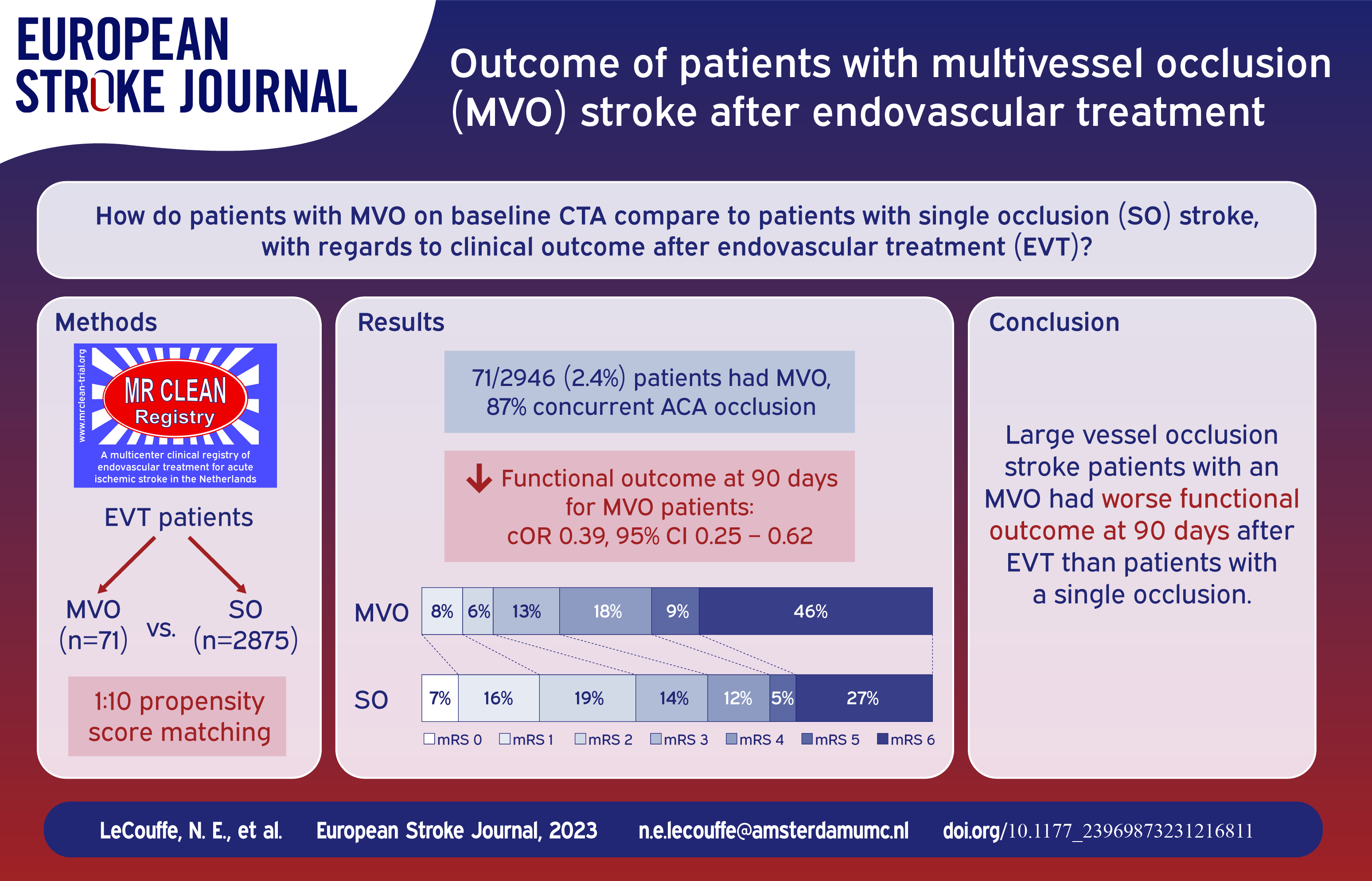
Of 2946 included patients, 71 patients (2.4%) had an MVO (87% concurrent ACA occlusion, 10% PCA occlusion, 3% ⩾3 occlusions). These patients were matched to 71 non-MVO patients. Before matching, MVO patients had a higher baseline NIHSS (median 18 vs 16, p = 0.001) and worse collateral status (absent collaterals: 17% vs 6%, p < 0.001) compared to non-MVO patients. After matching, MVO patients had worse functional outcome at 90 days (median mRS 5 vs 3, cOR 0.39; 95%CI 0.25–0.62). Mortality was higher in MVO patients (46% vs 27%, OR 2.11, 95%CI 1.24–3.57).
Discussion and conclusion:
MVOs on baseline imaging were uncommon in LVO stroke patients undergoing EVT, but were associated with poor functional outcome.
Introduction
Limited data are available on the clinical characteristics and outcomes of patients with acute ischemic stroke who have a multivessel occlusion (MVO) on baseline imaging (i.e., two or more concurrent intracranial artery occlusions on CT-angiography (CTA)) and who are eligible for endovascular treatment (EVT). In the current literature, the reported prevalence of MVO among large vessel occlusion stroke patients ranges between 0.35% and 10.7%.1–5 Importantly, when multiple vascular territories are involved, compensatory collateral circulation may be affected, leading to expedited infarction and worse functional outcome. Also, compared to single-territory infarcts, infarctions in multiple vascular territories are associated with an increased risk of recurrent stroke, and with etiologies such as cardioembolism, malignancy, and possibly coronavirus disease 2019.6–9 Thus, such patients may require a different strategy in work-up, secondary prevention and follow-up.
Furthermore, the optimal treatment for patients with MVOs has not yet been established. Multitarget EVT has been reported in a small series of MVO patients, however, functional outcome was poor despite successful recanalization of multiple vessels. 3 Administration of intravenous alteplase (IVT) might help dissolve occlusions that cannot be reached with a thrombectomy device.
We aimed to compare characteristics, clinical and radiological outcomes after EVT between patients with MVO and patients with single occlusion on baseline CTA.
Methods
Design and patients
We used data from the MR CLEAN Registry, previously reported.10,11 In short, the MR CLEAN Registry was a prospective, nationwide, observational cohort study of consecutive patients who underwent EVT in the Netherlands from March 2014 to January 1, 2019. Patients were treated according to national guidelines and received intravenous alteplase 0.9 mg/kg unless contraindicated. All imaging in the MR CLEAN Registry has been adjudicated by an independent core laboratory, whose members were blinded for clinical data except for symptom side. For the current study, we included adult patients with a primary target occlusion of the anterior circulation (internal carotid artery (ICA/ICA-T), middle cerebral artery (MCA; M1/M2) or anterior cerebral artery (ACA; A1/A2)) confirmed on CTA, who also had ⩾1 intracranial run on interventional digital subtraction angiography (DSA), treated March 16th, 2014–November 1st, 2017.
We defined an MVO as a case in which the patient had, in addition to the primary target occlusion, another large or medium vessel occlusion on baseline CTA that put a different arterial territory at risk than the territory of the primary target occlusion. For example, a patient with a proximal M1 occlusion and a concurrent ipsilateral A2 occlusion would be categorized as an MVO patient. Of note, patients with an initial ICA/ICA-T target occlusion with an additional A2 occlusion and a patent contralateral carotid terminus were also categorized as an MVO patient. A2 occlusions in these cases theoretically might have originated from the contralateral circulation and put the ACA territory at risk in addition to the MCA territory. Patients with a downstream concurrent occlusion (e.g., M3 occlusion downstream to an M1 occlusion) and patients with a tandem occlusion were not categorized as MVO.
MVOs were scored as follows: First, baseline CTA and interventional DSA images were scored separately by the core lab with standardized CRFs, which included the question if there were additional occlusions in any vascular territory other than that of the initial intracranial target occlusion. Hypoplasia of A1/A2 segments was scored separately. Second, if an additional occlusion was found on first run of DSA that had not initially been identified on baseline CTA, three authors (NL, MK and KT) re-evaluated the baseline CTA images, which were thereafter verified by an experienced interventional neuroradiologist (BE). If they were retrospectively identified on baseline CTA, they were also scored as an MVO. Of note, in the current registry, it is routine clinical practice to perform targeted DSA for emergent thrombectomy, as opposed to three- or four-vessel DSA, with rare exceptions. Finally, discharge letters were checked in all MVO cases for physician-reported MVOs identified during patient work-up at baseline.
Outcome measures were the modified Rankin Scale (mRS) score at 90 days; functional independence (mRS 0–2) at 90 days; change in score on the National Institute of Health Scale from baseline to 24–48 h post-treatment (ΔNIHSS); mortality at 90 days; proportion of patients with successful reperfusion (defined as an extended Thrombolysis in Cerebral Infarction scale 12 of 2B-3), and symptomatic intracranial hemorrhage (sICH). 13
Statistical analysis
Baseline characteristics are described using standard descriptive statistics. We performed a 10:1 propensity score matching analysis to compare outcomes of patients with an MVO to those of patients with a single occlusion. We first performed multiple imputation for missing data based on relevant covariates and outcome to reduce bias. 14 In each imputed dataset, we calculated the propensity score per patient with a logistic regression model based on the following potential confounders: age, initial intracranial target occlusion location, use of oral anticoagulation and pre-stroke mRS. 15 Patients with an MVO were then matched to patients with a single occlusion per imputed dataset in a 10:1 nearest-neighbor matching algorithm without replacement, with a caliper width of 0.20. To assess whether matching improved balance between MVO and single occlusion patients, we compared baseline characteristics in both groups in each matched, imputed dataset. We also visually evaluated the density of propensity scores graphically in the unmatched, imputed data versus the matched, imputed data. We then used (ordinal) logistic and linear regression analyses in the pooled unmatched and matched imputed datasets to compare functional, radiological and safety outcomes of MVO patients and single occlusion patients.
In a sensitivity analysis, we performed multivariable regression analyses without matching to compare outcomes of MVO patients with single occlusion patients, adjusting for age, initial intracranial target occlusion location, use of oral anticoagulation and pre-stroke mRS. For this analysis, we also used (ordinal) logistic and linear regression analyses to compare functional outcome on the mRS, functional independence, mortality, successful reperfusion, sICH and ΔNIHSS.
Lastly, we performed a subgroup analysis, assessing outcomes with descriptive statistics (due to the small sample size) for MVO patients who received IVT prior to EVT to those who underwent direct EVT.
(Common) odds ratios ((c)OR) and beta (β) coefficients are reported with 95% confidence intervals (CI). All p-values are 2 sided. Statistical analyses were performed with IBM Statistics for Windows, Version 25.0 and the computing environment R (R Development Core Team, 2013).
Results
Out of 3637 patients in the MR CLEAN Registry, 2946 patients met the inclusion criteria and had available imaging (Figure I in the Supplemental Appendix). Of these 2946 patients, 71 had an MVO on baseline CTA (2.4%). Sixty-two (87%) were identified by the core lab on the first run of DSA and retrospectively confirmed on baseline CTA by the authors. Nine (13%) were identified by the core lab on baseline CTA.
In eleven out of 66 (17%) available discharge letters of MVO patients, the local physician reported an MVO.
MVO patients had more severe neurological deficits at baseline (median NIHSS 18 vs 16, p = 0.001), worse pre-stroke mRS scores (pre-stroke mRS = 0: 37/70 (53%) vs 1920/2814 (68%), p = 0.05), worse collateral status on baseline CTA (absent collaterals 12/70 (17%) vs 161/2785 (6%), p < 0.001) and longer procedure times (median 66 vs 59 min, p = 0.01) than single occlusion patients (Table 1). In 62/71 patients (87%), the additional occlusion was located in the ACA territory; 5/71 patients (7%) had a concurrent, ipsilateral A1, 46/71 (65%) A2, and 11/71 (16%) A3 occlusion. Seven patients (10%) had an additional occlusion in the PCA territory. Two patients (3%) had ⩾3 intracranial occlusions.
Baseline characteristics.
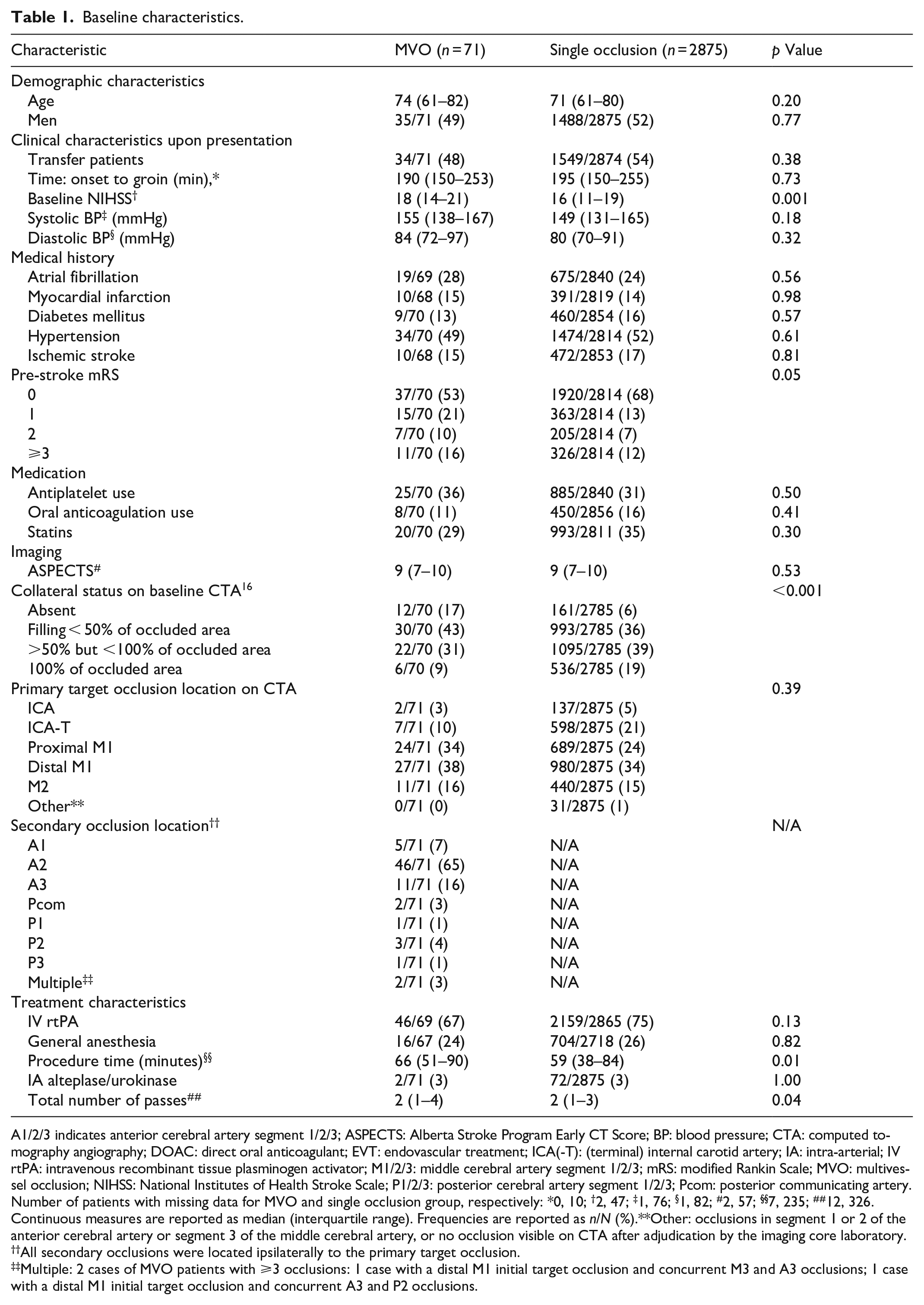
A1/2/3 indicates anterior cerebral artery segment 1/2/3; ASPECTS: Alberta Stroke Program Early CT Score; BP: blood pressure; CTA: computed tomography angiography; DOAC: direct oral anticoagulant; EVT: endovascular treatment; ICA(-T): (terminal) internal carotid artery; IA: intra-arterial; IV rtPA: intravenous recombinant tissue plasminogen activator; M1/2/3: middle cerebral artery segment 1/2/3; mRS: modified Rankin Scale; MVO: multivessel occlusion; NIHSS: National Institutes of Health Stroke Scale; P1/2/3: posterior cerebral artery segment 1/2/3; Pcom: posterior communicating artery.
Number of patients with missing data for MVO and single occlusion group, respectively: *0, 10; †2, 47; ‡1, 76; §1, 82; #2, 57; §§7, 235; ##12, 326.
Continuous measures are reported as median (interquartile range). Frequencies are reported as n/N (%).**Other: occlusions in segment 1 or 2 of the anterior cerebral artery or segment 3 of the middle cerebral artery, or no occlusion visible on CTA after adjudication by the imaging core laboratory.
All secondary occlusions were located ipsilaterally to the primary target occlusion.
Multiple: 2 cases of MVO patients with ⩾3 occlusions: 1 case with a distal M1 initial target occlusion and concurrent M3 and A3 occlusions; 1 case with a distal M1 initial target occlusion and concurrent A3 and P2 occlusions.
Overall, in the unadjusted analysis, MVO patients had worse functional outcome at 90 days than patients with a single occlusion (Figure 1; median mRS at 90 days 5 vs 3; crude cOR 0.42; 95%CI 0.27–0.66).
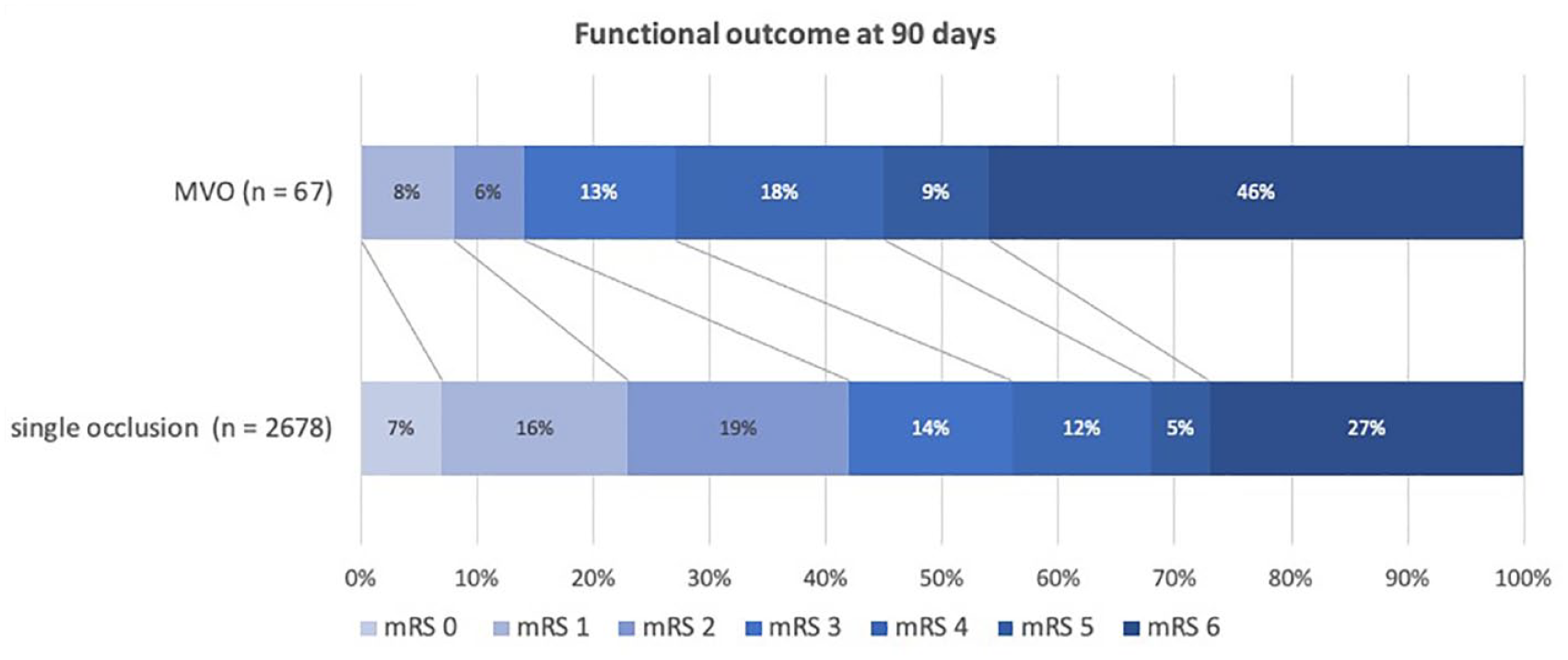
Distribution of the modified Rankin Scale score at 90 days in multivessel occlusion (MVO) versus single occlusion patients.
After propensity score matching, 71 MVO patients were matched with 710 single occlusion patients in each imputed dataset. Groups were fairly well balanced after matching, except for baseline NIHSS, collateral status, procedure time in 5/5 imputed matched baseline datasets, and use of IV tPA in 2/5 imputed matched baseline datasets (Table S1 in the Supplemental Appendix). Distribution of the propensity score showed more overlap between the groups per imputed dataset after matching, suggesting improved balance (Figure II in the Supplemental Appendix).
After matching, MVO patients had worse functional outcome (cOR 0.39; 95%CI 0.25–0.62) and lower odds of achieving functional independence at 90 days (OR 0.24; 95%CI 0.11–0.52) (Table 2). Mortality was also significantly higher in the MVO patient group (OR 2.11; 95%CI 1.24–3.57). The proportion of patients with successful reperfusion of the target occlusion was similar in both groups (47/71 (66%) vs 1803/2830 (64%), OR 1.10; 95%CI 0.63–1.93, for MVO vs single occlusion patients, respectively), as was the risk of sICH (4/71 (6%) vs 173/2875 (6%), OR 1.04; 95%CI 0.36–3.05).
Outcomes among patients with MVOs versus single occlusion, before and after propensity score matching.
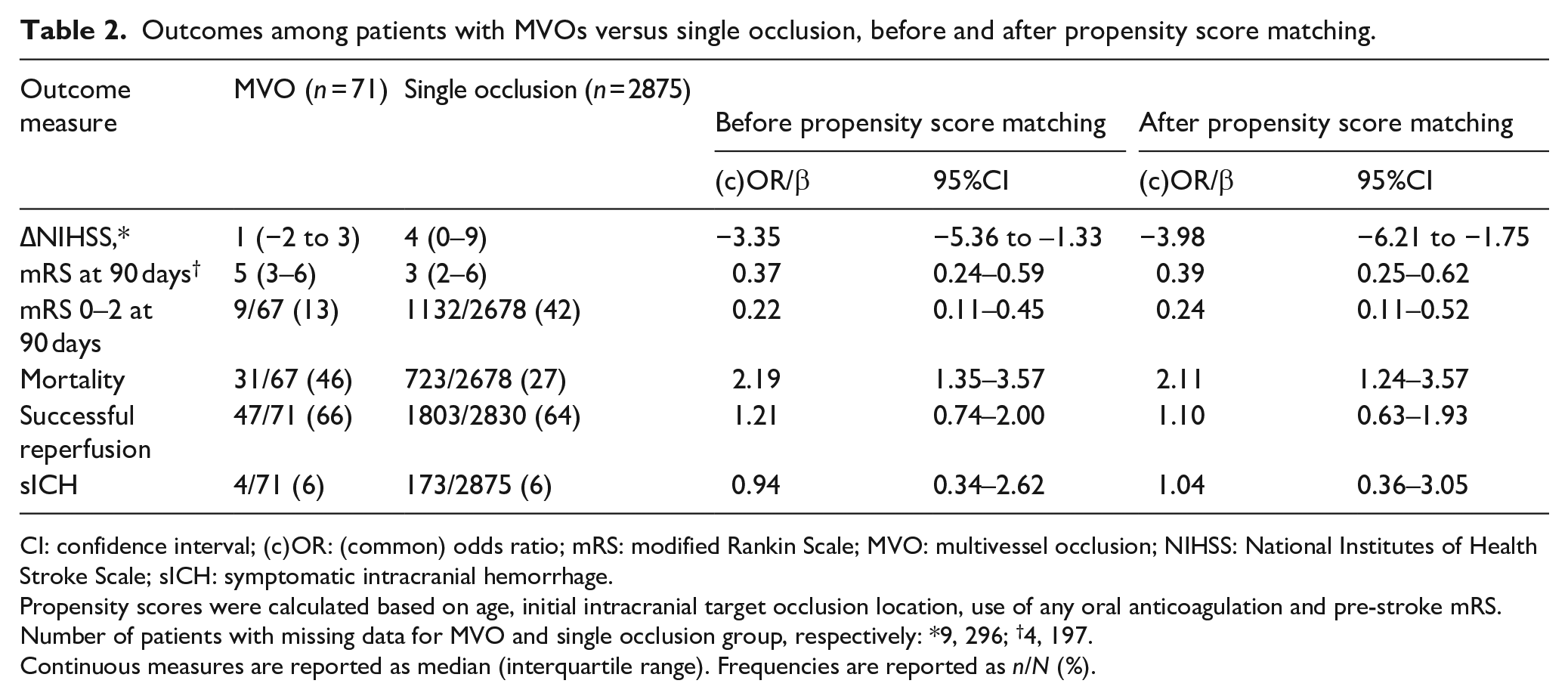
CI: confidence interval; (c)OR: (common) odds ratio; mRS: modified Rankin Scale; MVO: multivessel occlusion; NIHSS: National Institutes of Health Stroke Scale; sICH: symptomatic intracranial hemorrhage.
Propensity scores were calculated based on age, initial intracranial target occlusion location, use of any oral anticoagulation and pre-stroke mRS.
Number of patients with missing data for MVO and single occlusion group, respectively: *9, 296; †4, 197.
Continuous measures are reported as median (interquartile range). Frequencies are reported as n/N (%).
The sensitivity analysis, in which we assessed outcome measures using multivariable regression analyses instead of propensity score matching analysis, showed similar results (Table S2 in the Supplemental Appendix). The subgroup analysis comparing MVO patients who received IVT followed by EVT to those who underwent direct EVT showed overall worse outcomes for the direct EVT group and is reported in the Supplemental Appendix (Figure III and Table S3).
Discussion
In this nationwide registry of anterior circulation LVO patients undergoing EVT, we found that 2.4% of patients had an MVO on baseline CTA. Both in the unadjusted and adjusted analysis, the presence of an MVO was associated with a worse functional outcome at 90 days, despite similar frequencies of sICH in both groups. Almost half of all MVO patients had died at 90-day follow-up.
Previous studies also observed worse functional outcome in MVO patients.1–5 A Swiss multicenter study that reviewed 6081 EVT patients found that of 21 MVO patients, only 20% reached functional independence at 90 days, fairly similar to 13% in our study. 2 A Taiwanese, multicenter study of 602 EVT patients with 11 MVO patients, reported a mortality rate of 54% at 90 days for MVO patients. 3 Other studies report similar mortality rates for patients with MVOs (44%–60%).1,2 However, the definition of MVOs differs per registry (e.g., additional occlusions downstream from the initial target occlusion, or carotid tandem occlusions), making it difficult to compare results.
The reason why patients with an MVO have a poor prognosis is likely a combination of several factors. First, neurological deficits at baseline in this group of patients are worse, corresponding with a larger area of ischemic tissue at baseline. In the current study, we found a higher median NIHSS at baseline in MVO patients. Second, we found a higher percentage of absent collaterals in MVO patients in our study. Third, we found longer procedure times, likely leading to larger infarct volumes. Lastly, comorbidity may be different in MVO patients.7,17–20 We found no differences between groups for atrial fibrillation or previous myocardial infarction. We did, however, find that MVO patients less often had a pre-stroke mRS of 0 (53% vs 68%).
According to the definition we used, 2.4% of cases had an MVO. A recent post-hoc analysis of the ESCAPE-NA1 trial (NCT02930018), using a similar definition for MVOs to ours, reported a prevalence of 1.5%. 5 These prevalences are lower than those previously reported (10.7%), which can be partly explained by the fact that the earlier study also included downstream MVOs. 1 Whereas in the earlier study the majority of patients underwent MR imaging, downstream MVOs may be falsely identified as such on CTA and DSA, (the primary imaging modalities in the current study) as filling defects may only be due to slow flow distal to the target occlusion. Downstream MVOs could therefore not reliably be detected in the current study.
In an acute stroke population of EVT-eligible patients, there is likely underreporting of MVOs, possibly due to lack of awareness. 1 In our study, in only 17% of MVO cases with an available discharge letter, the local physician mentioned the presence of an additional occlusion in another territory. Further, the majority of MVO cases in this study were identified on targeted DSA and retrospectively confirmed on baseline CTA. This underscores the difficulty of MVO identification, both in an emergency and core laboratory setting. Physicians should be aware of phenomena such as satisfaction of search, previously reported amongst radiologists. 21 The use of CT perfusion or MR perfusion-weighted imaging helps to detect additional occlusions in the acute setting. 22 Unfortunately, in the current study, CT perfusion was not yet used in routine clinical practice in the Netherlands (2014–2017) and perfusion imaging was only available for 8% of the included patients. This may have contributed to an underestimation of the incidence of MVO patients in the study population.
We found a similar proportion of patients with successful reperfusion of the target occlusion in both groups, as opposed to Kaesmacher et al., who found a lower percentage of successful reperfusion in the MVO group. 1 This difference is likely because they included patients with downstream MVOs. As the eTICI score only takes the target downstream territory into account, 23 it underestimates reperfusion in cases with multiple occlusions in different vascular territories.
In an exploratory subgroup analysis of only MVO patients in which we compared patients who received IVT prior to EVT to those who did not, overall outcome was worse for MVO patients who underwent direct EVT. Although no conclusions can be drawn from this analysis, for MVO patients, it is conceivable that a direct EVT approach is less effective. Post-hoc subgroup analyses from recent randomized controlled trials investigating the added benefit of IVT prior to EVT may provide more insight on subgroups that may or may not benefit from IVT prior to EVT. 24
Our study has several limitations. First, most MVO cases were detected on DSA by core lab observers and retrospectively confirmed on baseline CTA. This indicates most cases were missed during the central core laboratory adjudication of the baseline CTAs. We also did not systematically re-evaluate all baseline CTAs after this finding, mainly due to the logistical challenges this would pose. This introduces two important caveats to our study: There might be an overrepresentation of patients with a “persistent” MVO, that is, those whose additional occlusions did not dissolve after IVT or auto-thrombolysis. Also, although a four-vessel DSA remains the reference standard to identify (multiple) occlusions, it is standard practice to only perform a targeted DSA for emergent thrombectomy. Thus, posterior and contralateral occlusions may have been missed, which is reflected in the fact that 87% of the concurrent occlusions were ACA occlusions. In contrast, a previous study, in which all patients underwent pre-interventional three-vessel angiography according to local protocol, reported 46% concurrent ACA occlusions for non-downstream MVOs.
Second, we have no data on whether the treating physician attempted thrombectomy of the additional occlusion. We therefore cannot make any conjectures on the added value of multi-targeted thrombectomy in MVO patients. Third, 10% of patients was excluded from the study population due to unavailable imaging. Fourth, it is possible that MVO patients, who have worse collateral flow and can therefore be classified as “fast-progressors,” are less likely to receive EVT, due to expansive marked hypodensity on baseline CT. Considering the MR CLEAN Registry only included patients who underwent EVT, we cannot exclude the possibility of selection bias. Finally, the group of MVO patients in the current study was small compared to the non-MVO group, possibly leading to a loss of statistical power. 25 Regardless, over 50% of MVO patients in our study were bedridden or had died at 90-day follow-up. Although these results must be interpreted with caution, they might provide an important consideration for physicians to take into account during decision-making in an acute stroke setting.
In conclusion, anterior circulation LVO stroke patients with an MVO had worse functional outcome at 90 days and had higher mortality rates after EVT than patients with a single occlusion. MVOs in EVT-eligible patients are likely underreported, even by expert observers. Although these results may be biased, physicians may take the presence of an MVO into consideration when selecting candidates for EVT.
Supplemental Material
sj-docx-1-eso-10.1177_23969873231216811 – Supplemental material for Outcome of patients with multivessel occlusion stroke after endovascular treatment
Supplemental material, sj-docx-1-eso-10.1177_23969873231216811 for Outcome of patients with multivessel occlusion stroke after endovascular treatment by Natalie E LeCouffe, Kilian M Treurniet, Manon Kappelhof, Ivo GH Jansen, Merel Boers, Henk A Marquering, Ludo FM Beenen, Jelis Boiten, Wim H van Zwam, Lonneke SF Yo, Charles BLM Majoie, Yvo BWEM Roos, Bart J Emmer and Jonathan M Coutinho in European Stroke Journal
Footnotes
Acknowledgements
We would like to thank Rob van de Graaf and Esmee Venema for their assistance and guidance on the statistical analyses.
Declaration of conflicting interest
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr LeCouffe, Dr Treurniet, Dr Kappelhof, Dr Emmer and Dr Coutinho were research coordinators for the MR CLEAN-NO IV trial (ISRCTN80619088). Dr Roos and Dr Majoie were principal investigators of the MR CLEAN-NO IV trial. Drs Roos, Majoie, Treurniet and Kappelhof are co-principal investigators for the IRIS study (![]() ). Dr Majoie reports grants from TWIN Foundation (related) and CVON/Dutch Heart Foundation, European Commission, Healthcare Evaluation Netherlands and Stryker, outside the submitted work (all paid to institution). In addition, Dr Majoie is shareholder of Nicolab, a company that focuses on the use of artificial intelligence for medical image analysis. Dr Roos reports stockholdings from Nicolab outside the submitted work. Dr Emmer received research support from Health Holland, Nicolab and The Netherlands Organisation for Health Research and Development. Dr van Zwam received speaker fees from Stryker, Cerenovus and Nicolab and consulting fees from Philips, all paid to Institution. Dr Coutinho received related research support from the Dutch Heart Foundation, Medtronic, unrelated research support from Bayer and Boehringer (all fees were paid to his employer), and is co-founder and shareholder of Trianect. Dr Jansen is Head of Clinical Development and shareholder of Nicolab. Dr Boers is CEO and shareholder of Nicolab. Dr Marquering is cofounder and shareholder in Nicolab.
). Dr Majoie reports grants from TWIN Foundation (related) and CVON/Dutch Heart Foundation, European Commission, Healthcare Evaluation Netherlands and Stryker, outside the submitted work (all paid to institution). In addition, Dr Majoie is shareholder of Nicolab, a company that focuses on the use of artificial intelligence for medical image analysis. Dr Roos reports stockholdings from Nicolab outside the submitted work. Dr Emmer received research support from Health Holland, Nicolab and The Netherlands Organisation for Health Research and Development. Dr van Zwam received speaker fees from Stryker, Cerenovus and Nicolab and consulting fees from Philips, all paid to Institution. Dr Coutinho received related research support from the Dutch Heart Foundation, Medtronic, unrelated research support from Bayer and Boehringer (all fees were paid to his employer), and is co-founder and shareholder of Trianect. Dr Jansen is Head of Clinical Development and shareholder of Nicolab. Dr Boers is CEO and shareholder of Nicolab. Dr Marquering is cofounder and shareholder in Nicolab.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded and carried out by the Erasmus University Medical Centre, the Amsterdam UMC, location AMC, and the Maastricht University Medical Centre. The study was additionally funded by the Applied Scientific Institute for Neuromodulation (Toegepast Wetenschappelijk Instituut voor Neuromodulatie), which played no role in trial design and patient enrollment, nor in data collection, analysis, or writing of the manuscript.
Ethical approval
The MR CLEAN Registry study protocol was evaluated by the ethics committee of the Erasmus MC University Medical Centre, Rotterdam, The Netherlands (MEC-2014-235), and permission to carry out the study as a registry was granted.
Informed consent
The need for individual patient consent has been waived by the ethics committee.
Guarantor
Jonathan Coutinho
Contributorship
NL researched literature, conceived the study, was involved in the data analysis and wrote the first draft of the manuscript. JC revised the manuscript. NL, MK, KT and BE reviewed the imaging. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
