Abstract
Introduction:
Neurology senior residents and stroke fellows are first to clinically assess and interpret imaging studies of patients presenting to the emergency department with acute stroke. The aim of this study was to compare the diagnostic accuracy of brain CT angiography (CTA) with and without CT perfusion (CTP) between neurology senior residents and stroke fellows.
Methods:
In this neuroimaging study, nine practitioners (four senior neurology residents (SNRs) and five stroke fellows (SFs)) clinically assessed and interpreted the imaging data of 50 cases (15 normal images, 21 large vessel occlusions (LVOs) and 14 medium vessel occlusions (MeVOs) in two sessions, 1 week apart in comparison to final diagnosis of experienced neuroradiologist and experienced stroke neurologist consensus. Interrater agreement of CTA alone and CTA with CTP was quantified using kappa statistics, sensitivity, specificity and overall accuracy.
Results:
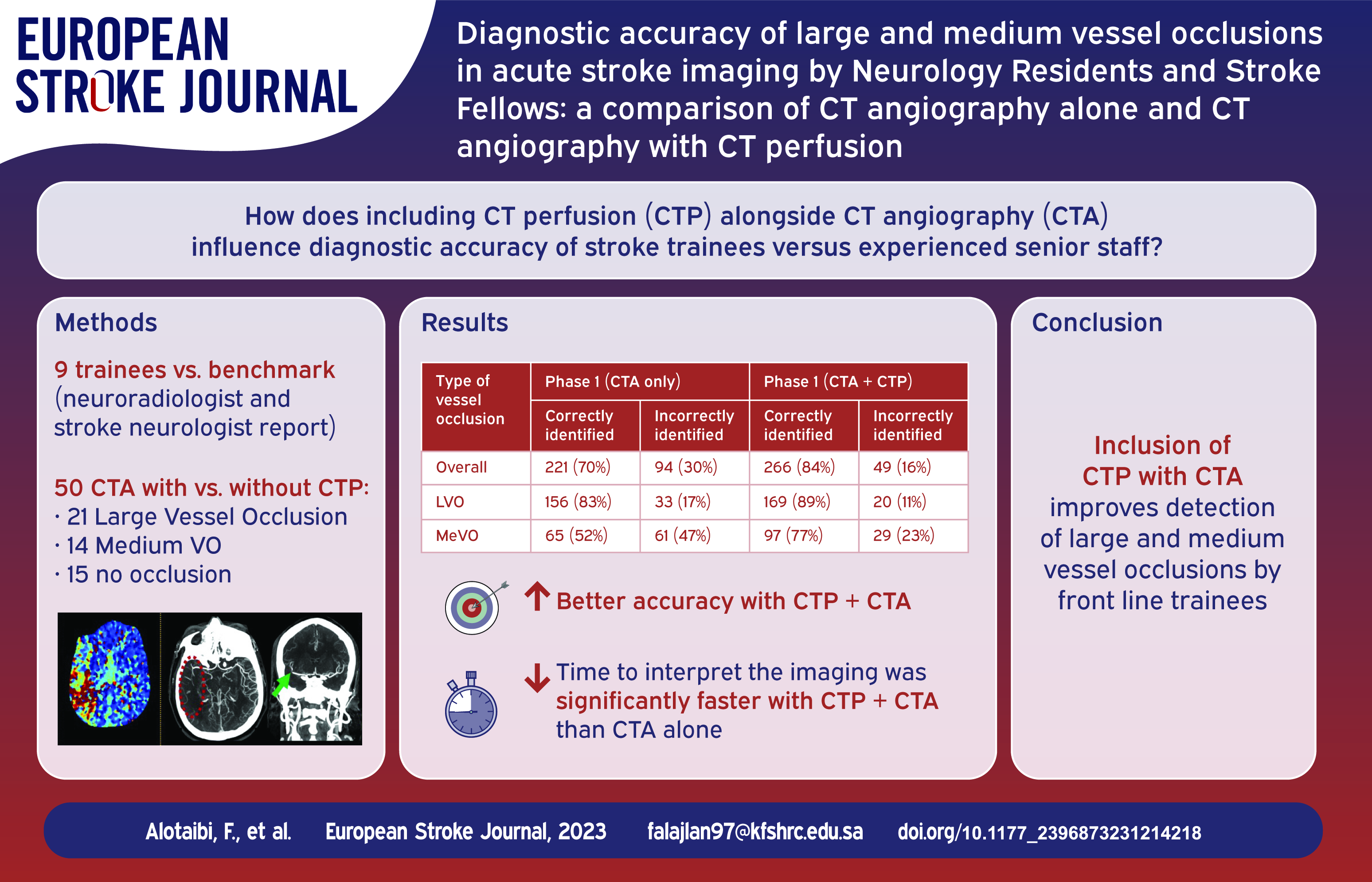
Overall, arterial occlusions were correctly identified in 221/315 (70.1%) with CTA alone and in 266/315 (84.4%) with CTA and CTP (p < 0.001). The sensitivity of overall arterial occlusions detection with CTA alone was 94.2% (95% CI: 90.8%–96.6%) while with addition of CTP was 98% (95% CI: 95.6%–99.3%), The specificity of CTA alone was 74.7% (95% CI: 67.2%–81.3%) which increased with CTP to 84.4% (95% CI: 77.7%–89.8%). The likelihood of correct identification with CTA alone was 156/189 (82.54%) for LVOs and 65/126 (51.59%) for MeVOs. This increased to 169/189 (89.42%; p = 0.054) for LVOs and 97/126 (76.98%; p < 0.001) for MeVOs when the CTA images with CTP were viewed. There was good overall interrater agreement between readers when using CTA alone (k 0.71, 95% CI, 0.62–0.80) and almost perfect (k 0.85, 95% CI, 0.76–0.94) when CTP was added to the image for interpretation. CTA and CTP had a significantly lower median interquartile range (IQR) interpretation time than CTA alone (114 [IQR, 103–120] s vs 156 [IQR, 133–160] s, p < 0.001).
Discussion:
In cerebral arterial occlusions, the rate of LVO and MeVOs detections increases when adding CTP to CTA. The accuracy and time for diagnosing arterial occlusion can be significantly improved if CTP is added to CTA. As MeVOs are commonly missed by front-line neurology senior residents or stroke fellows, cases with significant deficits and no apparent arterial occlusions need to be reviewed with neuroradiological expertise.
Introduction
Acute ischemic stroke is a medical emergency. Rapid and accurate diagnosis of the underlying intracranial large vessel occlusion (LVO) and appropriate treatment significantly improves patient outcomes. 1 Cerebral CT angiograms (CTAs) and CT perfusion (CTP) are being increasingly used to ensure the accurate identification of the arterial lesions that are responsible for acute stroke.1,2–7 Identification of occlusion on CTA can be challenging in emergency settings.8–15 The interrater agreement for accurately detecting a LVO on CTA may range from 0.48 to 0.97, depending on the clinical setting and the training and experience of the rater.12,14,16,17 Identifying a medium-sized vessel (MeVO) as the culprit for the occlusion is particularly difficult. 10 Although medical therapy remains the current standard of care in treating most MeVOs, with improvements in technology, we expect that EVT will likely be more frequently used to treat such occlusions. Failure to detect an arterial occlusion can lead to “missed opportunities” for treatment. In one recent study, 50% of patients in whom LVO was missed by the initial CTA evaluations were potential candidates for endovascular therapy (EVT). 9 Most such patients in whom the LVO is not treated have a poor outcome. 18
Specialty-trained neuroradiologists can accurately and rapidly diagnose an arterial occlusion in patients with acute stroke.8,11,14–16 However, such expertise is not always readily available, especially afterhours. Therefore, detection of the arterial occlusion in such situations may become the responsibility of the senior neurology residents (SNR) and stroke fellows (SF) who examine the patient in the emergency departments.19,20 The evaluation includes localizing the arterial occlusion and ensuring that such abnormalities are not overestimated, as this may result in unnecessary medical procedures.
Several recent studies have been performed to evaluate the ability of experienced radiologists or neuroradiologists to detect LVOs.8–12,15,17 Experienced radiologists may miss LVOs in 7%–20% of acute stroke patients.10,15 Missing arterial occlusions in smaller arteries, including the M2 and M3 segments, is especially challenging, and up to 38% may be missed by neuroradiologists when the CTA images of acute stroke patients are examined. 10
CT and MRI perfusion (CTP and MRP) techniques are used to examine changes in blood flow when an intracranial artery is occluded and may thus make the detection of intracranial arterial occlusions easier. This may be especially helpful when the occlusion is in second-order or distal arteries (MeVOs). Studies comparing the accurate detection of LVOs or MeVOs have shown that the addition of CTP to CTA significantly increases its sensitivity and specificity in the accurate detection of an intracranial occlusion.9,11,13,21
As SNRs and/or SFs are the initial physicians to evaluate acute stroke patients and interpret brain imaging in comprehensive stroke centers, it is important to determine the interpretation skills of these frontline trainees. To our knowledge, there are no studies that have evaluated the skills of SNRs and SFs in interpreting CTA images only or those combined with CTP.
We had three main objectives for this study. First, the aim was to assess the accuracy of SNRs and SFs to detect arterial occlusions on vignettes of CTA images. Additionally, we sought to identify any specific occlusions that were more likely to be missed by the trainees. Second, we aimed to determine if the additional information provided by CTP would increase the detection rate of intracranial occlusions. Finally, we aimed to evaluate the interpretation time and diagnostic accuracy of CTA or NCCT with and without CTP.
Methods
This study was conducted at the King Faisal Specialist Hospital and Research Center (KFSHRC) in Riyadh, Saudi Arabia. The Institutional Review Board approved the protocol for this study (DNS/865/44). The investigator-initiated study received no financial support. The data that support the findings are available from the senior author upon reasonable request.
This study was conducted in accordance with the Standards for Reporting Diagnostic Accuracy (STARD, https://www.equator-network.org/reporting-guidelines/stard/) guidelines.
The trainees who participated to evaluate the imaging studies included SNRs, postgraduate year (PGY) 4 and 5 (N = 4) and SFs, PGY 6 and 7 (N = 5). All the stroke trainees have extensive training and clinical experience in the interpretation of neuroimaging, including NCCT, CTA, and CTP studies.
Patient imaging selection
We retrospectively analyzed data from the RIyadh ThrombEctomy (RITE) registry. The RITE registry is a prospective citywide registry that includes all ischemic stroke patients who underwent EVT in Riyadh, Saudi Arabia. The registry is designed to collect data from a clinical cohort of stroke patients, study the natural history of ischemic stroke, and determine the impact of EVT in the Saudi population. It is a large real-world international registry that lists the outcomes after EVT. Information on demographic and clinical characteristics, medical history, physical examination, relevant laboratory parameters, and interval times was collected prospectively. The RITE registry has local ethics board approval.
For the present study, 50 patients with a final diagnosis of acute stroke were randomly selected from the registry. The diagnosis of acute stroke was confirmed between April 2019 and October 2022. The trainees were provided with brief clinical vignettes of all patients, including the age, side of the stroke on examination and their National Institute of Health Stroke Score (NIHSS).
For the purpose of this study, we included patients with single arterial occlusions in the intracranial and extracranial arteries. We also included patients in whom no occlusion was evident (N = 15). Arterial occlusions included isolated proximal internal carotid artery (ICA) occlusion with patent intracranial arteries (N = 3), ICA T-occlusion (N = 1), occlusion in the M1 segment of the middle cerebral artery (MCA) (N = 14), occlusion in the M2 segment of the MCA (N = 10), basilar artery occlusion (N = 2), and posterior cerebral artery occlusion (PCA) (N = 3). The trainees were aware of the mixed composition of occlusions and that some CTAs had no occlusion). The participants were, however, not aware of the number or location of the occlusions that were included in the study.
CT acquisition, reconstruction, and postprocessing
The standard of care for imaging at the KFSHRC includes NCCT followed by CTA and CTP images. CTA acquisition protocols were locally determined. In general, a single contrast bolus was given intravenously. Aortic contrast opacification was monitored using bolus tracking. After a certain threshold was achieved, a caudocranial CT scan was started from the aortic arch to the vertex.
Axial CTA images were reconstructed using a slice thickness of 0.6 mm. Images for multiplanar reconstruction were available in the axial, coronal and sagittal, with a 3 mm thickness. A maximal intensity projection (MIP) of 20 mm was also available for all patients. The CTP images were acquired axially and included craniocaudal coverage of 80 mm with reconstruction at 8 mm thickness and were processed using a commercially available software platform (RAPID 4.0, iSchemiaView, Monlo Park, CA, USA).
All images in the study were reviewed and approved unanimously by an experienced neuroradiologist and stroke neurologist for quality and accurate localization of the lesion site.
Experimental design
A reader study was undertaken with nine practitioners (four SNRs and five SFs), with each tasked with examining the same set of cases with and without CTP assistance. LVO was defined as any ICA occlusions intra- or extracranially, M1 segment, A1 segment, basilar artery, or P1 occlusion. MeVO was defined as an occlusion of the A2 segment, M2 segment (the postbifurcation branches of the M1 segment), or M3 segment (the location where the arteries rotate to the operculum and return in a horizontal position). For P1 (from the PCA origin at the top of the basilar artery to its side-to-end anastomosis with the posterior communicating artery, the P2, P3, and P4 began at the junction with the posterior communicating artery until the terminal branches.22–24
Setting for the evaluation
The trainees were given clinical vignettes of individual patients and access to quiet workstations in separate examination rooms. Trainees were evaluated on their skills in detecting arterial occlusions without knowing the composition of the occlusions. Each evaluation was recorded anonymously, and scores were analyzed statistically. Trainees with access to noncontrast computed tomography (NCCT) and computed tomography angiography (CTA) were evaluated twice, a week apart. CT perfusion (CTP) images were not provided in the first part.
The second part of the evaluation was performed with identical settings with the addition of CTP images of the patients. The selection and order of cases were randomized for each reader to avoid temporal and recall bias. Whereas the time taken for the detection of the individual occlusion was not determined, the time to complete diagnosis for the 50 cases was determined for individual trainees.
Statistical analysis
The level of agreement between each of the nine physicians and final diagnosis (experienced neuroradiologist and experienced stroke neurologist consensus) for the detection of any occlusion on the correct side, both with and without the use of CTP, was calculated. The agreement level was expressed as a percentage of agreement and evaluated according to Cohen’s kappa (κ) with a 95% confidence interval. A Cohen’s kappa statistic of κ = 0–0.20 is considered slight agreement; κ = 0.21–0.40 is fair agreement; κ = 0.41–0.60 is moderate agreement; κ = 0.61–0.80 is substantial agreement; and κ = 0.81–1 is optimal agreement. 25 The Kappa for the detection of LVO and MeVO on the correct side before and after CTP addition is summarized as box and whisker plots. Sensitivity and specificity with 95% confidence intervals were calculated. In addition, the Mann–Whitney test was used to compare the Kappa values of the nine readers. The chi-square test was used to compare the percentage of accurately identified cases of LVO and MeVO across the two phases. The median and interquartile range were calculated for interpretation time. A two-sided p-value <0.05 was considered significant. All analyses were performed using StataCorp. 2021. Stata Statistical Software: Release 17. College Station, TX: StataCorp LLC.
Results
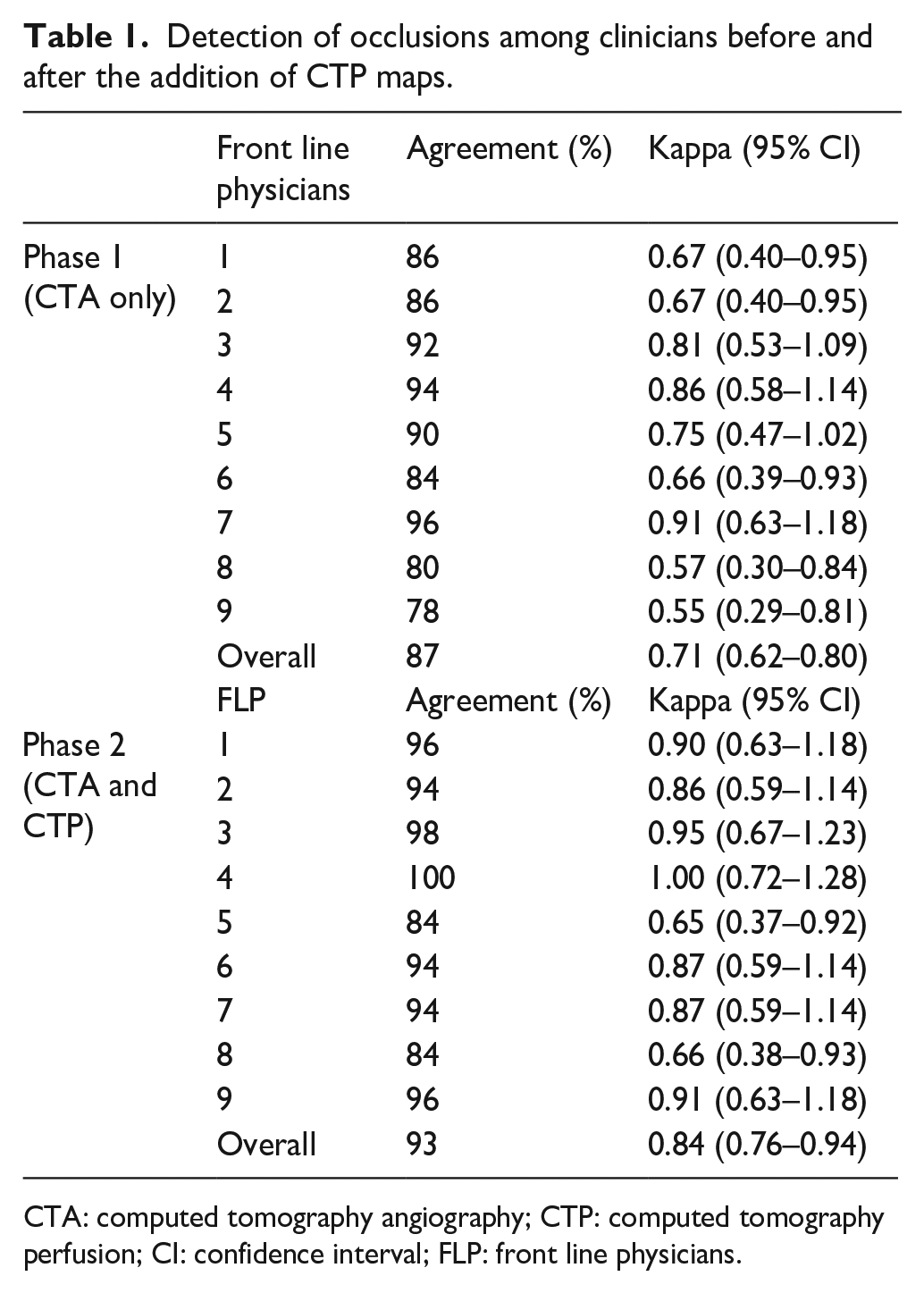
All nine participants completed the study. There were no technical problems in terms of access to imaging, and all the trainees were satisfied with the quality of the scans available for evaluation. There were 450 individual imaging results available for analysis. The overall interrater agreement was substantial between readers when using CTA alone (k 0.71, 95% CI, 0.62–0.80). Agreement improved to almost optimal (k 0.85, 95% CI, 0.76–0.94) when CTP was added to the image interpretation (Table 1). The sensitivity and specificity were 94.2% and 74.7%, respectively for the detection of arterial occlusion with CTA alone, which increased to 98% and 84.4%, respectively for the detection of arterial occlusion with CTA and CTP.
Detection of occlusions among clinicians before and after the addition of CTP maps.
CTA: computed tomography angiography; CTP: computed tomography perfusion; CI: confidence interval; FLP: front line physicians.
Objective 1: Accurate detection of arterial occlusion in patients using NCCT and CTA
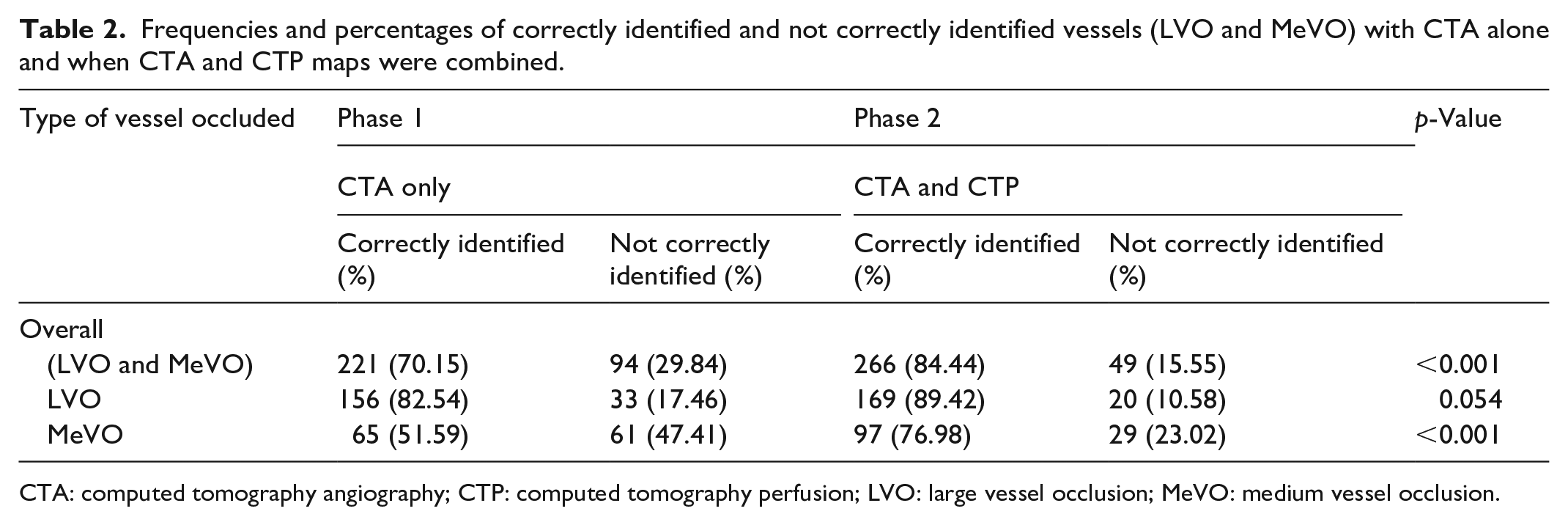
In 221 out of 315 cases (70.15%), the occlusions were accurately identified. Among patients with LVOs (ICA, M1, the P1, and basilar), 156/189 (82.5% of occlusions) were correctly identified by the participants. In 33/189 (17.46%) patients, the LVOs were not properly identified or were missed. MeVOs were correctly identified by only 65/126 participants (51.59%) (Table 2).
Frequencies and percentages of correctly identified and not correctly identified vessels (LVO and MeVO) with CTA alone and when CTA and CTP maps were combined.
CTA: computed tomography angiography; CTP: computed tomography perfusion; LVO: large vessel occlusion; MeVO: medium vessel occlusion.
In addition to missed diagnoses, we also evaluated the possibility of incorrect diagnoses. Lesions in the P2 segment of the PCA distribution (13/18, 72.2%) and the M2 segment of the MCA (41/90, 45.5%) were most likely to be missed. For the M2 segment of the MCA, occlusion was commonly missed if the occlusion segments of the MCA were at the bifurcation (origin of one of the M2 branches) or the distal M2 segments were at the mid-Sylvian point. The common reasons for missed diagnosis at various anatomical locations are shown in Figure 1.
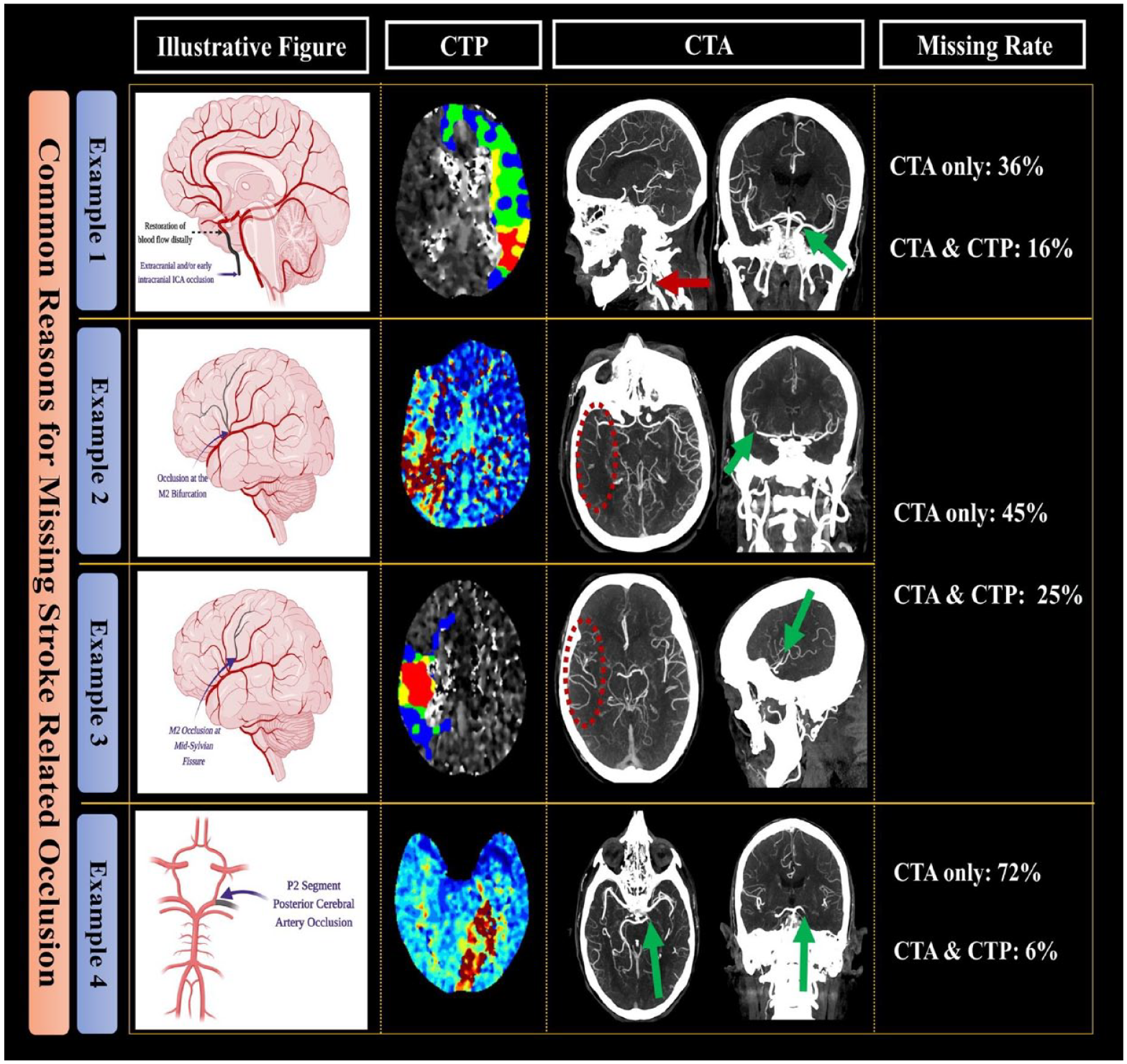
Illustrative figure showing the most common reasons for missing stroke-related occlusions with or without the addition of CTP.
Objective 2: Accurate detection of arterial occlusion in patients with NCCT, CTA, and CTP
About 1 week following the initial evaluation, the participants were reassessed. This time, they also had additional CTP information for the individual cases. The evident blood flow changes on CTP were easy to identify by the participants. This allowed the participants to focus on the appropriate anatomical region of the brain.
With the addition of the CTP information, the overall detection rate increased significantly, with 221 out of 315 cases (70.15%) initially being accurately diagnosed with CTA only to 266/315 cases (84.4%), p < 0.001, Table 2. LVOs were correctly identified in 169/189 (89.42%) patients. The ability to identify MeVOs correctly also improved significantly with the addition of CTP imaging. The participants identified 97/126 (76.98%) lesions correctly (p < 0.001) (Table 2). The interrater agreement for the correct and missed diagnoses also significantly improved, as shown in Table 1. This was most evident for MeVOs, where the agreement was only fair without the inclusion of CTP imaging, as shown in objective 1. The frequency of P2 lesions was identified correctly in 27.7% using NCCT and CTA. This significantly improved to 94.4% with the addition of CTP (p < 0.001). Detection of MCA M2 occlusion improved from 54.4% to 75.5% with the addition of CTP (p = 0.022). There was a reduction of incorrect diagnosis of ICA occlusion from 36.1% to 16.6% with the addition of CTP (p = 0.06).
Objective 3: Comparisons of the interpretation time when CTP is added to NCCT and CTA for diagnosis
CTA and CTP had a faster median (IQR) interpretation time than CTA alone (114 [IQR, 103–120] s vs 156 [IQR, 133–160] s, p < 0.001). This is shown in the box and whisker plot in Figure 2.
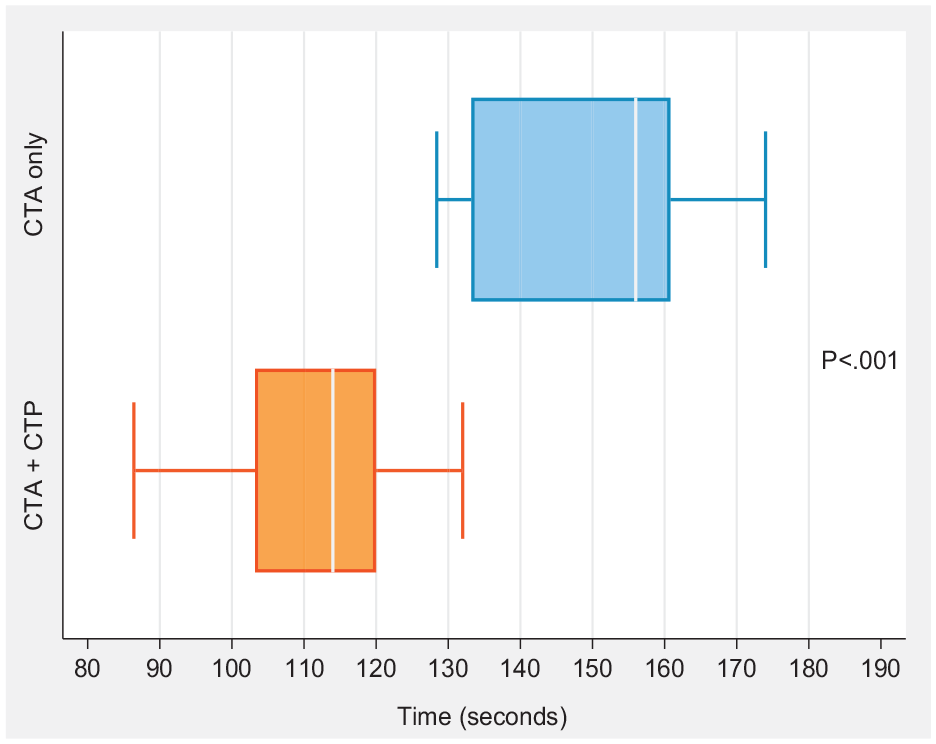
Box plot of time taken to evaluate stroke-related occlusion with CT perfusion and angiography (CTA) compared to CTA only. The interpretation time with CTA plus CTP was significantly faster (p < 0.001) than with CTA only. The box plot lines correspond from bottom of the box to top: 25th percentile, median percentile, and 75th percentile. The whiskers extend to the minimum and maximum values.
Discussion
To our knowledge, this study evaluating the ability of SNRs or SFs to accurately detect stroke-related occlusion (LVOs or MeVOs) is the first of its kind and simulates daily practice. In most academic and tertiary care centers, a SNR and/or a SF are most commonly on the acute stroke team. 26 The ability of these trainees to correctly diagnose stroke-related occlusion (LVO or MeVO) has not been previously tested. The results of the study provide comprehensive data on their ability to make the correct diagnosis and the reasons for common misdiagnoses. Similar to previous studies in which neuroradiologists or attending neurologists conducted the analysis (Table 3), we showed that the diagnosis can frequently be missed, especially with MeVOs. We also showed that the trainee’s ability and time to make the correct diagnosis improved significantly when CTP information was also available at the time of analysis. Our results are similar to those of previous studies in which CTP was utilized in addition to CTA and then compared to CTA alone by expert radiology residents and neuroradiology readers. 11 This may lead to better workflow efficiency and possibly better clinical outcomes. 27
Systematic literature review of detecting (LVO and/or MeVO) with CTA and/or CTP.
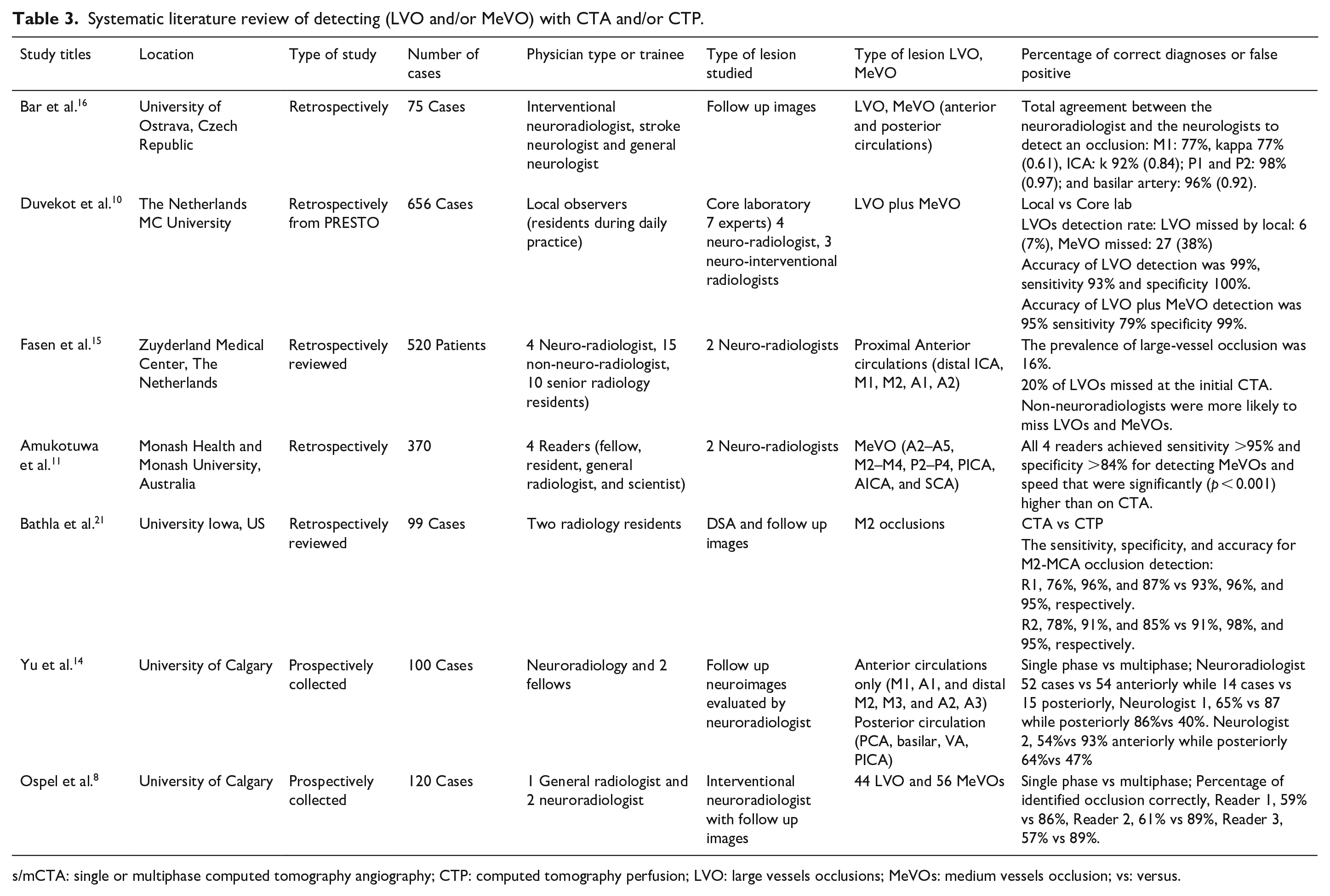
s/mCTA: single or multiphase computed tomography angiography; CTP: computed tomography perfusion; LVO: large vessels occlusions; MeVOs: medium vessels occlusion; vs: versus.
In a few previously published studies, the readers included radiology trainees.10,12,14,15 Most reports comprised of patients where the data was prospectively collected on consecutive stroke patients and the final analysis was conducted in a core laboratory, 10 retrospectively reviewed 12 or compared to the findings of CTP studies.9,11,13,21 The trainees took a longer time and were less likely to make a diagnosis, especially when the occlusions involved MeVOs. LVOs in the intracranial circulation are less likely to be missed. In the recent analysis of the PRESTO study, 7% of LVOs were not correctly diagnosed by the local observers. 10 However, the rate of misdiagnosis of MeVOs is higher. The local observers in the PRESTO study missed 38% of lesions, with the majority of lesions in the M2 site. 10 In at least 50% of cases, the missed diagnosis led to a failure to treat the acute stroke. 9 Although our rate of misdiagnosis of MeVOs with CTA is similar to that reported in the PRESTO study, it improved significantly when the CTP data were available to the participants. The PRESTO investigators noted that the common reasons for misdiagnosis included poor anatomical localization of clinical symptoms, milder symptoms, prolonged duration of symptoms and the presence of good collaterals. 10 An alternate strategy that has higher detection rates has longer CTA acquisition times. Studies using “multiphase CTA” have also shown an increased accurate MeVO detection rate.8,14 Our data also shows that likelihood of missing MeVO is high, therefore in the event of significant neurological deficits, and the absence intracranial occlusion, especially with an abnormal CTP, requires review with an experienced neuroradiologist. It was also noticed in a recent study that including CTP as part of a telestroke imaging protocol was associated with a definitive diagnosis of acute ischemic stroke in approximately one in five cases where NCCT and CTA were nondiagnostic. 28
Our study provides some common reasons that trainees miss stroke-related arterial occlusions (Figure 1). For example, such occlusions were most common in the PCA and M2 territories. Very few studies provide information on the accuracy and reliability of identifying PCA occlusions via CTA. Recent evidence suggests that the combination of CTA and CTP has greater sensitivity for detecting ischemic stroke-related arterial occlusions in the posterior circulation than CTA alone.29–32 In our study, the P2 occlusion identification with CTA alone was only 27.7%, which significantly improved to approximately 95% with the addition of CTP. This high degree of detection of occlusions in the PCA was possible when CTA was reformatted in thick slab MIP and was viewed in combination with CTP, as shown in Figure 1.
Similar to previous studies, in the current study, the detection rate of MCA M2 occlusion was low and was evident in only 50% of patients with CTA alone. This significantly improved to 75.5% with the addition of CTP. The pattern of occlusions that are commonly missed include M2 occlusion at the origin of the bifurcation of one of the M2s or when the M2 occlusion is in the distal M2 segments of MCA in the mid-Sylvian region. The likelihood of identifying these M2 occlusions improves with a focus on the region of the perfusion defects on CTP. Thick slab reformatted CTA, especially in the coronal view, can identify M2 occlusion at the bifurcation. Sagittal views can provide important information for identifying occlusions of distal M2 segments in the mid-Sylvian region, as shown in Figure 1, examples 2 and 3. Unexpectedly, the diagnosis of isolated proximal ICA occlusion with patent intracranial arteries was also high (13/36, 36.1%). This high rate of misdiagnosis may reflect the frequent tendency to focus attention on the intracranial vasculature, especially in cases where there is reformatting of the intracranial carotid artery. The misdiagnosis improved when CTP information was made available to the trainees. Data from a large registry indicate that this type of occlusion (i.e. isolated occlusions of the extra or intracranial segments of the ICA) occurs more frequently than tandem occlusions. 33 Fast identification of an isolated proximal or distal ICA occlusion with patent intracranial arteries is crucial, as EVT may be considered for patients with severe stroke or when there is progression of neurological symptoms.34–36 This high failure rate is difficult to understand and may reflect how trainees evaluate imaging in the acute setting, focusing mainly on the intracranial vessels, especially when there is reformatting of the intracranial internal carotid or middle cerebral arteries, as shown in Figure 1, example 1. Neuronal damage can occur very rapidly following LVO, especially in patients with poor collaterals.37,38 Therefore, awareness about these relatively common types of carotid occlusions that may require EVT is important. A systematic approach to evaluating the entire CTA, including reformatting of the CTA with MIP in multiple views, needs to be emphasized to the trainees. Our study shows that access to CTP data can also be very helpful in identifying the offending lesions in such situations.
Limitations
There are limitations to our study. This is a single-center retrospective study with a limited number of patients. The trainees were aware that they would be reviewing 50 cases with a mixture of normal and abnormal cases and therefore may have anticipated the lesions based on the clinical information provided to them. Furthermore, importantly, the evaluation was conducted in a quiet environment, which may not accurately reflect the often chaotic environment of the emergency department where fast diagnosis is crucial. Furthermore, although we have the total time for interpretations for each trainee, the individual time per each examination was not recorded. We therefore do not know whether the addition of CTP resulted in shorter times to diagnosis for any particular types of arterial occlusions. Additionally, CTA interpretations may be subject to interobserver variation, as demonstrated by the potential for discrepancies in interpreting terms such as “distal M1” as proximal “M2.” Such variability may have contributed to the differences observed between individual interpretations. Finally, it should be noted that multiphase CTA was not available for the participants, which may have improved the diagnosis of MeVOs.
Conclusions
In this study, we show that the use of CTP, in addition to non-contrast CT and CTA, improves the rate of detection of LVO and MeVO by front-line stroke trainees. Vessel occlusion interrater agreement improved from “substantial without the use of CTP” to “optimal with the use of CTP.” Furthermore, the use of CT perfusion decreased the time needed to detect vessel occlusions. This decreases the time required to interpret brain imaging in the acute setting and improve workflow allowing for more rapid decision-making in regards to reperfusion therapy. We also highlight some of the common pitfalls in stroke image interpretation that may lead to incorrect diagnosis. To our knowledge, this is the first study that specifically evaluated the stroke trainees ability to detect arterial occlusions based on clinical information, and neuroimaging. The high rates of missing MeVOs by our trainees suggests that imaging interpretation by experienced neuroradiologists should be done when the clinical presentation is highly suspicious for acute stroke and the trainee fails to detect an intracranial occlusion on imaging.
Footnotes
Acknowledgements
We would like to thank all coauthors for their contributions in the study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The Institutional Review Board of King Faisal Specialist Center and Research center approved the protocol for this study (DNS/865/44).
Informed consent
Written informed consent was obtained from the patients for their anonymized information to be published in this article.
Guarantor
FA.
Contributorship
FO and FA researched literature and conceived the study. AS, MG, MS, HB, AA, AH, SG, AN, MA, MQ, HA, MA, AR, AK, AH, MZRO, AS were involved in protocol development, gaining ethical approval. MG and AS was involved in data analysis. FO, FA, and AS wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
