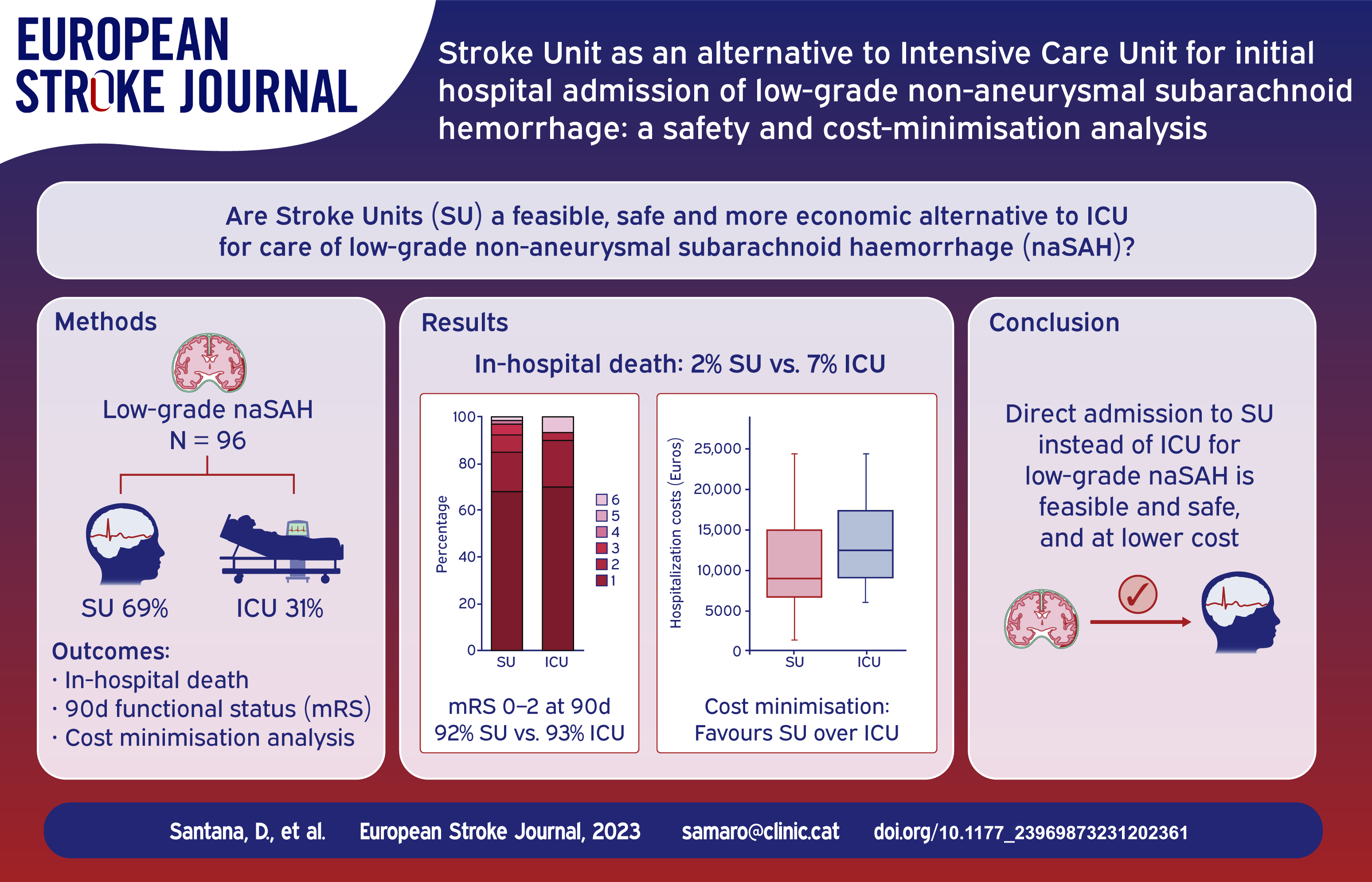
Abstract
Introduction:
Stroke Units (SU) have been suggested as an alternative to Intensive Care units (ICU) for initial admission of low-grade non-aneurysmal spontaneous subarachnoid haemorrhage (naSAH). We hypothesised that the incidence of in-hospital complications and long-term clinical outcomes in low-grade naSAH patients would be comparable in both settings, and that a cost-minimisation analysis would favour the use of SU.
Patients and methods:
Retrospective, single-centre study at a third-level stroke-referral hospital, including low-grade spontaneous naSAH patients with WFNS 1–2. Primary outcomes were death and functional status at 3 months. Secondary outcomes were incidence of in-hospital major neurological and systemic complications. Additionally, a cost-minimisation analysis was conducted to estimate the average cost savings that could be achieved with the most efficient approach.
Results:
Out of 96 naSAH patients, 30 (31%) were initially admitted to ICU and 66 (69%) to SU. Both groups had similar demographic and radiological features except for a higher proportion of WFNS 2 in ICU subgroup. There were no statistically significant differences between ICU and SU-managed subgroups in death rate (2 (7%) and 1 (2%), respectively), functional outcome at 90 days (28 (93%) and 61 (92%) modified Rankin Scale 0–2) or neurological and systemic in-hospital complications. Cost-minimisation analysis demonstrated significant monetary savings favouring the SU strategy.
Discussion and conclusion:
Initial admission to the SU appears to be a safe and cost-effective alternative to the ICU for low-grade naSAH patients, with comparable clinical outcomes and a reduction of hospitalisation-related costs. Prospective multicenter randomised studies are encouraged to further evaluate this approach.
Introduction
Spontaneous subarachnoid haemorrhage (SAH) can result in various neurological and systemic complications that have a major impact on prognosis.1–6 The incidence of complications varies widely according to several factors, including the neurological state and the amount of subarachnoid blood upon admission.7–10 Non-aneurysmal SAH (naSAH) accounts for about 10%–20% of all spontaneous SAH cases and is associated with a more benign course.11–14 However, the paradigmatic complications of SAH may still occur, especially in cases with a diffuse pattern of blood distribution in contrast with a perimesencephalic pattern.15,16
Conventionally, aneurysmal SAH patients are admitted to the Intensive Care Unit (ICU) for specialised care during the acute stage and are subsequently transferred to the neurosurgical ward once the high-risk period is over. 17 In contrast, recommendations regarding the most appropriate initial level of care unit for naSAH patients are scarce. In specialised centres, Stroke Units (SU) have emerged as a safe and potentially cost-effective alternative for selected SAH cases expected to have a benign course, such as those affected by naSAH. However, there are no clear recommendations indicating when ICU admission can be avoided in favour of SU in this subset of patients. The theoretical advantages of the straight to the SU protocol include reduced hospitalisation costs and a rationale distribution of ICU beds for neurocritical patients in higher need of intensive support.18–20 Still, these advantages are only theoretical, as no formal clinical and economic comparison between the ICU and the SU protocols has to date been performed.
We hypothesised that in-hospital complications and long-term outcome in low-grade naSAH would be comparable between ICU and SU settings, and that a cost-minimisation analysis would favour SU. To test these hypotheses, we first performed a retrospective evaluation of low-grade naSAH sufferers admitted to our centre and initially managed either in the ICU or in the SU, followed by a cost-minimisation analysis to estimate the average cost savings that could be achieved with the most efficient approach.
Patients and methods
The study protocol was approved by the institutional Clinical Research Ethics Committee and follows the national legislation of biomedical research, the protection of personal data (15/1999), the standards of Good Clinical Practice and the Helsinki Declaration (1975 and 1983 revisions). All data are available on reasonable request.
Study population
The study was held in a third-level stroke-referral hospital (Hospital Clínic of Barcelona), in a European high-income country, which provides assistance to a local population of 540,000 inhabitants and tertiary acute stroke care to >1,000,000 people. A prospectively collected database included all patients admitted to our institution with spontaneous SAH, from 2012 to 2022. Only low-grade spontaneous naSAH patients, defined as WFNS score <3, were included in the present analysis. Exclusion criteria were: intubation or advanced life support at hospital admission, WFNS 3–5, and an unstable clinical condition with short life expectancy.
Management protocol of low-grade non-aneurysmal spontaneous SAH
According to our institutional protocol, patients who arrive at the Emergency Department with the suspicion of spontaneous SAH are evaluated by a multidisciplinary team including Neurologists, Anaesthesiologists and Neurosurgeons. Following the initial stabilisation and neurological evaluation, a plain CT scan and CT angiography are done to confirm the diagnosis of SAH and to identify intracranial aneurysms. A digital subtraction angiography (DSA) is arranged in a timely manner for diagnostic and oftentimes treatment purposes. If no aneurysm is found in the DSA, a second vascular imaging is performed within 7–10 days. This second image is either aMR angiography or DSA, depending on the estimated probabilities of harbouring an aneurysm based on the initial bleeding pattern. Low-grade naSAH patients (WFNS 1–2) are initially admitted in the SU or in the ICU depending on bed availability. Clinical and neurological examinations are conducted daily by either stroke neurologists in the SU or anaesthesiologists in the ICU, as well as by trained nurses three additional times a day. Special attention is paid to common neurological and systemic complications, including re-bleeding, seizures, hydrocephalus, delayed cerebral ischaemia (DCI), stress cardiomyopathy, pneumonia, meningitis or ventriculitis. Screening for angiographic vasospasm is performed once or twice a day, according to the clinical condition, by means of transcranial Doppler ultrasound. Blood tests including an electrolyte panel are run daily, glucose levels are recorded every 6 h and non-invasive blood-pressure monitoring is run every hour. Fluid balance and external ventricular drain (EVD) output are registered in an 8-h basis. Intravenous antihypertensive and antiplatelet drugs can be administered in both units, as well as intrathecal antibiotics and fibrinolytics. A more detailed comparison between ICU and SU characteristics can be seen in Supplemental Table 1. Patients initially admitted to the SU are transferred to the ICU in case of haemodynamic instability, severe hydrocephalus, symptomatic vasospasm requiring vasopressor therapy and advanced haemodynamic monitoring or in the case of severe systemic complications that require intensive management.
Effectiveness and safety evaluation
Relevant data for clinical evaluation included epidemiological and premorbid information, Glasgow Coma Scale (GCS), WFNS score and modified Fisher score (mF) upon arrival, initial glycaemia, haemorrhage distribution, presence of intraparenchymal haemorrhage, development of acute hydrocephalus and the need for EVD placement.
Patients were classified according to their bleeding pattern in diffuse, perimesencephalic and focal SAH. Diffuse SAH was defined as widespread bleeding occupying most of the subarachnoid space bilaterally. Perimesencephalic SAH was defined as a bleeding pattern confined to the basal cisterns surrounding the brainstem; blood may extend to the basal portions of the sylvian and interhemispheric fissures but not into their distal portions; also, blood may be partially filling the occipital horns of the lateral ventricles without a frank intraventricular haemorrhage. Focal SAH was defined as the presence of blood confined to a specific subarachnoid cistern other than perimesencephalic or cortical sulci.
Major complications related to SAH were re-bleeding, angiographic vasospasm (an increase in Doppler velocities over predefined thresholds or a significant arterial narrowing >20%), DCI (neurological deterioration of ⩾2 points in GCS or NIHSS score, lasting at least 1 h and without other plausible causes), myocardial dysfunction (1/hypo- or dyskinesia in echocardiogram; or 2/cardiovascular dysfunction and elevated troponin I levels; or 3/cardiovascular dysfunction and EKG abnormalities), ventriculitis/meningitis (1/positive CSF cultures, or 2/clinical suspicion of CNS infection or fever in absence of other causes, plus compatible CSF profile), and pneumonia. Functional outcome was assessed by certified neurologists at hospital discharge and at 90 days, using the modified Rankin Scale (mRS) and following structured questionnaires in an in-person visit.
Economic assessment
The length of stay (LoS) in the different level-of-care units (ICU, SU and general ward) was extracted at an individual level from hospital charts. Health costs were calculated assuming a cost of €1175.9 per ICU day, €707.4 per SU day and €419 per general ward day, according to the rates provided by the local billing department updated by January 2020. Patients were categorised according to the unit of first admission. Crossover cases were defined as patients who were initially admitted to the SU but needed secondary transfer to the ICU. Meanwhile, step-down cases were defined as patients who were primarily admitted to the ICU and subsequently shifted to the SU due to clinical stability (see formulae below).
Cost in ICU = LoS ICU * €1,175.9/day + LoS SU * €707.4/day + LoS Ward * €419/day.
Cost in SU = LoS SU *€707.4/day + LoS ICU *€1,175.9/day + LoS Ward * €419/day.
Cost minimization = Cost ICU – Cost SU.
These costs included both in-hospital direct and indirect costs (nursing services, drugs, medical supplies, rehabilitation, food services, general administration, health records, information technology and human resources, among others).
Statistical analysis
Data is presented as mean ± standard deviation, median ± interquartile range (IQR) and percentages and group differences were assessed with Student t test, Mann-Whitney test, chi-squared or Fisher exact test as appropriate. Hypothesis testing was performed with a two-step approach. First, the ICU and SU protocols were compared in terms of safety (i.e. incidence of complications) and effectiveness (i.e. short- and long-term clinical outcomes). Primary outcomes were death and functional outcome (mRS) at 90 days; secondary outcomes were the major complications detailed above. A stratification analysis was also performed, in which the bleeding pattern (perimesencephalic or diffuse) was considered a potential confounding factor. In essence, the Cochran-Mantel-Haenszel method was used to compare a series of two-by-two tables showing the association between the unit of first admission and outcome at two levels of stratification (type of bleeding pattern). Second, a cost-minimisation analysis was performed, after proving the equal effectiveness of both protocols. This test evaluated the amount of monetary cost that could be afforded if the alternative strategy (initial SU admission) had been chosen over the conventional one (initial ICU admission). For this analysis, only health-related costs derived from the hospitalisation were taken into consideration. Costs were expressed in 2020 referenced euros (€). All the analyses were performed with SPSS v27.0 (IBM Corp., Armonk, NY.). The null hypothesis was that both interventions were equal and a p-value of <0.05 (two-tailed) was considered an indicator of statistical significance.
Results
Baseline characteristics of the cohorts
Between 2012 and 2022, a total of 516 patients with spontaneous SAH were admitted to our tertiary stroke-referral institution. After applying the inclusion and exclusion criteria, 96 naSAH patients were eligible for inclusion in the study, of whom 30 (31%) were initially admitted to the ICU and 66 (69%) to the SU (Figure 1). Clinical and radiographic features are summarised in Table 1. No statistical differences were found in demographic variables, cardiovascular risk factors, initial glucose levels, bleeding pattern, or incidence of acute hydrocephalus. EVD were equally managed in the ICU and SU. A significant predilection towards the ICU as the admission unit was seen in WFNS grade 2 patients, compared to WFNS grade 1.
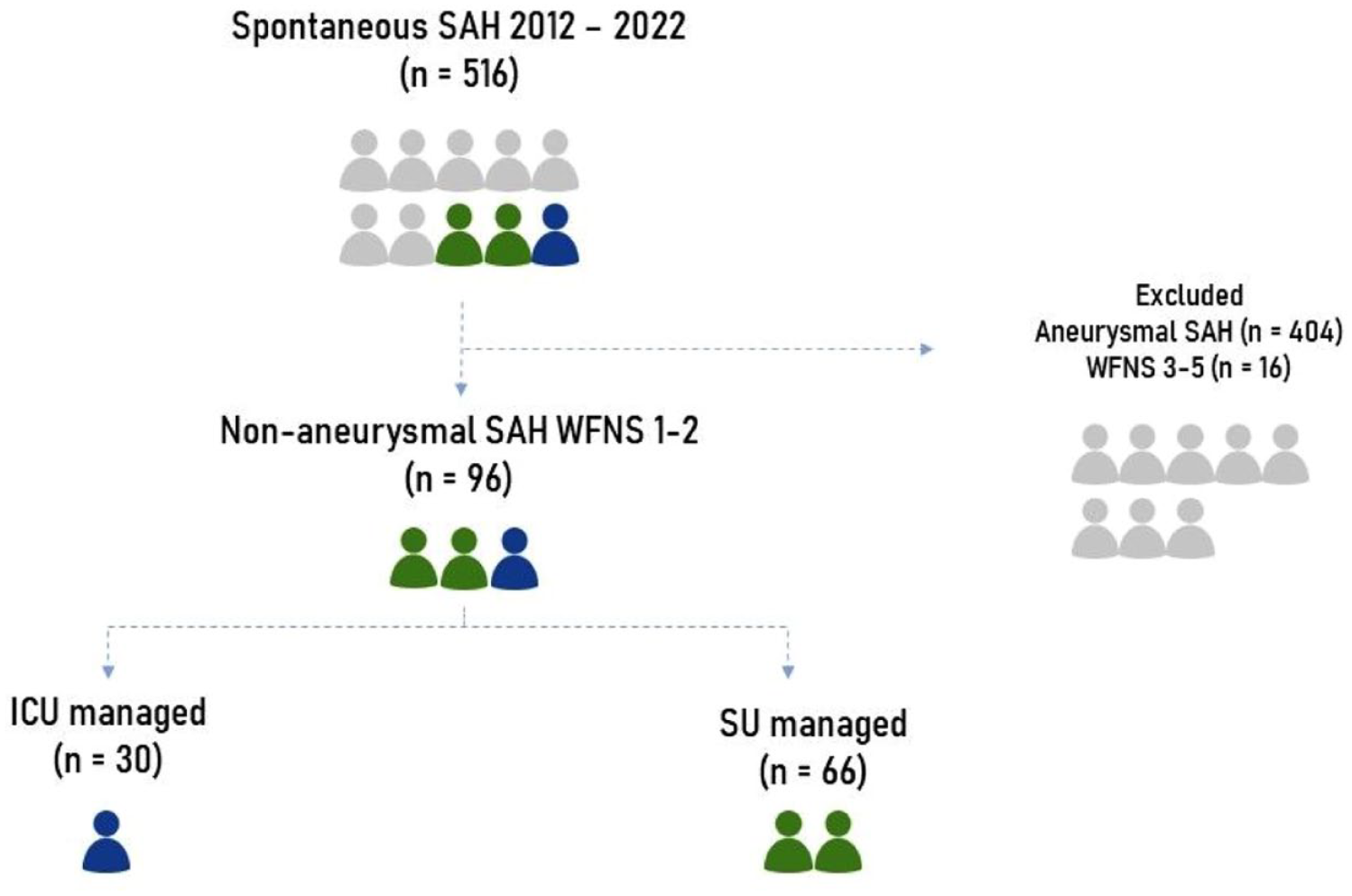
Patient flow chart: number of patients finally studied and their distribution.
Main features of the sample: low-risk non-aneurysmal spontaneous subarachnoid haemorrhage patients, grouped according to the initial unit at hospital admission.
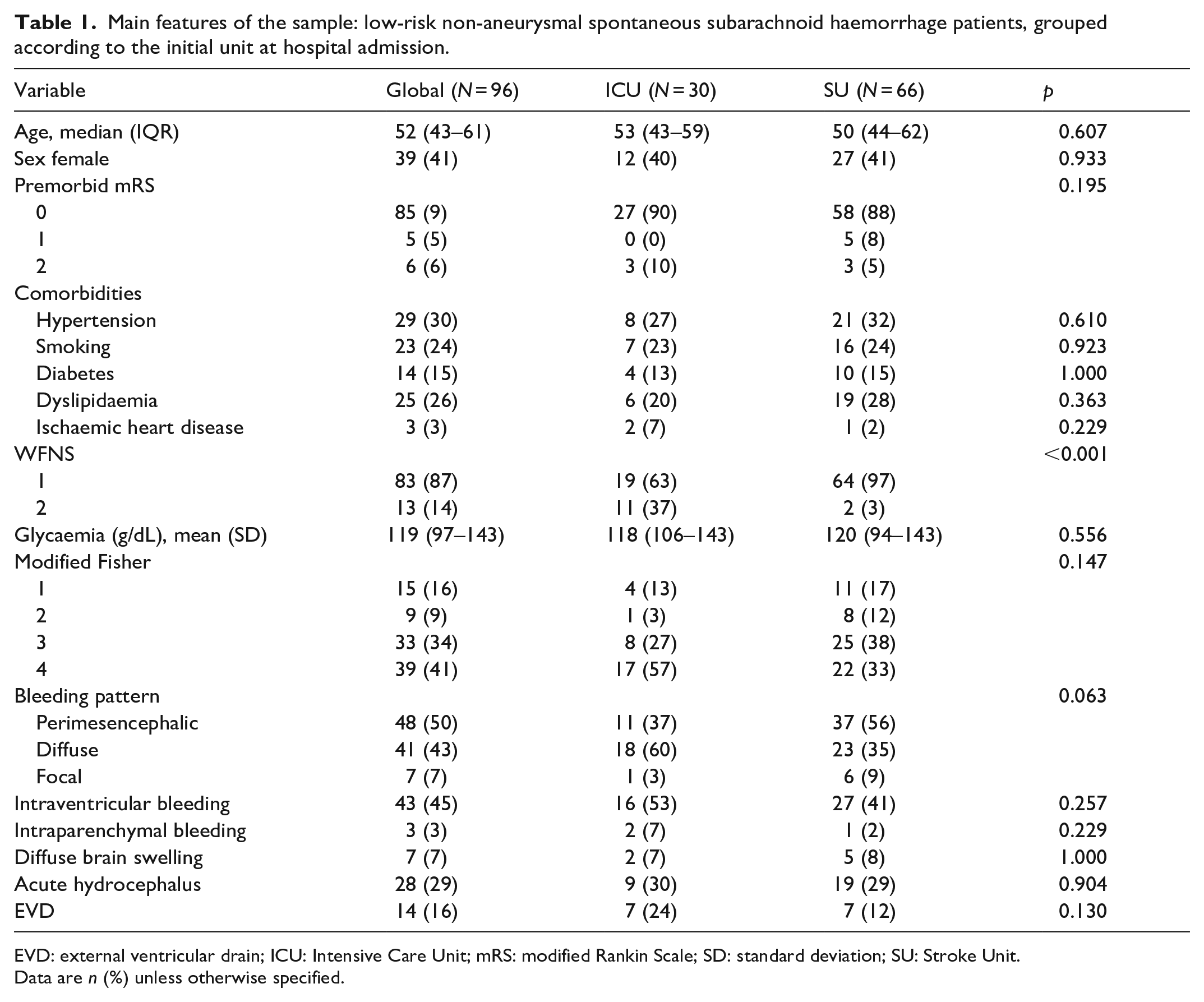
EVD: external ventricular drain; ICU: Intensive Care Unit; mRS: modified Rankin Scale; SD: standard deviation; SU: Stroke Unit.
Data are n (%) unless otherwise specified.
ICU versus SU: Safety and effectiveness comparison
As shown in Table 2, the mortality rate, the proportion of patients with good clinical outcome, and the incidence of complications was similar regardless of the unit of initial management. In this series, the most frequent complication was angiographic vasospasm, seen in 19 (63%) patients in the ICU and in 35 (53%) in the SU; still, only 2 (5%) and 1 (2%) of patients, respectively, developed DCI. The incidence of seizures, EVD-related infections and pneumonia was also similar between the two groups. No myocardial infarctions were observed in either group. Rebleeding occurred in two patients, one in each subgroup, due to re-rupture of blister-type aneurysms that were not identified in the initial vascular study. In-hospital death occurred in 2 (7%) and 1 (2%) patients in the ICU and SU, respectively. Two patients initially admitted to the SU required transfer to the ICU due to acute respiratory failure, but neither patient died during hospitalisation, and their mRS at 3 months were 3 and 0, respectively. Conversely, 13 (43%) of the patients admitted to the ICU were transferred to the SU after a median (IQR) of 3 (3–3) days of ICU management. After 90 days of the haemorrhagic event, more than 90% of the patients in both groups had good clinical recovery (mRS 0–2) and no differences were found between them. No deaths after discharge were seen in our sample.
Incidence of in-hospital complications, length of stay in the different units, long-term functional outcomes and total hospital costs in the study sample according to the initial level of care.
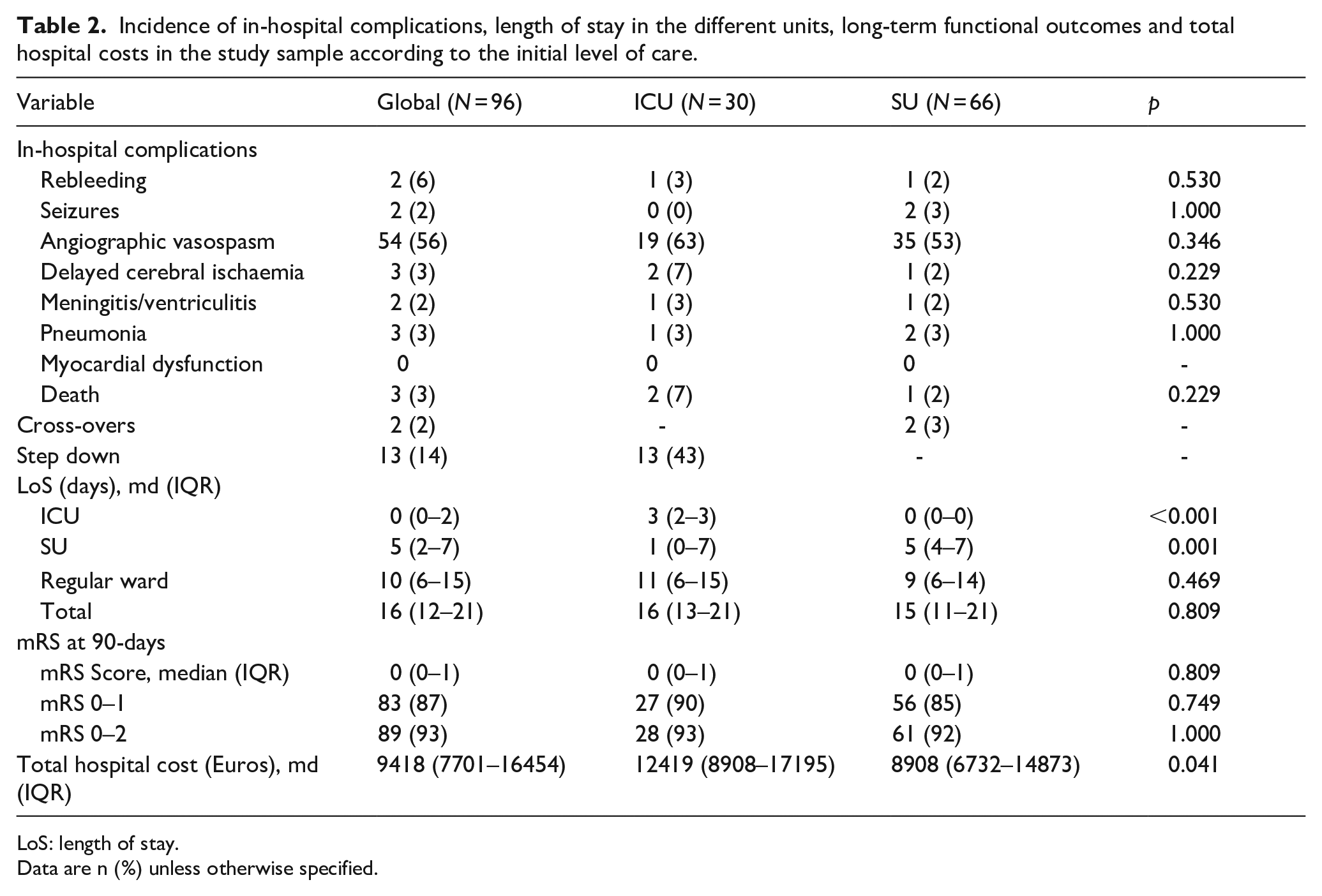
LoS: length of stay.
Data are n (%) unless otherwise specified.
Stratification analysis according to the bleeding pattern
An additional subgroup analysis was performed to investigate whether a perimesencephalic or a diffuse bleeding pattern accounted for more unfavourable outcomes if managed in the SU instead of the ICU. Patients with a focal bleeding pattern were excluded from the stratification analysis. According to our data, functional outcomes were similar regardless of the unit of initial admission (Table 3). No significant differences were seen in the incidence of complications between these two differential bleeding patterns.
Stratification analysis by type of subarachnoid bleeding pattern; perimesencephalic or diffuse.
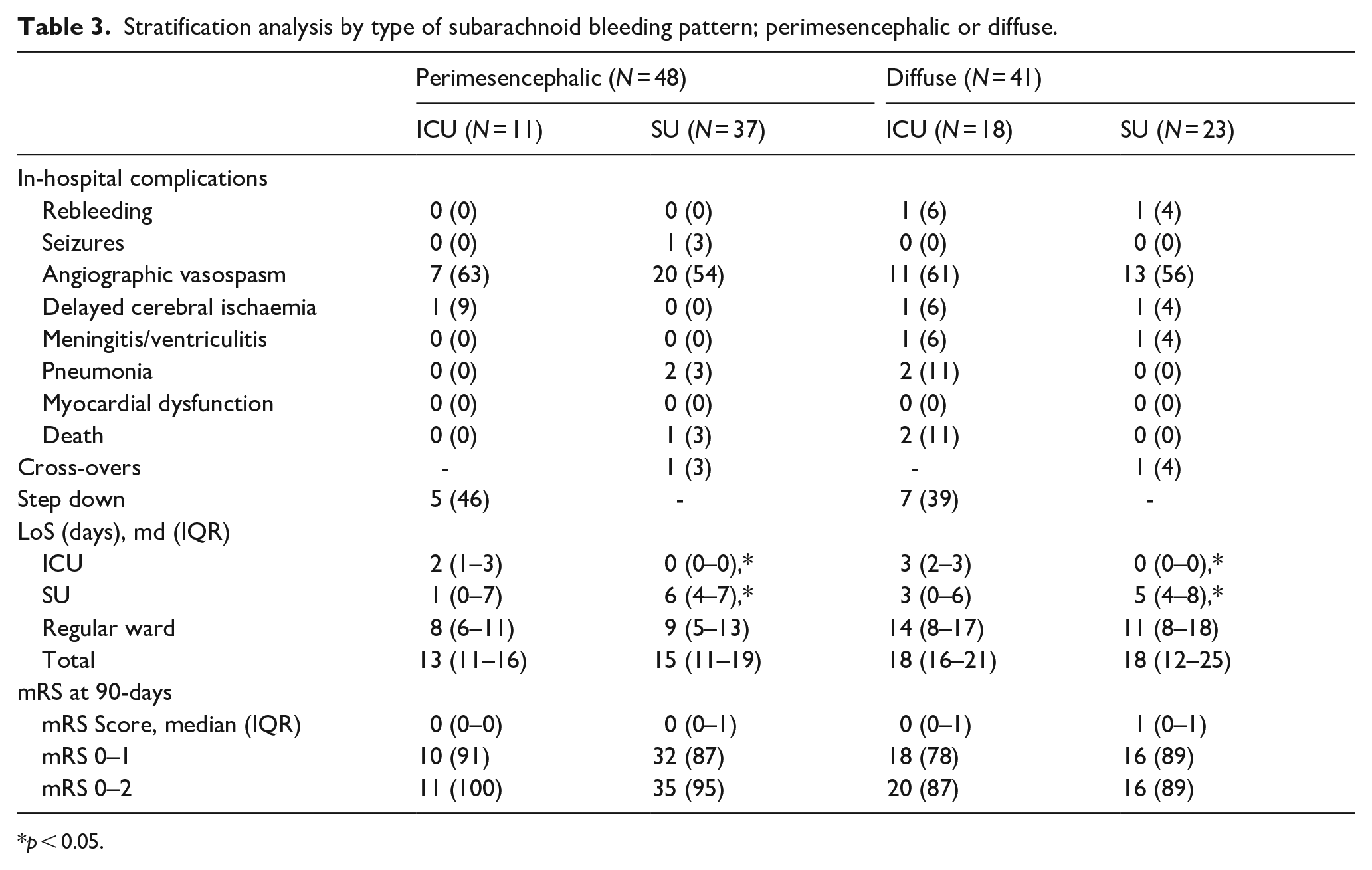
p < 0.05.
Cost-minimisation analysis
Given that the safety and effectiveness of the two management options were similar, a cost-minimisation analysis was performed, taking into account all the transitions across the different level-of-care units, from hospital admission to discharge. The analysis showed that the straight to the SU model was more cost-effective than the ICU model, with total hospitalisation costs being significantly lower in the SU group [median (IQR) 8908 (6732–14,873)] compared to the ICU group [median (IQR)12,419 (8908–17,195)] (p = 0.041) (Table 2).
Discussion
In this study we analysed the incidence of in-hospital complications and long-term clinical outcomes in low-grade naSAH patients according to two alternatives of specialised management in the acute stage: initial admission to an intensive care unit versus initial admission to an intermediate care unit in the SU. Two major findings can be highlighted. First, in our cohort, initial admission to the SU was similar to that of the ICU in terms of the incidence of complications and favourable short- and long-term clinical outcome. Second, a cost-minimisation analysis resulted in a reduction of hospitalisation-related costs favouring the straight to the SU protocol.
Non-aneurysmal SAH accounts for a non-negligible percentage (10%–20%) of all spontaneous SAH cases. Although conventionally considered a more benign form of SAH,11–14 the concern about the risk of paradigmatic complications of SAH usually leads to long hospitalisation periods. According to clinical guidelines, customary management involves admission to the ICU and subsequent transfer to a regular neurological or neurosurgical ward once the high-risk period of complications is over. However, given the natural history of the disease, the high-risk period could cover up to 2 or 3 weeks, meaning that patients could either be held in the ICU for a significant length of stay or prematurely moved to a conventional level of care, depending on the logistic resources and the healthcare overload at each institution. To alleviate expenses and optimise health resources, the SU has emerged as a feasible alternative to the ICU, either in the form of a step-down unit between the ICU and the regular ward, or as a first-line unit of admission. 5 Similarly to the ICU, the SU provides close clinical and neurological checks, serial assessment of angiographic vasospasm, and a variable degree of advanced support measures, like external ventricular drain (EVD) care, invasive arterial pressure monitoring or basic intravenous antihypertensive/vasopressor therapy, as appropriate.
In our tertiary hospital, a novel protocol was implemented in April 2013, thorough which the multidisciplinary management of low-grade SAH patients could be offered directly in the SU. The protocol included WFNS 1–2 patients with both treated and untreated ruptured intracranial aneurysms, as well as naSAH. Given the restricted respiratory and haemodynamic support that can be provided in the SU, in our centre only low-grade SAH were considered for the straight to the SU protocol, and a possibility of a rapid step-up into the ICU should always be available. Preliminary data suggest that the SU may provide similar in-hospital and long-term outcomes to the ICU in low-risk aneurysmal SAH patients, although the implications of the initial level-of-care unit of admission in hospital costs is unknown.21,22 Herein we found that in low-grade naSAH, the incidence of complications and the rate of good clinical outcome at 90 days were similar regardless of initial admission to the SU or ICU. These data build up on the idea of equal effectiveness and safety between the SU and the ICU previously observed in low-grade aneurysmal SAH also for naSAH. In this cohort of low-grade naSAH patients, given the reduced LoS in the ICU of the subgroup of patients initially admitted to the SU, the cost-minimisation analysis revealed a significant monetary saving in favour of the straight to the SU strategy. Importantly, the SU works with fewer human and material resources than the ICU, resulting in reduced direct and indirect costs. To the best of our knowledge, there are no other studies evaluating the advantages of SU care specifically for naSAH, and therefore an external comparison of our results could not be performed. Initiatives like ours aimed at reducing the LoS of SAH patients in ICU resources have recently been explored. Indeed, the implementation of rapid step-down or fast-track hospitalisation protocols for selected SAH patients without active complications has been shown to be feasible and safe, to reduce the ICU LoS, and to provide direct and indirect financial benefits.20,23
In this study, we also explored whether the bleeding pattern could modify the safety profile of the SU, based on previous reports claiming that patients with a diffuse pattern of bleeding show more aggressive behaviour than the perimesencephalic ones.15,16,24In our series, the incidence of complications was globally similar to those reported in general for naSAH 12,24–26; nonetheless, the more aggressive clinical course described for non-perimesencephalic (diffuse) SAH was not replicated here.15,16,24 This could be explained by the fact that only low-grade naSAH were included in our analysis.
Two major limitations can be attributed to our study. First, the comparison of effects between SU and ICU was performed on the basis of a retrospective non-randomised design, therefore a definite conclusion of equal outcome metrics cannot be extracted from our analysis. Indeed, a tendency to prioritise the ICU as the initial unit of admission for WFNS grade 2 patients was evident in our sample. Second, the study was conducted in a single third-level centre that has extensive experience managing SAH patients in both the ICU and SU. Consequently, the results can only be safely extrapolated to centres with similar clinical workflows. As an additional limitation, the economic evaluation in this study was performed in a cost-per-day basis rather than the increasingly used Diagnosis-Related Group (DRG) system. The DRG system reimburses hospitals based on the International Classification of Diseases diagnosis regardless of the initial level of care unit of hospital admission. Further studies are warranted to evaluate alternative methods for the assessment of the potential economic impact of a straight to the SU protocol. Overall, these limitations warrant further evaluation of the topic in prospective multicentre cohorts, preferably under randomised conditions, although such studies would be very difficult to organise in centres without dedicated SU with expertise in the management of SAH patients.
Conclusions
In low-grade (WFNS 1–2) non-aneurysmal spontaneous SAH, initial admission to a Stroke Unit might be comparable to an Intensive Care Unit in terms of the incidence of acute complications and the rates of favourable long-term clinical recovery. In monetary terms, a cost-minimisation analysis favoured the straight to the SU protocol. Although this study provides valuable insights into the management of low-grade non-aneurysmal spontaneous SAH and the potential benefits of a straight to the SU protocol, further evaluation is required to confirm our findings.
Supplemental Material
sj-docx-1-eso-10.1177_23969873231202361 – Supplemental material for Stroke Unit as an alternative to Intensive Care Unit for initial hospital admission of low-grade non-aneurysmal subarachnoid haemorrhage: A safety and cost-minimisation analysis
Supplemental material, sj-docx-1-eso-10.1177_23969873231202361 for Stroke Unit as an alternative to Intensive Care Unit for initial hospital admission of low-grade non-aneurysmal subarachnoid haemorrhage: A safety and cost-minimisation analysis by Daniel Santana, Alejandra Mosteiro, Laura Llull, Miquel Massons, Luigi Zattera, Gabriel Pujol-Fontrodona, Mariano Werner, Ramón Torné, Sergio Amaro and Angel Chamorro in European Stroke Journal
Footnotes
Acknowledgements
DS was supported by a grant from Hospital Clinic de Barcelona (Contracte Clínic de Recerca Emili Letang-Josep Font).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The study protocol was approved by the institutional Clinical Research Ethics Committee (HCB/2022/1117).
Informed consent
Informed consent was not sought for the present study, because it involved analysis of existing anonymised data, thereby maintaining participant confidentiality.
Guarantor
SA.
Contributorship
DS, AM and LLL researched literature, gained ethical approval, analysed the data and wrote the initial draft. MM, LZ, GP and MW participated in patient recruitment. RT, SA and AC were involved in protocol development and gave critical feedback to the initial draft. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
