Abstract
Background:
Previous studies have revealed that remote ischemic conditioning (RIC) may have a neuroprotective function. However, the potential benefit of RIC for patients with ICH remain unclear.
Objective:
The primary aim of this study is to assess the safety and efficacy of RIC for patients with ICH.
Methods:
The Safety and Efficacy of RIC for Spontaneous ICH (SERIC-ICH) is an ongoing prospective, randomized, multicenter, parallel-controlled, and blinded-endpoint clinical trial. The study will enroll an estimated 2000 patients aged ⩾18 years within 24 h after ICH onset, with National Institutes of Health Stroke Scale ⩾6 and Glasgow Coma Scale ⩾8 upon presentation. The patients will be randomly assigned to the RIC or control groups (1:1) and will be treated with cuffs inflated to a pressure of 200 or 60 mmHg, respectively, twice daily for 7 days. Each RIC treatment will consist of four cycles of arm ischemia for 5 min, followed by reperfusion for another 5 min, for a total procedure time of 35 min. The primary efficacy outcome measure is the proportion of patients with good functional outcomes (modified Rankin scale 0–2) at 180 days. The safety outcome measures will include all adverse events and severe adverse events occurring in the course of the study.
Discussion:
RIC is an inexpensive intervention and might be a strategy to improve outcomes in patients with ICH. The SERIC-ICH trial will investigate whether RIC treatment can be applied as an adjuvant treatment in the acute phase of ICH and identify safety issues.

Introduction and rationale
Spontaneous intracerebral hemorrhage (ICH) is a life-threatening and disabling subtype of stroke, which has global importance and a poor patient prognosis.1,2 One in three patients die within 30 days after ICH onset, and two-thirds of survivors show varying degrees of severe disability. 2 In patients who have large hematomas, surgery or minimally invasive surgery can be selected for treatment.3,4 However, for patients whose hematoma volume has not reached the surgical threshold, conservative treatment options remain limited.5–7 Notably, a large proportion of these patients have a poor prognosis and residual functional disability. Therefore, novel treatment approaches are needed for these patients with ICH. 1
Remote ischemic conditioning (RIC) is an intervention developed from research in cardiovascular disease. During RIC, brief doses of reversible episodes of ischemia and reperfusion are applied to a distant vascular bed, tissue, or organ to promote protective functions in remote tissues and organs. 8 The neuroprotective effect of RIC has been demonstrated in both experimental and clinical studies of cerebrovascular diseases.9–13 For patients with ICH, a previous proof-of-concept randomized controlled trial (RICH-1) found that RIC improved the hematoma resolution rate and reduced relative perihematomal edema. However, whether RIC can be used as an adjunct treatment to improve functional outcomes in patients with ICH remains unclear.
Therefore, we designed the Safety and Efficacy of Remote Ischemic Conditioning for Spontaneous Intracerebral Hemorrhage (SERIC-ICH) trial to explore the safety and efficacy of RIC in patients with acute ICH who receive conservative therapy.
Methods
Design
The SERIC-ICH trial is a prospective, randomized, multicenter, parallel-controlled, blinded-endpoint clinical trial conducted in China that aims to test the hypothesis that 7 days of RIC therapy promotes a neuroprotective function at 180 days and has good safety and tolerance in patients with ICH when applied within 24 h of symptom onset. The study design was registered (ClinicalTrials.gov identifier: NCT05609110) and approved by the Ethics Committee of the First Hospital of Jilin University (18K003) and by the ethics committees of each participating center. Standard medical management care for all enrolled patients are based on international guidelines. 14 RIC will be performed for 7 days after randomization. Cranial computed tomography (CT) will be performed at baseline, 24 h, and 7 days after randomization. The National Institutes of Health Stroke Scale (NIHSS) and Glasgow Coma Scale (GCS) scores will be assessed at baseline, 24 h, and 7 days after randomization by trained examiners blinded to the treatment assignment. The modified Rankin scale (mRS) score will be evaluated 90 and 180 days post-randomization in structured telephone interviews using a validated questionnaire. The mRS score ranges from 0 to 6, with lower values representing a better outcome.15,16 The flowchart of the study is shown in Figure 1.
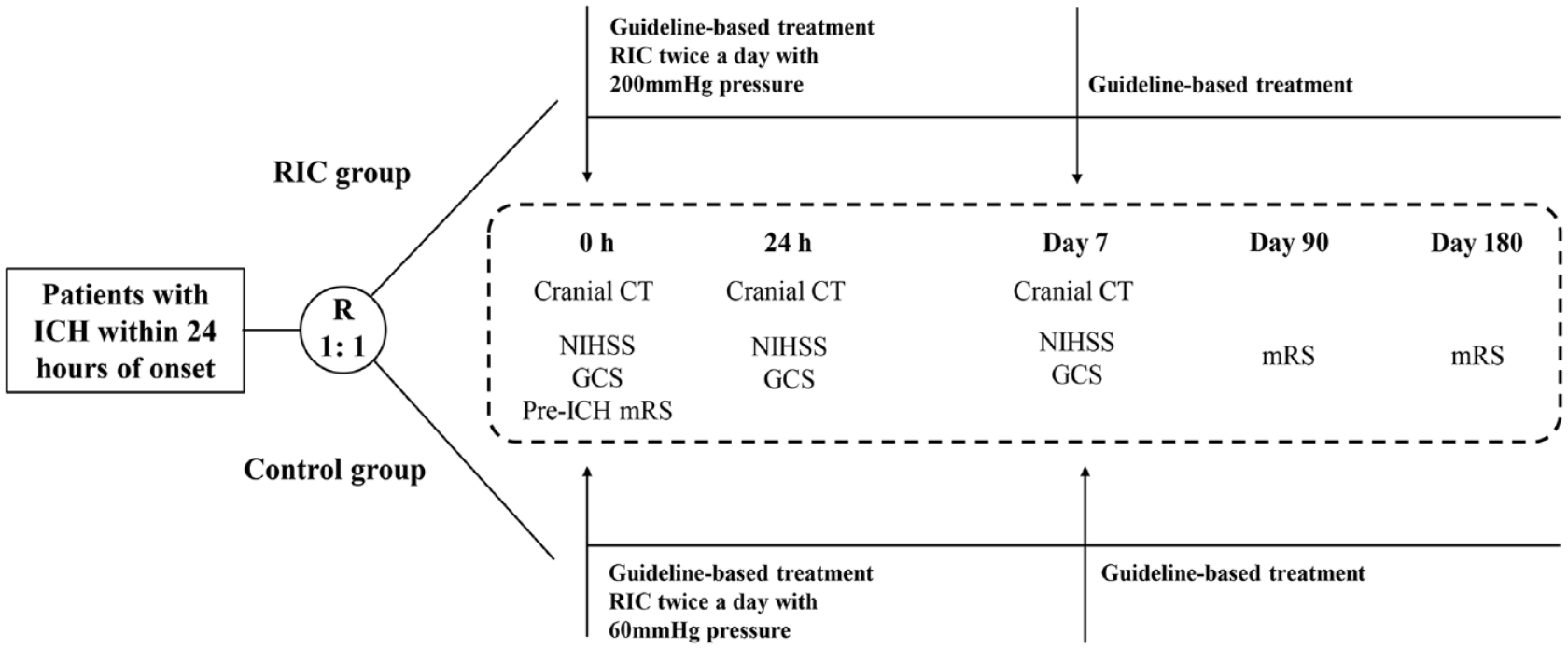
The flow of the trial.
Patient population
A total of 2000 patients with acute ICH within 24 h from onset will be enrolled in the trial according to the study criteria described in Table 1.
The inclusion and exclusion criteria.
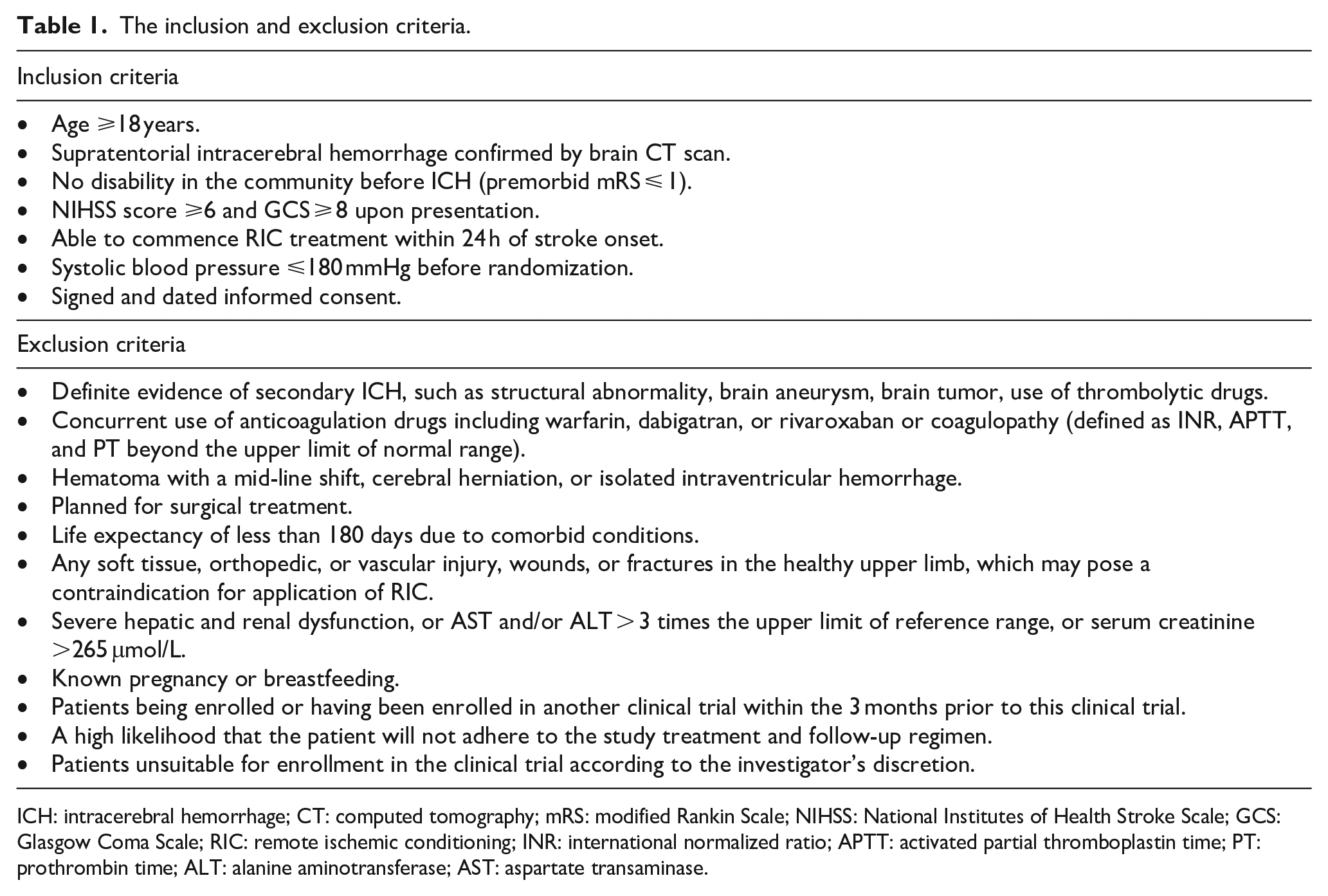
ICH: intracerebral hemorrhage; CT: computed tomography; mRS: modified Rankin Scale; NIHSS: National Institutes of Health Stroke Scale; GCS: Glasgow Coma Scale; RIC: remote ischemic conditioning; INR: international normalized ratio; APTT: activated partial thromboplastin time; PT: prothrombin time; ALT: alanine aminotransferase; AST: aspartate transaminase.
Randomization
Eligible participants will be randomly assigned to the RIC or control groups in a 1:1 ratio using a block randomization method with randomized blocks (4, 6, and 8) stratified by the centers. Randomization will be performed using a 24-h password-protected web-based program.
RIC procedure
After randomization, participants in both groups will undergo RIC treatment twice daily for 7 consecutive days using an automated device (BB-RIC-D5/LAPUL Medical Devices Co., Ltd, Beijing, China) placed on the upper arm of the unaffected side. Each RIC will consist of four cycles of arm ischemia with cuffs inflated to a pressure of 200 mmHg (RIC group) or 60 mmHg (control group) for 5 min, followed by reperfusion for another 5 min, for a total procedure time of 35 min. Hospital-based nurses will perform the RIC procedure.
Outcomes
The primary efficacy outcome measure is the proportion of patients with good functional outcomes, defined as mRS 0–2 at 180 days. The secondary efficacy outcome measure is the proportion of patients with mRS 0–2 at 90 days; an ordinal shift of the full range of mRS scores at 90 and 180 days; hematoma expansion at 24 h; NIHSS score at 7 days. Hematoma expansion is defined as an ICH volume increase of >6 mL or >33% as determined by image analysis of CT scans taken at 24 h compared with the baseline CT scan.17,18
Safety outcome measures include all adverse events and severe adverse events occurring in the course of the study. RIC-related adverse events include redness or swelling of the arms, skin petechiae on the arms, bruising, palpitations, dizziness, nausea, and headache not present at the beginning of the study.12,19
Data and safety monitoring
A Data and Safety Monitoring Board (DSMB) has been established to monitor the progress of the trial, perform data check every 6 months, and ensure patient safety, independently. The DSMB consists of clinicians and biostatisticians, and it can terminate the study unconditionally.
Sample size estimates
In the previous INTERACT3 study, approximately 42.7% of patients administered guideline-recommended treatment presented with good functional outcomes at 180 days (mRS 0–2). 20 The RICH-I study showed good functional outcomes (mRS 0–2) in 55% of patients in the RIC group (200 mmHg) at 90 days compared to 45% of patients in the control group (60 mmHg), indicating that RIC improved the proportion of patients with good functional outcomes by 10% at 90 days. 21 In this study, we estimated that RIC would improve the proportion of patients with ICH by 7% at 180 days. A sample size of 833 in each group would provide 80% power to detect a difference of 7% between the group proportions of 50% and 43% at a significance level (alpha) of 0.05, using a two-sided z-test with a continuity correction. These results assume that two sequential tests are performed after 50% and 100% of the subjects have completed follow-up, using the O’Brien-Fleming spending function to determine the test boundaries. To account for the dropout rate and maximize the efficiency of the study, we will recruit 2000 participants, with 1000 patients assigned to each group.
Statistical analyses
All statistical analyses will be performed using SPSS software version 26 (IBM) and R software version 4.1.3 (R Foundation for Statistical Computing). Efficacy outcomes will be analyzed using an intention-to-treat analysis set (ITT; all randomized patients). Safety outcomes will be analyzed in the safety analysis set (patients who received at least one RIC treatment).
Differences in outcome measures (categorical variables), including the proportions of patients with mRS 0–2 at 90 and 180 days and the proportions of patients with hematoma growth at 24 h, will be compared between the two groups using modified poisson regression. Scores of mRS at 90 days and 180 days will be analyzed using ordinary logistic regression. NIHSS score at 7 days will be analyzed using linear regression models. An interim analysis will be performed after 50% of subjects have been followed up completely. All tests will be two-tailed, with statistical significance set at p < 0.05, unless other specifications are stated. The exact list of all analyses to be performed was specified in the full Statistical Analysis Plan (Supplemental Material).
Current status
This study was registered on November 8, 2022, and recruitment began on February 2, 2023. At the time of the first submission of this article (July 17, 2023), 185 patients had been recruited. Recruitment will continue until a complete sample size is achieved, which is expected by June 2025 at the latest.
Discussion
The SERIC-ICH trial will investigate the safety and efficacy of RIC for patients with ICH.
Hematoma volume is an independent determinant of functional outcomes in patients with ICH, and the dynamics of perihemorrhagic edema and its peak volume have been verified to cause secondary injury after ICH.22,23 Therefore, clot resolution and perihematomal edema are considered therapeutic targets in ICH research. In previous studies, RIC significantly increased the hematoma resolution rate via AMPK-dependent immune regulation and reduced relative perihematomal edema.21,24 Hence, RIC may have a therapeutic effect on the outcomes of patients with ICH. However, whether RIC can improve the functional outcomes of patients with ICH remains unclear. Further studies are warranted, and the SERIC-ICH study will determine whether this is the case.
We selected patients with ICH within 24 h of symptom onset. One reason for this is that perihematomal edema is initiated in the acute phase and immediately after ICH onset. 25 Thus, the earlier that RIC is performed, the more significantly the increase in perihematomal edema may be inhibited, which may result in better outcomes. Another reason is that the secondary aim of the SERIC-ICH trial is to explore the effects of RIC on hematoma expansion. As nearly 100% of hematoma expansion after ICH occurs within 24 h of symptom onset, 26 we chose a time window of 24 h. Hematoma expansion is widely considered to be closely associated with increased blood pressure in patients with ICH. 18 The effect of RIC on lowering blood pressure has also been verified.27,28 Thus, we anticipate that the study will establish that RIC is effective in reducing hematoma expansion. In addition, we will strictly focus on safety outcome measures during follow-up because no study has performed RIC within 24 h in patients with ICH.
Summary and conclusions
RIC is an inexpensive intervention and may be a strategy to improve the functional outcomes in patients with ICH. SERIC-ICH will investigate whether RIC treatment can be applied as an adjuvant treatment in the acute phase of ICH and identify any safety issues.
Supplemental Material
sj-pdf-1-eso-10.1177_23969873231201712 – Supplemental material for Safety and efficacy of remote ischemic conditioning for spontaneous intracerebral hemorrhage (SERIC-ICH): A multicenter, randomized, parallel-controlled clinical trial study design and protocol
Supplemental material, sj-pdf-1-eso-10.1177_23969873231201712 for Safety and efficacy of remote ischemic conditioning for spontaneous intracerebral hemorrhage (SERIC-ICH): A multicenter, randomized, parallel-controlled clinical trial study design and protocol by Zhen-Ni Guo, Yang Qu, Reziya Abuduxukuer, Peng Zhang, Lijuan Wang, Ying Liu, Rui-Hong Teng, Jian-Hua Gao, Feng Jin, Hai-Feng Wang, Yu Cao, Yong-Quan Xue, Jun-Feng Zhao, Magdy H Selim, Thanh N Nguyen and Yi Yang in European Stroke Journal
Footnotes
Acknowledgements
We are very grateful to the Data and Safety Monitoring Board for their contributions to this study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was supported by the National Natural Science Foundation of China (Grant No. 82071291), the Norman Bethune Health Science Center of Jilin University (2022JBGS03), Science and Technology Department of Jilin Province (YDZJ202302CXJD061, 20220303002SF), and Jilin Provincial Key Laboratory (YDZJ202302CXJD017) to YY, and the Science and Technology Department of Jilin Province (YDZJ202201ZYTS677), and Norman Bethune Program of Jilin University (2022B02) to ZNG, and Graduate Innovation Fund of Jilin University (2023CX113) to YQ.
Ethical approval
This study involves human participants and was approved by the Ethics Committee of the First Hospital of Jilin University (18K003).
Informed consent
The authors declare that they consent for publication.
Guarantor
Yi Yang
Contributorship
YY, ZNG, MHS, and TNN contributed to the design of the study and contribute to its oversight. YQ, RA, PZ, LW, YL, RHT, JHG, FJ, HFW, YC, YQX, and JFZ coordinated the study. ZNG and YQ wrote the first draft of the manuscript, which was edited by all other authors. All authors revised the manuscript for relevant scientific content and approved the final version of the manuscript.
Trial registration
ClinicalTrials.gov identifier: NCT05609110.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
