Abstract
Purpose:
To propose a consensus-based definition and framework for motor rehabilitation after stroke.
Methods:
An expert European working group reviewed the literature, attaining internal consensus after external feedback.
Findings:
Motor rehabilitation is defined as a process that engages people with stroke to benefit their motor function, activity capacity and performance in daily life. It is necessary for people with residual motor disability whose goal is to enhance their functioning, independence and participation. Motor rehabilitation operates through learning- and use-dependent mechanisms. The trajectory of motor recovery varies across patients and stages of recovery. Early behavioral restitution of motor function depends on spontaneous biological mechanisms. Further improvements in activities of daily living are achieved by compensations. Motor rehabilitation is guided by regular assessment of motor function and activity using consensus-based measures, including patient-reported outcomes. Results are discussed with the patient and their carers to set personal goals. During motor rehabilitation patients learn to optimize and adapt their motor, sensory and cognitive functioning through appropriately dosed repetitive, goal-oriented, progressive, task- and context-specific training. Motor rehabilitation supports people with stroke to maximize health, well-being and quality of life. The framework describes the International Classification of Functioning, Disability and Health in the context of stroke, describes neurobiological mechanisms of behavioral restitution and compensation, and summarizes recommendations for clinical assessment, prediction tools, and motor interventions with strong recommendations from clinical practice guidelines (2016–2022).
Conclusions:
This definition and framework may guide clinical educators, inform clinicians on current recommendations and guidelines, and identify gaps in the evidence base.
Introduction
Stroke is the third leading cause of death and disability worldwide 1 and a leading cause of adult disability in Europe. 2 Forecasts for Europe from 2017 to 2047 predict a 17% decrease in stroke mortality, but a 27% increase in stroke prevalence. 2 These trends are expected to increase demand for stroke rehabilitation services.
The European Stroke Organisation (ESO) has released a European Stroke Action Plan (ESAP) 3 and defined 30 targets and 72 research priorities within seven domains to improve stroke services. One of these domains is stroke rehabilitation for improving management, outcome and quality of life after stroke in 2030. 3 Rehabilitation was defined following the WHO as “a set of measures that assist individuals, who experience or are likely to experience disability, to achieve and maintain optimal functioning in interaction with their environments.” 4 This general definition encompasses several neurological domains such as motor function, cognition and communication, but specific principles of motor rehabilitation were not addressed.
This article presents a definition of “motor rehabilitation” developed by expert consensus. The agreed definition is supported by a framework that synthesizes key literature to provide a state-of-the-art overview of the stroke motor rehabilitation domain. This framework is intended to guide educators who train stroke rehabilitation clinicians, to update clinicians about current recommendations and guidelines, and to enable researchers to identify gaps in the evidence base.
Development of a consensus-based definition of motor rehabilitation after stroke
The ESO Guideline Board invited a panel of 16 experts to collaborate on a definition of post-stroke motor rehabilitation, using standard ESO operating procedures. 5 The panel engaged in a three-round process. The first round consisted of online meetings in April and May 2022 where the panel agreed to structure the definition as a paragraph. A first draft of the definition paragraph was presented to panelists through an online survey, and panelists had the options to agree, suggest changes, request additional elements, or disagree with the paragraph. Results were collated (available from GV) and presented to the expert panel for discussion and revision at the end of June 2022. A second-round survey was held in July–August 2022, and panelists could agree or disagree with each part of the definition. A 75% agreement threshold was defined a priori for acceptance of each part of the definition, similar to formal Delphi processes.6,7 Results were collated (also available from GV) and presented in September to the expert panel, with further discussion and fine-tuning. The topic sentence at the beginning of the definition paragraph reached 91.7% agreement. The next three supporting sentences provide further explanation of the concept and reached 75%, 91.7%, and 81.8% agreement, respectively. The final concluding sentence reached 91.7% agreement. Along with the definition, a separate glossary related to motor rehabilitation was compiled (Box 1).
Glossary and definitions.

Expert panel members presented the agreed definition to a convenience sample of clinicians working in stroke rehabilitation through in-person and online consultation. Feedback was collated (available from GV) and discussed by the expert group in December 2022 with fine-tuning of the definition. The final agreed definition on motor rehabilitation after stroke is presented in Box 2. The definition was also discussed with two persons with lived experience of stroke, who confirmed that all elements of the definition were highly relevant and important for motor rehabilitation. They emphasized including maximizing health, well-being and quality of life, and the need to communicate that patients can improve, even long-term after stroke.
Agreed, expert-based definition of motor rehabilitation after stroke.

The definition paragraph describes key concepts. Further details are needed when using the definition in education, clinical practice and research. We addressed this by developing a framework that elaborates on each component of the definition. This framework is presented below to contextualize the motor rehabilitation definition.
Motor rehabilitation after stroke framework
Overarching concept: The International Classification of Functioning, Disability and Health
The first sentence of the definition positions it with respect to the International Classification of Functioning, Disability and Health (ICF). The ICF conceptualizes interactions between body functions and structures, activities, and participation in relation to environmental and personal factors (Figure 1). 8 Body functions can be impaired, activities can be limited, and participation can be restricted. Assessments in the body functions domain evaluate physiological functions of body systems, such as muscle strength and synergies, coordination, pain, and muscle tone. Assessments in the activity domain evaluate the execution of tasks such as reaching and grasping, self-care, mobility and walking. The primary goal for many patients is to return to participation in their home environment and life roles. Therefore, assessments in the participation domain evaluate involvement in everyday life situations such as shopping, working, and socializing. Capacity and performance are two constructs used in the activity and participation domains. Capacity relates to what an individual can do in a standardized environment or test situation, while performance relates to what a person actually does in their habitual environment. 8 Environmental and personal factors can be barriers or facilitators of the person’s functioning. Figure 1 illustrates the ICF in the context of motor rehabilitation after stroke.
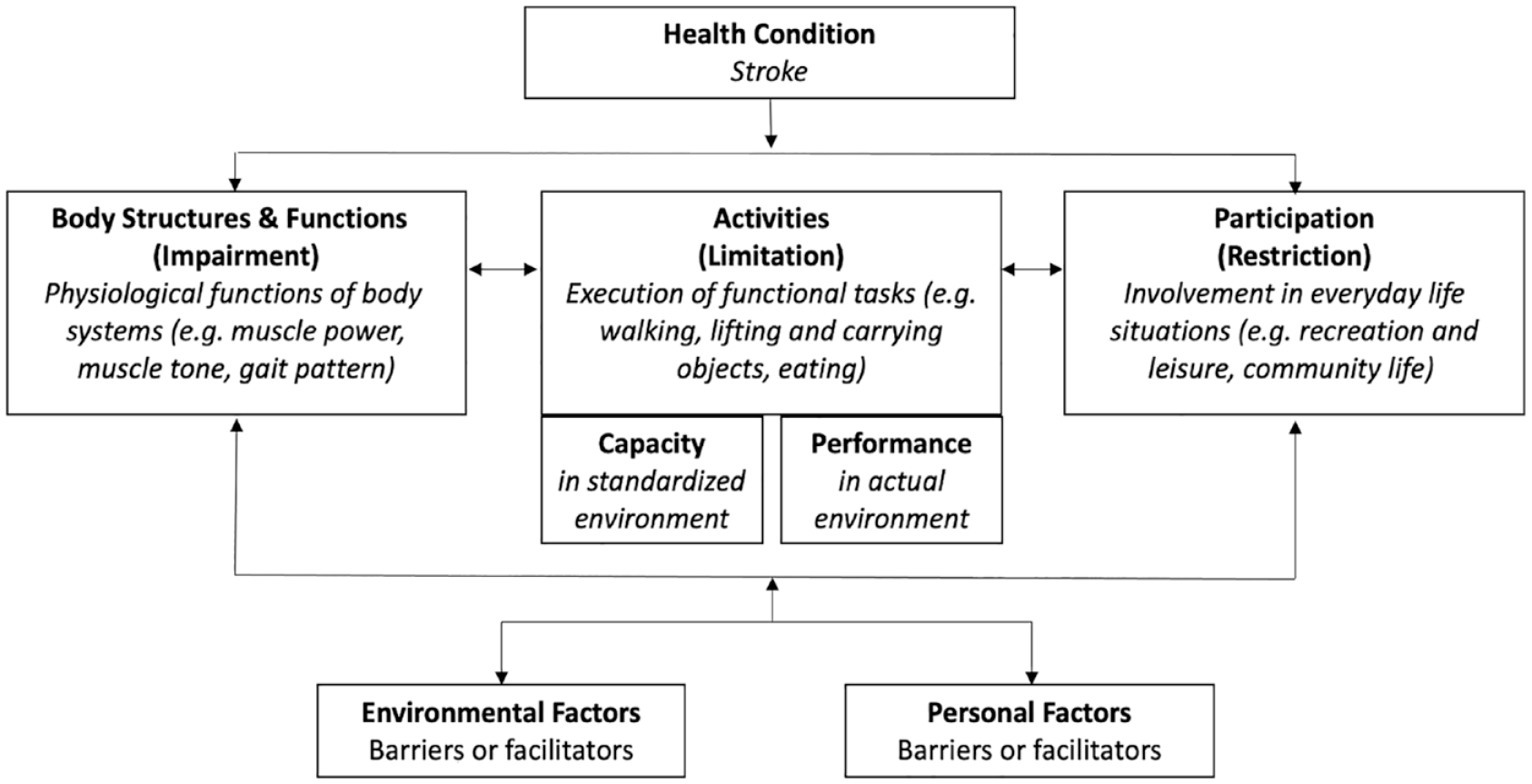
The international classification of functioning, disability and health. 8
Regaining motor function is critical for independence in activities of daily living, 9 which is associated with satisfactory quality of life. The concept of “Quality of Life” was not originally part of the ICF framework, however it has been defined by the World Health Organization 10 Quality of Life Group as an “individual’s perception of their position in life in the context of the culture and value systems in which they live, and in relation to their goals, expectations, standards and concerns” (p. 11). It can be thought of as an overarching concept related to all domains of the ICF. 11
It is important to note that the components of the ICF are interrelated in a non-linear way. An impairment in body function, such as leg muscle weakness, does not necessarily produce a limitation in walking or a restriction in participation. Furthermore, motor functions are closely related to and influenced by other body functions such as sensation, pain, cognition, mood and fatigue. All these aspects of functioning need to be considered in post-stroke motor rehabilitation.
Motor recovery
The second section of the definition reflects the biology of recovery and distinguishes between improvements at early versus later stages after stroke. Motor recovery probably occurs through a combination of spontaneous biological processes and “use-dependent” processes that include motor learning and skill acquisition.12,13 The interactions between spontaneous biological processes at molecular, cellular, and physiological levels and the mechanisms of learning in the first months post-stroke are still poorly understood. 13
Most stroke rehabilitation studies use the term “recovery” as a general expression of “change” or “improvement,” without distinguishing between behavioral restitution or compensation.12,14,15 In general, the term “motor recovery” indicates restitution of behavior after stroke, where movements or tasks are performed as they were before stroke. This “true recovery” is thought to reflect spontaneous biological recovery processes occurring during the initial days and weeks after stroke. In contrast, the term “compensation” encompasses performing movements or tasks with atypical movement patterns at the expense of movement quality, or through performing tasks with a different limb altogether. Adaptation and compensation are thought to result from motor learning processes that can continue indefinitely after stroke.
Findings from several longitudinal studies show that for most stroke patients spontaneous neurological recovery and behavioral restitution follow a logistic pattern that plateaus within the first 10 weeks post-stroke, regardless of their age or the type and amount of therapy they complete (Figure 2).9,16–18 Spontaneous neurological recovery is observed for the upper limb19–21 and lower limb,22,23 as well as somatosensory,24,25 visuospatial, 26 and language functions. 27 Patients with initially mild to moderate motor impairment typically exhibit early spontaneous neurological recovery of upper and lower limb motor functions. Some patients with initially more severe motor impairment can also exhibit early or slightly delayed spontaneous recovery, 21 while others remain severely impaired.17,20
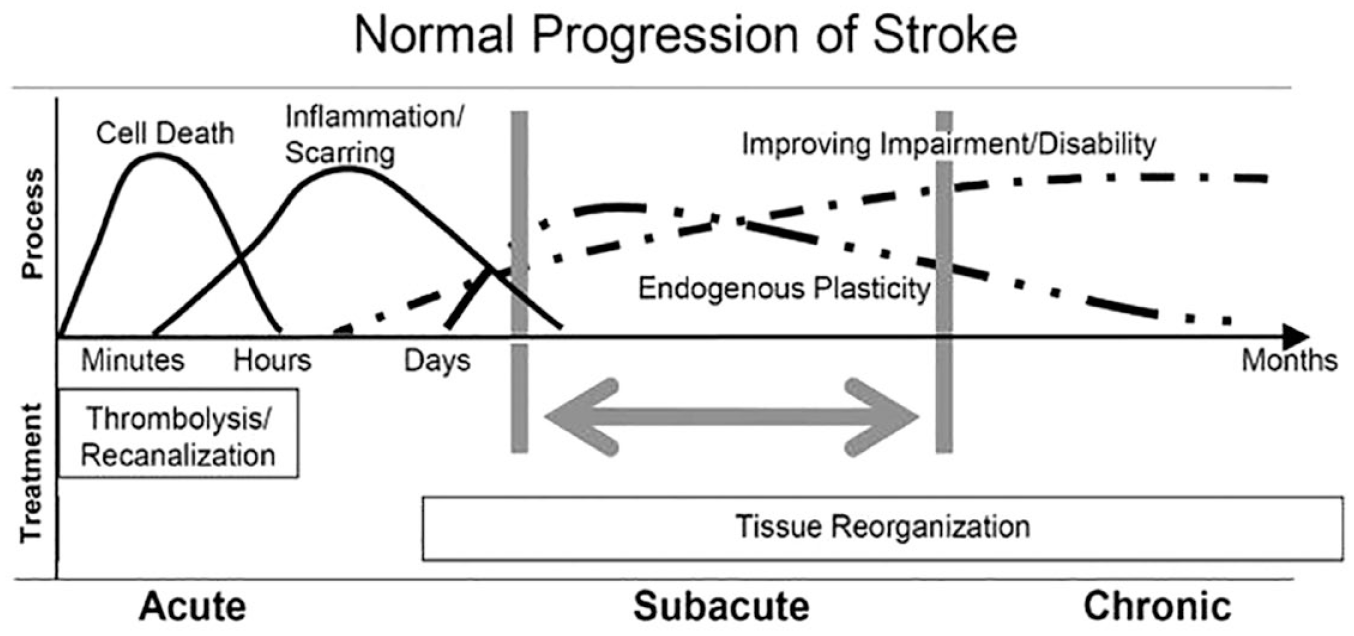
Patterns, processes and treatment opportunities post-stroke (adapted from Dobkin and Carmichael, 28 used with permission).
Spontaneous neurological recovery is initiated by the cascade of neurochemical processes resulting from focal ischemic brain injury. These involve bio-energetic failure leading to excitotoxicity and oxidative stress, mitochondrial failure, and ultimately to apoptosis and cell death. 29 These pathologic processes start within minutes after stroke onset, 30 expand quickly within hours, and may continue for days even if perfusion is restored. 29 Ultimately, the cascade of pathophysiological processes leads to permanent loss of neurons, microglia, astrocytes and endothelial cells in the infarcted area that includes damage to the blood-brain barrier as well as transsynaptic degeneration in remote, anatomically connected brain areas. 16
The mechanisms responsible for behavioral restitution are less well understood.13,31,32 The following time-restricted, overlapping processes contribute to behavioral restitution and compensation post-stroke.
Autoregulation of vascular collaterals can compensate for focal hypoperfusion and support the survival of penumbral tissue. A larger number of collaterals is associated with smaller final infarct volumes, faster and more extensive spontaneous neurological recovery in the first week post-stroke 33 and ultimately better outcomes. 34
Neuronal plasticity in perilesional areas is enhanced by a cascade of post-ischemic inflammation processes in the initial days and weeks post-stroke. At a cellular level, neuronal networks are (re)modeled by competition and selection in response to training and experience. 13 The sensitivity of cortical mapping with non-invasive serial neuroimaging 35 or transcranial magnetic stimulation36,37 techniques to these neuronal plasticity mechanisms is unclear.
Gradual peripheral effects can influence and constrain behavioral restitution and force patients to compensate for their motor performance after stroke. These include neural components such as spasticity, and mechanical effects such as loss of muscle volume and serial sarcomere number38,39 altering the length-tension relationship, 40 as well as higher passive stiffness in tendons and soft tissues.41,42
Behavioral restitution is driven by poorly understood mechanisms of spontaneous neurological recovery and plateaus within 10 weeks after stroke. 43 Unfortunately, there is currently insufficient evidence in humans that rehabilitation therapies interact with the biological mechanisms responsible for behavioral restitution.17,20 Fine-grained biomechanical measures of movement quality indicate that therapy-induced improvements are mainly adaptive, as patients learn to optimize the use of their limbs to accomplish a standardized task. For example, hand transport and orientation for grasping with the paretic upper limb is characterized by increased muscle synergies between shoulder abduction and elbow flexion.44,45 To compensate for the limitations imposed by these muscle synergies, patients learn to flex and rotate their trunk to expand their limited workspace and improve hand orientation.43,44,46 Similarly, faster walking speeds are achieved via compensation with the less-affected side post-stroke, and without significant improvements in paretic intralimb coordination. 47 Overall, the evidence indicates that improvements at the level of activities result from a combination of behavioral restitution of impaired body functions occurring mainly in the first 10 weeks post-stroke, 17 as well as learning to deal with residual impairments by using compensatory movement strategies. These findings support the growing point of view that motor rehabilitation interventions mainly help patients learn to optimize their performance by adaptation and compensation through appropriately dosed, repetitive, goal-oriented, progressive, task- and context-specific training. 48
Assessment of motor function and activity
The third section of the definition is supported by widely recommended motor assessments and prediction tools. Repeated motor assessments, preferably at specific timepoints, result in a better understanding of recovery. International consensus recommends early assessment within 1 week, followed by assessments at 4 weeks, and 3 and 6 months after stroke, as these times relate to key transitions in the biological processes underlying recovery. 16 Standardized assessments at specific times post-stroke provide transparency in stroke care pathways and allow comparison of stroke rehabilitation outcomes at the national, regional and global level. Aggregated standardized assessment data are also expected to reveal which interventions are effective in clinical practice, including information about dose, timing, and setting. 49
Recent evidence- and consensus-based recommendations for motor assessments after stroke have several common features and are summarized in Table 1. In clinical practice, motor assessment should be performed at least before and after rehabilitation programs, using standardized measures with sound psychometric properties and established validity, reliability, responsiveness, and interpretability.49–51 The Fugl-Meyer motor assessment, Action Research Arm Test, and 10 m walk test are commonly recommended for both clinical practice and research. Standardized measurement of upper limb movement quality using kinematic methods has also been recommended for both clinical practice 50 and research. 51
Recent consensus-based recommendations for motor assessments in stroke rehabilitation.

CAULIN: Clinical Assessment of Upper Limb in Neurorehabilitation; FMA: Fugl-Meyer Assessment; ARAT: Action Research Arm Test; BBT: Box & Block Test; CAHAI: Chedoke Arm and Hand Activity Inventory; WMFT: Wolf Motor Function Test; NHPT: Nine-Hole Peg Test; MI: Motricity Index; CMSA: Chedoke-McMaster Stroke Assessment; STREAM: Stroke Rehabilitation Assessment of Movement; FAT: Frenchay Arm Test; MAS: Modified Ashworth Scale; 10MWT: 10-Meter Walk Test; TUG: Timed Up & Go; BBS: Berg Balance Scale; SIS: Stroke Impact Scale; SRRR: Stroke Recovery and Rehabilitation Roundtable; NIHSS: National Institutes of Health Stroke Scale; UE: upper Extremity; LE: Lower Extremity; mRS: Modified Rankin Scale; EQ-5D: European Quality of Life 5 Dimensions.
Patients’ actual functioning should be assessed at different ICF levels, encompassing impaired motor function, limited activity capacity, and limited activity performance. 11 Patient-reported outcome measures relating to activity and participation should be considered to complement data collected by technology or clinician observation. 50
Agreed assessments are important for predicting and monitoring recovery and outcomes at different levels of the ICF. 8 Motor assessment results should be discussed with patients and their caregivers, together with assessments of other domains, such as cognition and communication, to establish a shared understanding of the patient’s current status. Assessment results can also be used to gauge the patient’s likely outcomes, and these expectations can be combined with the patient’s personal goals to agree on the rehabilitation plan. 55
The treatment plan for post-acute stroke patients is influenced by clinicians’ expectations of motor recovery and outcome.55,56 Patients with initially moderate to severe motor impairment are the most difficult to make accurate predictions for based on clinical assessments alone, yet accurate prognosis is most important for these patients. 57 Clinicians’ prognoses are a source of variation that can produce inequitable access to rehabilitation services.55,57 Variation can be reduced when clinicians use objective prediction tools that combine standardized assessments to predict an individual patient’s likely outcome. Prediction tools can be used to guide rehabilitation goal setting and tailor therapy, and doing so may improve rehabilitation efficiency.58,59
Several tools have been developed to predict motor activity after stroke. 55 Validated tools that predict outcomes at specific time points for individual patients are summarized in Table 2. Most predict the probability of achieving a categorical activity capacity outcome for the upper limb or walking, defined using recommended clinical assessments. There are also tools available that can be used to predict recovery trajectories at any time in the first year post-stroke, for upper limb activity capacity 60 and independence in ADLs. 61 Implementation, 62 clinical impact, 59 and long-term accuracy 63 have been evaluated for one tool that predicts upper limb activity outcome. 64 Impact studies are needed to investigate the clinical benefits of prediction tools, 65 and evidence-based strategies are needed to support their successful and sustainable implementation. 62
Cross-validated tools that predict outcomes at specific time points for individual patients.
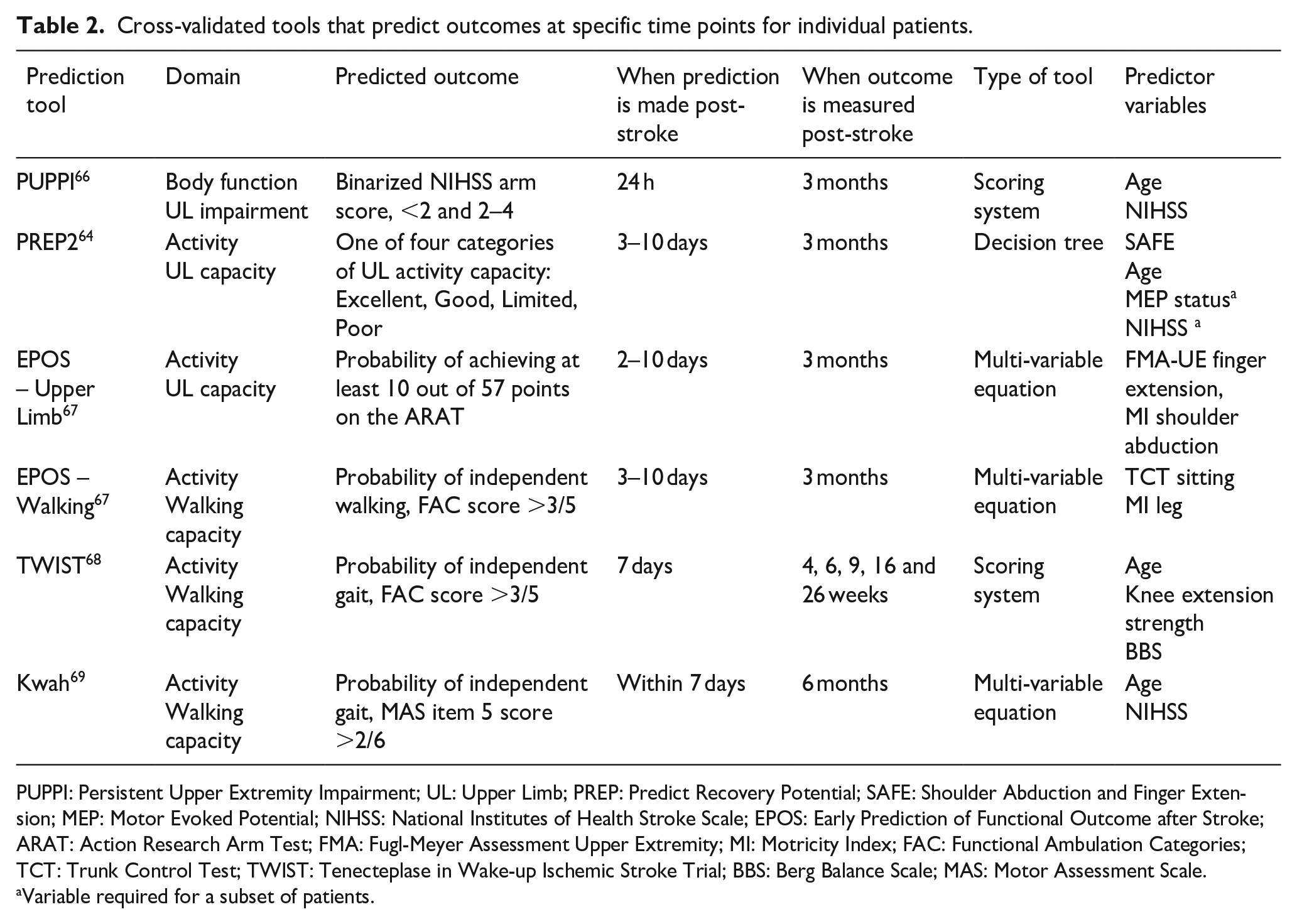
PUPPI: Persistent Upper Extremity Impairment; UL: Upper Limb; PREP: Predict Recovery Potential; SAFE: Shoulder Abduction and Finger Extension; MEP: Motor Evoked Potential; NIHSS: National Institutes of Health Stroke Scale; EPOS: Early Prediction of Functional Outcome after Stroke; ARAT: Action Research Arm Test; FMA: Fugl-Meyer Assessment Upper Extremity; MI: Motricity Index; FAC: Functional Ambulation Categories; TCT: Trunk Control Test; TWIST: Tenecteplase in Wake-up Ischemic Stroke Trial; BBS: Berg Balance Scale; MAS: Motor Assessment Scale.
Variable required for a subset of patients.
Standardized assessments and prediction tools are expected to inform shared decision-making and goal-setting between clinicians, patients, and families. They can be used to guide the rehabilitation plan so that it maximizes the patient’s chances of the best possible outcome. Standardized assessment of motor function at baseline is an essential element of all available prediction tools and needs to be repeated during recovery to understand progress toward predicted outcomes. Standardized assessment schedules are therefore required for consistently effective rehabilitation planning and monitoring.
Motor rehabilitation interventions
The fourth section of the definition is supported by strongly recommended interventions from recent motor rehabilitation guidelines. National clinical practice guidelines for stroke rehabilitation were reviewed to summarize the current evidence-base on interventions targeting motor rehabilitation. We included guidelines written in English and Dutch since three of the core writing group members were Dutch-speaking. Guidelines needed to contain a section that specifically addressed “rehabilitation after stroke” to be considered. An overview of existing stroke guidelines was recently published summarizing “strong” recommendations for the broad field of stroke care. 70 The definition and framework here focus on motor rehabilitation and summarize motor-specific recommendations.
Five high-quality, evidence-based clinical practice guidelines published between 2016 and 202271–76 were reviewed to identify common strong recommendations. The Australian and New Zealand guidelines 75 were developed using the GRADE methodology (Grading of Recommendations, Assessment, Development and Evaluation) 77 and strong recommendations mean that the evidence supports a clear balance toward a desirable effect. The Canadian guidelines 73 defined strong recommendations as evidence from a meta-analysis of randomized controlled trials or consistent findings from two or more randomized controlled trials. The American guidelines 76 made strong recommendations based on multiple randomized clinical trials or meta-analyses. The National Clinical Guidelines of the UK 72 did not adopt a hierarchical grading system for the “strength” of recommendations. Instead, a formal consensus approach was used to identify the key recommendations in terms of their wider impact on stroke. Last, the guidelines from the Netherlands71,74 made strong recommendations based on RCTs with good methodological quality, sufficient size and consistency, 78 and systematic reviews with at least some RCTs that met the aforementioned criteria.
Interventions for motor rehabilitation with strong recommendations were extracted independently by two researchers (BE, MMN), compiled, and discussed in case of disagreement. Recommendations “in favor of” that were included in at least three of the screened guidelines are presented in Table 3, providing an overview of strongly recommended interventions for the motor rehabilitation domain with international consensus. The recommendations presented in Table 3 cover: (i) timing of rehabilitation delivery, (ii) general principles of motor control and motor learning, (iii) interventions for functions and activities involving the lower limb, postural control, and walking and (iv) upper limb functions and activities. Guidelines and recommendations are continuously updated based on available evidence, and therefore it is important for clinicians to consult recent and updated or living guidelines.
Overview of motor interventions with strong recommendations in at least three international clinical practice guidelines published between 2016 and 2022.
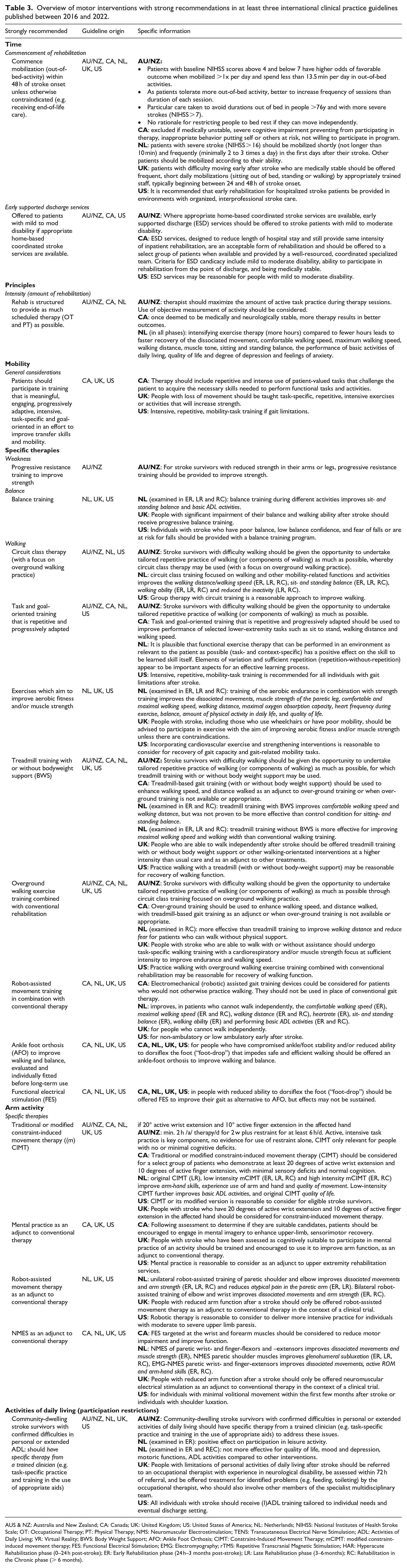
AUS & NZ: Australia and New Zealand; CA: Canada; UK: United Kingdom; US: United States of America; NL: Netherlands; NIHSS: National Institutes of Health Stroke Scale; OT: Occupational Therapy; PT: Physical Therapy; NMS: Neuromuscular Electrostimulation; TENS: Transcutaneous Electrical Nerve Stimulation; ADL: Activities of Daily Living; VR: Virtual Reality; BWS: Body Weight Support; AFO: Ankle Foot Orthosis; CIMT: Constraint-Induced Movement Therapy; mCIMT: modified constraint-induced movement therapy; FES: Functional Electrical Stimulation; EMG: Electromyography; rTMS: Repetitive Transcranial Magnetic Stimulation; HAR: Hyperacute Rehabilitation phase (0–24 h post-stroke); ER: Early Rehabilitation phase (24 h–3 months post-stroke); LR: Late Rehabilitation phase (3–6 months); RC: Rehabilitation in the Chronic phase (> 6 months).
Discussion and conclusion
Motor rehabilitation is a key element of the stroke care pathway for people with persisting movement and mobility deficits. An expert panel agreed on the first definition of motor rehabilitation after stroke, which is supported by a contemporary motor rehabilitation framework for clinicians, educators and researchers. Figure 3 presents the definition and core elements of the motor rehabilitation process after stroke in an abbreviated pictorial format.
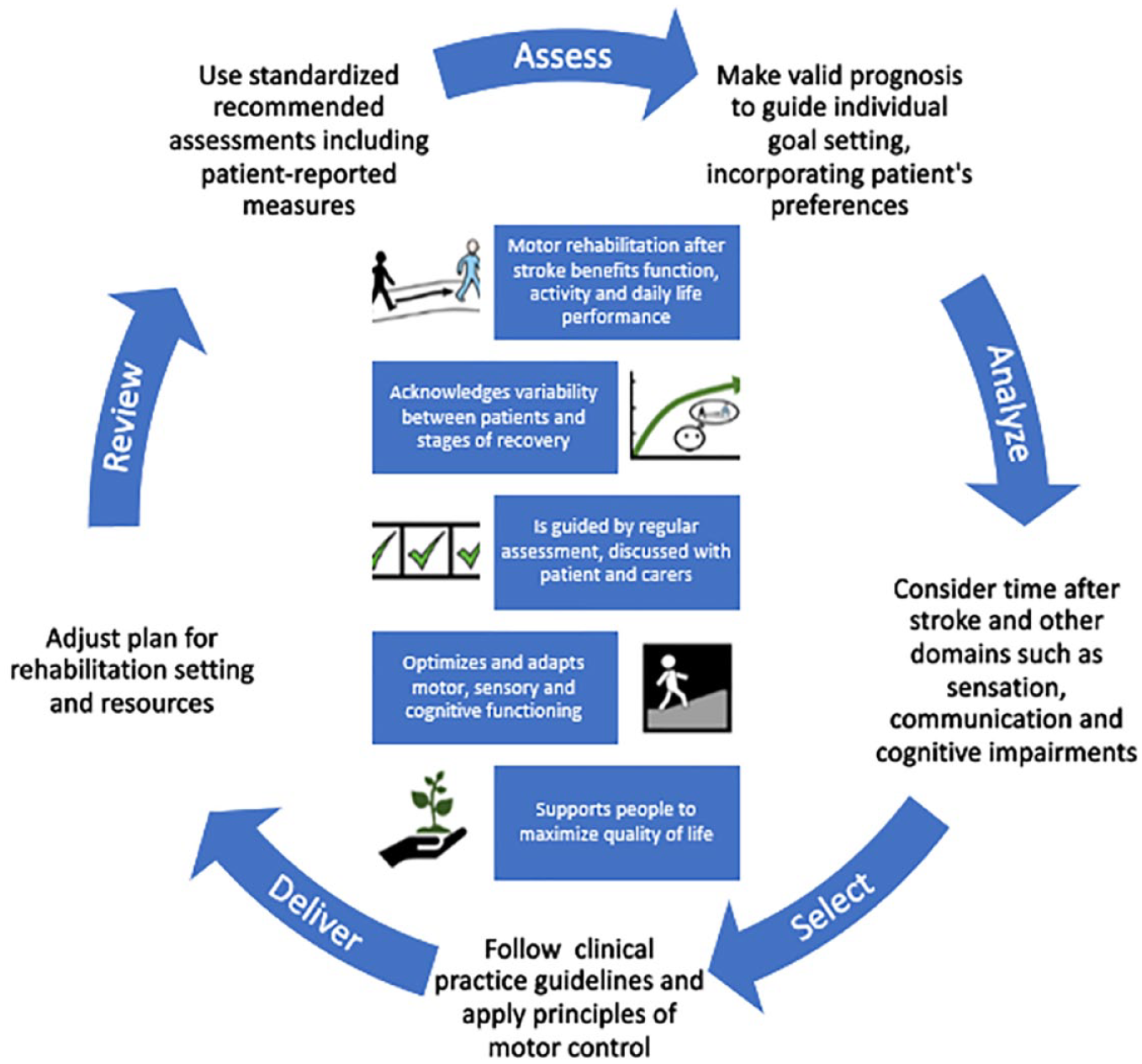
Pictorial abbreviated motor rehabilitation after stroke definition (center) and summarized process of motor rehabilitation after stroke.
The strengths of this work include the expertise of the panel, the three-round process used for producing the definition, and the consultation with clinical stakeholders, integrating their feedback in final group discussions. Working group members were primarily European, in response to the assignment provided by the ESO, and also included colleagues from three global regions. The definition focuses on fundamental concepts which are likely similar across countries. The WHO rehabilitation definition is one sentence, 4 however the panel constructed a definition paragraph which is more appropriate for multi-faceted concepts such as motor rehabilitation after stroke. It is rare for one definition to serve all stakeholders. Here we focused on the needs of educators, clinicians and researchers, rather than laypersons. Nevertheless, we obtained feedback from two people with lived experience, and a lay version with visual supporting elements could help to communicate the concepts to patients and carers.
The formal definition of motor rehabilitation forms a base for future research and guideline development in the domain of motor rehabilitation. The working group is now developing motor rehabilitation guidelines based on the summary of interventions with strong recommendations presented here. Interdisciplinary rehabilitation teams need to follow evidence-based recommendations and integrate the views of patients and carers to improve life after stroke.
Footnotes
Acknowledgements
The working group would like to acknowledge the valuable feedback received from all clinicians consulted in various clinical settings. More specifically, we are grateful for the input from: The stroke unit and neurorehabilitation ward team from Sahlgrenska University Hospital Sweden; 13 physiotherapists, 8 occupational therapists and 7 physicians; Neurologist and specialized neuro physiotherapist from University Hospital Brussels, Belgium; Neurological physiotherapists at Auckland City Hospital, New Zealand; Two clinician physiotherapists, specialized in subacute and chronic stroke care from the Hospital de Santa Creu de Vic and Clínica de Neurorehabilitación, Spain; Three specialized neuro physiotherapists working at the acute stroke unit and three at the rehabilitation unit from University Hospitals Leuven, Belgium; Highly specialized occupational therapist (1) and physiotherapists (3) from stroke services at Gateshead, Northumbria and South Tyneside and Sunderland NHS Foundation Trusts, UK; Two physiatrists from Department of physical medicine and rehabilitation, University Hospital Mostar, Bosnia and Herzegovina; Physiotherapist (1) from Unit of Occupational Medicine, IRCCS Azienda Ospedaliero-Universitaria di Bologna, Bologna, Italy; Physiotherapists (2) from Department of Neurology, University Hospital of Zurich, and cereneo Center for Neurology and Rehabilitation, Vitznau, Switzerland; Physicians (2) from Service de médecine physique et réadaptation, CHU de Toulouse, Toulouse, France. Special thanks to the patient partners Anders Andersson and Johanna Persson who provided valuable input to the definition.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Not applicable.
Ethical approval
Not applicable.
Guarantor
G.V.
Contributorship
Geert Verheyden (GV) and Margit Alt Murphy (MAM) are co-chairs of this European Stroke Organisation (ESO) working group and supported by two junior faculty; Bea Essers (BE) and Maria Munoz Novoa (MMN). Together with Gert Kwakkel and Cathy Stinear, they formed the core writing group as part of the working group, preparing the draft manuscript of this review, revising and finalizing the work based on the reviewer comments received. The working group further consists of Meret Branscheidt, Rosa Cabanas-Valdés, Sandra Lakičević, Sofia Lampropoulou, Andreas R Luft, Philippe Marque, Sarah A Moore, John M Solomon, Eva Swinnen and Andrea Turolla. GV, MAM, BE and MMN prepared all the meetings and the surveys, collected the results and led the discussions about the survey results and definition with all working group members.
All working group members reviewed and commented upon the manuscript, leading to the final draft before submission. Date received: 27 March 2023; accepted: 16 July 2023.
