Abstract
Background:
Seizures are a frequent complication of strokes. The initial severity of the stroke is a risk factor for both seizure occurrence and poor functional recovery.
Aim:
To determine whether epilepsy has a negative impact on functional recovery or is just a proxy for the initial severity of the stroke.
Patients and methods:
We conducted a monocentric retrospective case-control study in 408 consecutive patients hospitalized in the neurological rehabilitation department of the Pitié-Salpêtrière Hospital for rehabilitation of a recent stroke between 1999 and 2019. We matched 1:1 stroke patients with and without seizures according to numerous variables that may influence the outcome: type of stroke (ischemic vs hemorrhagic (ICH)), type of endovascular treatment performed (thrombolysis, thrombectomy), exact location of the stroke (arterial territory for ischemic strokes, lobar territory for ICH), extent of the stroke, side of the stroke, and age at the time of stroke. Two criteria were used to judge the impact on neurological recovery: the change in modified Rankin score between entry and the discharge from the rehabilitation department, and the length of stay. Seizures were divided into early (within 7 days of stroke) and late (after 7 days) seizures.
Results:
We accurately matched 110 stroke patients with and without seizures. Compared to seizure-free matched stroke patients, stroke patients with late seizures had a poorer neurological functional recovery in terms of Rankin score evolution (p = 0.011*) and length of stay (p = 0.004*). The occurrence of early seizures had no significant impact on functional recovery criteria.
Conclusion:
Late seizures, that is, stroke-related epilepsy, have a negative impact on early rehabilitation, whereas early symptomatic seizures do not negatively impact functional recovery. These results reinforce the recommendation not to treat early seizures.
Introduction
Though seizures are a common complication after stroke, there is only little scientific evidence available about the impact of seizures on early neurological recovery. Two main types of seizures in relation to strokes can be individualized: early seizures (or acute symptomatic seizures) occurring within 7 days of the stroke and late seizures occurring after 7 days. The new definition of epilepsy means that even a single late seizure is now considered to be stroke-related epilepsy 1 with an estimated incidence of 10%–12%.2,3 The risk factors for seizures after stroke have long been known: cortical location, hemorrhagic nature of the stroke, severity of stroke. 4 However, only a few studies have looked at the impact of seizures on initial recovery, with discordant results. Paolucci et al. conducted a prospective study of 306 consecutive patients admitted to a rehabilitation hospital for sequelae of their first stroke and found that seizures did not affect rehabilitation therapy. 5 On the other hand, more recent studies have found that stroke-related seizures or epilepsy can negatively influence functional recovery.6–8 This is an expected result, as stroke severity is a risk factor for stroke-related epilepsy. Most of these studies included an adjustment for stroke severity in some way, but it remains difficult to disentangle the exact contribution of seizures to rehabilitation difficulties. To better address the exact impact of seizures on early recovery during the active rehabilitation period, we conducted a case-control study that matched stroke patients with and without seizures according to numerous variables that may influence the outcome such as: the type of stroke (ischemic vs hemorrhagic), the type of endovascular treatment performed (thrombolysis, thrombectomy), the exact location of the stroke, the extent of the stroke, the side of the stroke, and the age at the time of stroke. The goal was to clarify the impact of epilepsy on early rehabilitation.
Material and methods
Case-control study
This retrospective comparative study, which followed STROBE guidelines, analyzed a cohort of stroke patients from the Pitié-Salpêtrière Hospital rehabilitation unit (Paris, France) between 1999 and 2019.
Stroke patients admitted to the rehabilitation unit at Pitié-Salpêtrière Hospital are admitted at least 7 days after the stroke, with no age or severity of symptoms limit.
We first identified stroke patients who had seizures during the hospital stay. Seizures were defined as: (i) acute early symptomatic seizures, that is, seizures occurring within 7 days of the stroke, and (ii) late-onset seizures (i.e. stroke-related epilepsy), that is,seizures occurring during the hospital stay for neurological rehabilitation of the stroke with a minimum delay of 7 days post-stroke. Seizures occurring after the discharge from the rehabilitation unit were not taken into account.
In a second step, we performed a 1:1 matching of the stroke patients with and without epileptic seizures according to the following parameters: (i) stroke type (ischemic vs intracerebral hemorrhage (ICH)), (ii) for ischemic strokes only: existence of endovascular treatment (thrombolysis and/or thrombectomy) or not ; (iii) territory (arterial territory for ischemic stroke and lobar or subcortical regions for ICH); (iv) extent (moderate extent: lacunar stroke; intermediate extent: involvement of a single arterial territory (ischemic stroke) or a single lobe (hematoma); major extent: involvement of several arterial territories (ischemic stroke) or several lobes (ICH)); (v) lesion side; and (vi) age ± 15 years. Existence of specific lobar localization in cases of ICH, hemorrhagic conversions and gender were also taken into account when possible, in this hierarchical order. When an exact match could not be found, we sought the best match regarding stroke type, territory, extent, lesion side, endovascular treatment, and age.
Two criteria were used to judge the impact on neurological recovery:
− Change in modified Rankin score between entry and discharge from the rehabilitation department
− Length of stay (defined as the number of days between admission to and discharge from the rehabilitation unit)
For all stroke patients (with or without seizures), inclusion and exclusion criterion were the following:
- Inclusion criteria: consecutive patients hospitalized in the neurological rehabilitation department of the Pitié-Salpêtrière Hospital for rehabilitation of a recent ischemic stroke or ICH less than 3 months old between 1999 and 2019.
- Exclusion criteria:
○ History of epilepsy
○ Progressive comorbid pathology that may influence neurological recovery:
■ Comorbid brain tumor pathology
■ Comorbid neurodegenerative disease (Parkinson’s disease, Alzheimer’s disease, multiple sclerosis)
○ Antiseizure treatment for other pathology (psychiatric, pain)
○ Initiation of antiseizure treatment for preventive purposes during hospitalization
The study was approved by the medical ethical committee of Sorbonne-Université (CER-2023-Epivasc).
Statistical analysis
Patients with seizures were compared to matched patients without seizure using Wilcoxon signed-rank test numerical variables and McNemar’s test for binary variables. Same tests were performed on paired early seizure patients and on paired late-onset seizure patients independently.
Patients with early and late-onset seizures were compared using Wilcoxon-Mann-Whitney test for numerical variables and Fisher’s exact test for categorical variables.
Statistical analyses were performed using R 4.2.2. (R Foundation for Statistical Computing, Vienna, Austria. URL: https://www.R-project.org/.)
Results
Global population
Over the period 1999–2019, 408 patients meeting the inclusion and exclusion criteria were admitted to the neurological rehabilitation department for stroke rehabilitation. Among them, 59 patients experienced epileptic seizures, of which four patients were excluded due to lack of a proper match according to age with control cases.
The occurrence of seizures did not change the rehabilitation management of the patients and did not require transfer to an intensive care unit. The specificity of the Pitié-Salpêtrière neurological rehabilitation unit is that the physicians working there are almost all neurologists and therefore competent in the management of potential neurological complications.
All patients met the matching criteria concerning: stroke type, territory (arterial/lobar/sublobar), extent, lesion side, and age.
By refining the territory criterion to the exact location (precise arterial territory, precise lobe, precise subcortical territory): only 54 patients with seizures were correctly matched for all criteria (see Table 1): one patient with seizures and a right frontal ICH was matched with a seizure-free patient with a right fronto-parietal ICH.
Descriptive and matching parameters: stroke type, early endovascular stroke treatment, location, extent, side, age, sex.
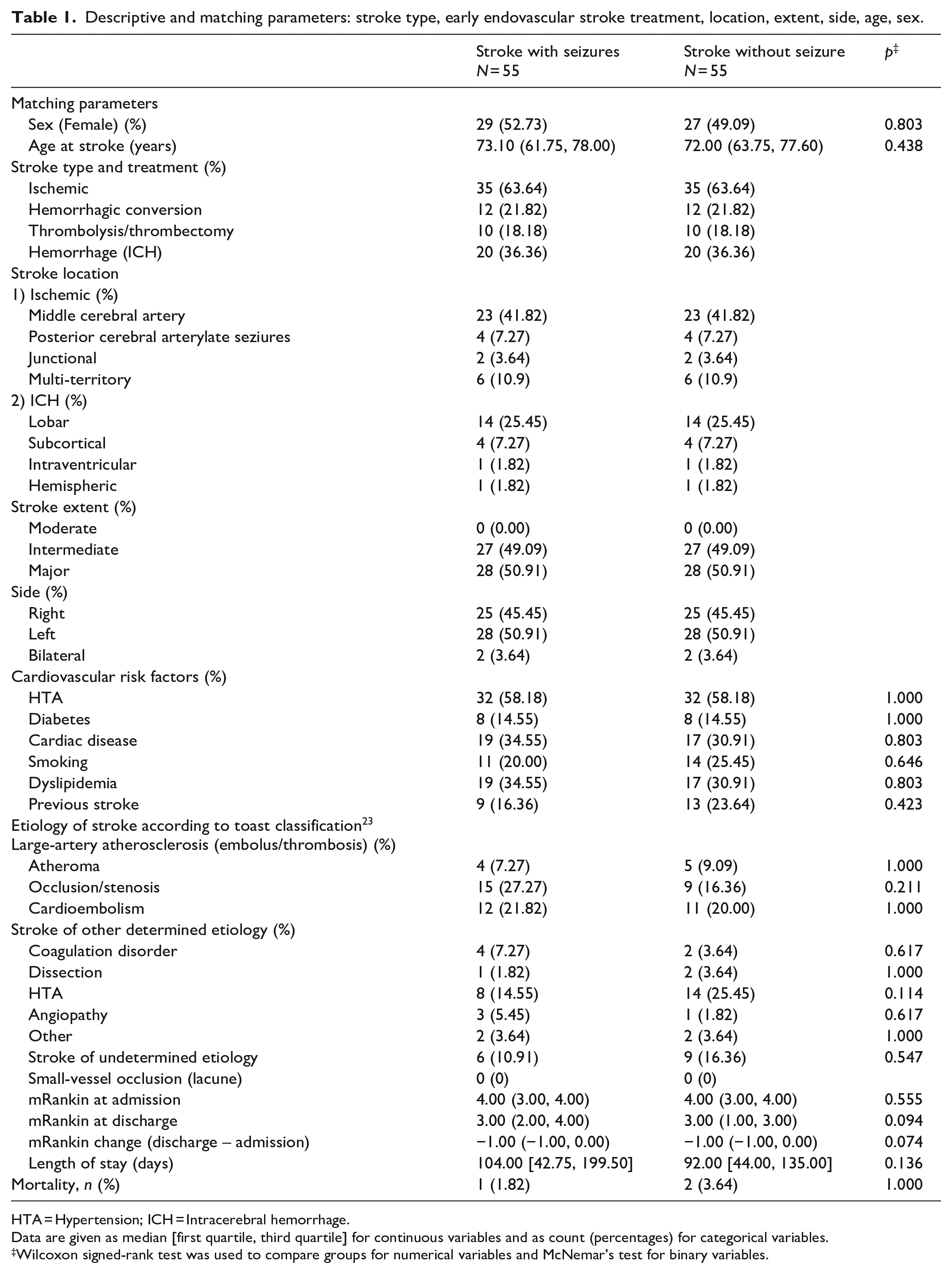
HTA = Hypertension; ICH = Intracerebral hemorrhage.
Data are given as median [first quartile, third quartile] for continuous variables and as count (percentages) for categorical variables.
Wilcoxon signed-rank test was used to compare groups for numerical variables and McNemar’s test for binary variables.
Stroke patients with seizures and stroke patients without seizures were identical in terms of mean age, proportion of females, cardiovascular risk factors, etiologies of strokes, and mean modified Rankin score at admission (Table 1).
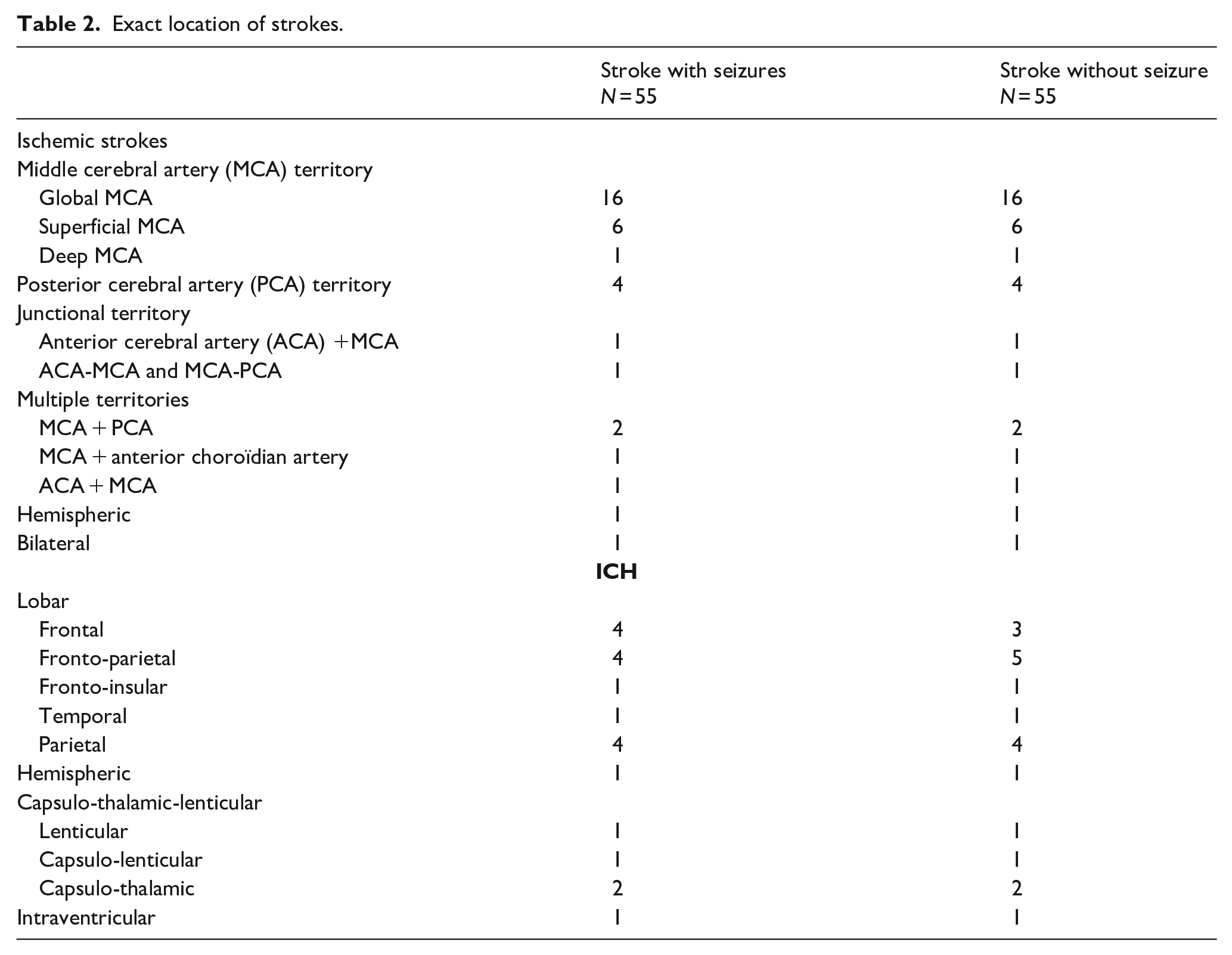
The exact location of strokes is described in Table 2.
Exact location of strokes.
Three patients died during the rehabilitation stay (one in the seizure group and two in the non seizure group) and were thus removed from the analysis.
Stroke patients with seizures
Among patients with seizures, there were 15 cases of early seizures occurring within 7 days of the stroke and 36 cases of late seizures occurring during hospital stay in the neurological rehabilitation unit with a minimum delay of 7 days post-stroke. Four patients had both early and late seizures (Table 3).
Seizures characteristics.
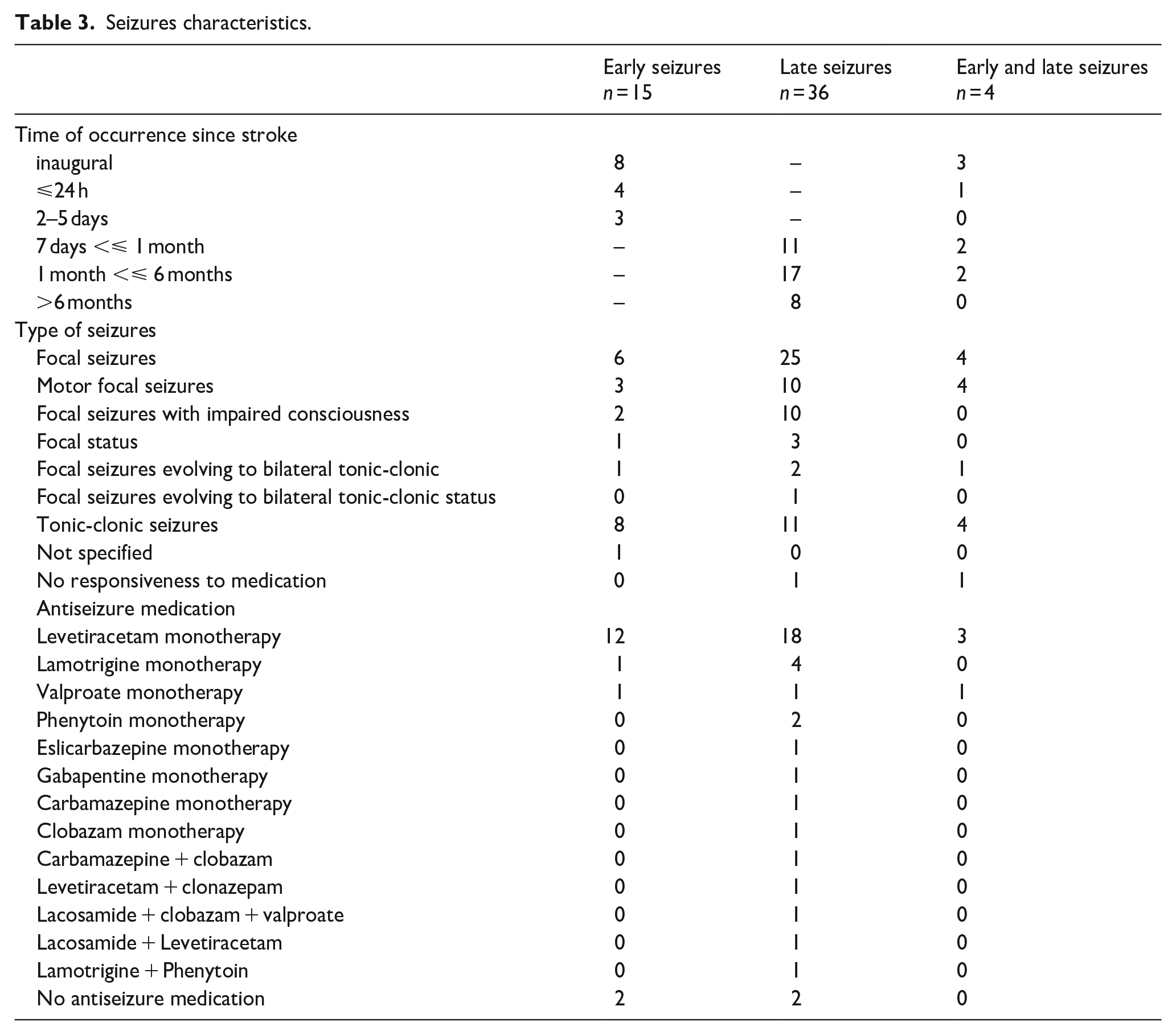
The most frequent seizure type was focal seizure (60%), especially motor seizures (29%). Most stroke patients with seizures responded well to antiseizure medications and were on monotherapy, mainly by Levetiracetam. Only five patients with late-onset seizures needed an antiseizure polytherapy (three patients with focal late status) or a medication reinforcement (relapse in two patients). One patient with early seizure was not treated at all with antiseizure medication. For all other patients with early seizures, anti-seizure treatment was initiated in the acute vascular unit and continued in the neurological rehabilitation unit discharge with the exception of one patient in whom the treatment was stopped during rehabilitation. Antiseizure medication was thus stopped during hospital stay in one patient with early seizure and in two patients with late seizures.
Impact of post-stroke seizures
Global population
In the overall analysis, stroke patients with and without seizures did not show a significant difference in the two parameters tested (change in modified Rankin score, length of stay) (see Table 1). Stroke patients with seizures had a more negative change in Rankin score but the difference was not statistically significant (median difference (Q1,Q3) = 0 (−0.25,1.00); p = 0.074).
Stroke patients with seizures had a higher average length of stay in hospital (104 days) than stroke patients without seizures (92 days) but the difference was not statistically significant (median difference (Q1, Q3) = 8.0 (–29.7,100.5) ; p = 0.136).
Late and early seizures stroke patients versus matched seizure-free stroke patients
When differentiating between the early and late seizure populations (excluding the four patients who had both types of seizures), there were statistically significant differences between the late-onset seizures stroke group and the seizure-free matched stroke population in terms of both evolution of Rankin score (better evolution for seizure-free stroke patients (median difference [Q1, Q3] = −0.5 [−1.0,0]; p = 0.011*) and length of stay (shorter length of stay for seizure-free stroke patients (median difference [Q1, Q3] = −17 [−158.2, 4.5]; p = 0.004*).
Early seizures stroke patients did not differ from seizure-free stroke patients in terms of Rankin score evolution and length of stay.
Late seizures versus early seizures stroke patients
When the populations of stroke patients with early and late onset seizures were compared between each other (excluding the four patients who had both types of seizures), there were statistically significant differences in terms of both evolution of Rankin score (better evolution in stroke patients with early seizures (median difference (Q1, Q3): early = -1.00 (-1.00, -1.00) versus late = −1.00 (−1.00, 0.00), p = 0.024*)) and length of stay (shorter length of stay in stroke patients with early seizures (median difference [Q1, Q3]: early = 64.5 [27.0, 129.2] vs late = 135.0 [60.2, 215.0], p = 0.044*)).
Discussion
This case-control study allowed us to demonstrate that stroke-related epilepsy has a negative impact on early rehabilitation, whereas early symptomatic seizures do not negatively impact functional recovery.
Epidemiology of early stroke-related epilepsy
With the world population aging and the resulting increased prevalence of post-stroke survivors, the number of patients with stroke-related epilepsy is expected to increase.
Impact of the new ILAE definition of epilepsy
Stroke-related epilepsy represented 9.8% of our complete stroke cohort, which is in accordance with recent studies that examined long term stroke-related epilepsy prevalence according to the new ILAE definitions. This means that a single unprovoked post-stroke seizure occurring more than 7 days after the stroke qualifies as stroke-related epilepsy due to the high (greater than 60%) risk of recurrence within the next 10 years.9,10
Recruitment bias
In our case-control study, we were only interested in seizures occurring during hospitalization in the neurological rehabilitation unit. Our average length of stay in hospital was 104 days for stroke patients with seizures versus 92 days for stroke patients without seizures. It is clear that our estimated prevalence of 9.8% stroke-related epilepsy is an underestimate and does not reflect the true prevalence of stroke-related epilepsy in our population, as seizures may have occurred after discharge. Dedicated studies have shown that the majority of stroke-related epilepsy develops within 24 months after the stroke with a doubling of numbers between 6 months and 2 years and then a plateau. 11 The prevalence measured on a longer-term follow-up of our cohort should therefore be significantly higher, and thus discordant with the literature studies. This is likely due to our case series selected patients with a need for rehabilitation, usually because of a permanent language and/or motor deficit, indicating a higher severity of stroke and thus a greater risk of stroke-related epilepsy.
Time window for impact of seizures on functional recovery
Pathophysiological mechanisms of early and late stroke seizures
The temporal cut-off for acute (⩽7 days) versus remote (> 7 days) symptomatic seizures after stroke has been set on the assumption of fundamentally different pathophysiological mechanisms. Early seizures are assumed to be caused by potentially transient processes in the acute phase after stroke, including hypoxia, cerebral perfusion shift, neuronal metabolic dysfunction, and excitotoxicity as well as blood brain barrier disruption.12,13 Late seizures, that is, stroke-related epilepsy, are assumed to be caused by (i) the permanent brain damage, and (ii) a subsequent later process of epileptogenesis triggered by a neuroinflammatory cascade.12,14,15 Studies of stroke recovery show that post-stroke motor recovery mostly follows a nonlinear trajectory that reaches asymptotic levels a few months after the injury.16,17 A dysfunctional process, such as an epileptic activity, will logically disturb the recovery processes, whereas early seizures may be considered as a symptom of stroke, reflecting its severity.
State of the literature
Similarly to our retrospective study, prospective studies have already shown a negative impact of late seizures on functional recovery. In a prospective study on 151 patients with an acute anterior circulation ischemic stroke, Bentes and Peralta found that late seizures, but not early seizures were independent predictors of functional outcome in the first year after stroke. 6 In this study, stroke severity was estimated by stroke size on CT imaging. In another prospective cohort study among 537 stroke young survivors (aged 18–50 years) with a first ischemic or hemorrhagic stroke, multiple regression analysis revealed that epilepsy was an independent predictor of poor functional outcome after ischemic stroke, assessed by modified Rankin Score. 7 In this later study, stroke severity was assessed by NIH Stroke Scale (NIHSS) and modified Rankin score. Covariates in the multiple regression analysis model used to eliminate potential confounders were: sex, age at follow-up, follow-up duration, modified Rankin score, and recurrent stroke. Neither stroke location nor extent were included in this model. In the few studies that did not show a negative impact of seizures on functional recovery, patients with seizures had a greater severity of stroke at admission than those without seizures, which obviously represents a major bias.5,18
Potentially confounding effects
The recovery processes of post-stroke patients are extremely heterogeneous, due, as already mentioned, to the extent of stroke, the degree of spontaneous regeneration, the phenomenon of neuroplasticity, but also to selection of drugs, and appropriate rehabilitation. 19 Thus, the potential confounding factors in our study could be : (i) neurological rehabilitation techniques that differ between the two groups with and without seizures and (ii) the administration of antiseizure drugs (ASDs) to stroke-related patients, which is known to negatively influence the functional outcome. 20
Effect of rehabilitation techniques
Concerning the first potential confounding factor, as our study has a monocentric design, the type of rehabilitation, the frequency of sessions and the rehabilitation techniques used did not differ between the groups.
Effect of antiseizure drugs
Concerning the second potential confounding factor, the side effects of ASDs have effectively been discussed as potential threats to successful rehabilitation, as those side effects may hamper participation in physical exercises as well as cognitive function. 8 Indeed, the impact of antiseizure medications is still uncertain in post stroke epilepsy. In our study, both stroke patients with early (contrary to the recommendations not to treat acute symptomatic seizures) and late seizures were treated by antiseizure drugs, mainly Levetiracetam monotherapy, and only stroke-related epilepsy patients exhibited a worse functional recovery. This suggests that the use of ASDs might be a proxy for more severe seizures or stroke rather than the drugs themselves having an adverse effect.
Clinical implications
The fact that no potentially detrimental effect of early seizures on neurological recovery has been found supports the current recommendations not to routinely treat acute symptomatic seizures following stroke. 21 These recommendations are based on a potential low risk of recurrence of early seizures, even though this risk of recurrence remains controversial.3,22
Conversely, the negative effect of the occurrence of late seizures, mostly isolated, argues in favor of introducing an antiseizure drug treatment as soon as the first late seizure occurs. This is in line with the revision of the ILAE definition of epilepsy.
Limitations and strength of the study
Our analysis was retrospective, based on medical and seizure charts and we acknowledge the limitations of a retrospective study. Furthermore, due to the retrospective nature of the study, functional recovery was judged on the evolution of the modified Rankin score, which is less relevant than the NIHSS score for a detailed assessment of this recovery. Unfortunately, although we had a NIHSS score for all the patients established at the time of their admission to the neurovascular unit, the score was missing at the time of discharge from the rehabilitation unit and could not be reliably reconstructed using only the data collected in the clinical records. Considering the matching criterion, a recent multivariable prediction model of the risk of late seizures, that is, the SeLECT score, identified five main clinical predictors, including the etiology of stroke, showing that etiology may also have an impact on the development of seizures. 22 However, the etiology used to create the SELECT score, that is, large-artery atherosclerotic aetiology, was equally distributed in our study between the two groups, with and without seizures. In the present study, taking into account the precise etiology for the matching procedure would have excluded too many cases, rendering the analysis useless on a statistical level. A detailed etiologic analysis could be interesting in the future.
The main strength of our study is in spite of everything the quality and accuracy of the matching. The single-center design also allowed us to collect information systematically and to ensure that all patients, with and without seizures, received the same rehabilitation techniques, reducing the risk of bias.
Conclusion
The impact of seizures on early post-stroke functional recovery is of growing importance due to the aging of the world population and the resultant increased prevalence of post-stroke survivors. To avoid the circular reasoning that patients with severe stroke have more seizures and that seizures are a marker of stroke severity, we used strict matching and demonstrated that late, rather than early, seizures had a negative impact on neurological functional recovery. This is one of the first unbiased studies to show that stroke-related epilepsy plays a negative role in the early rehabilitation phase of stroke patients, and thus requires appropriate treatment.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
All persons or their relatives gave their informed consent within written treatment contract on admission.
Ethical approval
The study was approved by the medical ethical committee of Sorbonne-Université (CER-2023-Epivasc).
Guarantor
Sophie Dupont had full access to all the data in the study and takes responsibility for its integrity and the data analysis.
Authors’ contribution
SD conceived the study; SD wrote the first draft of the manuscript; MH performed the statistical analysis; BS and CZ did the data collection; All authors contributed to interpretation of the results. All authors reviewed and edited the manuscript and approved its final version.
