Abstract
Objectives:
To determine the cancer incidence after the first-ever cerebrovascular event (CVE) and compare it to the cancer incidence in the population from the same region.
Methods:
We evaluated 1069 patients with a first-ever CVE (Ischaemic or haemorrhagic stroke and Transient Ischaemic Attack) from a prospective population registry of stroke and transient focal neurological attacks, diagnosed between 2009 and 2011. We conducted a structured search to identify cancer-related variables and case-fatality for a period of 8 years following CVE. Cancer incidence in CVE patients was compared to the North Region Cancer Registry (RORENO).
Results:
We found that 90/1069 (8.4%) CVE patients developed cancer after a first-ever CVE. Overall cancer annual incidence rate was higher after a CVE (820/100,000, 95%CI: 619–1020) than in general population (513/100,000, 95%CI: 508–518). In the 45–54 age group cancer incidence post-CVE was 3.2-fold (RR, 95%CI: 1.6–6.4) higher compared to the general population, decreasing gradually in older age-groups. Median time between CVE and cancer was 3.2 years (IQR = 1.4–5.2). Lower respiratory tract and colorectal were the most frequent cancer types. In univariable models, male sex (sHR = 1.78, 95%CI: 1.17–2.72, p = 0.007), tobacco use (sHR = 2.04, 95%CI: 1.31–3.18, p = 0.002) and peripheral artery disease (sHR = 2.37, 95%CI: 1.10–5.13, p = 0.028) were associated to higher cancer risk after CVE. After adjustment, tobacco use (sHR = 1.84, 95%CI: 1.08–3.14, p = 0.026) remained associated to a higher risk of cancer.
Conclusions:
At the population level, patients presenting a first-ever CVE have higher cancer incidence, that is particularly prominent in younger age-groups. Higher cancer incidence, delayed cancer diagnosis and increased mortality post-CVE warrants further research on long-term cancer surveillance in first-ever CVE survivors.
Introduction
Cerebrovascular diseases and cancer occur frequently in the general population, constituting leading causes of morbidity and mortality, particularly in developed countries. 1 About 15% of cancer patients have a concomitant cerebrovascular disease. 2 As the incidence of both conditions increases with age, it is expected that the number of stroke patients with malignancy will rise.3,4
The association between cancer and venous thromboembolism has been well established since the 19th century, with an expected rate of 13% of occult malignancy to be found in the screening of venous thrombosis. 5 However, until recently, it was unclear if cancer also increased arterial thromboembolism risk.3,6 In cohort studies incident cancer was associated with a significant increase of short-term risk of arterial thromboembolism, including ischaemic stroke.3,7,8 In addition, after a cerebrovascular event it has estimated a 8%–10% rate of subsequent cancer.5,9,10 Importantly, most of these studies involved hospital-based cohorts, that frequently have an over-representation of cryptogenic strokes3,11,12 in cancer patients, and only few analysed the incidence of cancer in stroke survivors.9,13,14 Population-based prospective studies are scarce with short follow-up period, limited to ischaemic strokes 13 and lack comparison with population-based registries. 9
It has not been established whether stroke is a long-term cancer risk factor or an early manifestation of occult malignancy. Proposed cancer-specific mechanisms include direct tumour effects,3,7 hypercoagulable state by overexpressing procoagulant factors,3,8 mucin production3,7 and non-bacterial thrombotic endocarditis,8,15,16 or anti-neoplastic treatments.3,9 These mechanisms, may emerge at early stage cancers and cause or contribute with other causes or risk factors, to the occurrence of a stroke.
Considering the association between thrombotic events and cancer, we hypothesize that the incidence of cancer in stroke or TIA patients is higher than the general population, and some patients may have an underlying malignancy at the cerebrovascular event onset. If confirmed, such finding could open the way to a cancer screening or active malignancy surveillance in a specific set of stroke patients, aiming at personalized health care.
Thus, in the present study, we aim to determine the cancer incidence after the first-ever cerebrovascular event and compare it to the cancer incidence in the population from the same region.
Material and methods
ACIN2 study population and design
ACIN2 is a population-based study, that identified and registered all patients with a first-ever-in-a-lifetime stroke and transient focal neurological attacks (TNA), including Transient Ischaemic Attacks, that occurred between October 2009 and September 2011, in the northern region of Portugal.17,18 Study population included patients from five Health Centres (HC) of western Porto main city (190,000 individuals) and two HCs from rural areas, Mirandela and Vila Pouca de Aguiar (46,000 individuals). 19 Multiple sources of information were used to identify ACIN cases, using a record-linkage methodology based on the National Health Number, a unique identifier for residents in Portugal to contact the National Health Service. 19 Hot-pursuit encompassed daily review of emergency admissions and referrals to the project out-patient clinic at Hospital Santo António – Porto Hospital Centre, a tertiary referral hospital. Cold-pursuit included weekly, monthly or quarterly based review of emergency registers at other three hospitals within and near Porto and 21 hospitals in the northern region; inpatient discharges with diagnostic coding (ICD 9th revision codes 430–438, 342 and 781), and brain/vascular imaging lists.17,18
The patients were observed as soon as possible after the event, by a study neurologist who established the diagnosis (with meeting discussion when the diagnosis was not clear) and then were followed-up until 3 months after the index event. The World Health Organization’s ‘stroke’ definition and Sudlow’s and Warlow’s stroke pathological types – ischaemic stroke (IS), intracerebral haemorrhage (ICH) and subarachnoid haemorrhage (SAH) – were considered for the corresponding concepts. Neuro imaging (computerized tomography scan/magnetic resonance imaging) was used to confirm stroke types.17,19 Trial of Org 10172 in Acute Stroke Treatment (TOAST) criteria 20 were used to determine the aetiology for ischaemic stroke.
TIA was defined as a clinical syndrome characterized by acute loss of focal cerebral or monocular function (amaurosis fugax) lasting less than 24 h whose aetiology was attributed to inadequate cerebral or ocular blood supply as a result of arterial thrombosis or embolism associated with arterial, cardiac or haematological disease. Patients not fulfilling this definition were classified as TNA non-TIA. The TIA’s aetiology was established based on the initial routine evaluation at the emergency department and in the outpatient clinic follow-up consultations. 21 Study senior neurologists reviewed clinical information from all patients to ascertain first-ever stroke and TIA diagnosis.
Demographic characteristics of the study population, and prior vascular risk factors for stroke, were obtained from the ACIN2 study, which included Arterial Hypertension, Diabetes Mellitus, Dyslipidaemia, Tobacco use, Peripheral Artery Disease, Coronary Disease, history of thrombotic events and Atrial Fibrillation. Vascular risk factors were deemed present if diagnosed by a physician previously to the cerebrovascular event, and defined by the following criteria 17 : previous diagnosis of high blood pressure or antihypertensive treatment; use of oral anti-diabetic agents/insulin or history of known diabetes (fasting blood glucose ⩾126 mg/dL; 2 h post prandial glycaemia ⩾200 mg/dL; random blood glucose ⩾200 mg/dL with symptoms of hyperglycaemia; HbA1c ⩾6.5%); previous diagnosis/treatment of dyslipidaemia; current smokers if history of tobacco use in the past 12 months; atrial fibrillation documented in patients’ records; prior superficial and deep venous thrombosis or pulmonary thromboembolism.
All patients or legal representatives gave informed consent.
Stroke and cancer study design
A retrospective cohort study was conducted to determine cancer incidence in the following 8 years of the cerebrovascular event for all ACIN patients presenting ischaemic or haemorrhagic stroke (ICH or SAH) and Transient Ischaemic Attacks.
E-health records (including hospital-based information sources and general practitioners’ records), and a structured Case Report Form were used to register cancer related variables for a period of 8 years following the index event. The variables included were prior history of cancer diagnosis, definite diagnosis by a physician, date of diagnosis, primary location of cancer, cancer staging and evidence of metastatic disease. The primary location of cancer was subsequently divided into organ systems: Respiratory System (Larynx; Trachea, Bronchi, Lung), Haematopoietic System (Hodgkin Lymphoma; Non-Hodgkin’s Lymphoma; Lymphoid Leukaemia; Myeloid Leukaemia; Myelodysplastic Syndrome; Chronic Myeloproliferative Neoplasms; Multiple Myeloma), Gastrointestinal System (Oral Cavity and Pharynx; Oesophagus; Stomach; Small Intestine; Colon and Rectum; Liver; Gallbladder and Biliary Tract; Pancreas), Genitourinary System (Cervix; Uterus Body; Ovary; Prostate; Testicle; Kidney; Ureter and Bladder), Breast, Skin Melanoma and Others (Central Nervous System; Carcinoma of Unknown Primary Origin; Bones and Joints; Thyroid Gland; Salivary Gland; Adrenal Gland). Non-Melanoma Skin Cancer (Basal cell and Squamous cell Carcinoma) were not contemplated in the analysis.
Additionally, data pertaining to the vital status of the patient as date, cause and place of death, along with succeeding recurrent cerebrovascular events was also registered.
Data from the North Region Cancer Registry (RORENO) including the years 2009–2014, was used to compare the incidence of cancer in stroke and TIA patients with cancer incidence from the general population. This population-based cancer registry, with high completeness 22 was set up in 1988 and covers the entire northern region of Portugal, around 3.6 million inhabitants, nearly 30% of the Portuguese population.
The present study was approved by Ethics Committees from Centro Hospitalar Universitário do Porto/Instituto de Ciências Biomédicas Abel Salazar, and Instituto Português de Oncologia do Porto.
Statistical methods
The index cerebrovascular event patients were dichotomized as all stroke/TIA patients diagnosed with cancer after the cerebrovascular event onset (Incident Cancer group; IC), and cerebrovascular event patients who had no history of cancer after the cerebrovascular event (Non-Incident Cancer group; NIC). Patients with a previous history of cancer were not included in the analysis because of a higher risk of a second cancer, and the role of cancer treatment in stroke pathogenesis.
Continuous variables were reported as median and interquartile range or as mean and standard deviation (SD). Categorical variables were reported as counts or percentages. Comparisons between groups were performed, using independent samples t-tests (or Mann–Whitney tests) for continuous variables. Chi-squared or Fisher’s exact tests were used to evaluate the association between two categorical variables. Estimates of cumulative incidence function taking competing risks into account were calculated. Regression models taking competing risks into account were carried out to analyse the effect of covariates in cancer occurrence and in the patient survival. This analysis was performed using de approach proposed by Fine and Gray 23 to model the subdistribution hazard.
Incidence was reported as crude and age-standardized rates (ASR) for the 2013 European standard population 24 based on person-years. Person-years were calculated from the time at which subjects were included in the study until first diagnosis of cancer, death, latest contact with public health services or the end of the study period. The 95% confidence intervals (CI) were estimated from Poisson distribution applying the Wilson score method.25,26
Statistical analyses were performed using IBM SPSS Statistics v26 and R software using the package cmprsk. Significance level was set at p-value <0.05.
Data availability statement
All de-identified participant data from the current study can be made available after review of requests for overlap with ongoing analyses and according to site-specific policies for data access. Data sharing requests should be sent to LM or MC at luismaia.neurologia@chporto.min-saude.pt or mcorreia@chporto.min-saude.pt.
Results
ACIN2 patients
We identified 1069 eligible patients, from the ACIN2 study, who had a first-ever cerebrovascular event and met the study criteria, between October 2009 and September 2011, (Figure 1). Among these, 90 (8.4%) had a diagnosis of cancer following the index cerebrovascular event, through the 8 years of follow-up.
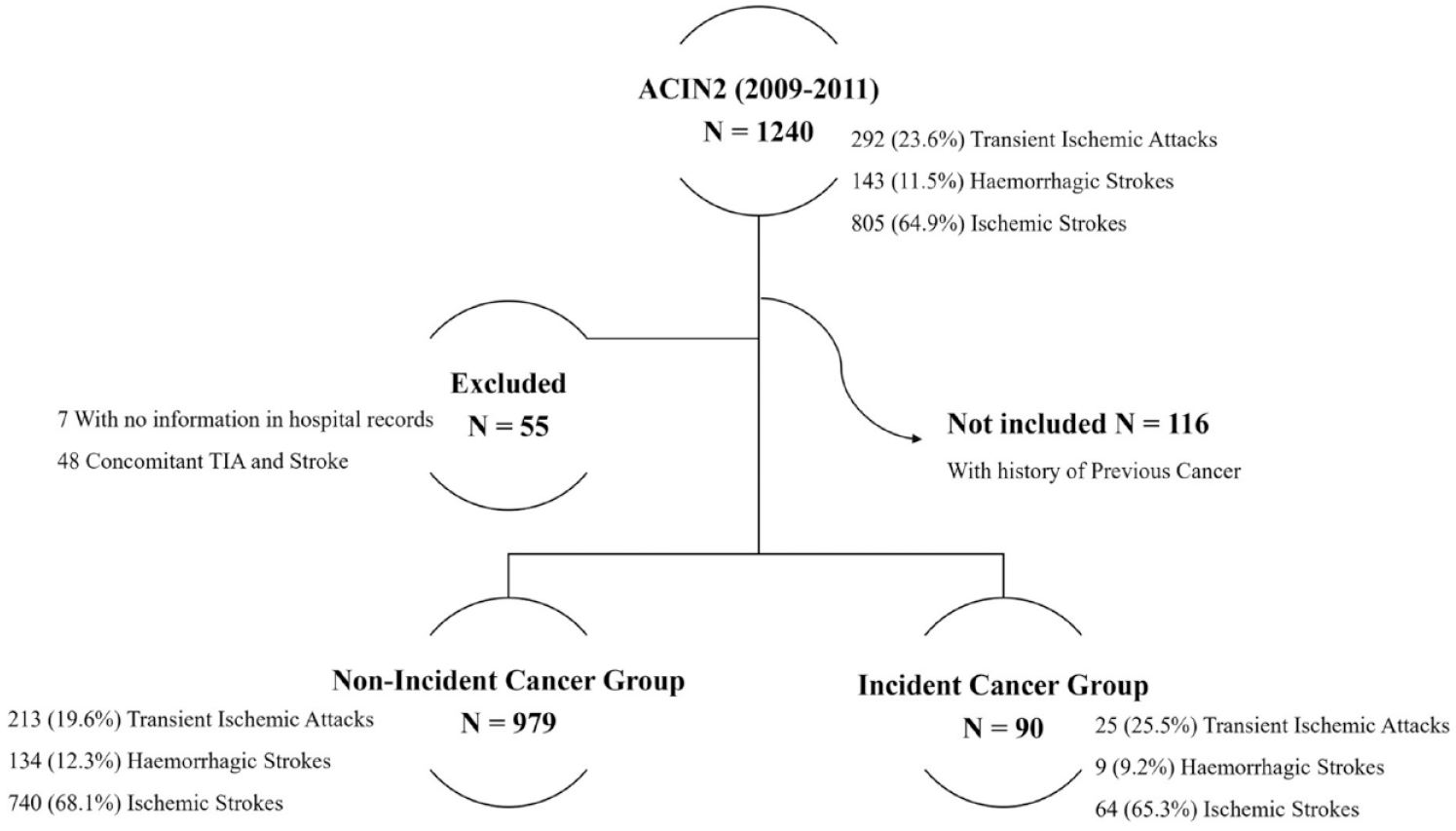
Flowchart describing study design and patient selection.
Patients with incident cancer had a mean age of 71.5 ± 10.4 years compared to 70.8 ± 14.6 years (p > 0.05) and 60% were men, compared to 45% in the Non-Incident Cancer group (p = 0.006) (Table 1). Arterial hypertension and dyslipidaemia were the most frequent vascular risk factors for stroke in both groups (Table 1). There were no differences between IC and NIC groups concerning recurrent cerebrovascular events. Tobacco use (p = 0.002) and peripheral artery disease (p = 0.019) were more prevalent in the incident cancer group. By the end of follow-up, 510 (47.7%) patients had died. Patients with incident cancer displayed higher mortality (66.3% vs 40.1%; p < 0.001).
Demographics, vascular risk factors and cerebrovascular characteristics in ACIN2 stroke and TIA patients according to incident cancer.
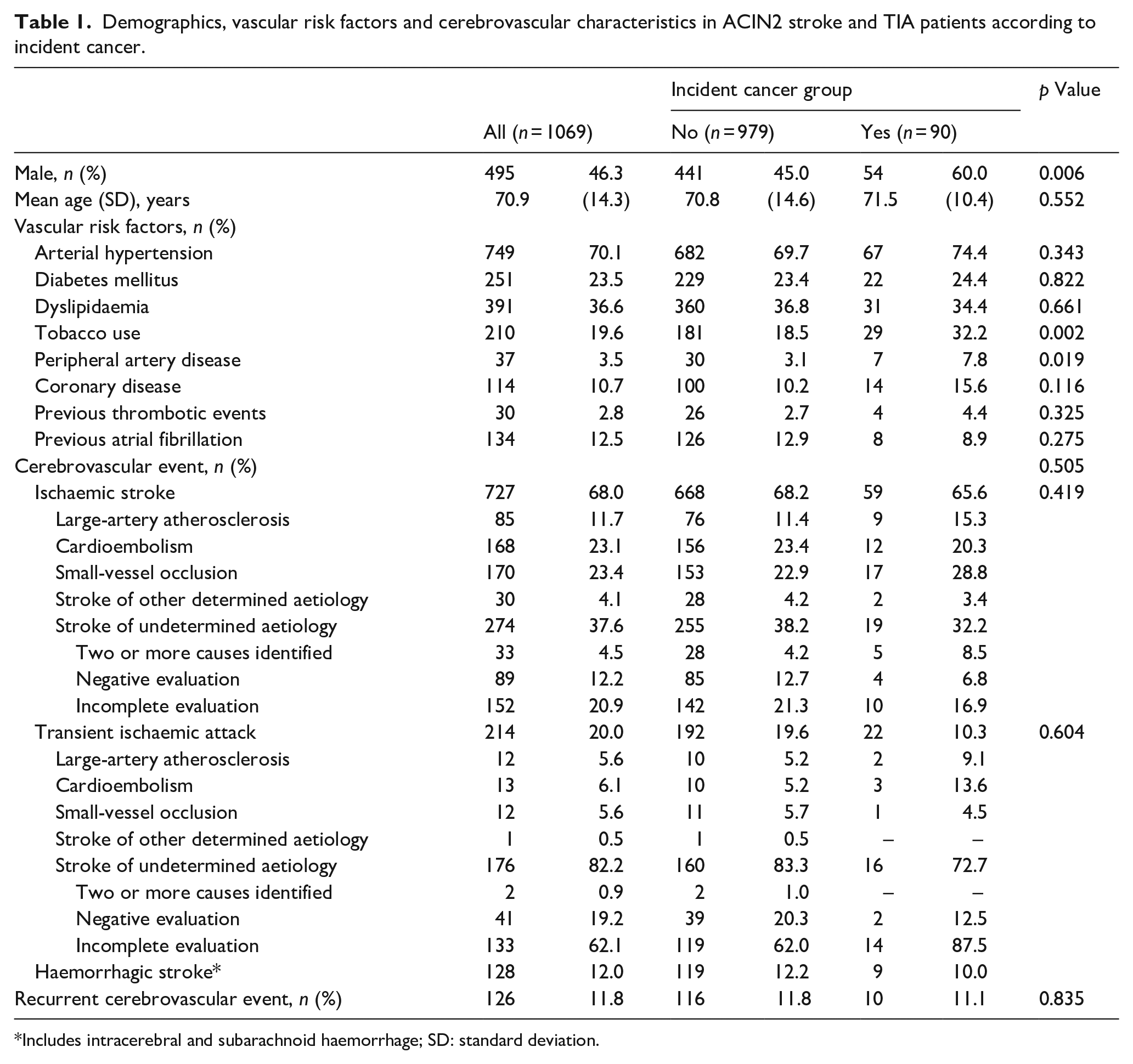
Includes intracerebral and subarachnoid haemorrhage; SD: standard deviation.
Figure 2 summarizes the cumulative incidence estimates for the two possible outcomes (cancer occurrence vs death) taking competing risks into account. In univariable model (Supplemental Table A1), male sex (sHR 1.78, 95%CI: 1.17–2.72, p = 0.007), tobacco use (sHR 2.04, 95%CI: 1.31–3.18, p = 0.002) and peripheral artery disease (sHR 2.37, 95%CI: 1.10–5.13, p = 0.028) were associated with cancer occurrence. Although in multivariable analysis, only tobacco use (sHR 1.84, 95%CI: 1.08–3.14, p = 0.025) was consistently identified as a risk factor.
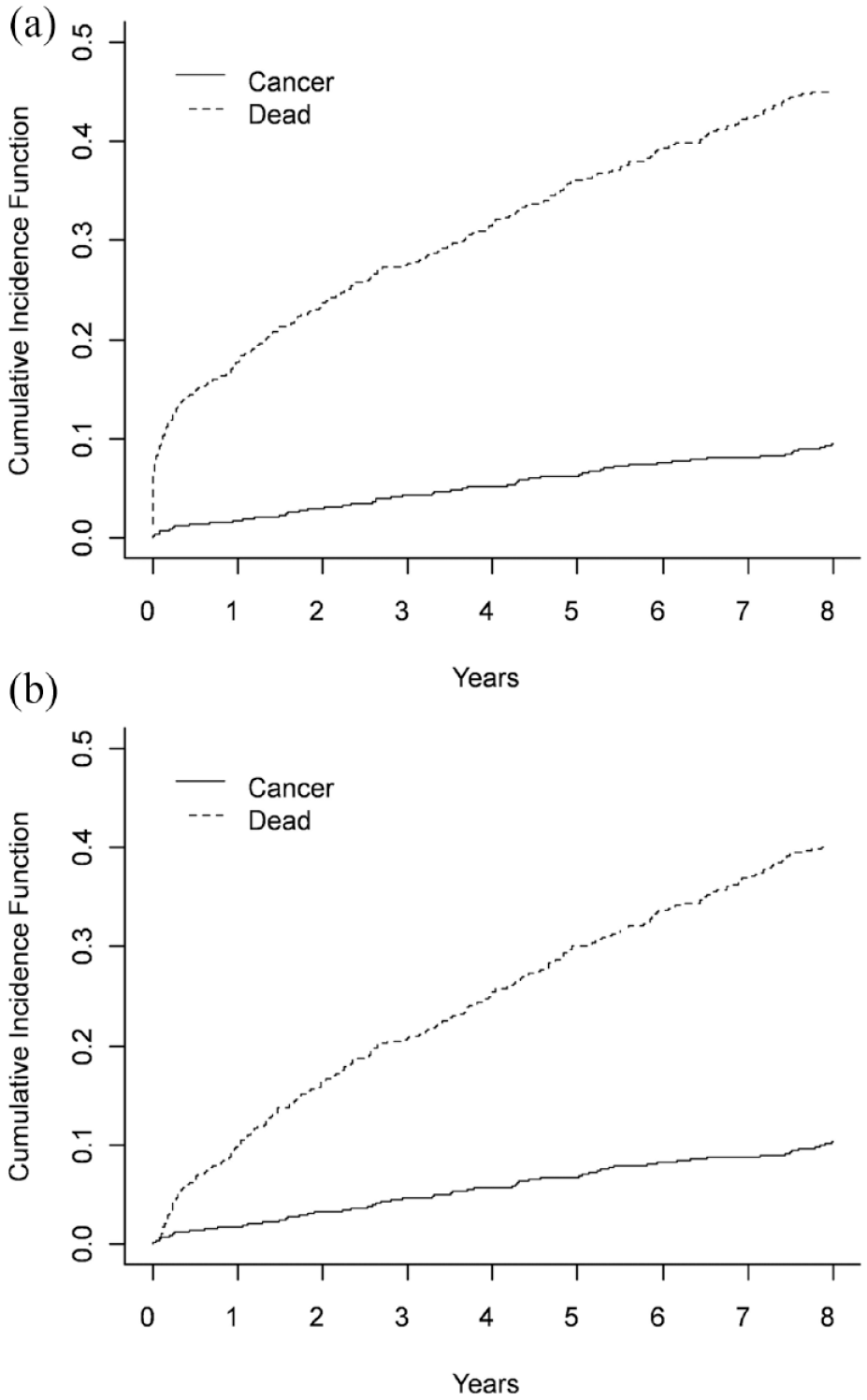
Cumulative incidence functions for all possible outcomes (Cancer or Death) taking competing risks into account: (a) including stroke-mortality (⩽30 days), n = 1069 and (b) excluding stroke-mortality, n = 984.
Cancer incidence in ACIN2 patients
The overall annual crude incidence rate of a first cancer diagnosis after a cerebrovascular event (all strokes or TIA) per 100,000 was 1821 (95%CI: 1465–2239), and 820 (95%CI: 619–1020) adjusted for the European standard population (Table 2). The annual crude incidence per 100,000 was 1400 (95%CI: 981–1939) for women and 2278 (95%CI: 1712–2973) for men in all Stroke and TIA patients. Comparing it to RORENO data (Table 2 and Figure 3), age-adjusted rates for European population in the IC group, and particularly, in males were higher than the general population (1051/100,000 vs 646/100,000). In females, despite the marginally coincident confidence intervals, adjusted rates remained higher than regional cancer registries (606/100,000 vs 419/100,000).
Age- and sex-specific annual incidence per 100,000 for cancer in ischaemic stroke/TIA and all strokes and TIA patients.
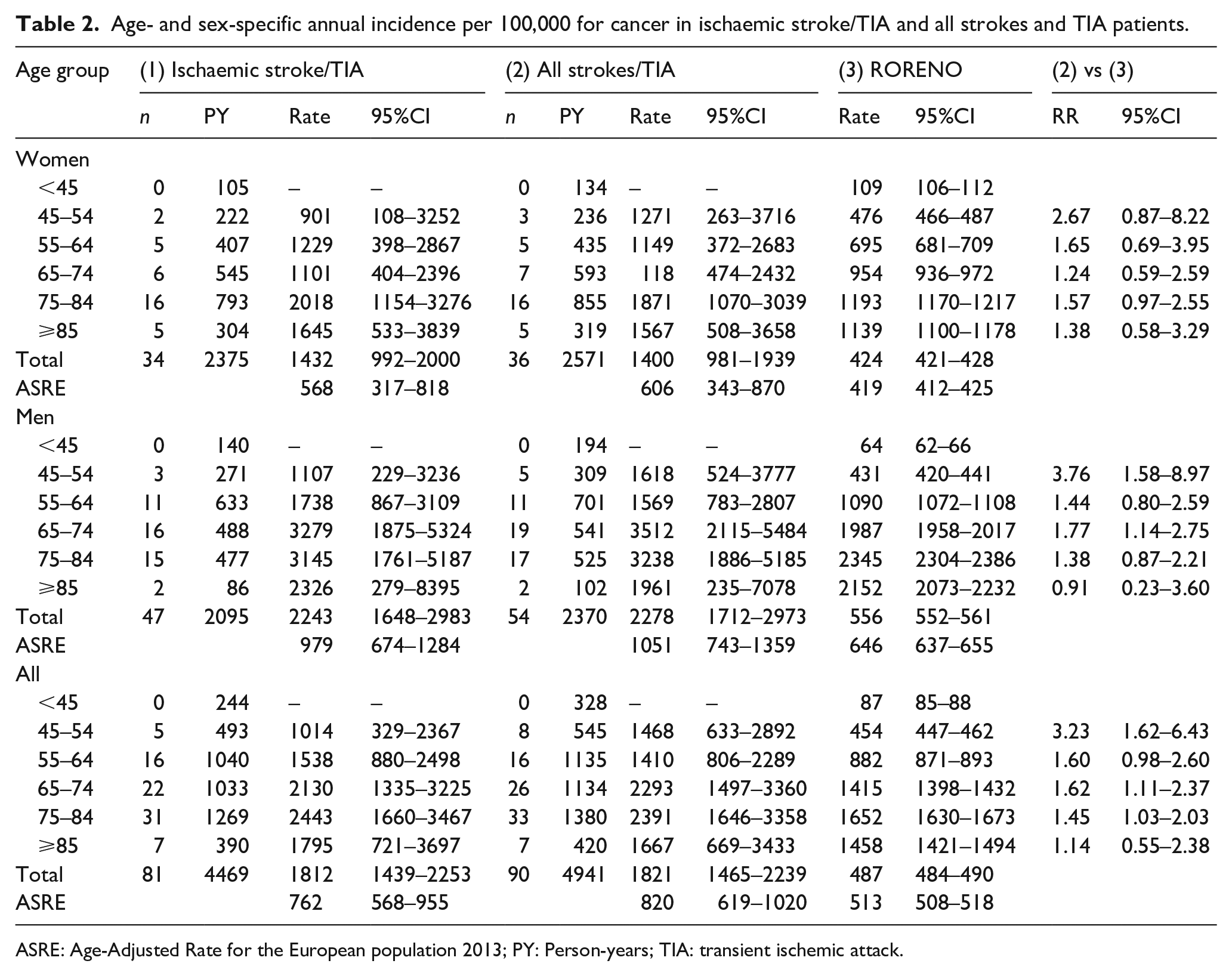
ASRE: Age-Adjusted Rate for the European population 2013; PY: Person-years; TIA: transient ischemic attack.
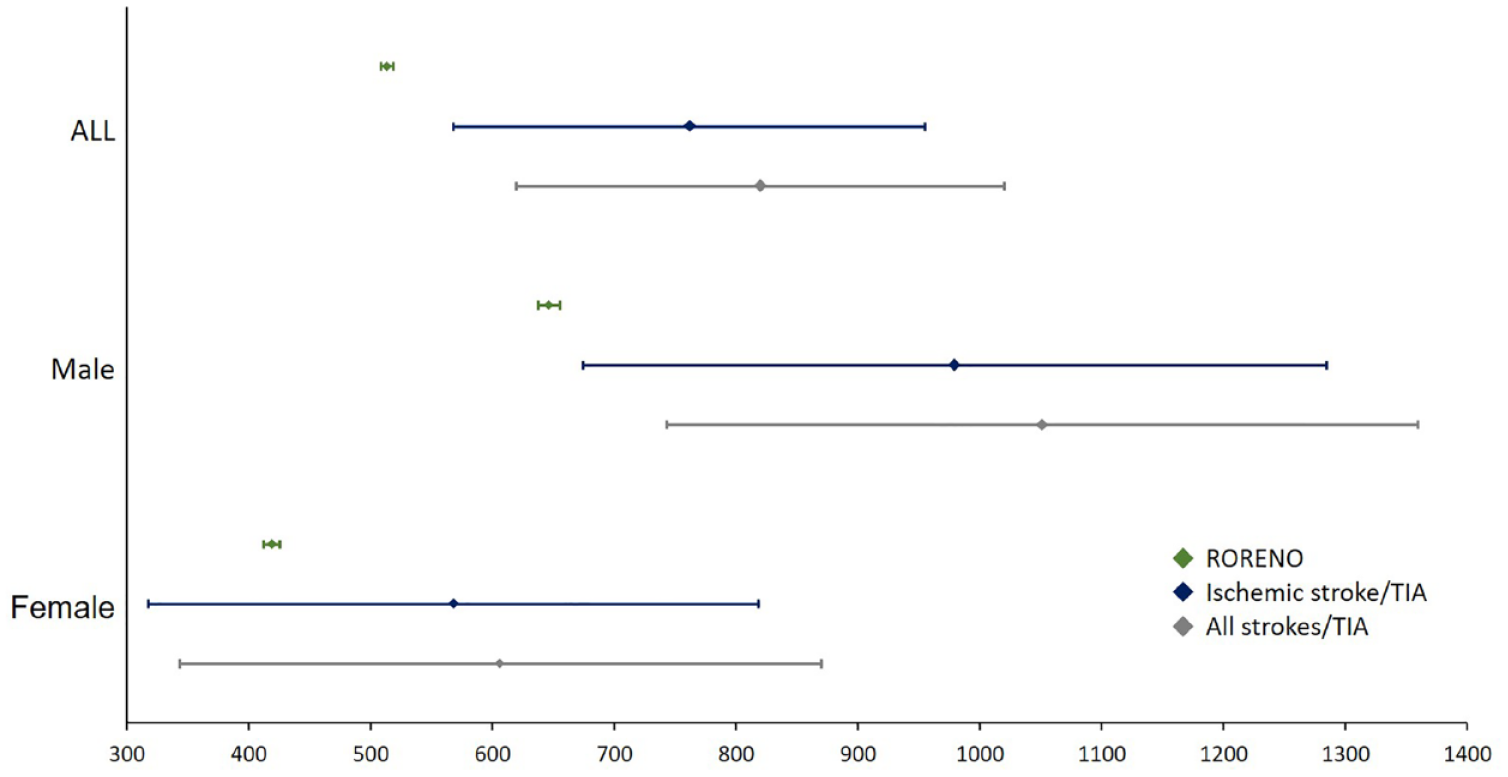
Sex-specific annual incidence rates for Cancer in ACIN2 patients and RORENO population. In green cancer incidence in the general population (RORENO); In Blue cancer incidence in Ischaemic strokes and TIAs; In Grey cancer incidence in all strokes and TIAs.
The risk of cancer was higher in ACIN patients compared to the general population (RORENO registry) across age groups of 45–54, 65–74 and 75–84. In fact, in the youngest age-group (45–54) the relative risk (RR) of cancer was the highest compared to the corresponding group in the general population (RR = 3.23, 95%CI: 1.62–6.43). Curiously, the relative risk declined gradually in older age-groups: 55–64 (RR = 1.60; 95%CI: 0.98–2.60), 65–74 (RR, 1.62; 95%CI: 1.11–2.37); 75–84 (RR, 1.45; 95%CI: 1.03–2.03); and >84 (RR, 1.14; 95%CI: 0.55–2.38). In male patients, we also observed a significant higher risk of cancer compared to the corresponding groups in the population: age groups of 45–54 (RR, 3.76; 95%CI: 1.58–8.97); and 65–74 (RR, 1.77; 95%CI: 1.14–2.75) (Table 2).
Stroke and tumour characteristics
The most frequent primary incident tumour location was Gastrointestinal (n = 32/90, 35.6%), followed by Genitourinary (n = 21/90, 23.3%) and Trachea, Bronchi, Lung (n = 11/90, 12.2%) (Supplemental Table A2). Metastatic disease was diagnosed in 34/90 (37.8%) of the incident cancer patients. Tumours from Trachea, Bronchi, Lung (n = 9/11, 81.8%), Colorectal (n = 6/11, 54.5%) and Pancreas (n = 3/5, 60%) were more frequently associated with disseminated malignancy compared to other tumour types (p = 0.035).
The mean age at cancer diagnosis was 75.4 ± 10.5 years. The median time interval between cerebrovascular event and the diagnosis of cancer was 3.2 years (IQR = 1.4–5.2). Eighteen out of 90 (20%) patients were diagnosed within 1 year, 14 of which within the first 6 months after cerebrovascular event. Trachea, Bronchi, Lung tumours had the shortest median time from cerebrovascular event onset, 1.1 years (IQR = 0.3–4.2), followed by Genitourinary, 2.6 years (IQR = 1.5–5.1), and Gastrointestinal Cancer exhibited the longest interval, 3.8 years (IQR = 1.8–5.5).
Regarding age-adjusted incidence rates (Table 3), Trachea, Bronchi, Lung and Gastrointestinal Tract Tumours were overall higher than in general population aged 45 or older. Age-adjusted rates for Genitourinary Tract Tumours in all strokes/TIA cohort were not significantly different from RORENO data, except for female stroke/TIA patients that were higher than in the general population. Given the low incident number, comparison between other tumours was not done.
Age- and sex-specific annual incidence per 100,000 for cancer, stratified by type of tumour, in ischaemic stroke/TIA and all stroke and TIA patients.
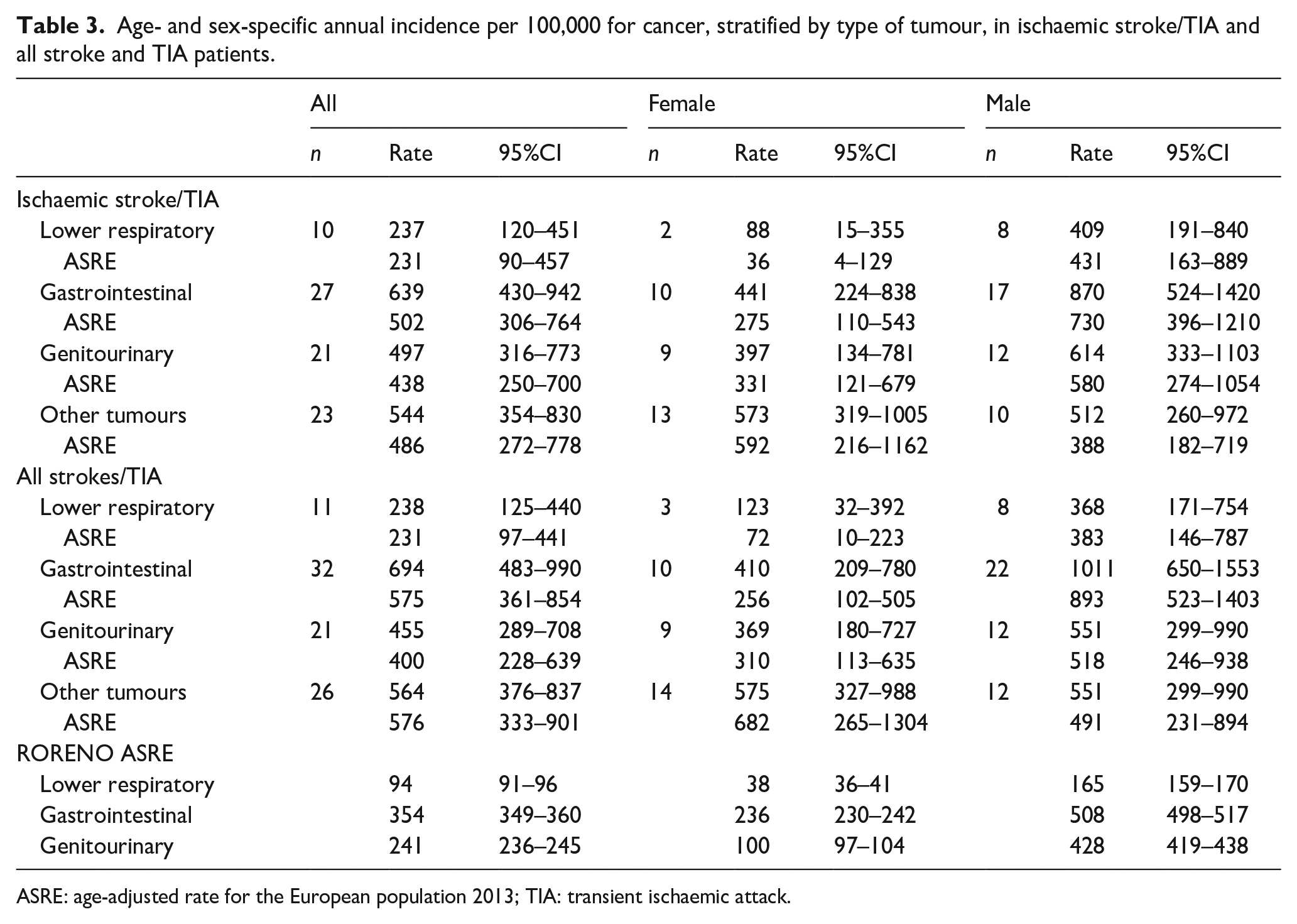
ASRE: age-adjusted rate for the European population 2013; TIA: transient ischaemic attack.
Stroke aetiology in ACIN2 patients
In ACIN2 cohort ischaemic stroke patients, cardioembolic stroke (23.1%) and small vessel disease (23.4%), were the most frequent identifiable aetiologies and the aetiology was undetermined in 37.6% of ischaemic stroke patients (Table 1). The aetiological investigation was incomplete in 62.1% of TIA patients.
Cerebrovascular event aetiology was not different among IC and NCI groups (p = 0.419; p = 0.604). Specifically, stroke of undetermined aetiology and was not associated to an increased risk of cancer (p = 0.293).
Discussion
This population-based retrospective cohort study provides unique data on cancer annual incidence over an 8-year period after the first stroke or TIA at a population level by comparing age- and sex-specific incidence rates to the national cancer registry (RORENO). We found that the overall incidence of cancer was higher than in the general population from the same geographical region. Moreover, this difference was mostly prominent in the younger age-group (3.2-fold-higher).
In fact, higher cancer incidence was particularly striking in male patients aged between 45–54 years and 65–74 years, suggesting that such patients are at higher risk of cancer after the first stroke/TIA. This finding is in line with the fact that male sex is a known risk factor for most types of cancer and consistent with the data from RORENO. Tobacco use and Peripheral Artery Disease were also associated with higher malignancy risk, but only tobacco use showed an independent association. These findings support the relation between arterial cerebrovascular diseases and cancer, are in line with and provide added value to previous findings documented in cohort and population studies.5,9,13
Classic vascular risk factors frequencies were similar in patient with or without incident cancer, except for tobacco use and peripheral artery disease, that were higher in incident cancer group. In fact, multivariable analysis considering death without cancer as a competing risk, tobacco use has a clear association with cancer occurrence. This is consistent with previous studies,3,7 highlighting the overlap of risk factors in both pathologies. The observed higher incidence of cancer in stroke and TIA patients supports that malignancy-specific mechanisms may co-exist and eventually contribute to cerebrovascular disease occurrence.6,7,15 Actually, this may be particularly relevant for younger patients, as we found that the incidence rate of cancer in age group of 45–54 years is three-fold higher than the age- and geographical region-matched population. This is in line with recent studies where younger patients have a two to five-fold increased long-term risk of malignancy after an ischaemic stroke.5,27 It has been demonstrated that in younger patients, specially under 49 years, classic stroke aetiologies play a minor role in cerebrovascular events and a prothrombotic state is frequently identified,28 –30 adding to the relevant clinical question whether a first-ever stroke in a malignancy naïve patient can be the initial presentation of an occult cancer.6,31
The median time observed from cerebrovascular event to cancer diagnosis was 3.2 years, which contrasts with 1.2 years observed in the Norwegian population-based study (NORSTROKE). 13 This may be due to a shorter follow-up time in NORSTROKE compared to our study (5.5-year vs 8 years). In fact, in the first 6 months after stroke, the cancer frequency in ischaemic stroke patients from the NORSTROKE study (1.0%) 13 was similar to what we observed in ACIN2rpc patients (1.3%). We observed that the cumulative cancer incidence increased steadily suggesting that the incidence was constant throughout 8-year follow-up period. Thus, a longer period of follow-up may have contributed to further cancer diagnosis suggesting that longer cancer surveillance may be considered in stroke patients. Whether the index stroke or TIA was the first manifestation of an underlying cancer is not possible to determine in this study, but available evidence on pre-symptomatic tumour progression over the course of years may support this possibility, either as a unique or a cumulative factor. Curiously, we observed a lower median time amongst Lung tumours (1.1 years; IQR 0.3–4.2), in opposition to Gastrointestinal Cancer (3.8 years; IQR 1.75–5.5). This may suggest that the tumour may have been present at the time of the stroke but remained undiagnosed for a longer period, which may be justified by the different cancer cell behaviour regarding tumour cell proliferation and tumour systemic impact.32,33
We observed higher case fatality in Incident Cancer group, consistent with cancer-related stroke being frequently associated with worst outcomes, specifically stroke mortality,34,35 poorer functional outcomes and longer hospital stays.3,31
Unlike previous cohort studies,36,37 we have not found an association between a specific stroke aetiology and cancer occurrence. Stroke aetiology in Incident Cancer group, did not differ from the Non-Incident Cancer group, as observed in population based-studies.9,13 This contrasts to what is observed in hospital-based cohort studies that frequently display selection bias towards younger patients and cryptogenic strokes.10,11,15 In the current study, we included both ischaemic and haemorrhagic strokes and our results are in line with a recent registry-based study in the Netherlands, 27 that found an higher cancer incidence associated with both types of stroke. Verhoeven et al. demonstrated an association between haemorrhagic strokes and haematological and lung cancers. We did not find an association between haemorrhagic stroke and a specific cancer type in our population-based cohort. Mechanisms underlying the association between haemorrhagic stroke and incident cancer remain to be elucidated.
The tumour type has been shown of relevance to explain stroke pathophysiology in cancer patients.3,7,13 In our study, Respiratory tract, Colorectal and Genitourinary tumours were the most frequent cancer types, which is consistent with previous studies.12,13,38 This can be partially explained by the higher prevalence of tobacco use in Incident Cancer group as smoking-related cancers (i.e. Lung, Bladder, Pancreas and Colorectal tumours) are more frequent in our study and show increased incidence comparing to population data. Although, the prevalent use of antithrombotics in stroke patients could, in theory, anticipate cancer diagnosis by increasing GI bleeding, several studies have shown that aspirin use can reduce the incidence of polyps, which can be precursors of colorectal cancer. 39 Notably, the organs most frequently affected by tumours in our study (Lung, Colorectal, Pancreas) are more frequently associated with mucin producing adenocarcinomas, known to potentiate an hypercoagulable state.3,7
Compared to previous studies, the strength of our study is the population-based design and the exclusion of patients with a prior diagnosis of cancer, minimizing the higher risk of a subsequent cancer diagnosis and the role of antineoplastic treatments in stroke pathophysiology.9,40 Still, the retrospective collection of cancer related data that was obtained through medical patient records, may have underestimated a potential higher incidence of cancer in stroke and TIA patients. Secondly, due to an increased case-fatality linked to cerebrovascular events and the aged population, by the end of the study, half of the patients had died. This may have led occult malignancies that couldn’t have been recognized and reported. In addition, our study population may have been under closed medical surveillance due to the cerebrovascular event, raising the possibility of early cancer detection. The relative low number of study subjects has limited subgroup statistical analysis, especially of tumour types, as well as the comparison with more robust national records. Nevertheless, crude rates were consistently higher overall. Additionally, the incomplete aetiological work-up may have limited the association between cancer occurrence and specific stroke aetiology.
Despite unequivocal higher incidence of cancer in stroke and TIA patients, it is unclear to what extent it contributes to cerebrovascular events pathogenesis and its subsequent initial clinical manifestation. Further research is necessary to fully understand this causal relation as previous studies have focused specifically on cryptogenic strokes and have not acknowledge the potential synergistic role of cancer in conventional stroke mechanisms.
Conclusions
The incidence of cancer in our population cohort of stroke and TIA patients is higher than the general population from the same geographical region. This was particularly striking in the youngest age-group (45–54) and for gastrointestinal, genitourinary and lower respiratory tract tumours. A specific stroke aetiology was not associated with malignancy occurrence. Further studies are necessary to clarify this contribution to stroke pathogenesis.
Importantly, clinicians should be aware of this heightened risk and consider an approach comprising a selection of a subgroup of patients who would benefit from active malignancy surveillance, particularly male sex and tobacco users, after a Stroke or TIA. However, further research is necessary to assess the cost-benefit of long-term cancer surveillance in first-ever Stroke or TIA survivors.
Supplemental Material
sj-docx-1-eso-10.1177_23969873231181628 – Supplemental material for The crosstalk between Stroke and Cancer: Incidence of cancer after a first-ever cerebrovascular event in a population-based study
Supplemental material, sj-docx-1-eso-10.1177_23969873231181628 for The crosstalk between Stroke and Cancer: Incidence of cancer after a first-ever cerebrovascular event in a population-based study by Catarina Guedes Vaz, Jéssica Rodrigues, Diogo Pereira, Ilda Matos, Carla Oliveira, Maria José Bento, Rui Magalhães, Manuel Correia and Luis F Maia in European Stroke Journal
Footnotes
Acknowledgements
We would like to thank Drs. Miguel Veloso from CHVNG, Vitor Cruz from ULS Matosinhos, Pedro Abreu from CHUSJ and Mário Rui CHTMAD for their contribution in the ACIN2 registry.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was partially financed by FEDER – Fundo Europeu de Desenvolvimento Regional funds through the COMPETE 2020 – Operacional Programme for Competitiveness and Internationalisation (POCI), Portugal 2020, by Fundação para a Ciência e a Tecnologia/Ministério da Ciência, Tecnologia e Ensino Superior in the framework of the project POCI-01-0145-FEDER-031674 (PTDC/MEC-NEU/31674/2017). JR is supported by a PhD fellowship from Fundação para a Ciência e a Tecnologia, Portugal (UI/BD/152282/2021).
Ethical approval
The present study was approved by Ethics Committees from Centro Hospitalar Universitário do Porto/Instituto de Ciências Biomédicas Abel Salazar (2020.42(188-DEFI/189-CE)), and Instituto Português de Oncologia do Porto (CES IPO:21/021).
Informed consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
Guarantor
Luis Maia.
Contributorship
C.G.V. – Conception and Study design, acquisition and data analysis and drafting the first version of the manuscript or figures; J.R. – Acquisition, data analysis and manuscript review; D.P. – Acquisition and data analysis; I.M. – Acquisition and data analysis; C.O. – Conception and Study design, data analysis and manuscript review; M.J.B. – Study design, data analysis and manuscript review; R.M. – Conception and Study design, statistical analysis and manuscript draft and review; M.C. – Conception and Study design, data analysis and manuscript review; L.M. – Conception and Study design, data analysis and manuscript draft and review.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
