Abstract
Background:
Hemorrhagic stroke (HS) is a sudden-onset disease with high mortality and disability rates, and it is crucial to explore the triggers of HS. In this study, we analyzed individual triggers for HS to provide a basis for HS prevention and intervention.
Methods:
A systematic search of five databases was conducted until December 2022. Studies on HS-related individual triggers conducted using a case-crossover study or self-controlled case series design were included in the descriptive summary and comprehensive evidence synthesis of each trigger.
Results:
A total of 39 studies were included after the screening, and 32 trigger factor categories were explored for associations. Potential trigger factors for HS were as follows: Antiplatelet (odd ratio (OR), 1.10; 95% confidence interval (CI), 1.00–1.21) and anticoagulant (OR, 5.43; 95% CI, 2.04–14.46) medications, mood stabilizers/antipsychotics (OR, 1.33; 95% CI, 1.07–1.65), infections (OR, 2.15; 95% CI, 1.73–2.67), vaccinations (relative risk, 1.11; 95% CI, 1.02–1.21), physical exertion (OR, 2.08; 95% CI, 1.58–2.74), cola consumption (OR, 5.45; 95% CI, 2.76–10.76), sexual activity (OR, 7.49; 95% CI, 2.23–25.22), nose blowing (OR range, 2.40–56.40), defecation (OR, 16.94; 95% CI, 3.40–84.37), and anger (OR, 3.59; 95% CI, 1.56–8.26). No associations were observed with illicit drug use (OR, 2.05; 95% CI, 0.52–8.06) or cigarette smoking (OR, 0.81; 95% CI, 0.52–1.24) and HS.
Conclusions:
Individual triggers, including several medications, infections, vaccinations, and behaviors, may trigger HS onset. Direct control measures for behavioral triggers can play a crucial role in preventing HS. High-risk populations should receive personalized therapies and monitoring measures during the medication treatment to balance the risk of acute HS and the basic diseases.
The incidence of hemorrhagic stroke (HS) is 81.52/100,000 person-years worldwide, and despite the fact that HS accounts for only 15%–36% of strokes, the second leading cause of death and disability worldwide, it has a higher rate of death, disability, and economic burden.1,2 Intracerebral hemorrhage (ICH) and subarachnoid hemorrhage (SAH), subtypes of HS, account for 27% and 9.7% of all strokes, respectively. 2 A tremendous amount of pressure has been put on families and society as 37.3% of patients with HS die within 90 days and survivors suffer from long-term cognitive impairment. 3 Therefore, understanding the mechanism of HS onset is crucial for its prevention.
As an acute event, the onset of HS is associated with certain short-term exposures (trigger factors) that lead to a rapid increase in arterial blood pressure, heart rate, or vasoconstriction over a short period of time, such as strenuous exercise and anger. 4 As the proximate cause in the causal chain of the disease, these short-term exposure factors can suddenly increase the risk of disease in a short period of time and thus may directly contribute to the onset of HS. They are closer to the occurrence of the outcome event than other chronic factors, including demographic characteristics (age, sex, etc.), long-term life habits, chronic diseases, and environmental and genetic factors.5,6 Behavioral factors, which are relatively easier to modify, accounted for 66.3% of the stroke burden, which was much higher than the 28.1% for chronic environmental factors. 7 Thus, it is important to investigate these intervenable trigger factors for disease prevention.
Existing study designs include case-crossover study (CCOS) and self-controlled case series (SCCS), which are suitable for assessing the transient effects of trigger factors on acute events, and several potential trigger factors have been identified.8,9 However, the effects of these factors remain unclear due to the large heterogeneity of the results. This study systematically reviewed the literature on HS trigger factors using these two study designs, aiming to provide a comprehensive summary and a basis for preventing HS scientifically and effectively.
Methods
This systematic review was conducted according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) and registered in PROSPERO (registration number: CRD42022327934).
Search strategy and selection criteria
PUBMED, Web of Science, Embase, China National Knowledge Internet (CNKI), and Wanfang were searched for the keywords “hemorrhagic stroke,” “cerebral hemorrhage,” “intracranial hemorrhage,” “subarachnoid hemorrhage,” “case-crossover,” and “self-controlled case series” until December 2022. References from relevant reviews and articles were also screened to identify candidate studies.
Eligibility criteria and study selection
The inclusion criteria were as follows: (1) patients with clearly diagnosed HS according to the World Health Organization definition, confirmed by computed tomography, magnetic resonance imaging, or autopsy, or defined using the International Classification of Diseases (ICD-9, 430–432.9, 437.2; ICD-10, 160–162.9, 167–169.2) 10 ; (2) all primary HS, such as hypertension or amyloid angiopathy, or secondary HS due to pathological changes in the vascular structures caused by aneurysms, arteriovenous malformations, cavernous aneurysms, etc. 11 ; (3) a CCOS or SCCS design was used; (4) the outcome was HS, ICH, or SAH inpatient, emergency, or outpatient; (5) the factors studied were individual factors of transient exposure, such as medications, physical activity, infection, etc.; (6) quantitative results, such as risk ratios (RRs) and confidence intervals (CIs), were provided; and (7) publication in English or Chinese.
The exclusion criteria were as follows: (1) studies that did not report detailed data on HS, ICH, or SAH; (2) studies reporting outcomes other than the onset of HS, ICH, or SAH; (3) studies examining long-term risk factors, such as environmental factors of heavy metals, organic pollutants, and radiation, as well as age, hypertension, diabetes, and atherosclerosis; (4) ecological studies, animal studies, or simulation studies; and (5) reviews, letters, or case reports.
Data extraction
Two investigators searched and screened articles in parallel, and articles that met the inclusion criteria were extracted separately for the study region, study year, case definition, sample size, mean age, study design, data source, exposure information (definition and assessment methods), case period, control period, effect values, and other information that would affect the quality and results of the study. The decision was discussed or made by a third person when there was a disagreement.
Quality assessment
There are no formal scales to assess the methodological quality of the CCOSs and SCCSs. As CCOS and SCCS are similar to case-control or cohort studies, we assessed the quality of the literature using the Newcastle–Ottawa Scale. 12 Studies with a score ⩾6 were considered to have low bias.
Statistical analysis
Comprehensive evidence synthesis was conducted using STATA 14.0 software (Stata Corporation). The combination of the effective values (odds ratio (OR) or relative risk (RR)) and 95% CIs from included studies were analyzed using random-effect models (Mantel–Haenszel heterogeneity), and a two-sided
Results
General characteristics of included studies
A total of 441 potentially relevant articles were screened. Finally, 39 candidate articles4,13-50 met the eligibility criteria (Figure 1), including 28 CCOSs, 10 SCCSs, and 1 using both designs. Associations with the onset of HS were assessed among 32 categories of trigger factors (Table 1, Figure 2), which were classified into four main categories: medications (
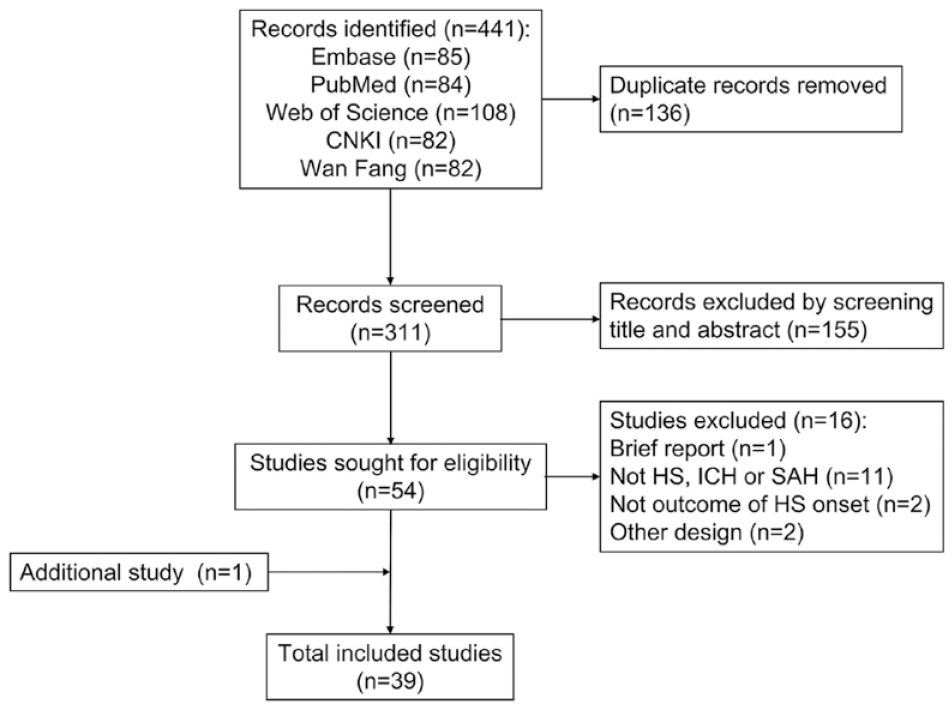
PRISMA flow diagram of the study selection.
Characteristics of included studies.
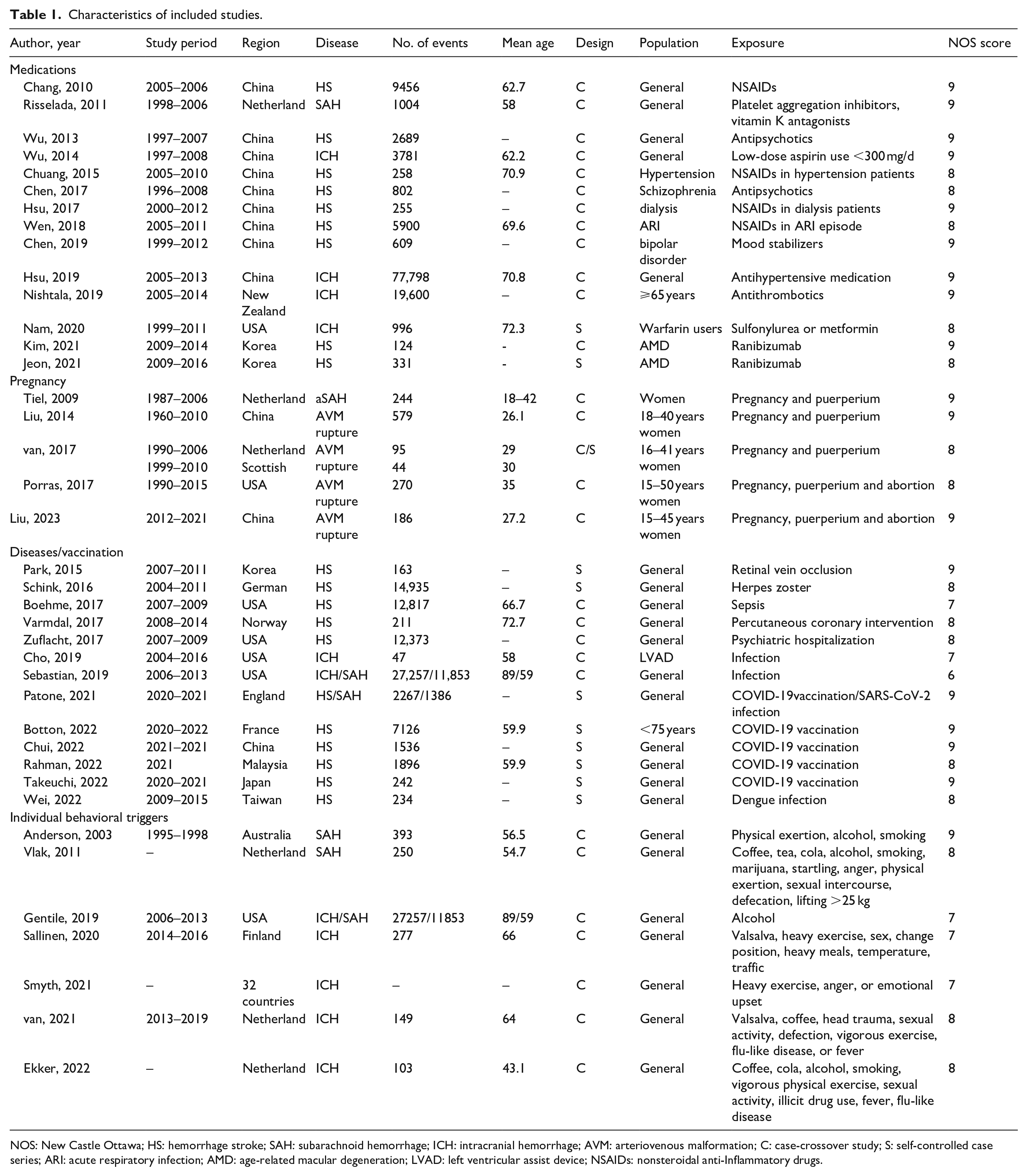
NOS: New Castle Ottawa; HS: hemorrhage stroke; SAH: subarachnoid hemorrhage; ICH: intracranial hemorrhage; AVM: arteriovenous malformation; C: case-crossover study; S: self-controlled case series; ARI: acute respiratory infection; AMD: age-related macular degeneration; LVAD: left ventricular assist device; NSAIDs: nonsteroidal anti-Inflammatory drugs.
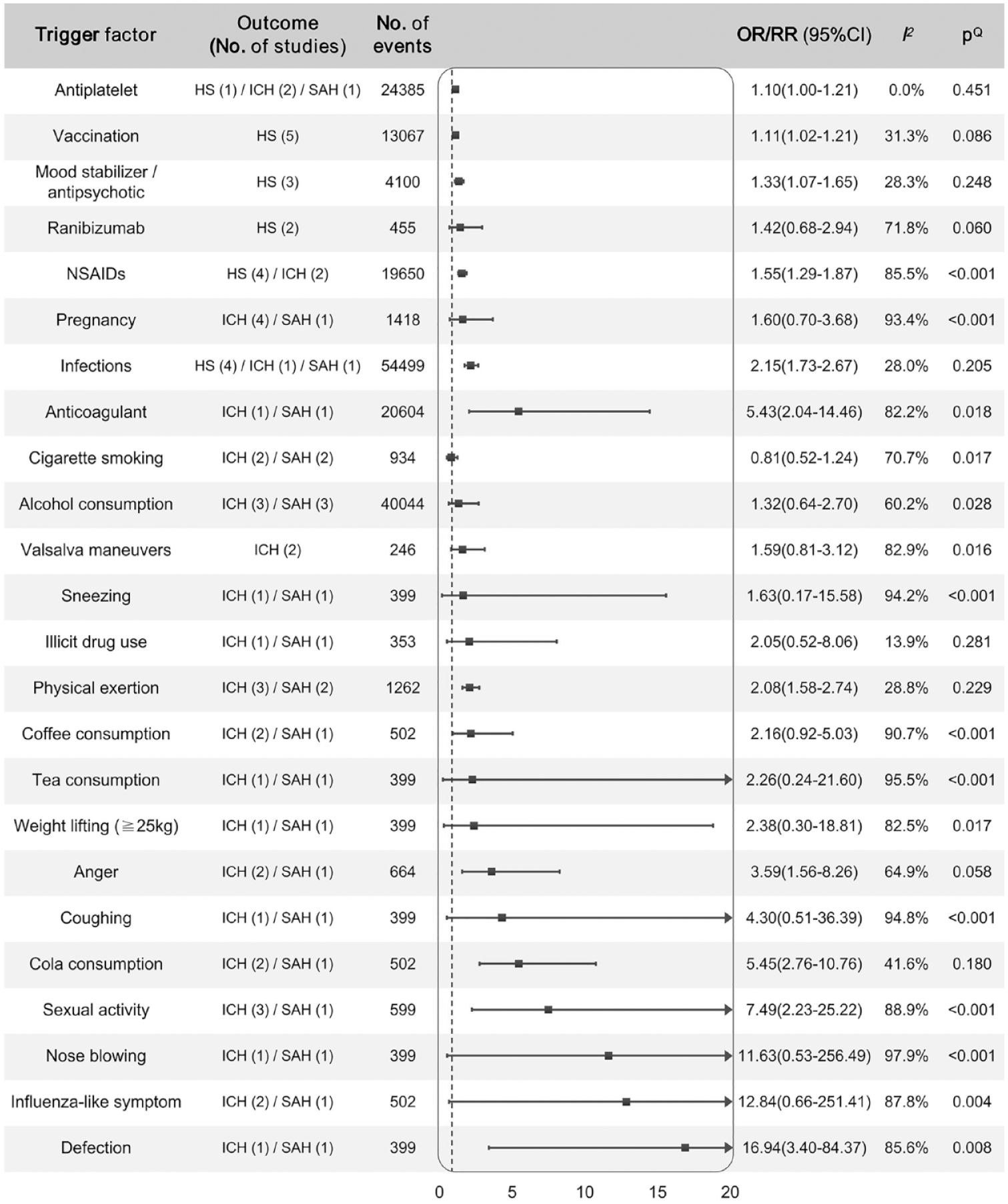
Summarized evidence synthesis of associations between potential trigger factors and hemorrhage stroke.
Medication use and treatments
Sixteen studies examined whether medication use could trigger the onset of HS, including non-steroidal anti-inflammatory drugs (NSAIDs), antiplatelets, anticoagulants, ranibizumab, mood stabilizers/antipsychotics, antihypertensive and hypoglycemic drugs, hospitalization for psychiatric disorders, and percutaneous coronary intervention.13–28 Among them, 14 studies were CCOSs and two were SCCSs (Supplemental Table S1 and Figure S1).
Six CCOSs explored whether NSAID use increased the risk of HS over a short period.13–18 Among Chinese patients, the use of NSAIDs, either orally or by injection, significantly increased the risk of HS within 1 or 3 months of concomitant NSAID intake for acute respiratory infections.13,14 Moreover, two Dutch studies reported an increased risk of HS within 1 month when patients with hypertension or on dialysis were treated with NSAIDs.15,16 Aspirin is an NSAID and antiplatelet agent. A significant association was found between the risk of HS and aspirin use within 7 days in a study of 19,600 patients with ICH in New England,
17
while similar results were not found for aspirin use within 56 days in a study of 3781 Chinese patients
18
or for antiplatelet use within 30 days.
19
The pooled results showed significant associations of HS with both NSAIDs (OR, 1.55; 95% CI, 1.29–1.87) and antiplatelets (OR, 1.10; 95% CI, 1.00–1.21;
Three studies attempted to explore the association between the use of mood stabilizers/antipsychotics and HS in Taiwan, China.20,21 Two studies did not identify the trigger effect of antipsychotics, whereas the other illustrated an increased risk of HS upon the exposure of mood stabilizers (OR, 1.46; 95% CI, 1.28–1.66),
22
consistent with the pooled result (OR, 1.33; 95% CI, 1.07–1.65;
Whether ranibizumab, an anti-angiogenic agent, increases the risk of HS in the short term remains controversial. Two studies were conducted in Korea to determine the association between ranibizumab injection and HS in patients with age-related macular degeneration.23,24 The CCOS showed that ranibizumab treatment was associated with the onset of HS within 2 months (OR, 2.25; 95% CI, 1.07–4.75), 23 while the SCCS did not report a positive result (OR, 1.05; 95% CI, 0.80–1.38). 24
Hsu et al. 25 studied 77,797 Chinese patients and found that the use of short-acting antihypertensive drugs, such as nifedipine, labetalol, and captopril, was associated with the risk of developing ICH (ORs range, 1.99–3.24). As hypoglycemic drugs, neither sulfonylureas nor metformin was associated with an increased risk of HS within 30 days in 996 warfarin users in the United States. 26 An American CCOS identified that the risk of HS was associated with hospitalization for psychiatric disorders within 15 days (OR, 4.29; 95% CI, 2.37–7.76). 27 A CCOS in 211 Norwegian patients with HS did not reveal an association between percutaneous coronary intervention and the risk of HS onset within 28 days. 28
Diseases and vaccination
Several diseases, including infections in different organs, infection with dengue, herpes zoster or SARS-CoV-2 virus, retinal vein occlusion, psychiatric disorders, and percutaneous coronary intervention, and vaccination were evaluated for their association with the onset of HS via the results of three CCOSs and eight SCCSs (Supplemental Table S2 and Figure S2).29–39 An increased risk of HS was identified within 3 months of herpes zoster infection in 14,935 German patients,
29
and one American CCOS found an increased risk of ICH within 14 days of urinary tract infection, 60 days of septic infection, 7 days of respiratory infection, and 15 days of sepsis, but not within 30 days of skin infections.30,31 In addition, a significant association with SAH was observed within 14 days of respiratory infection.
30
Infection within 2 weeks of treatment with a left ventricular assist device was associated with ICH onset in the American population.
32
Patone et al.
33
showed 2.01 (95% CI, 1.29–3.15) and 4.17 (95% CI, 2.59–6.71) times higher risks of HS and SAH, respectively, within 7 days of SARS-CoV-2 infection in England. The pooled results also illustrated a higher risk of HS after infections with low heterogeneity (OR, 2.15; 95% CI, 1.73–2.67;
Recently, five SCCSs33,36–39 attempted to detect whether COVID-19 vaccination would trigger the onset of HS among populations from England, France, Hong Kong, China, Malaysia, and Japan. Significant associations were reported for the Pfizer BioNTech vaccine and HS: the first dose by Patone et al.
33
(RR, 1.24; 95% CI, 1.07–1.43) and Rahman et al.
38
(RR, 1.29; 95% CI, 1.05–1.59); the second dose by Chui et al.
39
(RR, 2.69; 95% CI, 1.54–4.69). Rahman et al.
38
also reported an increased risk of HS after the first dose of CoronaVac (RR, 1.31; 95% CI, 1.02–1.68). However, other studies did not identify this association. Evidence synthesis demonstrated a higher risk of HS within 7–21 days of COVID-19 vaccination (RR, 1.11; 95% CI, 1.02–1.21;
Pregnancy, puerperium, and abortion
Five studies examined whether pregnancy, puerperium, and abortion increased the risk of HS (Supplemental Table S3 and Figure S5).40–44 A study using both the CCOS and SCCS designs in 95 Finnish and 44 Scottish patients reported that pregnancy, delivery, and puerperium were associated with the risk of ICH in the Finnish population but not in the Scottish population.
42
In addition, a significant association was identified in a study of 270 American women with pregnancy, puerperium, and abortion.
43
Two Chinese studies41,44 presented controversial opinions, and an increased risk of HS was not found in Dutch populations
40
or the pooled result (RR, 1.60; 95% CI, 0.70–3.68;
Individual behaviors
The association between 19 behavioral trigger factors and HS has been studied using CCOS among North American and European populations (Supplemental Table S4 and Figure S6).4,45–50 These triggers were divided into three parts: behavioral activities (physical exertion, sexual activity, Valsalva maneuvers, anger, cigarette smoking, and influenza-like symptoms), specific diet changes (alcohol, coffee, tea, and cola consumption, illicit drug use, and heavy meals), and head or traffic trauma.
Except for Sallinen et al.’s study in Finland, studies reported an increased risk of SAH or ICH within 1–2 h after physical activity with a metabolic equivalent (MET) ⩾ 6, which was consistent with the pooled analysis result (OR, 2.08; 95% CI, 1.58–2.74;
The use of illicit drugs were not identified as potential triggers for the onset of HS in the Dutch populations.46,49 Except for the study by Ekker et al.
49
(alcohol consumption: OR [95% CI], 3.4 [1.2–9.5]; cigarette smoking: OR [95% CI], 0.6 [0.5–0.7]), studies did not reveal the association of alcohol consumption46,48,50 or cigarette smoking45,46,48,49 with HS. Coffee and tea consumption and influenza-like symptoms were tested for their trigger effects on HS in Dutch populations.46,48,49 An increased risk of HS was shown within 1 h of coffee consumption by two studies compared with the usual frequency in the past year,46,48 but a significant result was not obtained in a recent study
49
or the pooled analysis (OR, 2.16; 95% CI, 0.92–5.03;
Discussion
This study systematically reviewed 32 categories of individual triggers of HS from 39 studies and performed an evidence synthesis. The results illustrated that the use of antiplatelets and mood stabilizers/antipsychotics, infection, vaccination, physical exertion, and cola consumption were potential triggers for the onset of HS. Although large heterogeneity existed in the pooled results, all the included original studies reported the role of anticoagulants, defecation, nose blowing, and anger in triggering the onset of HS. Other medications, physiological or pathological states, and behavioral factors need to be studied further to gather more evidence. As a multifactorial disease, HS involves a number of factors during the process, including genetic, environmental, and several cerebrovascular factors, besides the individual trigger factors.5,51,52 Their interactions can be involved in promoting the process of HS. Since short-term triggers are relatively easier to prevent, it is essential to avoid such exposures to reduce the risk of acute HS events.
Medication and treatment
The use of antiplatelets and mood stabilizers/antipsychotics was significantly associated with increased HS risk, with less heterogeneity. Although large heterogeneity existed, the use of anticoagulants should be a potential trigger, given that all the included studies reported a significant association with HS. The use of antihypertensives, psychiatric hospitalization, and coronary intervention have been reported to be associated with the onset of HS, but more evidence should be provided to enhance reliability.25,27,28,35 The use of NSAIDs was associated with HS but with a large heterogeneity. Despite that most included studies reported consistent results, more evidence should be obtained for further verification.
Antihypertensive drugs may induce an abrupt blood pressure change and cause hemodynamic cataclysm, finally leading to arterial rupture, whereas antiplatelet and anticoagulant drugs can inhibit the platelet and blood coagulation systems, then promote hemorrhage (Figure 3). Although studies have reported a higher risk of HS following the use of mood stabilizers/antipsychotics, the underlying mechanism remains unclear. Despite the HS risk, patients still benefit from these medications because of their protection against diseases. For example, antiplatelet and anticoagulant drugs marginally increase the risk of bleeding while also contributing to the prevention of new cardiovascular events.53,54 Physicians should promote personalized treatment, and patients should have regular follow-up visits and drug monitoring to balance the risk of acute HS with the benefits of managing basic diseases. 55
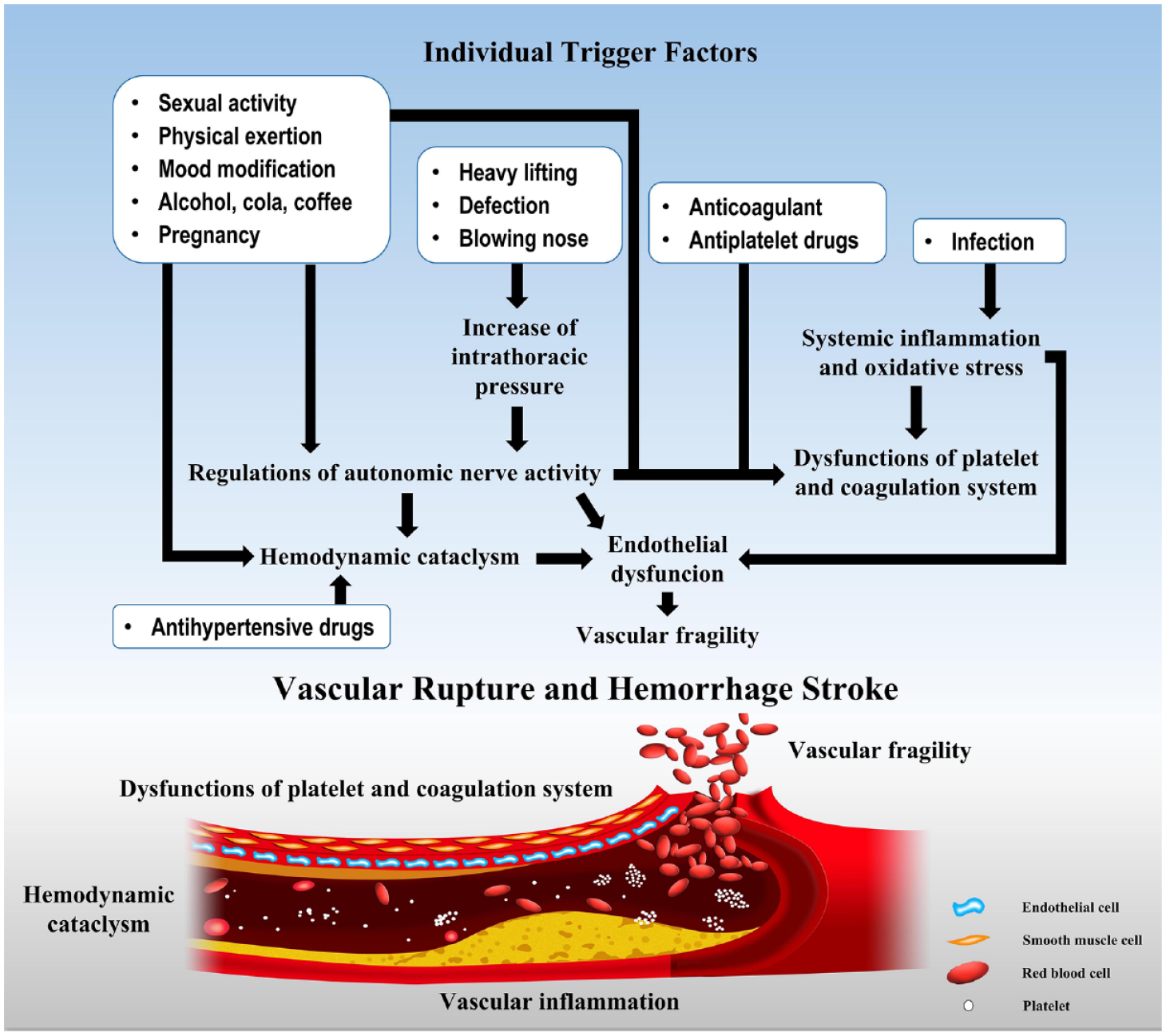
Potential risk factors associated with the onset of HS and underlying mechanisms.
Infections and vaccination
Initially, non-negligible heterogeneity existed regarding the trigger effect of infections in our results due to the inclusion of the study by Sebastian et al. 30 on skin infections. We suspected that infections localized in the skin might have less potential to spread and cause systemic symptoms than infections in other systems. Heterogeneity reduced after eliminating the study, and both the pooled result and each remaining study showed a significant association between infection and HS risk. Considering that patients with a specific infection of the dengue virus usually show intensive symptoms of fever and platelet dysfunction, we distinguished it from other infections for evidence synthesis.
Infections or vaccination, a synthetic artificial infection, can promote systematic inflammation and oxidative stress and induce multisystem dysfunction, including in vascular systems and endothelial cells. Then, the vascular wall weakens and can get broken due to the impact of blood flow, while platelet and blood coagulation systems are affected to aggravate bleeding (Figure 3).5,6 Given the increased risk of HS following infections in different organ systems by various pathogens, the high-risk populations should enhance their immunizations. During the infection, taking active therapeutics may help patients pass the risk period and reduce the risk of HS. The included studies using the SCCS design compared the risk of HS among each vaccinated individual; however, evidence from cohort studies is also needed to compare the incidence of HS between populations with and without vaccination. It should not be ignored that humans benefit from vaccines that induce immunity against corresponding infectious diseases. Thus, further investigation regarding the COVID-19 vaccination is needed for a comprehensive evaluation and recommendation.
Females and pregnancy
We could not conclude whether the state of pregnancy, puerperium, and abortion increase the risk of HS due to large heterogeneity among the studies40–44; however, a recent study successfully summarized the association of these factors with brain arteriovenous malformation-related ICH. 44 Evidence synthesis was conducted combining their original data by subgroups according to different study designs, and a relatively moderate risk was obtained during female childbearing state. They also explained a potential mechanism by which females experience turbulent blood volume and coagulation changes induced by increased secretion of progesterone during pregnancy and puerperium, finally leading to a higher risk of HS. 44 When making clinical decisions for high-risk females contemplating pregnancy, clinicians should comprehensively consider their experience as well as the characteristics and intentions of their patients.
Behaviors and living habits
Among the 19 included factors, the results of three factors had less heterogeneity: physical exertion and cola consumption were identified as potential trigger factors of HS, whereas the association between use of illicit drugs and HS was not statistically significant. Although large heterogeneity existed in the results for sexual activity, defecation, and anger, they should also be considered candidate triggers because almost the original studies reported significant associations with a higher risk of HS. A similar situation was observed among studies on the use of illicit drugs, where both the original studies and pooled result denied its association with the onset of HS. Cigarette smoking is generally acknowledged as a hazard behavior, but one study 49 reported that it was a protective factor of SAH. Although the pooled result did not show the association between smoking and HS, considering the included studies with a small sample size, further investigation is needed to enhance the robustness. However, several behavioral triggers have a controversial role in the onset of SAH and ICH in Dutch populations,46,48 including tea consumption, some Valsalva maneuvers, and influenza-like symptoms, and further studies are needed. Influenza-like symptoms, but not the viral infection, were considered a behavioral trigger and separated from the Valsalva maneuvers of coughing, sneezing, and nose-blowing because the original research was based on questionnaires provided by patients and not on diagnoses from the clinics, and symptoms usually lasted for several days.46,48,49
Behavioral activities and mood modifications can induce an abrupt blood pressure change via enhanced sympathetic activity, and increased consumption of caffeine and alcohol may also cause hemodynamic cataclysm, finally leading to arterial rupture and HS (Figure 3).5,6 At the individual level, targeting measures to avoid short-term changeable triggers and minimizing exposure would be efficient ways to reduce the burden of HS, particularly in high-risk populations.
Strengths and limitations
This study comprehensively searched for studies that used CCOS and SCCS designs to investigate the transient trigger factors of HS since these two designs treat the cases as the controls at different periods (risk and control periods). Thus, both selection bias of control and confounding bias were eliminated to confirm comparability, and many advantages have been shown in the analysis of transient exposure factors and the elimination of confounding bias from most chronic risk factors.8,9 Then, evidence synthesis was conducted to test the robustness of the 32 trigger factors from 39 studies.
However, this study had several limitations. First, most of the pooled results showed large heterogeneity owing to different populations and definitions of case and control periods for each factor. Most of the studies had small sample sizes, resulting in low robustness of the results. Another reason for low robustness could be that the included studies evaluated the associations between triggers and HS by calculating the RRs or ORs, between which there were still several differences. Some studies have performed the analyses without separating the subtypes of SAH and ICH, which might have resulted in low specificity of the results. Although we conducted a subgroup analysis, heterogeneity could not be reduced, and 95% CIs were relatively wide. Thus, multicenter cohorts with larger sample sizes are needed to comprehensively explore the triggers of specific HS subtypes. Second, recall and information biases existed because the studies relied on patient self-reports. In addition, within-person confounding factors exist when a patient is exposed to multiple triggers with temporal trends. However, none of the existing studies adjusted them using a multivariate analysis. 56 Additionally, the included studies were mainly on North American and European populations; however, explorations focusing on populations of various regions and races are also necessary because trigger factors might vary in distinct regions because different populations behave with their specific features.
Conclusion
Associations between the onset of HS and several trigger factors were identified, including medications (NSAIDs, anticoagulants, and antihypertensive drugs), definite states of diseases (infections and retinal vein occlusion), hospitalization for psychosis, and individual behaviors (heavy physical exercise, sexual activity, cola consumption, nose blowing, defection, and anger). Evidence synthesis showed an increased risk of HS associated with the use of antiplatelets and mood stabilizers/antipsychotics, infections, vaccinations, physical exertion, and cola consumption. Avoiding exposures to behavioral triggers is crucial to prevent HS. At the medical level, individualized treatments should be introduced, and measures should be monitored, especially for high-risk populations. Further studies with larger sample sizes are needed to evaluate the interactions among various risk factors and enhance effective stroke prevention.
Supplemental Material
sj-docx-1-eso-10.1177_23969873231173285 – Supplemental material for Individual trigger factors for hemorrhagic stroke: Evidence from case-crossover and self-controlled case series studies
Supplemental material, sj-docx-1-eso-10.1177_23969873231173285 for Individual trigger factors for hemorrhagic stroke: Evidence from case-crossover and self-controlled case series studies by Junyu Liu, Chun Luo, Yuxin Guo, Fang Cao and Junxia Yan in European Stroke Journal
Supplemental Material
sj-pdf-2-eso-10.1177_23969873231173285 – Supplemental material for Individual trigger factors for hemorrhagic stroke: Evidence from case-crossover and self-controlled case series studies
Supplemental material, sj-pdf-2-eso-10.1177_23969873231173285 for Individual trigger factors for hemorrhagic stroke: Evidence from case-crossover and self-controlled case series studies by Junyu Liu, Chun Luo, Yuxin Guo, Fang Cao and Junxia Yan in European Stroke Journal
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the grants from the Hunan Province Nature Science Foundation, China (2021JJ30911); Central South University Case Database Construction Project for Graduate Students, China (2020ALK24).
Informed consent
Not applicable.
Ethical approval
Not applicable.
Guarantor
JY, MD, Department of Epidemiology and Health Statistics and Hunan Provincial Key Laboratory of Clinical Epidemiology, Xiangya School of Public Health, Central South University, Shang Mayuanling, Changsha, 410078, China. E-mail:
Contributorship
JL: data curation, statistical analysis, validation, resources and software, writing for original draft & review & editing; CL: data curation, statistical analysis, investigation, validation, resources and software, writing for original draft; YG and FC: data curation, statistical analysis; JY: project administration and supervision, methodology, funding acquisition, statistical analysis, writing for original draft & review & editing.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
