Abstract
Background:
The potential benefit of intraarterial tenecteplase in acute basilar artery occlusion (BAO) patients with successful reperfusion following endovascular treatment (EVT) has not been studied.
Aims:
To explore the efficacy and safety of intraarterial tenecteplase in acute BAO patients with successful reperfusion after EVT.
Sample size estimates:
A maximum of 228 patients are required to test the superiority hypothesis with 80% power according to a two-side 0.05 level of significance, stratified by center.
Design:
We will conduct a prospective, randomized, adaptive-enrichment, open-label, blinded-end point, multicenter trial. Eligible BAO patients with successful recanalization after EVT [modified Thrombolysis in Cerebral Infarction (mTICI) 2b-3] will be randomly assigned into the experimental and control group with a 1:1 ratio. Patients in the experimental group will receive intraarterial tenecteplase (0.2–0.3 mg/min for 20–30 min), while patients in the control group will receive routine treatment according to the usual practice of each center. Patients in both groups will receive standard guideline-based medical treatment.
Outcome:
The primary efficacy endpoint is a favorable functional outcome, defined as the modified Rankin Scale 0–3 at 90 days after randomization. The primary safety endpoint is symptomatic intracerebral hemorrhage, defined as National Institutes of Health Stroke Scale score increase ⩾4 caused by intracranial hemorrhage within 48 h after randomization. Subgroup analysis of the primary outcome will be performed by age, gender, baseline NIHSS score, baseline pc-ASPECTS, intravenous thrombolysis, time from estimated symptom onset to treatment, mTICI, blood glucose, and stroke etiology.
Conclusions:
The results of this study will provide evidence of whether adjunct use of intraarterial tenecteplase after successful reperfusion with EVT is associated with better outcomes for acute BAO patients.
Keywords
Introduction and rationale
Basilar artery occlusion (BAO) accounts for approximately 1% of ischemic strokes and causes high morbidity and mortality in up to 70% of patients.1,2 Early reperfusion treatments including intravenous thrombolysis and endovascular treatment (EVT) are typically demonstrated to be associated with favorable functional outcomes for anterior circulation patients with large vessel occlusions (LVO).3,4 The benefit of EVT for acute BAO patients was controversial with two early neutral trials (BEST and BASICS).5–8 Although, two recent randomized controlled trials (BAOCHE and ATTENTION trials) have demonstrated that EVT was associated with better functional outcomes and survival at 90 days for acute BAO patients,9,10 only 46% of patients achieved good neurological outcomes (mRS 0–3) at 90 days.
In contrast to anterior circulation stroke, acute posterior circulation stroke has distinct characteristics such as more perforator branches, vulnerability to neurologic deterioration, yet a relatively good response to intravenous thrombolysis.11–13 Moreover, there may be many small branch artery occlusions or distal microembolism from proximal thrombi, which may be one of the reasons for the poor prognosis of acute BAO patients after successful recanalization. Recently, the CHOICE (Chemical OptImization of Cerebral Embolectomy) trial revealed that adjunct intraarterial alteplase may improve functional outcomes without an increased risk of symptomatic intracranial hemorrhage (sICH) following successful reperfusion for anterior LVO stroke patients. 14 Moreover, prior studies showed that intravenous tenecteplase (TNK) is associated with higher reperfusion rates and better functional outcome than alteplase in bridging EVT patients with LVO both in anterior and posterior circulation,15,16 although this was not confirmed in the ACT trial. 17 In a Chinese population of ischemic stroke patients not eligible for EVT, intravenous TNK was shown to be non-inferior to alteplase. 18 Moreover, intra-arterial TNK in acute stroke was found to be safe and result in comparable favorable outcomes to alteplase. 19 Our recent study (BRETIS-TNK trial, NCT 04202458, unpublished) also showed that intra-arterial TNK during the first pass of EVT seemed safe, may increase first-pass reperfusion and improve good outcome in patients with LVO.
In this context, we hypothesize that intraarterial TNK after successful reperfusion may be effective and safe in EVT patients with acute BAO. Thus, the Improving Neurological outcome for acute baSilar artery occlusIon with Sufficient recanalization after Thrombectomy by Intraarterial Tenecteplase (INSIST-IT) trial is designed to explore this hypothesis.
Methods
Design
INSIST-IT is a prospective, randomized, adaptive-enrichment, open label, blinded-end point, multi-center study in China, aiming to estimate the efficacy and safety of intraarterial TNK treatment following successful reperfusion in patients with acute BAO treated with EVT. The trial flowchart is shown in Figure 1.
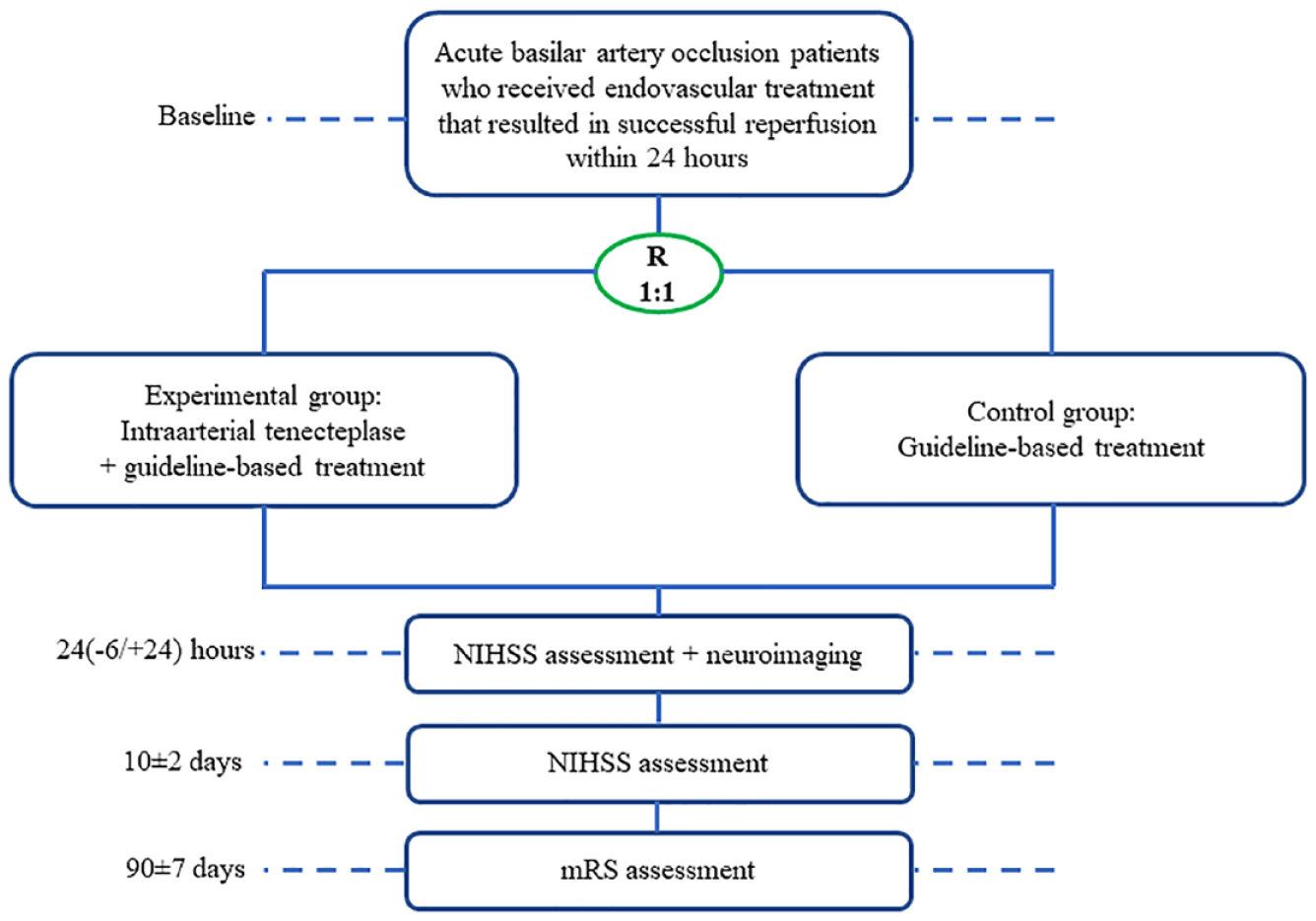
Study flowchart.
Study population
In the INSIST-IT trial, eligible participants are patients with acute BAO (National Institute of Health Stroke Scale, NIHSS ⩾ 6) following EVT resulting in a modified Thrombolysis in Cerebral Infarction (mTICI) score 2b-3, as judged by the investigator. The patients will be enrolled at approximately 20 comprehensive stroke centers in China between March 2023 and March 2025. A detailed description of the inclusion/exclusion criteria is provided in Table 1.
The inclusion/exclusion criteria.

mTICI: modified Thrombolysis in Cerebral Infarction; CT: computed tomography; MRI: magnetic resonance imaging; INR: international normalized ratio; ALT: alanine aminotransferase; AST: aspartate aminotransferase.
Standard protocol approvals, registrations, and patient consents
The protocol of the INSIST-IT trial and related data collection have or will be approved by the ethics committee of the General Hospital of Northern Theater Command and all participating sites. All patients or their legally authorized representatives will provide signed informed consent. The INSIST-IT trial is registered on www.clinicaltrial.gov (NCT05657457).
Randomization and intervention
Eligible patients will be divided into an experimental and control group at a 1:1 ratio by using central, computerized random sequence generation as stratified by center. Intravenous alteplase (0.9 mg/kg, maximum dose 90 mg), TNK (0.25 mg/kg, maximum dose 25 mg), or urokinase (1,000,000–1,500,000 U) can be used before EVT according to institutional guidelines, but the actual dose will be recorded due to potential difference among different sites. EVT will be carried out according to the usual practice of each center, including EVT with stent retrievers, contact aspiration, balloon angioplasty, stenting, or a combination of these approaches. The patients in the experimental group will receive intraarterial TNK for 20–30 min from the guide or intermediate catheter in the vertebral artery: (1) TNK at a rate of 0.3 mg/min if there is neither intravenous thrombolysis nor salvage balloon angioplasty and/or stenting; (2) TNK at a rate of 0.25 mg/min if there is either intravenous thrombolysis or salvage stent deployment; (3) TNK at a rate of 0.2 mg/min if there is both intravenous thrombolysis and salvage stent deployment. All stroke centers are required to start intraarterial TNK within 10 min after randomization. If mTICI 3 is achieved before intraarterial TNK, TNK will be given for 20 min. If mTICI 3 is not achieved, DSA will be performed 20 min after intraarterial TNK infusion: if mTICI 3 is achieved, the IA TNK infusion will be stopped; if mTICI 3 is not achieved, IA TNK infusion will be given for an additional 10 min. DSA will be performed after the end of intraarterial TNK administration. If contrast extravasation occurs during the intraarterial TNK, which is suspected to be related to active bleeding, TNK infusion will be stopped immediately. Patients in both groups will receive standard medical therapy, in accordance with guidelines for the management of acute ischemic stroke. 20
To minimize the risk of reperfusion-related intracranial hemorrhage, systolic blood pressure is recommended to maintain below 160 mmHg.21,22
The antiplatelet strategy is recommended as follows. If the patient has not received intravenous thrombolysis, patients will take antiplatelet therapy (aspirin and/or clopidogrel) when CT exclude cerebral hemorrhage after the endovascular treatment. If the patient received intravenous thrombolysis, antiplatelet therapy (aspirin and/or clopidogrel) will begin 24 h after the administration of intravenous thrombolysis and confirmation of absence of intracranial hemorrhage on a preceding CT scan. After endovascular treatment, aspirin and/or clopidogrel tablets were administrated orally. If patients underwent angioplasty/stenting, an intra-arterial bolus (0.4–0.6 mg) followed by intravenous maintenance dose of 0.1 μg/kg/min tirofiban for 12–24 h. Dual antiplatelet therapy (aspirin 100 mg and clopidogrel 75 mg) was given before the end of tirofiban. The actual antiplatelet therapy will be determined by the operator based on their clinical and neuroimaging judgment, which will be recorded in detail.
Outcomes
The primary efficacy endpoint is the proportion of patients achieving a favorable functional outcome, defined as a modified Rankin Scale (mRS) score of 0–3 at 90 days.
The secondary efficacy endpoints are a comparison between the active and control groups as follows: (1) the proportion of patients with an improved mTICI score (from 2b/c to 3) after intraarterial TNK administration or at the end of procedure; (2) the proportion of patients with mRS 0–1 at 90 days; (3) the proportion of patients with mRS 0–2 at 90 days; (4) ordinal distribution of mRS at 90 days; (5) change in mRS at 90 days compared with premorbid mRS; (6) change in NIHSS at 24 (−6/ + 24) h, compared with the NIHSS at baseline; (7) the proportion of early neurological improvement (ENI), defined as a NIHSS decrease ⩾4 within 24 (−6/ + 24) h; (8) change in the cerebral circulation time after treatment, defined as the time from the appearance of contrast at the V2 segment of the vertebral artery to the end of the basilar artery during DSA; (9) the composite of recurrent stroke, cardiovascular or cerebrovascular events within 90 days after randomization.
The primary safety endpoint is sICH, which is defined as a NIHSS increase ⩾4 caused by intracranial hemorrhage within 24 (−6/ + 24) h after randomization. 23
The secondary safety endpoints are: (1) the rate of intraparenchymal hemorrhage (PH1 and PH2) within 24 (−6/ + 24) h after randomization; (2) the percentage of severe adverse events within 24 (−6/ + 24) h after randomization; (3) cerebral edema within 24 (−6/ + 24) h after randomization; (4) all-cause mortality within 10 ± 2 days of randomization or during hospitalization, whichever is earlier; (5) the number of TNK infusions interrupted due to suspected active bleeding.
Follow-up procedure
All included patients will have an in-person or telephone interview at baseline, 24 (−6/ + 24) h, 10 ± 2 days, and 90 ± 7 days post-randomization, respectively (Table 2). To avoid bias, the follow-up NIHSS scores will be obtained by the same neurologist, and the 90-day clinical assessments including mRS will be evaluated by one qualified personnel blinded to treatment allocation according to a standardized procedure manual in each study center. We will have a training course for all investigators at each center. The primary endpoint will be evaluated by in-person or by telephone interview if an in-person evaluation is not possible. The duration of this research is expected to be approximately 24 months.
Timing of all procedures in INSIST-IT.

CT: computed tomography; MRI: magnetic resonance imaging; NIHSS: National Institute of Health Stroke Scale; mRS: modified Rankin Scale; mTICI: modified Thrombolysis in Cerebral Infarction; TOAST: Trail of Org 10172 in Acute Stroke Treatment.
Treatment arm only.
CT + CTA or MRI + MRA.
At the end of procedure.
Data management and quality control
All patient data and case report forms will be entered in MedSci (http://INSIS-IT.medsci.cn). The data will be downloaded from MedSci with a dedicated person for statistical analysis. An independent Data Safety and Monitoring Committee (iDSMC) will perform data checking in this study in an unblinded manner for safety reasons, such as hemorrhagic events and other adverse events (AEs), etc. A central core lab for imaging will judge intracranial bleeding events and assess neuroimaging data. Sequential interim analyses will be performed after every 50 enrolled patients who have been followed up for safety analysis by a statistician of iDSMC, who is not involved in managing the trial. Based on this analysis, iDSMC can recommend to the Steering Committee of the INSIST-IT trial to adjust the sample size, enrich the study, remove a patient subgroup (e.g. a subgroup at high-risk of sICH who received intravenous thrombolysis before EVT and antiplatelet for rescue angioplasty/stenting), or early termination of the study. No formal analysis of the primary endpoint will be performed at interim analysis, to avoid a type-1 error.
Adverse event monitoring
An AE is any adverse medical event that occurs during the study. All information about AEs will be recorded. The AE will be adjudicated as associated with intraarterial TNK treatment, the procedure, the index stroke, intercurrent condition, incidental finding, or other reasons and will be further adjudicated by iDMSC.
Sample size determination
According to a previous basilar artery registry, 8 the proportion of good functional outcome at 90 days (mRS 0–3) in the control group was 32%. Based on CHOICE, 14 we estimate an absolute increase in good functional outcome of 19% in the experimental compared to the control group. After a one-sided test was performed with a power of 80% and an α set at 0.025, the calculated sample size to test the superiority hypothesis is 204 cases. Considering a loss to follow-up rate of 10%, the sample size was therefore readjusted to 227 cases. Therefore, this study was designed to include 228 patients, with 114 patients in each group.
Statistical analysis
An intention-to-treat (ITT) analysis will be used to analyze the therapeutic effects of the two groups and all data will be analyzed with SPSS 23.0 Software. The mean ± standard deviation (SD) will be used if the data are normally distributed. The median and quartile range (IQR) will be used if the data are not normally distributed. Count data are expressed as n (%). Differences in the primary and secondary endpoints such as mRS (0–3) at 90 days, mRS (0–2) at 90 days, mRS (0–1) at 90 days, improved mTICI, ENI, the percentage of recurrent stroke, cardiovascular and cerebrovascular events within 90 days, proportion of sICH within 24 (−6/ + 24) h, incidence of intraparenchymal hemorrhage (PH1 and PH2) within 24 (−6/ + 24) h, incidence of severe adverse events within 24 (−6/ + 24) h, and occurrence of all-cause death within 10 days will be compared using binary logistic regression. Distribution of the mRS at 90 days between the two groups will be compared using the ordinal logistic regression. Change in NIHSS score, cerebral edema, and the cerebral circulation time between the two groups will be compared using a general linear model. Time-to-events of stroke recurrence and other vascular events will be compared using Cox regression. There is statistical significance if the p value is <0.05.
The primary endpoint in INSIST-IT will be further analyzed by the following subgroups: (1) intravenous thrombolysis (yes vs no); (2) estimated onset of symptoms to TNK time (⩽12 h vs 12–24 h); (3) NIHSS score on admission (4–10 vs >10); (4) age (<60 vs ⩾60); (5) gender (men vs women); (6) stroke etiology (large-artery atherosclerosis vs cardioembolism); (7) baseline pc ASPECTS (6–7 vs 8–10; (8) mTICI (2b vs 2c/3); (9) blood glucose (⩽100 mg/dl vs >100 mg/dl).
Study organization and funding
The protocol was designed by Hui-Sheng Chen and discussed by the trial steering committee. The trial steering committee is comprised of external scientific advisors, who will monitor the research regularly. The trial steering committee will organize teleconference or physical meetings to provide recommendations on the trial. Neuroimaging associated with clinical events will be collected centrally and interpreted by two independent neuroradiologists. The trial is initiated by the Cerebrovascular Disease Collaboration & Innovation Alliance (CDCIA) of Liaoning and supported by grants from the Science and Technology Project Plan of Liao Ning Province and CSPC Recomgen Pharmaceutical (Guangzhou) Co., LTD.
Current status
At the time of the submission of this article, the trial has not begun patient recruitment. Recruitment will be continued until the complete sample size is achieved, which is expected to be in March 2025.
Discussion
The evidence to support the benefit of endovascular therapy in patients with basilar artery occlusion has only been demonstrated recently.24,25 Two prior multicenter randomized trials did not detect favorable outcomes in BAO patients receiving EVT compared with those receiving standard medical therapy.5,6 Two multicenter, registry studies suggested the benefit of EVT for BAO patients.7,8 Even so, another challenge that is often encountered in BAO patients receiving EVT is the relatively high rate of reperfusion without functional independence: only about half of patients have a good functional outcome at 90 days (mRS 0–3) although with a > 85% sufficient reperfusion rate.9,10 Nonetheless, a pharmacological adjunct such as intraarterial thrombolysis that could enhance the clinical benefit of the endovascular procedure is of importance. In this context, we conduct this trial aiming to estimate the efficacy and safety of intraarterial TNK treatment in acute BAO patients treated with EVT that resulted in successful reperfusion on digital subtraction angiography.
In contrast to the CHOICE trial, 14 which was designed to evaluate the efficacy and safety of adjunct intraarterial alteplase in anterior LVO stroke patients treated with EVT, the INSIST-IT trial will use TNK as the intraarterial thrombolytic agents and target patients with acute BAO. We choose TNK, based on several considerations. TNK has greater fibrin specificity and a longer half-life compared with alteplase. In a meta-analysis of four randomized trials, TNK was associated with better recanalization and good functional outcome for patients with LVO compared with alteplase. 26 A retrospective analysis of the Basilar Artery Treatment and Management (BATMAN) registry and the Tenecteplase versus Alteplase before Endovascular Therapy for Ischemic Stroke (EXTEND-IA TNK) trial showed that acute ischemic patients with BAO receiving intravenous TNK before EVT have significantly better recanalization and clinical outcome in comparison with alteplase. 16 In China, data in support of TNK has now been shown to be non-inferior to alteplase in an ischemic stroke population that is not eligible for or refused EVT via the TRACE-2 randomized trial. 18 Moreover, the safety of intraarterial TNK in acute stroke was confirmed in the previous study. 19 We hypothesize that adjunct intraarterial TNK would produce a comparable or better efficacy and safety profile in acute BAO patients, given the distinct characteristics of the posterior circulation such as more branch vessels, which may be susceptible to the formation of small artery thrombi or micro-thrombi.
The different doses of intra-arterial TNK in different subgroups were chosen based on discussion with the Steering Committee due to lack of available references regarding IA dosing, aiming to minimize sICH risk. Furthermore, the positive results of the recent BAOCHE and ATTENTION trials9,10,27 will encourage physicians to perform thrombectomy for BAO in patients with moderate to severe stroke, which will help enroll patients during the 2-year trial cycle.
Conclusions
The results of INSIST-IT will provide important evidence for the efficacy and safety of intraarterial TNK, as a new promising adjuvant therapy for acute BAO stroke population following endovascular treatment in clinical practice.
Supplemental Material
sj-docx-1-eso-10.1177_23969873231164790 – Supplemental material for Improving neurological outcome for acute basilar artery occlusion with sufficient recanalization after thrombectomy by intraarterial tenecteplase (INSIST-IT): Rationale and design
Supplemental material, sj-docx-1-eso-10.1177_23969873231164790 for Improving neurological outcome for acute basilar artery occlusion with sufficient recanalization after thrombectomy by intraarterial tenecteplase (INSIST-IT): Rationale and design by Liang Liu, Wei Li, Jing Qiu, Thanh N Nguyen, Ming Wei, Feng Wang, Di Li, Huai-Zhang Shi, Shou-Chun Wang and Hui-Sheng Chen in European Stroke Journal
Footnotes
Acknowledgements
We are grateful to the steering committee and iDSMC for their contributions to this study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The work was supported by grants from the Science and Technology Project Plan of Liao Ning Province (2022JH2/101500020).
Ethical approval
This retrospective study was approved by the Institutional Review Board of General Hospital of Northern Theater Command (IRB: Y (2022) 185).
Informed consent
The authors declare that they consent for publication.
Guarantor
I will take full responsibility for the article, including for the accuracy and appropriateness of the reference list.
Contributorship
L.L. and W.L. wrote the first draft of the manuscript. J.Q., T.N.N., M.W., F.W., D.L., H.Z.S., and S.C.W. critically revised the manuscript. H.S.C. designed the study and critically revised the manuscript. All the authors have carefully read and approved the article.
Data availability statement
Not Applicable
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
