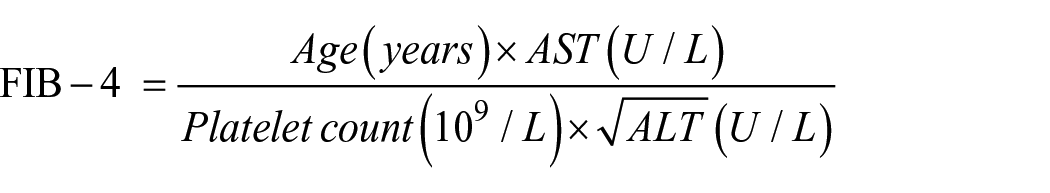Abstract
Background:
Several blood biomarkers have been identified as predictors for poor outcome after ischemic stroke. However, recent studies mainly focused on single or experimental biomarkers and considered rather short follow-up intervals limiting their value for daily clinical practice. We, therefore, aimed to compare various clinical routine blood biomarkers for their predictive value on post-stroke mortality over a 5-year follow-up period.
Patients and methods:
This data analysis of a prospective single-center study included all consecutive ischemic stroke patients admitted to the stroke unit of our university hospital over a 1-year period. Various blood biomarkers of inflammation, heart failure, metabolic disorders, and coagulation were analyzed from standardized routine blood samples collected within 24 h of hospital admission. All patients underwent a thorough diagnostic workup and were followed for 5 years post-stroke.
Results:
Of 405 patients (mean age: 70.3 years), 72 deceased (17.8%) during the follow-up period. While various routine blood biomarkers were associated with post-stroke mortality in univariable analyses, only NT-proBNP remained an independent predictor (adjusted odds ratio 5.1; 95% CI 2.0–13.1; p < 0.001) for death after stroke. NT-proBNP levels ⩾794 pg/mL (n = 169, 42%) had a sensitivity of 90% for post-stroke mortality with a negative predictive value of 97% and was additionally associated with cardioembolic stroke and heart failure (each p ⩽ 0.05).
Conclusion:
NT-proBNP represents the most relevant routine blood-based biomarker for the prediction of long-term mortality after ischemic stroke. Increased NT-proBNP levels indicate a vulnerable subgroup of stroke patients in which early and thorough cardiovascular assessment and consistent follow-ups could improve outcome after stroke.
Background
Ischemic stroke is a leading cause of death and disability worldwide.1–3 Apart from classical predictors of post-stroke outcome (such as initial stroke severity and concomitant chronic diseases), 4 various blood biomarkers were reported to identify patients at increased risk for complications and poor clinical prognosis after stroke. In this context, markers of (sub)clinical inflammation (C-reactive protein (CRP), neutrophil-to-lymphocyte ratio (NLR)),5–7 activation of the coagulation system (D-dimer, fibrinogen),5,8–10 liver fibrosis (Fibrosis-4 index (FIB-4)),11,12 and heart failure (N-terminal pro-brain natriuretic peptide (NT-proBNP))13–15 were associated with post-stroke mortality. However, most recent studies primarily focused on single biomarkers or investigated experimental markers (such as growth/differentiation factor 15, resistin, and von Willebrand factor) typically not available in clinical routine, which limits their use in the management of ischemic stroke patients.6,7,9,16–18 In addition, the impact of such markers on the long-term prognosis after stroke remains unknown, as earlier studies mainly examined outcomes over follow-up periods of 3–12 months.6,9–11,16,18,19
This study, therefore, aimed to compare various clinical routine blood biomarkers for their predictive value on post-stroke mortality and as a secondary outcome on recurrent vascular events over a long-term follow-up period of 5 years.
Patients and methods
Data were collected from a prospectively designed single-center cohort study. All consecutive patients with ischemic stroke who were admitted to the stroke unit of our primary and tertiary care university hospital over a 1-year period between April 2017 and April 2018 were included.
Demographics, medical history, cerebrovascular risk factors, pre-stroke modified Rankin scale (mRS) scores, and stroke severity according to the National Institute of Health Stroke Scale (NIHSS) at stroke unit admission were documented. We only included acute stroke patients with a visible ischemic infarction on cerebral imaging (mostly based on magnetic resonance imaging (MRI): 89%). All ischemic stroke patients underwent a standardized detailed diagnostic work-up including laboratory tests, 12-lead electrocardiography (ECG) at admission, continuous 3-lead ECG monitoring at the stroke unit for at least 48 h, and duplex sonography of the intra- and extracranial vessels. All patients that underwent brain MRI were investigated with time-of-flight MR-angiography and selected patients were also examined with contrast-enhanced MR-angiography of extra- and intracranial brain-supplying arteries. Transthoracic echocardiogram (TTE) was routinely performed, whereas transesophageal echocardiography (TEE) was additionally conducted in younger patients (<60 years of age) or if specific clinical signs of cardiac disease/embolism (such as suspected endocarditis) were present. Stroke etiology was defined according to the Trail of Org 10172 in Acute Stroke Treatment (TOAST) classification. 20 A detailed description on etiological workup of this cohort has been previously reported. 21
Based on patient’s clinical signs and symptoms, TTE/TEE results, and NT-proBNP levels heart failure (HF) was diagnosed according to current guideline recommendations and classified into (a) HF with preserved ejection fraction (HFpEF: left ventricle ejection fraction (LVEF) ⩾50%); (b) HF with mildly reduced ejection fraction (HFmrEF: LVEF 41%–49%); and (c) HF with reduced ejection fraction (HFrEF: LVEF ⩽40%). 22 HFpEF was diagnosed if HFA-PEFF scores ⩾5 points were present. 23
Approval of this study was granted by the Ethics Committee of the Medical University of Graz. All patients gave written informed consent (approval number: 29–285ex 16/17).
Blood biomarkers assessment
Clinical routine blood markers for the work-up of stroke patients (blood count, CRP, lipid profile, glucose, HbA1c, electrolytes, liver and renal function tests, hemostatic parameters, as well as D-dimer, fibrinogen, and NT-proBNP levels), were analyzed from uniform (pre-defined) peripheral venous blood samples drawn within 24 h from hospital admission.
Blood samples were collected either in ethylenediaminetetraacetic acid (EDTA), serum, heparin, or sodium citrate Vacuette blood collection tubes (Greiner Bio-One, Kremsmünster, Austria), centrifuged at 2300×g for 10 min and quantified in various methods (photometric, coagulometric, immunoturbidimetric, electrochemiluminescence, chromatographic, and potentiometric; for details see Supplemental Table 1).
NT-proBNP had a normal reference range between 0 and 100 pg/mL, was quantified with electrochemiluminescence using the Cobas 8000 immunoassay analyzer (Roche Diagnostics, Mannheim, Germany) and had an inter-assay coefficient of variation (CV) range 1.5%–4.8% and an intra-assay CV range of 1.9%–5.7%.
D-dimer was collected in a sodium citrate Vacuette tube and measured with an immunoturbidimetric method using Atellica Coag 360 system (Siemen Healthineers, Erlangen, Germany). Inter-assay CVs were 7.8%, 3.4%, and 1.5% for the normal-, low-, and high plasma pool, respectively and intra-assay CVs were 7.9%, 4.5%, and 2.6%, respectively.
The normal reference range of D-dimer for patients <50 years of age was 0–0.50 µg/mL. For patients ⩾50 years old age-adjusted D-dimer (AADD) cut-off was calculated (AADD = age (years) × 10 µg/L).
Neutrophil-to-lymphocyte ratio (NLR) derived from admission neutrophil and lymphocyte count (each ×109/L) and the liver fibrosis screening instrument fibrosis-4 index (FIB-4) was calculated as follows:
Follow-up
All patients underwent a clinical routine follow-up investigation by a vascular neurologist 3 months post-stroke at our outpatient clinic. Moreover, 5-year follow-up data were collected via the medical and nursing documentation and communication network of Styria (MEDOCS), which covers medical information collected in all public hospitals in the province of Styria. 24 Further relevant follow-up data were retrieved from the Austrian electronic health record (ELGA), an electronic information system documenting medical records from non-public hospitals and healthcare facilities in Austria.
All-cause mortality, vascular death and recurrent vascular events were recorded. Vascular death was defined as death due to a venous or arterial vascular event (i.e. ischemic stroke, myocardial infarction, or pulmonary embolism).
In addition, malignant tumors were manually assessed from patients’ health records including malignant blood cancer disease. Active cancer was defined as the presence of an in situ malignant tumor or an ongoing chemo- or radiotherapy within 6 months post-stroke. 25
Statistical analysis
Statistical analysis was carried out using the statistical software IBM SPSS Statistics, version 26 (IBM Corp, Armonk, NY, USA). Nominal variables were expressed with count and percentage and ordinal data with median and interquartile range (IQR). Comparison of these data was done using contingency tables, either with Pearson’s chi-square test or with Fisher’s exact test if the sample size of subgroups were small.
Quantitative continuous variables were presented with mean and standard deviation or median with IQR. Normal distribution of analyzed data was tested with histogram, Q-Q plot, Shapiro-Wilk test, and Kolmogorov-Smirnov test. Comparison of normally distributed independent continuous variables was performed with parametric analysis using the unpaired student’s test. Not normally distributed continuous variables were tested with the nonparametric Mann-Whitney-U test.
A multivariable binary logistic regression model was calculated for all-cause post-stroke mortality as the target variable and adjusted for age, sex, stroke severity (according to NIHSS), and variables that were significantly associated with mortality in the univariable analysis after applying the Bonferroni correction method (p < 0.0024). In a second model, early post-stroke mortality within 30 days was set as the target variable. Logs base 10 of NT-proBNP and D-dimer were used to account for the large variance of these parameters.
Receiver operating characteristic (ROC) curves of single blood biomarkers (NT-proBNP, D-dimer, CRP, NLR, eGFR, glucose, and FIB-4) were plotted to evaluate their accuracy for predicting all-cause mortality. By using Youden’s-index, the cut-off with highest sensitivity and specificity of each biomarker was estimated. In a further step, biomarkers with the highest sensitivity and specificity for post-stroke mortality (NT-proBNP, D-dimer, and NLR) were included in a 2 or 3-marker model to evaluate if they would improve the predictive accuracy for post-stroke death.
Multivariable binary logistic regression analysis was performed for the association of the estimated NT-proBNP cut-off (⩾794 pg/mL) with comorbidities and outcome and adjusted for clinical covariates and blood markers that were significantly associated with increased NT-proBNP in the univariable analysis after applying the Bonferroni correction method (p < 0.0024).
The association between NT-proBNP values and time from ischemic stroke to death during the 5-years follow-up was demonstrated with a scatter plot using SPSS.
Results
Baseline characteristics
Four hundred five ischemic stroke patients with a mean age of 70.3 ± 13.4 years (42.5% female sex) were included in this study. Median NIHSS at admission was 4 (IQR: 2–9) and 95 (23.5%) patients underwent acute recanalization therapy (intravenous thrombolysis: n = 73, 18.0%; mechanical thrombectomy: n = 33, 8.1%).
Cardioembolism was the most common stroke etiology (n = 113, 27.9%; of those atrial fibrillations: n = 101, 89.4%), followed by large vessel disease (n = 103, 25.4%) and small vessel disease (n = 45, 11.1%). Seventy-nine patients (19.5%) had a concomitant heart failure, mostly HFpEF (n = 32, 7.9%). Baseline data including blood biomarkers are presented in Table 1.
Demographics, clinical data, and laboratory parameters of ischemic stroke patients according to their 5-year mortality.
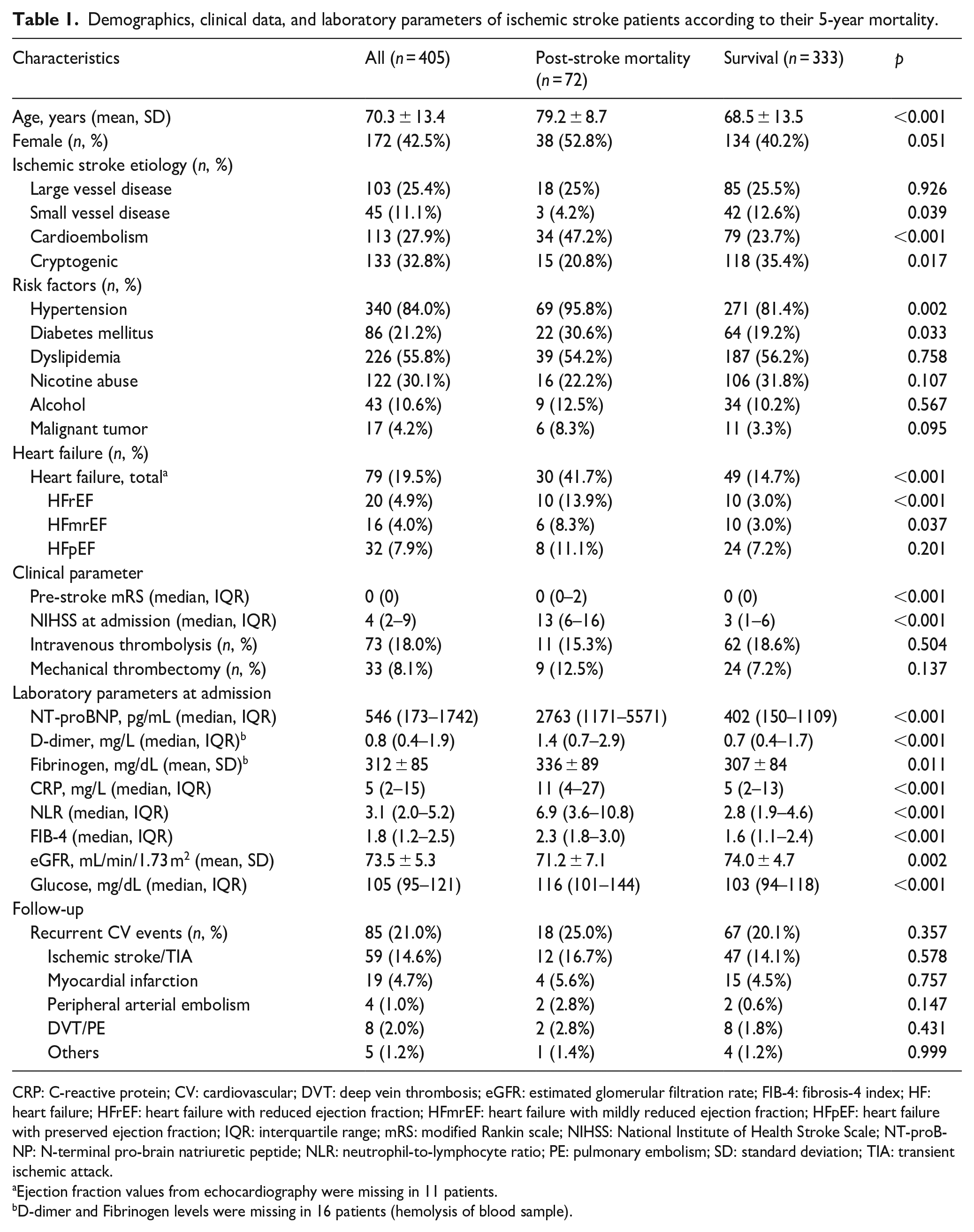
CRP: C-reactive protein; CV: cardiovascular; DVT: deep vein thrombosis; eGFR: estimated glomerular filtration rate; FIB-4: fibrosis-4 index; HF: heart failure; HFrEF: heart failure with reduced ejection fraction; HFmrEF: heart failure with mildly reduced ejection fraction; HFpEF: heart failure with preserved ejection fraction; IQR: interquartile range; mRS: modified Rankin scale; NIHSS: National Institute of Health Stroke Scale; NT-proBNP: N-terminal pro-brain natriuretic peptide; NLR: neutrophil-to-lymphocyte ratio; PE: pulmonary embolism; SD: standard deviation; TIA: transient ischemic attack.
Ejection fraction values from echocardiography were missing in 11 patients.
D-dimer and Fibrinogen levels were missing in 16 patients (hemolysis of blood sample).
Blood biomarkers and mortality
Over a 5-year follow-up period, 72 deaths (17.8%) were identified at a median time from hospital admission to death of 123 days (IQR 14–632 days). Among them, 37 (51.4%) patients died within the first 90-days following the index stroke. Most prevalent causes of death were stroke-related complications (n = 23, 31.9%), infections (n = 19, 26.4%), heart failure (n = 9, 12.5%), and cancer (n = 6, 8.3%).
Of all included blood biomarkers, NT-proBNP (median 2763 vs 402 pg/mL, p < 0.001), D-dimer (median 1.4 vs 0.7 mg/L; p < 0.001), CRP (median 11 vs 5 mg/L; p < 0.001), NLR (median 6.9 vs 2.8; p < 0.001), and glucose (median 116 vs 103 mg/dL; p < 0.001) were significantly associated with post-stroke mortality as were markers of liver fibrosis (FIB-4: median 2.3 vs 1.6; p < 0.001) and impaired kidney function (eGFR: 71.2 vs 74.0 mL/min/1.73 m2; p = 0.002) (Table 1, Supplemental Table 1).
In the multivariable analysis, NT-proBNP remained the only independent predictor for mortality after stroke with an adjusted odds ratio of 5.1 (95% CI 2.0–13.0; p < 0.001) (Table 2).
Multivariable regression analysis for all-cause post-stroke mortality during a 5-year follow-up period.
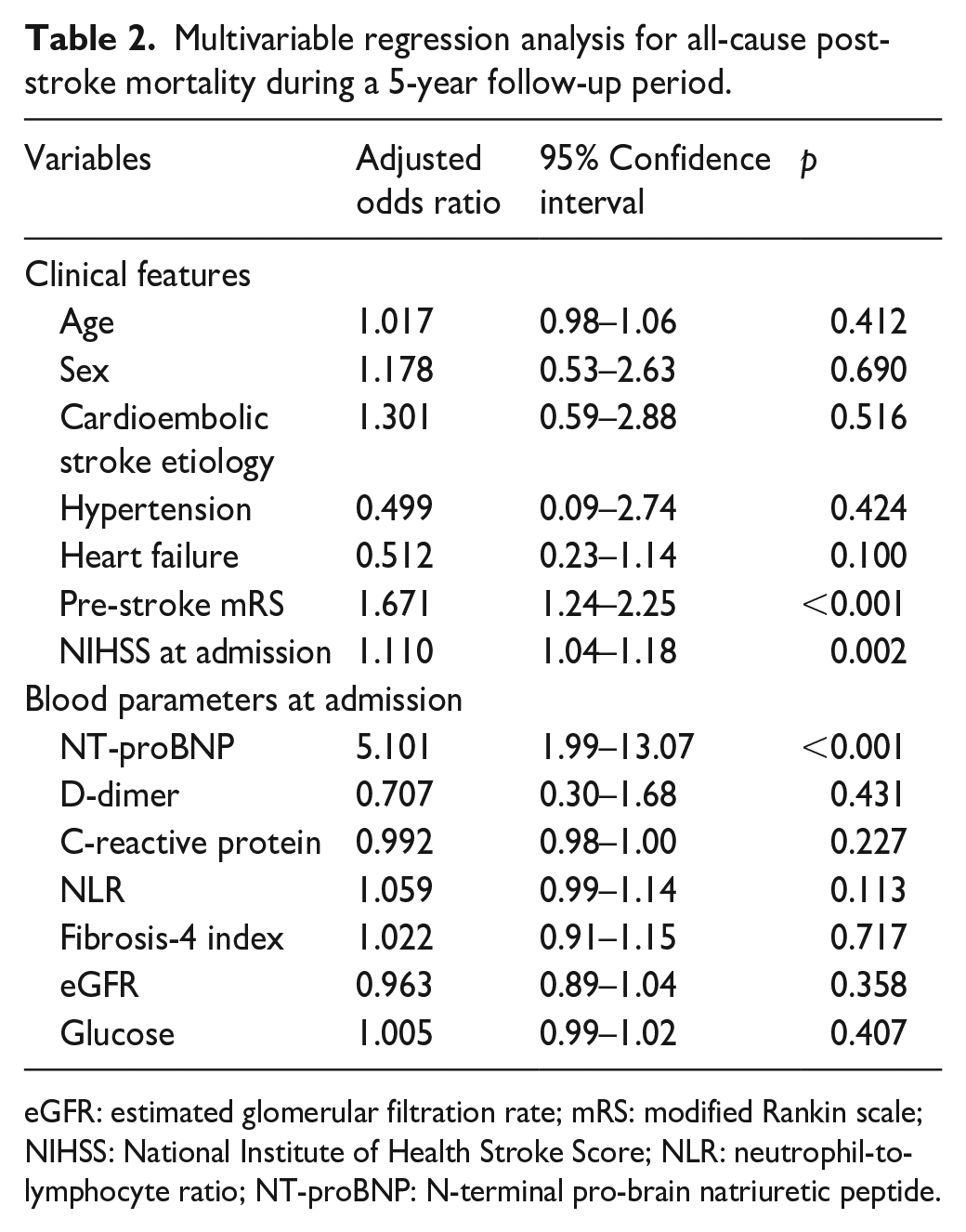
eGFR: estimated glomerular filtration rate; mRS: modified Rankin scale; NIHSS: National Institute of Health Stroke Score; NLR: neutrophil-to-lymphocyte ratio; NT-proBNP: N-terminal pro-brain natriuretic peptide.
NT-proBNP levels were comparable between different causes of death and did not significantly correlate with the time interval from stroke admission to death (all p > 0.1, multivariable analysis). The association between NT-proBNP and mortality did also not differ when analyzing its levels in relation to mortality within 30 days versus long term mortality (p = 0.604, multivariable analysis).
NT-proBNP and post-stroke mortality
In ROC analysis, the cut-off value of NT-proBNP with the highest sensitivity and specificity for all-cause mortality was estimated at 794 pg/mL (area under curve: 0.84, 90.1% sensitivity, and 67.5% specificity Figure 1). A two- or three-marker strategy (such as D-dimer, NT-proBNP, and NLR) did not increase the diagnostic accuracy.
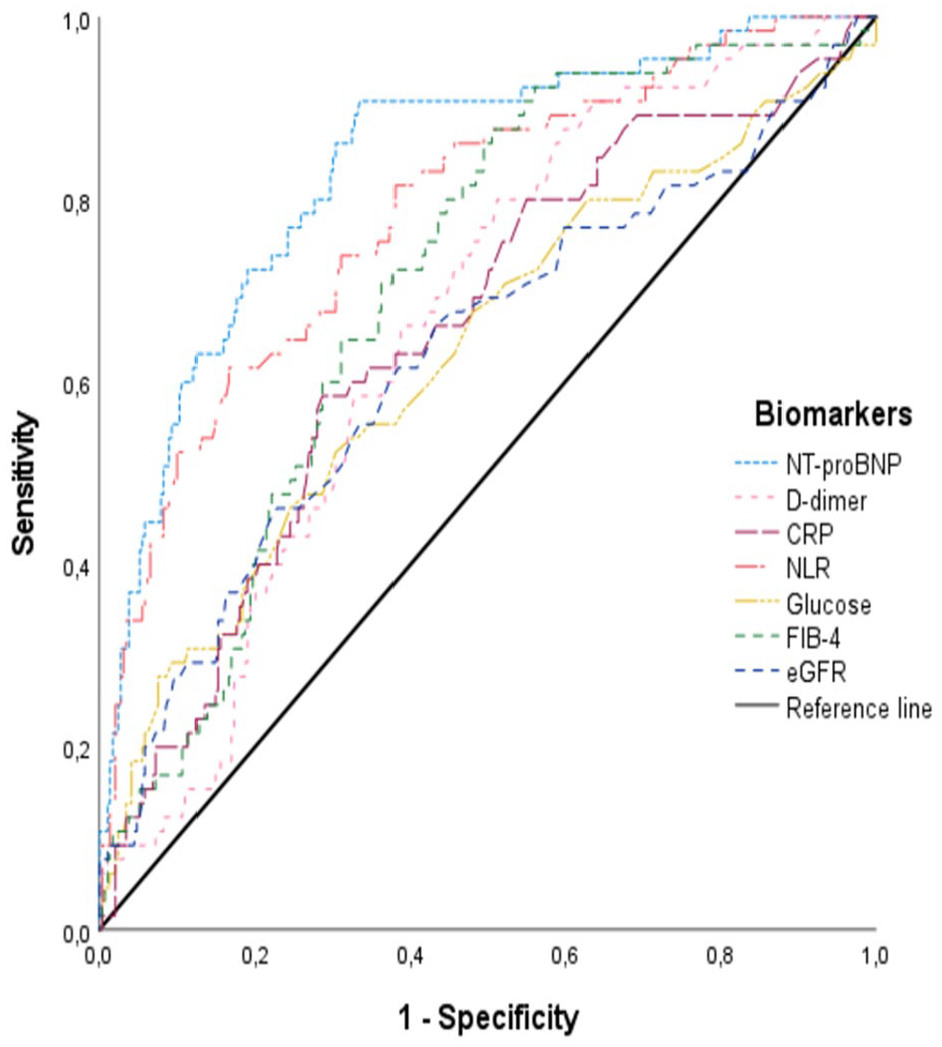
Receiver operating characteristic curve of admission blood biomarkers for 5-year mortality after ischemic stroke.
NT-proBNP ⩾794 pg/mL at admission (n = 169, 42%) was associated with cardioembolic stroke, and heart failure and was predictive for post-stroke mortality (all p < 0.001, multivariable analysis). Detailed results are presented in Table 3.
Correlation of NT-proBNP with clinical, laboratory, and follow-up data.
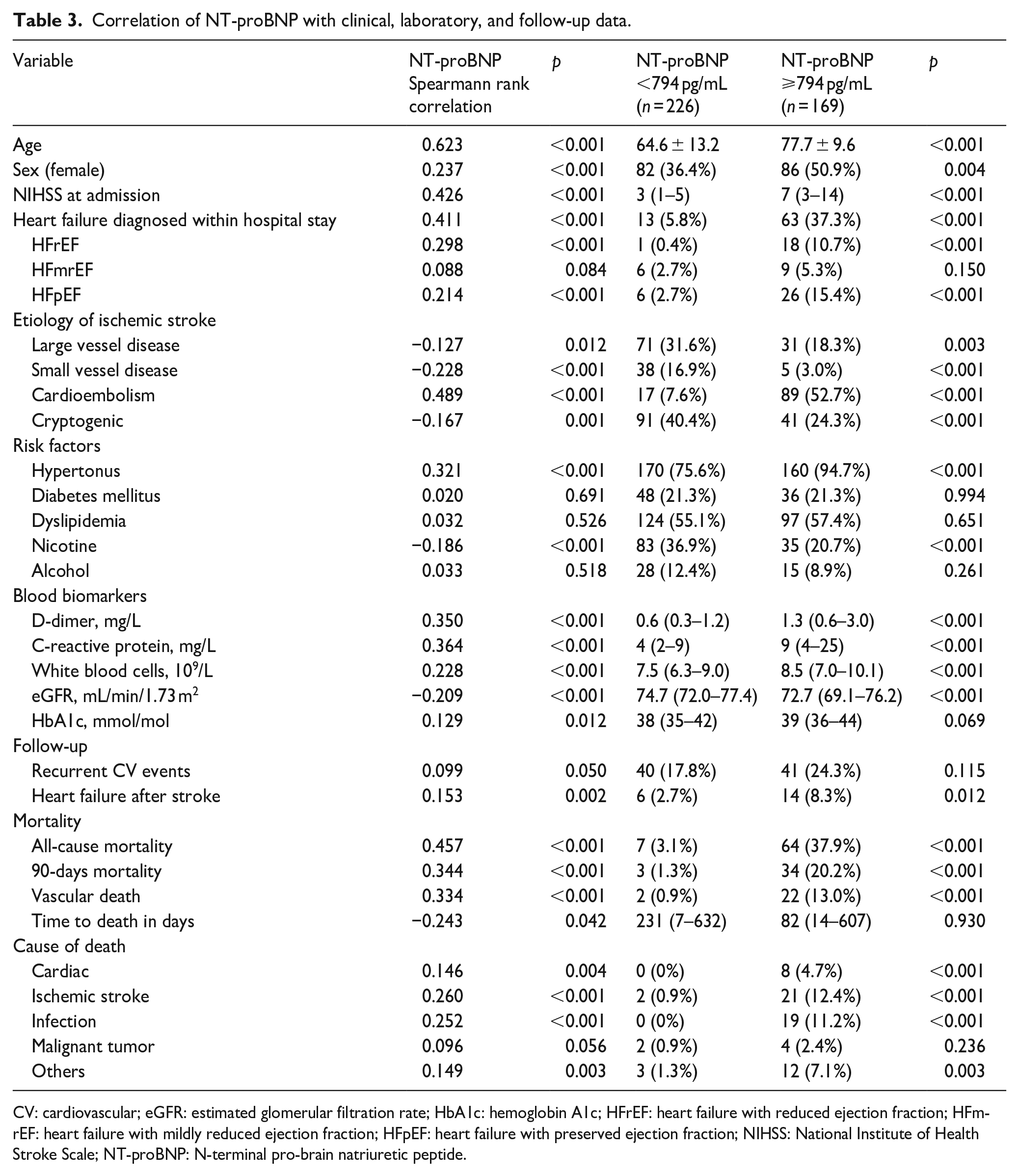
CV: cardiovascular; eGFR: estimated glomerular filtration rate; HbA1c: hemoglobin A1c; HFrEF: heart failure with reduced ejection fraction; HFmrEF: heart failure with mildly reduced ejection fraction; HFpEF: heart failure with preserved ejection fraction; NIHSS: National Institute of Health Stroke Scale; NT-proBNP: N-terminal pro-brain natriuretic peptide.
Of note, only seven out of 72 deceased patients (9.7%) had an NT-proBNP-value <794 pg/mL at admission (negative predictive value 97%) Figure 2.
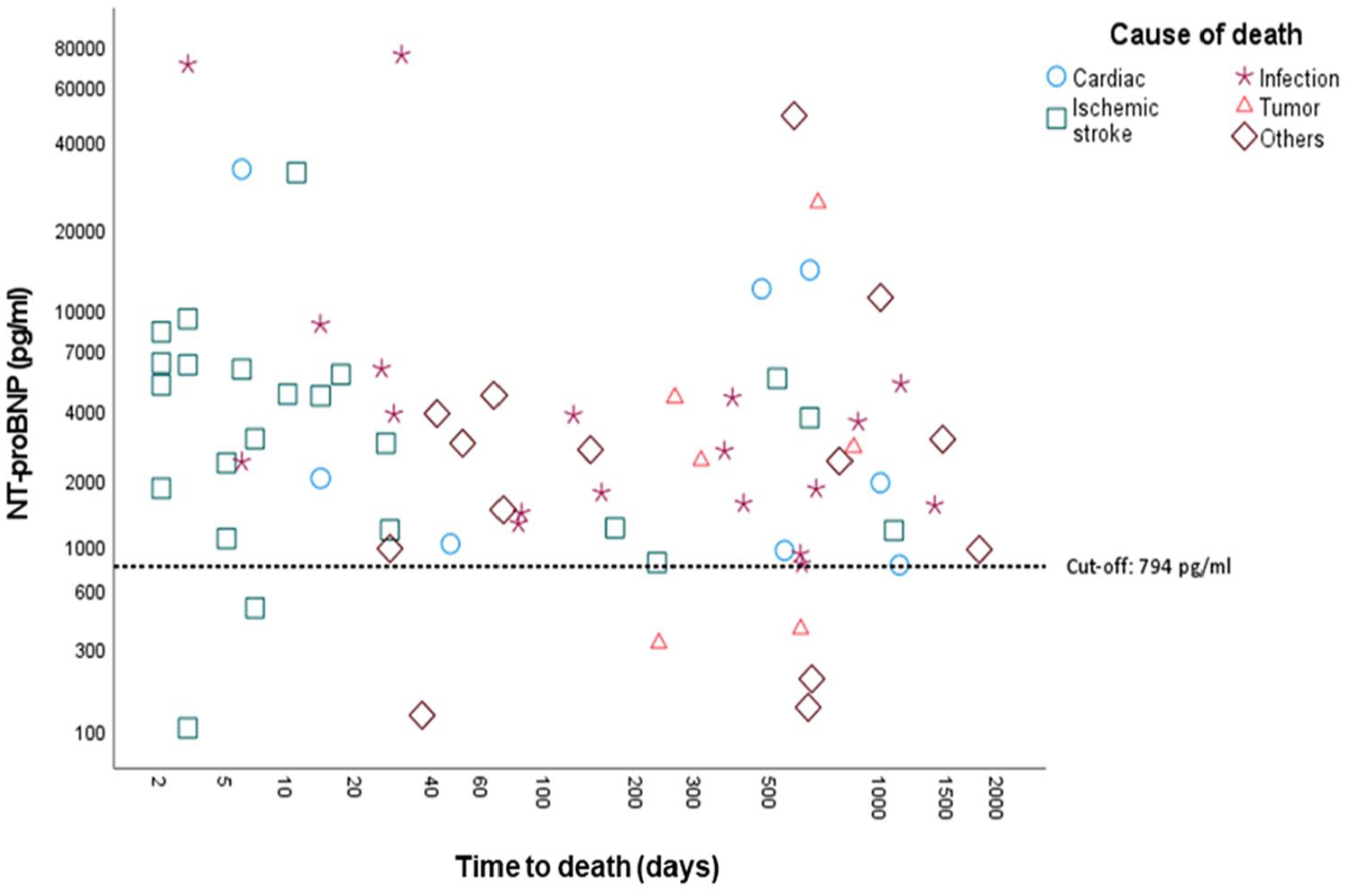
Association of NT-proBNP with causes of death within a 5-year follow-up period.
Recurrent vascular events
A total of 85 individuals (21%) suffered from a recurrent vascular event at a median time of 466 days after the index stroke (IQR 178–888 days). Most prevalent recurrent vascular events were ischemic stroke (n = 46, 54.1%) and myocardial infarction (n = 19, 22.4%). Of all tested blood biomarkers, only NT-proBNP (median, IQR: 797 (320–1550) vs 483 (155–1742) pg/mL; p = 0.05) tended to be associated with recurrent vascular events in the univariable analysis.
Discussion
Identifying patients at increased risk for poor clinical outcomes after stroke by routinely available blood biomarkers is a compelling field of research. In this study, we analyzed the prognostic value of various clinical routine blood biomarkers with all-cause mortality after stroke and identified NT-proBNP as the most valuable marker for predicting post-stroke death over a 5-year follow-up period. Moreover, we could show that admission NT-proBNP levels <794 pg/mL indicated a subgroup of stroke patients with a high chance of long-term survival after stroke.
The association of various blood biomarkers with post-stroke mortality is not new. While experimental multimarker strategies are cost-intensive procedures and difficult to be applied in clinical practice,16,17 recent studies presented approachable markers of inflammation (NLR), and liver fibrosis (FIB-4) calculated from routine laboratory data that might help to identify high-risk patients for a poor 90-day prognosis after stroke.6,7,11 The same applies to parameters of blood clotting (D-dimer, fibrinogen) and heart failure (NT-proBNP), which are also routinely accessible in most stroke centers and have been found to have a high predictive value for early post-stroke mortality.5,8,9,13,15,18
A major limitation of most presented studies was their focus on a single pathophysiological process (such as inflammation) neglecting competing pathways with a potentially higher impact on post-stroke prognosis.6,7,9,12,18 This might explain why the univariable association between NLR, FIB-4, D-dimer and fibrinogen with post-stroke mortality disappeared in our multivariable model, whereas NT-proBNP remained the sole independent laboratory predictor of post-stroke mortality over a 5-year follow-up in our cohort. Moreover, a two or three-marker strategy did not improve the predictive accuracy.
Compared to earlier studies reporting on the predictive value of NT-proBNP for mortality up to 12 months after stroke,13–15,26 our results are strengthened by the unique long-term follow-up of 5 years. Moreover, previous studies did not include heart failure – a major factor of increased NT-proBNP levels – into their analyses. The inclusion of such important comorbidities in our study and the analysis of a routine stroke unit cohort should facilitate the translation of our findings into daily clinical practice.
Our results might be explained by different factors: First, NT-proBNP levels strongly correlate with the presence of heart failure,13,15,26 which itself is an important predictor of poor post-stroke survival. Nevertheless, it is interesting to note that NT-proBNP remained an independent predictor of post-stroke mortality after adjustment for heart failure and cardioembolic stroke etiology.
Consequently, we identified several patients with increased NT-proBNP values, who had neither a diagnosed heart failure (clinically as well as signs on TTE) nor any kind of known heart disease. This might be explained by a significant number of undetected heart failure patients at the time of stroke, that is, HFpEF type. Despite new effective treatment strategies (such as SGLT 2 inhibitors), the recognition of HFpEF with recent non-invasive methods remains a challenge in clinical practice. 22 According to our data, patients with NT-proBNP levels above the cut-off of 794 pg/mL could present a vulnerable group of patients that may profit from timely cardiac work-up and corresponding treatment.
Second, high NT-proBNP levels might indicate an underlying atrial fibrillation in ischemic stroke patients, who remained cryptogenic after the initial workup. 21 As (untreated) cardioembolic stroke etiology has a high rate of recurrent vascular events and often leads to severe stroke syndromes with larger infarct size,1,27 this could in part explain the higher rate of post-stroke mortality in patients with high NT-proBNP levels at admission. These considerations are further supported by a trend toward a higher number of recurrent vascular events in patients with NT-proBNP levels above 794 pg/mL in our cohort.
Third, NT-proBNP is produced and secreted by an increase of wall stress in the myocardium. This may not only be present with cardiomyopathy or atrial fibrillation, but also in other situations with high wall stress, for example, loading conditions (volume overload, hypertension), infection and sepsis, liver or kidney failure.14,22,28,29
A major limitation of this study is the single-center design, which limits generalizability of our data. However, this ensures a highly standardized and systematic approach applied to all patients. The presented NT-proBNP cut-off must be interpreted cautiously, but still offers a reference for future studies in this field. Moreover, laboratory follow-up investigations were not performed, but could have contributed to a better understanding of the biomarker dynamics and their influence on post-stroke outcomes – still, for clinical applicability a one-time measurement that is temporally standardized for example, at the time of admission seems most amenable to clinical implementation. Finally, long-term follow-up data were retrieved retrospectively from clinical records. We can therefore not exclude that single patients that deceased at home or did not seek medical attention during the follow-up period were not identified. However, based on the detailed data documentation including public and non-public Austrian healthcare facilities and the fact that mortality outcomes of our study were similar to the outcome results of two prospective cohort studies investigating the 5-year mortality in a routine stroke cohort,30,31 this should not have affected our results to a significant extent.
Conclusion
Among numerous routine blood biomarkers, NT-proBNP represents the most relevant prognostic marker of long-term mortality after ischemic stroke. Increased NT-proBNP levels might indicate a vulnerable subgroup of stroke patients in which early and thorough (cardiac) assessment and consistent follow-up investigations could improve the prognostic outcome after stroke.
Supplemental Material
sj-docx-1-eso-10.1177_23969873231162125 – Supplemental material for The value of clinical routine blood biomarkers in predicting long-term mortality after stroke
Supplemental material, sj-docx-1-eso-10.1177_23969873231162125 for The value of clinical routine blood biomarkers in predicting long-term mortality after stroke by Isra Hatab, Markus Kneihsl, Egbert Bisping, Peter P Rainer, Simon Fandler-Höfler, Sebastian Eppinger, Melanie Haidegger, Natalie Berger, Harald Mangge, Reinhold Schmidt, Christian Enzinger and Thomas Gattringer in European Stroke Journal
Footnotes
Acknowledgements
None.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval for this study was obtained from the Ethics Committee of the Medical University of Graz (approval number: 29-285ex 16/17).
Informed consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
Guarantor
MK
Contributorship
IH: data acquisition and interpretation, manuscript preparation. MK: study design, data acquisition and interpretation, manuscript preparation. EB: critical revision of the manuscript content. PR: critical revision of the manuscript content. SF-H: acquisition of data, critical revision of the manuscript content. SE: acquisition of data. MH: critical revision of the manuscript content. NB: critical revision of the manuscript content. HM: critical revision of the manuscript content. RS: critical revision of the manuscript content. CE: critical revision of the manuscript content. TG: study design, acquisition and interpretation of data, manuscript preparation, critical revision of the manuscript. All authors have read and approved the final manuscript and agreed to be accountable for all aspects of the work.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.