Abstract
Introduction:
Marfan syndrome (MFS) is the most common inherited connective tissue disorder and its association with intracranial aneurysms (ICAs) has been debated for more than two decades. Here, we report the prevalence of ICAs at screening neuroimaging in a population of genetically confirmed MFS patients and present the results of a meta-analysis including our cohort of patients and those of previous studies.
Patients and methods:
We enrolled 100 consecutive MFS patients, who underwent screening with brain magnetic resonance angiography at our tertiary center between August 2018 and May 2022. We did a PubMed and Web of Science search to retrieve all studies on the prevalence of ICAs in patients with MFS published before November, 2022.
Results:
Of the 100 patients included in this study (94% Caucasians, 40% females, mean age 38.6 ± 14.6 years), three had an ICA. We pooled the current study with five previously published studies, including a total of 465 patients, 43 of which harbored at least one unruptured ICA, leading to an overall ICA prevalence of 8.9% (95% CI 5.8%–13.3%).
Discussion and conclusion:
In our cohort of genetically confirmed MFS patients, the prevalence of ICAs was 3%, which is substantially lower compared to previous studies based on neuroimaging. The high frequency of ICA found in previous studies could be explained by selection bias and lack of genetic testing, which may have led to the inclusion of patients with different connective tissue disorders. Further studies, including several centers and a large number of patients with genetically confirmed MFS, are needed to confirm our results.
Keywords
Introduction
Marfan syndrome (MFS) is an autosomal dominant systemic disorder of connective tissue characterized by a variable combination of cardiovascular, musculoskeletal, and ophthalmic manifestations. It is caused by pathogenetic variants in FBN1, which encodes fibrillin-1, a major structural component of the extracellular matrix. 1
MFS is the most common inherited connective tissue disorder and its association with intracranial aneurysms (ICAs) has been debated for more than two decades. Early reports based on autopsy series and clinical manifestations of ICAs yielded conflicting results, with estimated prevalence ranging from 0% to 29%.2 –4 More recent retrospective studies, evaluating neuroimaging findings, reported ICAs in up to 14% of patients with MFS.5 –7
However, due to heterogeneous inclusion criteria and lack of genetic testing, none of these studies allowed to establish an association between ICAs and FBN1 mutation in MFS. Here, we report the prevalence of ICAs at screening neuroimaging in a population of MFS patients with FBN1 pathogenic variants. Additionally, we present the results of a meta-analysis pooling prevalence data from our cohort of patients and those of previous studies.
Methods
Cross-sectional study
We enrolled in this study 100 consecutive patients with genetically confirmed MFS who underwent brain magnetic resonance angiography (MRA) at our institution (Hub Center for heritable connective tissue diseases of the University of Bologna, Italy) between August 2018 and May 2022. The reason for MRA examination was only to screen for the presence of ICA, and no patients had a previous history of neurovascular pathology or neurological symptoms. The study was approved by the local ethical committee (CE – AVEC, reference code: EM624-2019_246/2016/O/Oss/AOUBo), and informed consent was obtained.
Source MRA images as well as maximum intensity projections and 3D volume rendered angiograms were independently examined by two neuroradiologists who were blinded to any clinical information. The presence of an ICA, defined as an abnormal focal outpouching of a cerebral artery, was determined in a consensus reading.
MFS diagnosis was established according to the most recent guidelines 1 and only patients with likely pathogenic or pathogenic FBN1 variants (classes 4 and 5 according to the American College of Medical Genetics guidelines 8 ) were included in this study. Variants were categorized as either in-frame (including missense variants and small in-frame insertions/deletions) or protein-truncating (including nonsense, splice-site and frameshift variants). Genetic testing was performed in all patients with suggestive features of MFS which were referred to our center either as a proband (n = 82) or after the identification of an affected relative (n = 18). Clinical and demographic data were obtained from medical records.
Meta-analysis
We did a PubMed and Web of Science search to retrieve all studies on the prevalence of ICAs in patients with Marfan syndrome published before November, 2022. We used the keywords “Marfan AND aneurysm AND (cerebral OR brain OR intracranial OR berry OR basilar OR saccular OR communicating).” We searched the reference lists of all relevant publications for additional studies.
We included studies presenting crude numbers on patients with ICA and on the study population, or that allowed recalculation of these crude numbers; unruptured ICAs had to be reported separately from ruptured ICAs. Case reports and literature reviews were excluded from the analysis, as well as studies estimating ICA prevalence on the basis of clinical symptoms or the absence thereof.
For each study we extracted data on mid-year of study, type of investigation (autopsy or neuroimaging), size of study population, number of patients with unruptured ICA, total number, maximum diameter ad site of unruptured ICAs. When available, we also collected demographic and comorbidities data of the study population and of patients with and without ICA.
ICA location was classified as the internal carotid artery (including the ophthalmic artery and the posterior communicating artery), anterior cerebral artery (including the anterior communicating artery), middle cerebral artery, posterior cerebral artery and vertebrobasilar artery.
Statistical analysis
For the cross-sectional study sample size was estimated using the formula
For the meta-analysis we computed the crude prevalence of unruptured ICAs for each study. We pooled the prevalences from previous studies, as well as from our own cohort of patients by means of a generalized linear mixed model, with the number of unruptured ICAs and the total number of included patients for each study as variables. We assessed sex differences by pooling the prevalence ratios for females (with males as a reference) by means of a fixed effects model using Mantel-Haenszel method with continuity correction. We assessed age differences by pooling the mean age differences between patients with and without ICA. The mean aneurysm size across all studies was calculated using the inverse variance method.
Statistical analysis was performed using R version 4.2.2 (The R Foundation for Statistical Computing, Vienna, Austria, 2022) and the meta package (v6.0-0).
Results
Cross-sectional study
Of the 100 patients included in this study (94% Caucasians, 40% females, mean age 38.6 ± 14.6 years), three had an ICA, resulting in a prevalence of 3% (95% CI 1.0%–8.4%). All three aneurysms had a saccular morphology, and the average diameter was 3.2 ± 0.8 mm (range 2.5–4 mm). All aneurysms were located at the supraclinoid segment of the internal carotid artery, two on the right and one on the left.
A total of 76 different FBN1 variants were identified in our cohort of patients, belonging to 82 unrelated families. Variants were scattered along the gene. Demographic, clinical, and genetic characteristics of our patient population, as well as their association with ICA, are detailed in Table 1.
Demographic, clinical, and genetic characteristics of our cohort of patients.
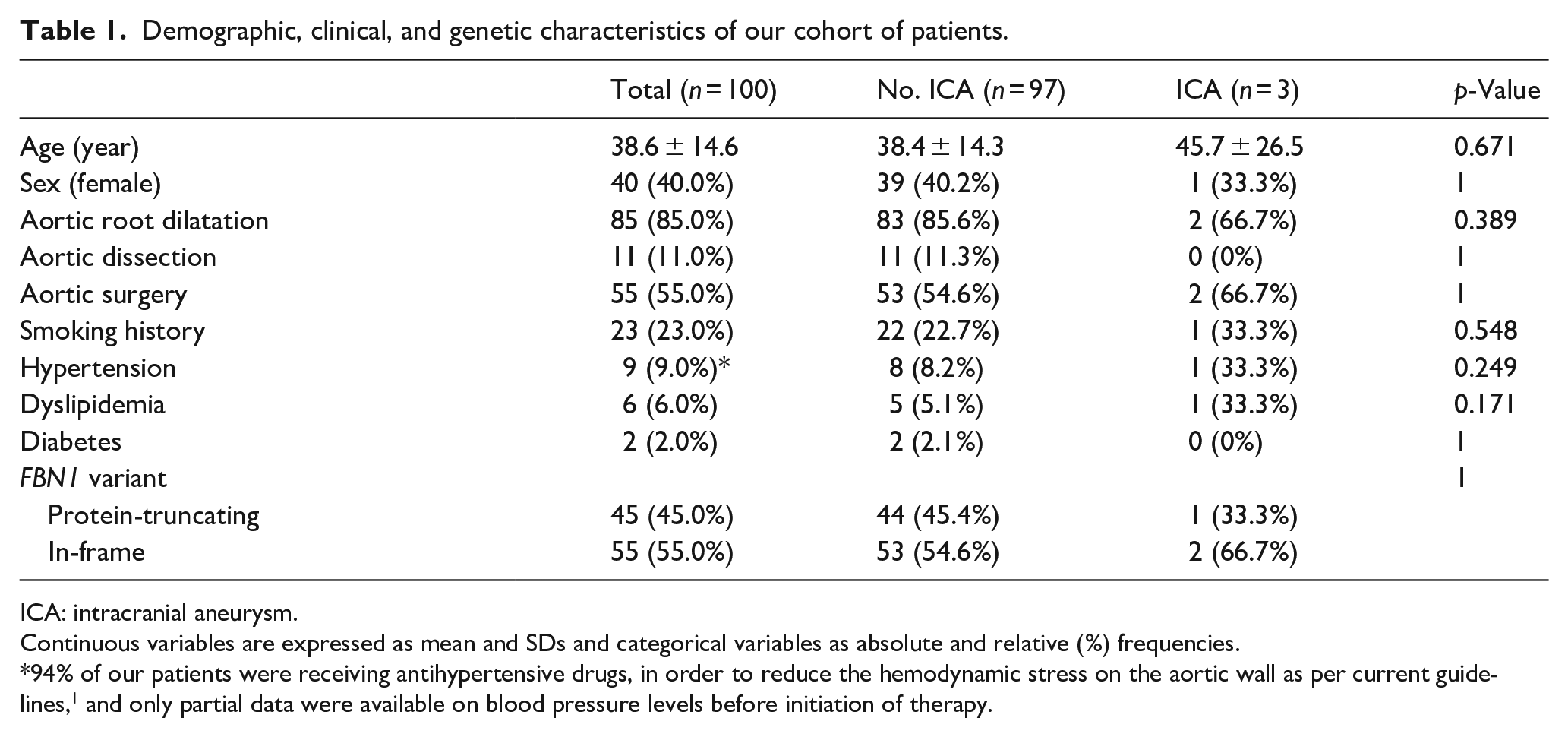
ICA: intracranial aneurysm.
Continuous variables are expressed as mean and SDs and categorical variables as absolute and relative (%) frequencies.
94% of our patients were receiving antihypertensive drugs, in order to reduce the hemodynamic stress on the aortic wall as per current guidelines, 1 and only partial data were available on blood pressure levels before initiation of therapy.
No significant differences were found in demographic and clinical data between patients with and without ICA, including age, sex, cardiovascular risk factors, presence of aortic root dilatation, previous aortic dissection and aortic surgery. There was no significant difference in the frequency of ICA by type of FBN1 variant. Nonetheless, these results were limited by the small number of patients in the ICA group.
None of our patients underwent ICA treatment and no subarachnoid hemorrhages were reported during a mean follow-up period of 22 ± 10 months.
Meta-analysis
Five studies met our inclusion criteria3 –7 (Figure 1 and Table 2), two were autopsy studies3,4, three were based on neuroimaging5 –7; all studies had a retrospective design and the mid-year of study ranged from 1969 to 2013. Four studies were conducted in the USA3 –6, one in Korea. 7 Both the lowest and highest prevalence of unruptured ICA (4.0% and 16.7% respectively) were reported in autopsy studies.
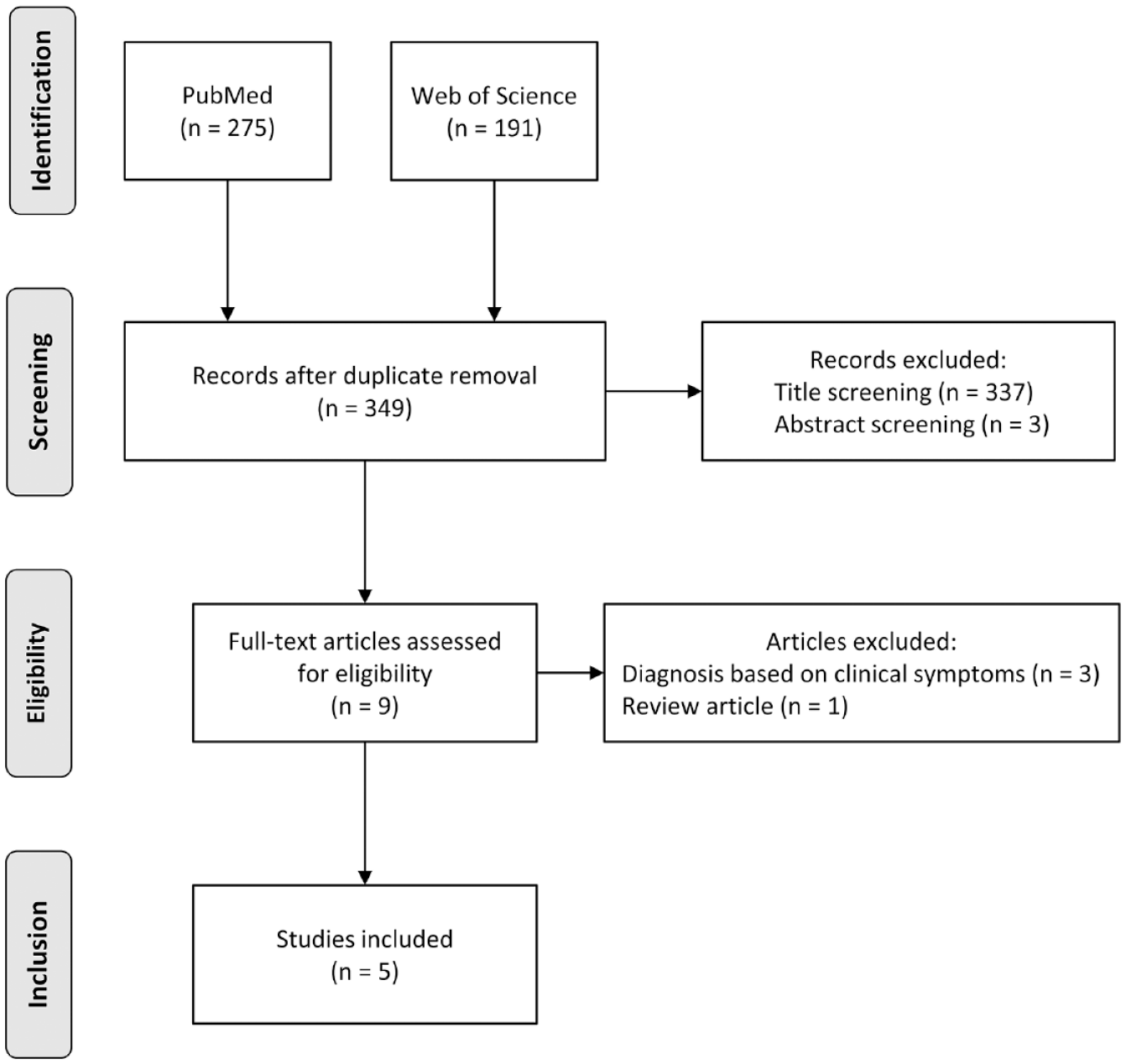
Selection of studies included in the meta-analysis.
Overview of studies included in the meta-analysis.
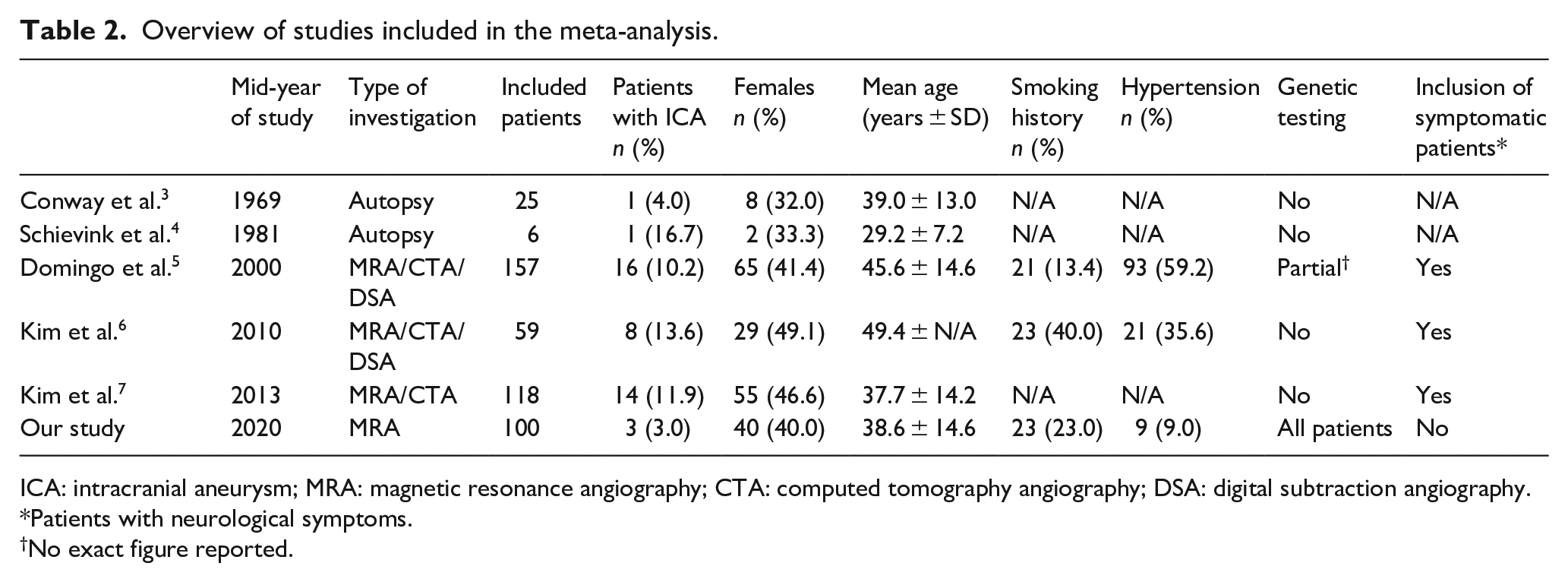
ICA: intracranial aneurysm; MRA: magnetic resonance angiography; CTA: computed tomography angiography; DSA: digital subtraction angiography.
Patients with neurological symptoms.
No exact figure reported.
We pooled the current study with previous studies, including 465 patients, 43 of which harbored at least one unruptured ICA, leading to an overall ICA prevalence of 8.9% (95% CI 5.8%–13.3%) (Figure 2). A total of 54 aneurysms were reported in the 43 affected patients, 49 saccular (90.7%) and 5 fusiform (9.3%) with an average maximum diameter of 3.6 mm (95% CI 2.9–4.1 mm). Forty-six aneurysms (85.2%) were located in the anterior circulation and 8 (14.8%) in the posterior circulation (Table 3).
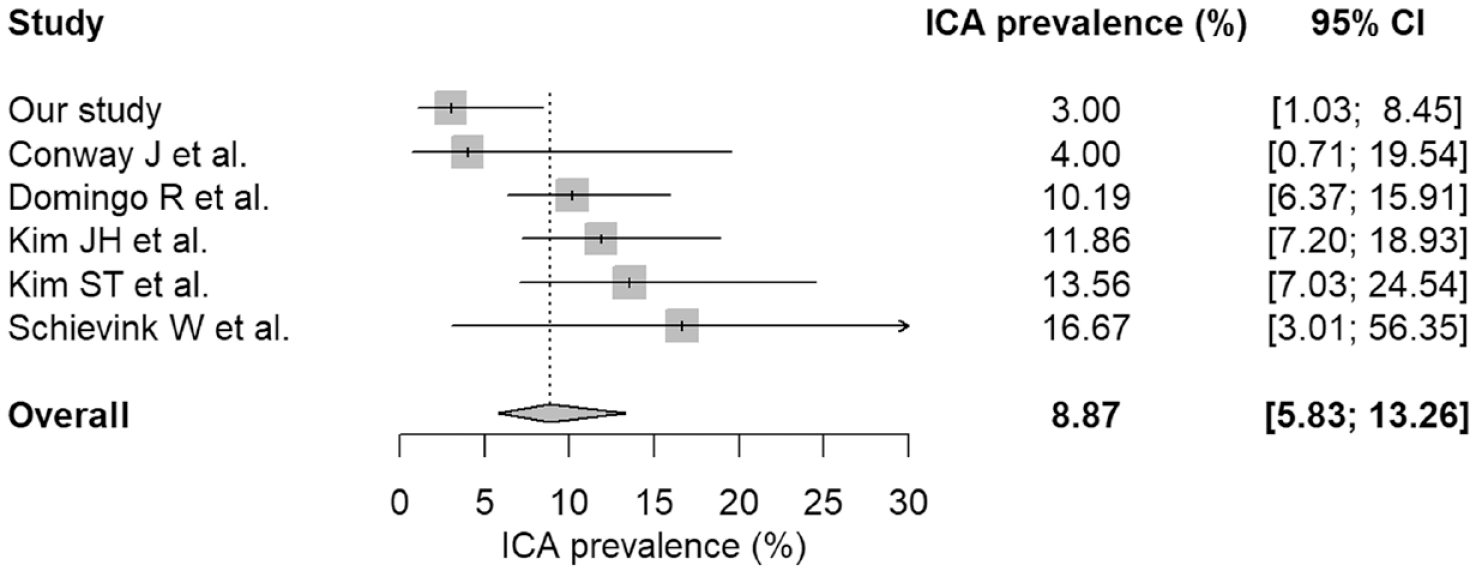
Prevalence of unruptured ICAs.
Characteristics of ICAs.
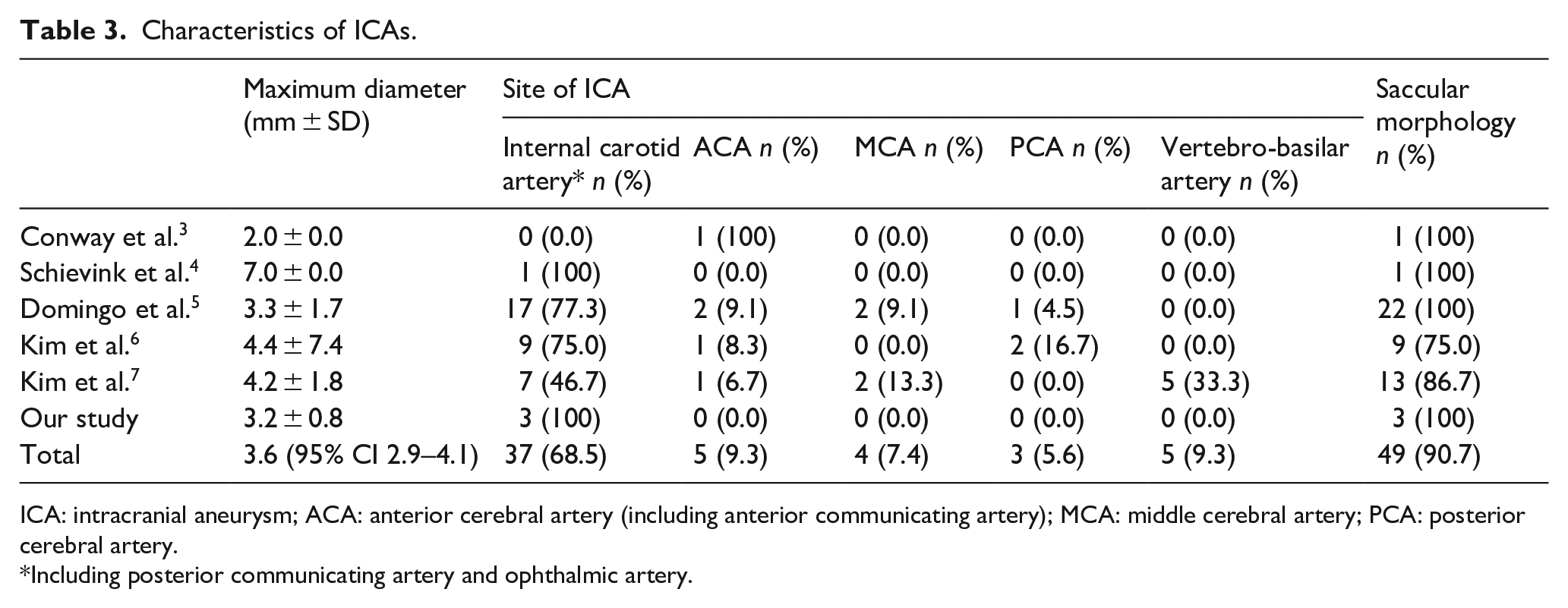
ICA: intracranial aneurysm; ACA: anterior cerebral artery (including anterior communicating artery); MCA: middle cerebral artery; PCA: posterior cerebral artery.
Including posterior communicating artery and ophthalmic artery.
The pooled prevalence of ICA in females (11.4% [95% CI 6.1%–20.5%]) was significantly higher compared to males (6.8% [95% CI 4.3%–10.5%]), with a prevalence ratio of 1.8 (95% CI 1.0–3.2, p = 0.048). The mean age of patients harboring an ICA was 48.5 years (95% CI 40.6–56.4 years), while the mean age of patients without an ICA was 38.1 years (95% CI 33.6–42.7 years), with a pooled mean age difference of 8.3 years (95% CI 6.2–10.5, p = 0.0036); one study 6 was excluded from the analysis because only the aggregate mean age of the entire study population was reported.
Discussion
In our cohort of genetically confirmed MFS patients, the prevalence of unruptured ICAs was 3% (95% CI 1.0%–8.4%), which is substantially lower compared to previous studies based on neuroimaging (ranging from 10.2% to 13.6%),5 –7 as well as to the pooled prevalence of the studies included in the meta-analysis (8.9% [95% CI 5.8%–13.3%]).
Several factors may explain the higher frequency of ICAs found in previous reports, including: (a) the possible enrollment of patients with other connective tissue disorders misdiagnosed as MFS for lack of genetic testing; (b) the potential for selection bias due to the inclusion of patients who underwent neuroimaging for neurological symptoms; (c) a different distribution of demographic characteristics and comorbidities.
Among previous studies, only Domingo etal. 5 mention the use of genetic testing in some of their patients, without reporting the exact figure, while all other studies only employed clinical diagnostic criteria. Additionally, these studies retrospectively included patients over a long-time frame, ranging from 10 to 60 years. In all studies based on autopsy3,4 and in two of those employing neuroimaging,5,6 at least part of the study period predated the first description of Loeys-Dietz syndrome (LDS), 9 and even in the most recent study 7 some patients were diagnosed before the publication of the revised Ghent nosology. 10 The use of more inclusive clinical criteria, such as the Berlin nosology, together with the lack of genetic testing, may have led to the inclusion of patients with different connective tissue disorders, notably LDS, whose high prevalence of ICAs has been widely reported.11,12
Indeed, LDS is an autosomal dominant condition that includes many features of MFS. Recent studies showed that quantification of cervical and intracranial arterial tortuosity may be a useful tool in differentiating these two entities.13,14 Nonetheless, many of the features included in the systemic score of the revised Ghent nosology 10 can also be found among LDS patients, including scoliosis, pes planus, anterior chest deformity, spontaneous pneumothorax, joint hyperextension, mitral valve prolapse, and dural ectasia. 15 Furthermore, craniofacial anomalies which are absent in MFS, such as bifid uvula, cleft palate and hypertelorism, are not always found in patients with LDS. 9
Notably, Domingo etal. 5 report a high prevalence of extra-aortic aneurysms among their patients with ICA, with involvement of renal, mesenteric, and extracranial carotid arteries, as well as vessels of the upper and lower extremities. These findings are very unusual for MFS, 1 but are actually a distinguishing feature of LDS and their finding should prompt genetic testing to exclude this diagnosis.15,16
An additional confounding factor, which may have led to a higher frequency of ICAs in previous reports on MFS patients, is the possible occurrence of selection bias due to the enrollment of patients who underwent neuroimaging for the presence of neurological symptoms. Although patients who had available neurovascular studies were included consecutively, in one report 6 half of the patients with ICA underwent neuroimaging due to cranial nerve palsy. Also in the other two neuroimaging studies5,7 the authors admittedly included patients who were examined for the evaluation of neurological symptoms or previous subarachnoid hemorrhage, leading to a possible overestimation of the prevalence of ICAs compared to the general MFS population.
In our cohort, the presence of ICA was not associated with demographic or clinical characteristics, including cardiovascular risk factors, although these findings were limited by the small number of patients in the ICA group. The meta-analysis showed a significantly higher prevalence of ICAs in female patients, as already reported by Domingo etal. 5 and patients harboring an ICA were significantly older compared to controls, confirming the results of previous studies.5 –7
Therefore, we cannot exclude that the low prevalence of ICAs found in our patients may be partially explained by differences in demographic characteristics. In our cohort the proportion of female patients (40.0%) was lower compared to previous studies based on neuroimaging (41.4%, 5 49.1%, 6 and 46.6% 7 respectively), while it was higher compared to the autopsy studies (32.0% 3 and 33.3% 4 ). The mean age of our cohort (38.6 ± 14.6 years) was lower compared to the studies by Conway etal. 3 (39.0 ± 13.0 years), Domingo etal. 5 (45.6 ± 14.6) and Kim etal. 6 (49.4 years), while it was higher compared to the studies by Schievink etal. 4 (29.2 ± 7.2 years) and Kim etal. 7 (37.7 ± 14.2).
In the meta-analysis we could not evaluate the effect of cardiovascular risk factors on the prevalence of ICAs, since only the study by Domingo etal. 5 reported these data separately for patients with and without ICA, finding an increased risk of aneurysm in smokers. Compared to the study by Domingo etal., 5 in our cohort the number of smokers was higher (23.0% vs 13.8%), while the prevalence of hypertension was considerably lower (9.0% vs 59.1%), although the magnitude of this difference could be significantly overestimated since 94% of our patients were receiving antihypertensive drugs at last follow-up, in order to reduce the hemodynamic stress on the aortic wall as per current guidelines, 1 and only partial data were available on blood pressure levels before initiation of therapy. Nonetheless, no association was reported between hypertension and ICA in previous studies on MFS. 5
Our study shares some of the limitations of previous reports, including a relatively small number of subjects and a possible overrepresentation of severe cases in patients referred to a tertiary center. Nonetheless, in contrast to previous studies, our patients have been enrolled over a short time-frame, employing homogeneous inclusion and diagnostic criteria, with genetic data available for all patients.
Conclusions
In our cohort of genetically confirmed MFS patients, the prevalence o ICAs was 3%, which is substantially lower compared to previous studies based on neuroimaging, as well as to the pooled prevalence of the studies included in the meta-analysis (8.9% [95% CI 5.8%–13.3%]). The higher frequency of ICA found in previous studies could be explained by selection bias and lack of genetic testing, which may have led to the inclusion of patients with different connective tissue disorders. Obtaining an unbiased estimate of the prevalence of ICAs in MFS patients has important consequences for counseling and clinical management. Further prospective case-control studies, including several centers and a large number of patients with genetically confirmed MFS, are needed to confirm our results.
Footnotes
Acknowledgements
We thank the Italian National BiblioSan Consortium for funding the open access publication fees.
Correction (January 2023)
Article updated to correct the second author’s affiliations.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The open access publication of this article was funded by the Italian National BiblioSan Consortium.
Ethical approval
ethical approval for this study was obtained from by the local ethical committee (CE – AVEC, reference code: EM624-2019_246/2016/O/Oss/AOUBo).
Informed consent
Written informed consent was obtained from all subjects before the study.
Guarantor
GV.
Contributorship
All authors contributed to the study conception and design. Data collection and analysis were performed by GV, SRMD, MCB, and CR. The first draft of the manuscript was written by GV and LS. All authors reviewed and edited previous versions of the manuscript. All authors read and approved the final manuscript.
Data availability statement
The data presented in this study are deposited in Zenodo repository (https://zenodo.org) and are accessible with the following link: ![]() .
.
