Abstract
Purpose:
Oropharyngeal dysphagia is a common and complication-prone symptom after stroke and is assumed to increase medical expenses. The purpose of this study was therefore to examine acute hospitalization costs associated with post-stroke dysphagia.
Method:
This retrospective study included patients with acute stroke who had been examined by Flexible Endoscopic Evaluation of Swallowing (FEES). Health insurance expenditures were determined for the patient cases according to the 2021 revenue criteria. Multiple linear regression was used to examine predictors of health insurance spending including age, sex, stroke severity, stroke characteristics, comorbidity, therapeutic interventions, duration of artificial ventilation, length of hospital stay, and severity of dysphagia, as assessed by the Fiberoptic Endoscopic Dysphagia Severity Scale (FEDSS), ranging from 1 (best) to 6 (worst).
Findings:
Six hundred seventy four patients (men/women: 367/307; mean age: 71.1 ± 12.8 years; mean National Institute of Health Stroke Scale: 11.2 ± 6.2; FEDSS 1/2/3/4/5/6: 113/73/144/119/124/101; mean health-insurance costs 11,521.5 ± 12,950.5€) were included in the analysis. Advanced age (p = 0.007; B = 57.6), catheter interventions (p < 0.001; B = 4105.6), tracheotomy (p = 0.006; B = 5195.2), duration of artificial ventilation (p < 0.001; B = 388.6), length of hospital stay (p < 0.001; B = 441.9), and severe dysphagia with an FEDSS of 6 (p = 0.004, B = 2554.3) were independent predictors of increased health insurance expenditures (p < 0.001, R-squared = adjusted-R-squared = 0.83).
Discussion and conclusion:
The results of this study show an association between severe dysphagia and health care costs for acute hospitalization from a health-insurance perspective. Therefore, therapies that target severe dysphagia with impaired secretion management may have the potential to reduce costs.
Introduction
Dysphagia is derived from the Greek prefix “dys” and the word “phagein” and literally translates as disturbed eating. In medical context, dysphagia refers to a swallowing disorder, that is, impaired bolus transport from the oral cavity to the gastrointestinal tract. Swallowing impairment is common in most neurological disorders such as stroke, Parkinson’s disease, dementia, neuroinflammatory diseases, and neuromuscular disorders, but can also occur in healthy older people.1–10 Often, the oral and pharyngeal phases of swallowing are affected simultaneously, which is characterized as oropharyngeal dysphagia.
Oropharyngeal dysphagia after stroke occurs in the majority of patients during the acute phase of the disease. 8 Swallowing impairment not only represents a major psychological burden for patients, but also leads to serious complications. In particular, aspiration pneumonia 11 and malnutrition 12 may occur as a result of dysphagia, worsening functional outcomes and contributing to premature mortality. 13
Dysphagia is not always noticed by patients, and silent aspiration may be present that is not apparent on clinical examination. Therefore, instrumental procedures that visualize swallowing are necessary to reliably detect swallowing impairment. Both, Flexible Endoscopic Evaluation of Swallowing (FEES) and Videofluoroscopic Swallowing Study are considered equivalent gold-standard diagnostic methods for neurogenic dysphagia. 14
In addition to the medical complications and compromised quality of life, post-stroke dysphagia is assumed to increase costs in the healthcare system. In particular, prolonged hospitalization was reported in different studies. A meta-analysis combining six studies found that the average length of hospital stay increased by 4.7 days in patients with post-stroke dysphagia. 15 However, there are few studies available that have conducted an analysis of dysphagia-related costs in stroke patients.16–21 Complicating matters further, the methodology used to diagnose dysphagia and to determine costs was highly heterogeneous, and study results are partly contradictory. For example, some authors conclude that dysphagia is not associated with additional costs, 18 while other studies report that costs double by several thousand dollars. 16 To date, no study has examined dysphagia using gold-standard instrumental diagnostics or differentiated the severity of dysphagia in a cost analysis. In line with this, a recent systematic review on healthcare-related costs of post-stroke dysphagia concludes that further studies are needed in which early dysphagia screening and assessment is performed. 22
Therefore, the aim of this study was to investigate the acute hospitalization costs associated with oropharyngeal dysphagia assessed with FEES from the perspective of health-insurance. A differentiated picture of cost distribution depending on dysphagia severity was drawn.
Material and methods
Patient cohort
This study represents a secondary analysis of a previously collected retrospective patient cohort from University Hospital Muenster between 2004 and 2014, published elsewhere. 2 For the initial cohort, a random sample of 1000 cases treated between 2004 and 2014 was drawn. Only patients who had an acute stroke confirmed by brain imaging and who had undergone FEES in the acute phase within the first 96 hours after stroke were included in the study. In addition, the patient record had to contain sufficient information on patient demographics, stroke characteristics, and National Institute of Health Stroke Scale (NIHSS) on admission. Patients were excluded if they had documented dysphagia before stroke or had other conditions associated with dysphagia.
For this secondary financial analysis, we further excluded patients for whom documentation in the DRG department was insufficient to determine cost data. Patient demographics (age and sex), stroke characteristics (ischemia, hemorrhage, lesion localization, and lesion hemisphere), stroke severity on admission (NIHSS, modified Rankin Scale (mRS)), therapeutic interventions (catheter-based therapy, neurosurgery, intravenous thrombolysis, intubation, tracheotomy) and outcome parameters (length of stay, length of ventilation, pneumonia rate, other infections (e.g. urinary tract infection, fever of unclear origin), death) were recorded from patient documentation.
Dysphagia assessment
As a screening examination, all patients underwent a standardized water-swallowing test by trained staff. In case of abnormal findings or the presence of other clinical predictors for dysphagia such as severe dysarthria, aphasia, or facial palsy, FEES was performed. FEES was conducted by a trained neurologist together with a speech-language pathologist according to a standardized protocol. 23 Dysphagia severity was assessed using the Fiberoptic Endoscopic Dysphagia Severity Scale (FEDSS), an ordinal score validated for stroke, grading dysphagia severity from 1 (no relevant dysphagia) to 6 (severe dysphagia with impaired secretion management).23,24 The FEES-examination protocol, major dysphagia findings, and clinical implications of each ordinal FEDSS level are illustrated in Figure 1.
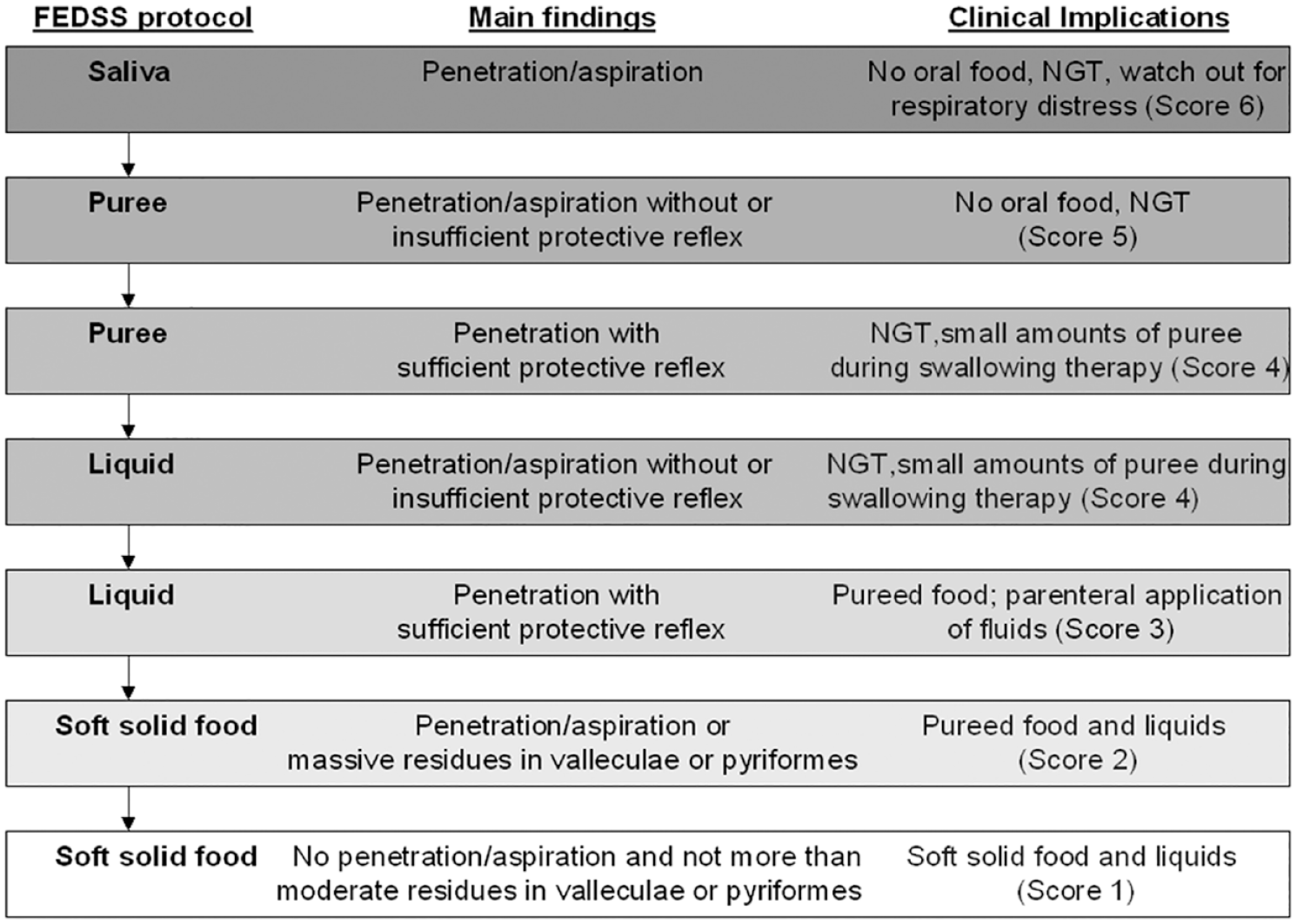
Determination of expenditures by health insurers
The amount that the hospital is required to bill health insurers for each patient case was determined in accordance with the 2021 billing regulations. 25 In Germany, inpatient hospital stays are billed based on Diagnostic Related Groups (DRG), a performance-related and fixed-priced remuneration system. Each patient case is assigned to a DRG according to the main diagnosis that caused hospitalization. In addition, Operation and Procedure Codes (OPS), periods of artificial ventilation, and secondary diagnoses are also relevant for DRG assignment. Severity levels for complications and comorbidities are assigned in the DRG system using the Patient Clinical Complexity Level (PCCL) based on secondary diagnoses. The PCCL thus reflects the comorbidity level and is derived using a complex formula. The total length of stay in the hospital is also relevant for billing. Surcharges or discounts are due if the lower, medium or upper limit length of stay is exceeded or not reached. These limits in length of stay are based on the average length of stay of patients with the corresponding DRG. The DRGs are calculated by the “Institut für Entgeltsystem im Krankenhaus” (Institute for Hospital Remuneration) and are based on the average costs of comparable patient cases at reference hospitals. There are detailed guidelines on how revenues are to be allocated for a patient case according to DRGs. 25 To calculate the health insurance costs, the applicable DRGs for billing were determined for each patient case. Only the costs for acute hospitalization at University Hospital Muenster were taken into account and no subsequent costs for rehabilitation stays, outpatient treatment or hospital readmission.
Statistical analysis
A two-step approach was used to analyze the data. In a first step, average health insurance costs were calculated for each FEDSS dysphagia severity level and visualized using box-plots. The Kruskal-Wallis test was used to test whether health insurance costs differed between FEDSS levels. In a post-hoc test, all individual FEDSS levels were compared.
In a further step, a multiple linear regression using backward elimination analysis was performed to identify appropriate predictors of health insurance costs. Here, age, sex, DRG comorbidity level according to the PCCL, ischemic versus hemorrhagic stroke, infratentorial lesion location versus supratentorial lesion location, supra- and infratentorial or undefined lesion location versus supratentorial lesion location, left-sided lesion location versus right-sided lesion location, bihemispheric or undefined lesion location versus right-sided lesion location, NIHSS, mRS, catheter-based therapy, neurosurgery, intravenous thrombolysis, intubation, tracheotomy, length of hospital stay, duration of artificial ventilation, pneumonia, other infections, and death were included as possible predictors in the model. The FEDSS was further included as a dysphagia severity parameter, as was the distinction between FEDSS level 6 versus all other FEDSS levels. The reason for the latter step was that the Kruskal-Wallis test in the previous analysis (see Results section “health insurance costs depending on dysphagia”) had shown indications that a binary distinction between FEDSS level 6 and all other levels might be more appropriate than ordinal dysphagia scaling. After the relevant predictors were determined using the backward elimination method, a regression analysis was performed again, using only the previously determined relevant predictors, but further addressing violated assumptions for a regression analysis. Due to heteroscedasticity and non-normally distributed residuals, a wild-bootstrapping and exclusion of all outliers with standardized residuals outside of three standard deviations was performed. Multicollinearity was tested using a bivariate correlation of all predictors (for ordinally scaled variables the Spearman’s-Rho correlation coefficient, for metrically scaled variables the Person’s correlation coefficient was used). For correlation coefficients above 0.7, which is considered a critical threshold, 26 one of the two variables was excluded, namely the one whose exclusion resulted in a larger R-squared value for the prediction in the model. Furthermore, the presence of multicollinearity was now assessed for the remaining variables using the variance inflation factors (VIF), using a cut-off value of 10. 26
Results
Patient cohort
Of the 687 patients in the original study cohort, 2 13 patients did not have sufficient information in the medical record to determine health insurance costs. Thus, 674 patients remained in the cost analysis. The demographic, clinical and financial parameters of the patient cohort are shown in Table 1.
Patient characteristics of the cohort.

SD: standard deviation; NIHSS: National Institute of Health Stroke Scale, mRS: Modified Rankin Scale; FEDSS: Fiberoptic Endoscopic Dysphagia Severity Scale; PCCL: Patient Clinical Complexity Level.
Frequencies of the DRGs billed
Most patients were billed with the DRG “Apoplexy” (~65% of patients) or the DRG “Complex neurological treatment of acute stroke with specific operating room procedure” (~12% of patients). The majority of the remaining cases were billed with intensive care or artificial ventilation DRGs. The detailed distribution of the billed DRGs with illustration in a bar chart is visualized in Figure 2.
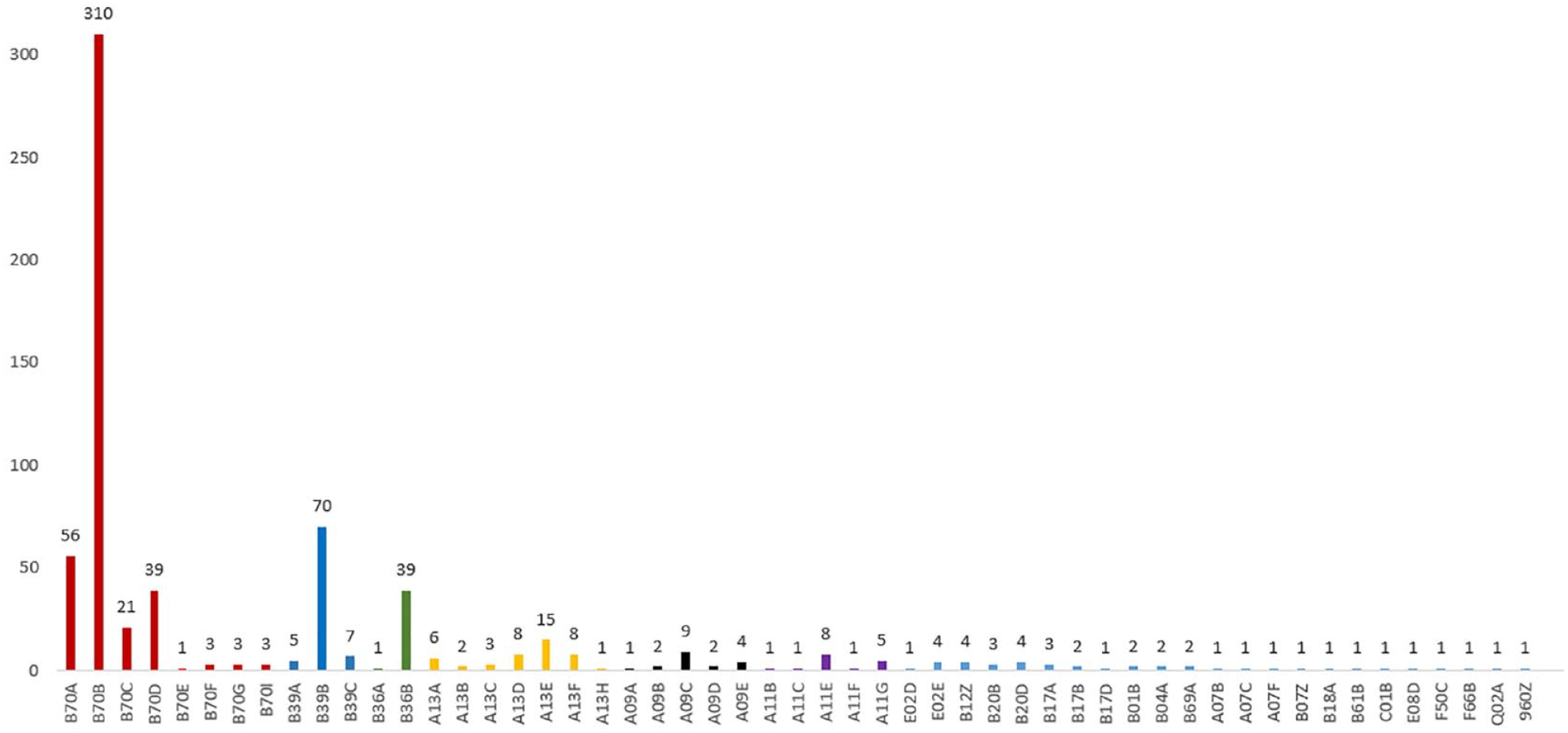
DRG frequencies in the cohort: B70 (red): Apoplexy; B39 (dark blue): Neurological complex treatment of acute stroke with specific OR procedure; B36 (green): Intensive care complex treatment >1176/1104/1104 expense points or >588/552/552 expense points with specific OR procedure for diseases and disorders of the nervous system or specific high-cost implants; A13 (yellow): Ventilation >95 h, without intensive care complex treatment >1764/1656/1656 expenditure points; A09 (black): A09 – Ventilation >499 h or >249 h with intensive care complex treatment >2352/1932/2208 expenditure points; A11 (purple): A11 – Ventilation >249 h or >95 h with intensive care complex treatment >1764/1656/1656 effort points; The last letter in the 4-digit DRG represents the severity/resource use (A: highest severity to Z: no severity subdivision).
Health insurance costs depending on dysphagia
The Kruskal Wallis Test revealed differences in the distribution of health insurance costs depending on the FEDSS-Level [H(5) = 105.7, p(asymp.) < 0.001]. Average health insurance costs ranged from 7580€ to 11,641€ for FEDSS severity levels 1–5, whereas for FEDSS severity level 6, the mean costs increased to 25,643€ (Table 2 and Figure 3). The post-hoc test (Dunn-Bonferroni) showed higher costs for FEDSS level 6 (severe dysphagia and impaired secretion management) compared with all other FEDSS levels. In addition, higher costs were shown for FEDSS level 5 compared with FEDSS level 2. Beyond that, there were no significant differences among the FEDSS levels (Supplemental Table 1). This finding suggested that instead of ordinal scaling, a binary subdivision between FEDSS = 6 versus all other FEDSS levels might be better suited to predict health care costs. Therefore, FEDSS level 6 versus all other FEDSS-levels was inserted as a further possible predictor in the backward elimination regression model (Methods section “statistical analysis”).
Results of the multiple linear regression for the prediction of hospital costs from a health insurance perspective. Left column: Results of the backward elimination regression modal. Right column: Results of the wild bootstrapping model.
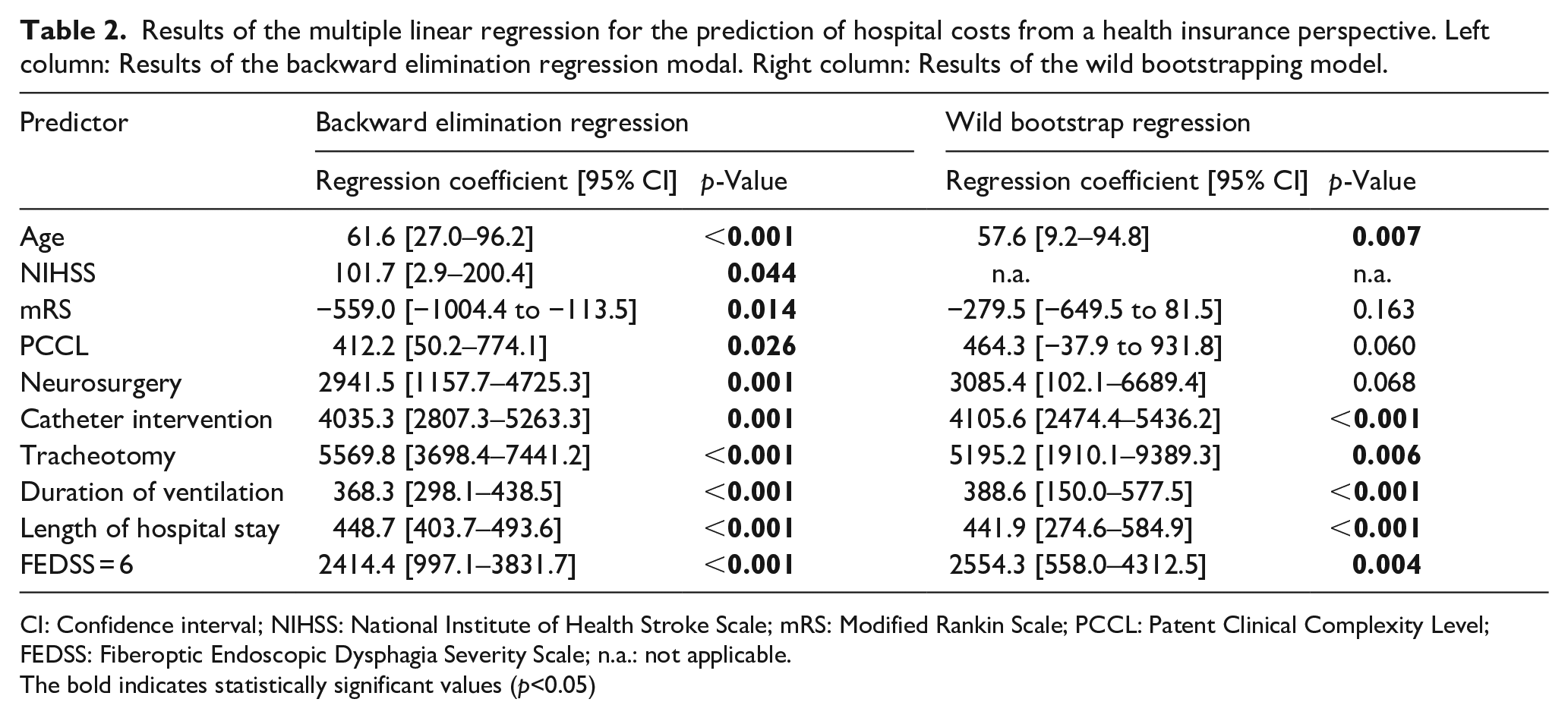
CI: Confidence interval; NIHSS: National Institute of Health Stroke Scale; mRS: Modified Rankin Scale; PCCL: Patent Clinical Complexity Level; FEDSS: Fiberoptic Endoscopic Dysphagia Severity Scale; n.a.: not applicable.
The bold indicates statistically significant values (p<0.05)
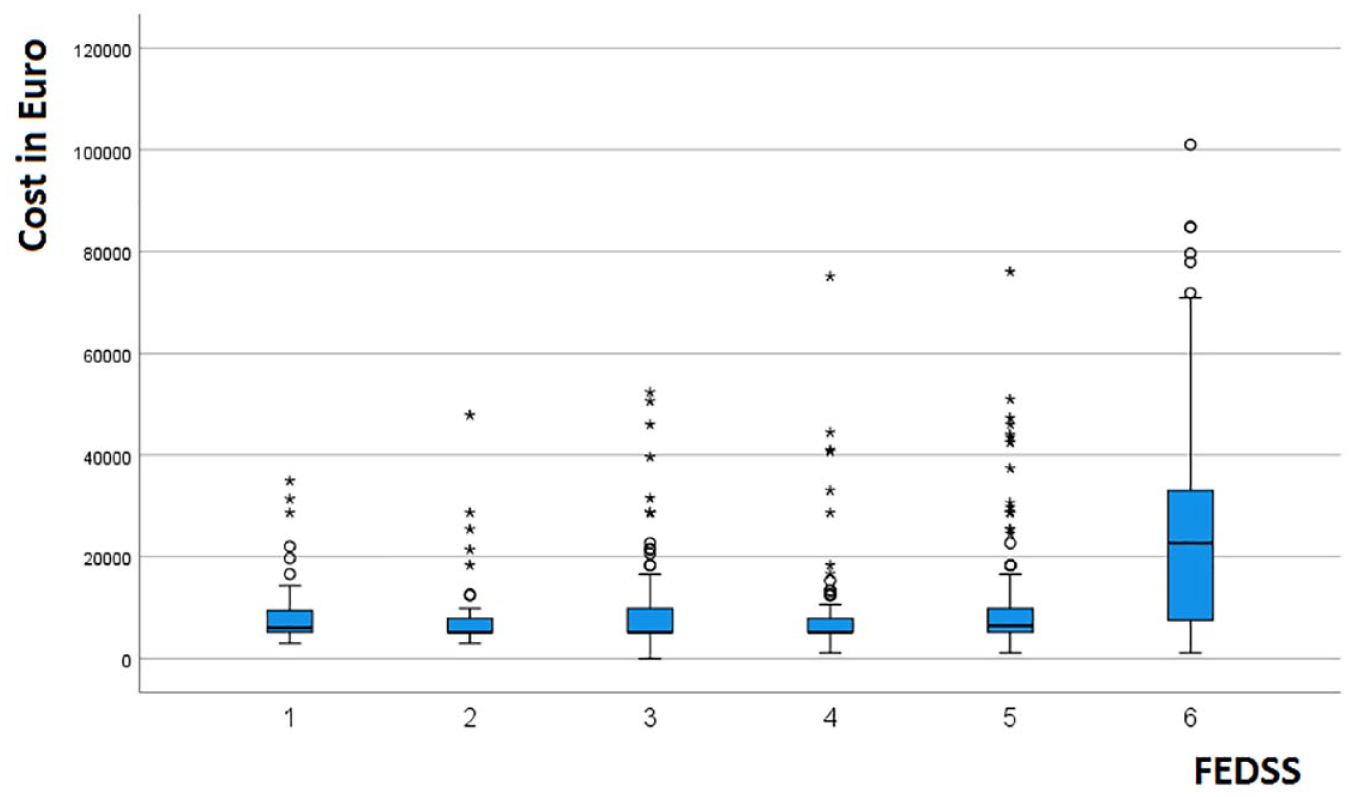
Box plots for health insurance costs depending on Fiberoptic Endoscopic Dysphagia Severity Scale (FEDSS).
Multiple linear regression using the backward elimination method revealed a significant regression line (F[10,632] = 316.7, p < 0.001, R-squared = adjusted-R-squared of 0.83), whereas higher age, higher NIHSS, lower mRS, higher DRG comorbidity level according to de PCCL, catheter-based therapy, neurosurgery, tracheotomy, length of hospital stay, duration of artificial ventilation, and FEDSS level 6 were identified as relevant predictors of higher hospital costs (Table 3, left column). The bivariate correlation analysis of the predictors showed a correlation coefficient of 0.72 for the association of NIHSS with mRS. In addition, there was a comparatively high correlation between the duration of artificial ventilation and length of stay in hospital with a correlation coefficient of 0.69, which, however, was just below the cut-off value of 0.7 (Supplemental Table 2). Apart from that, there was no evidence of significant multicollinearity in the data set. Therefore, the previously identified predictors from the backward elimination regression were used for the anew regression analysis, but the NIHSS was excluded due to multicollinearity (this allowed for a larger R-squared in model estimation than excluding the mRS). Due to heteroscedasticity and non-normally distributed residuals, the further regression analysis was performed with wild-bootstrapping and exclusion of outliers (15 patients were excluded). A significant regression line was found (F[9,619] = 330.3, p < 0.001, R-squared = adjusted-R-squared = 0.83). Again, higher age, catheter-based therapy, tracheotomy, length of hospital stay, duration of artificial ventilation, and FEDSS level 6 were significant predictors for higher costs. However, DRG comorbidity level according to the PCCL and neurosurgery narrowly missed the significance level and lower mRS was no longer a significant predictor (Table 3, right column). Repeated multicollinearity analysis yielded VIF values of less than 3 for all parameters, thus showing no evidence of significant multicollinearity (Supplemental Table 3).
Mean costs depending on dysphagia severity.
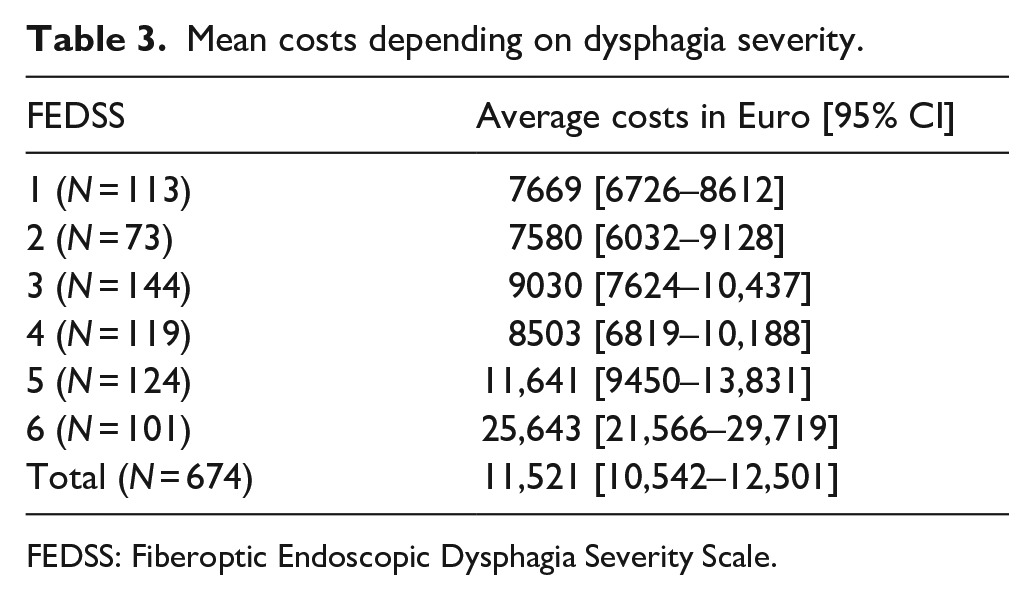
FEDSS: Fiberoptic Endoscopic Dysphagia Severity Scale.
Discussion
The results of the study show that dysphagia is associated with additional costs for health insurances during acute hospitalization. In fact, sever dysphagia with impaired secretion management was an independent predictor of higher costs. A differentiated analysis considering dysphagia severity revealed a cost-driving effect only for severe dysphagia (corresponding to an FEDSS of 6), whereas no increased costs could be shown for the ordinal dysphagia severity scaling with the severity levels 1–5.
Severe forms of dysphagia with an FEDSS score of 6 due to impaired secretion management have been shown to independently predict endotracheal intubation in stroke patients.23,24 Accordingly, close respiratory monitoring is recommended for severe dysphagia with an FEDSS of 6 . 23 Thus, additional costs due to dysphagia might be associated with the need for intubation, artificial ventilation, and intensive care treatment. Consistent with this, the intubation rate in the cohort was more than twice as high in patients with dysphagia (34%) compared to patients without dysphagia. There were also longer length of artificial ventilation and increased length of stay in the hospital in the dysphagic patients. 2 In line with this and further highlighting the role of dysphagia in stroke intensive care, a recent study demonstrated that dysphagia was the only predictor of extubation failure in critically ill stroke patients. 27 However, it should be noted that in the analysis of this study, severe dysphagia had a cost-increasing effect independently of ventilation time and length of hospital stay. The need for intubation and pneumonia were also included in the initial backward elimination regression model but were removed from the analysis in favor of other factors with stronger explanatory effects (including severe dysphagia). This can be seen as a hint that severe dysphagia might increase costs via its effect on these respective parameters.
The results of our study may be interpreted as an indication that therapies targeting severe forms of dysphagia might have the potential to reduce costs. To date, there are only few studies available that examine the effect of therapies on dysphagia with impaired secretion management. One of the few available therapies in this context is Pharyngeal Electrical Stimulation (PES), a neurostimulation method that can be used in the intensive care setting. This procedure promotes neurorehabilitation of swallowing almost independently of the patient’s ability to cooperate by delivering a pulsed current to the pharynx via catheter-mounted electrodes. A recent guideline of the European Stroke Association and the European Society for Swallowing Disorders on diagnoses and therapy of post-stroke dysphagia concludes that PES can increase the decannulation rate in tracheotomized stroke patients. 28 This conclusion is supported by a multicenter randomized controlled trial in which decannulation was feasible from a FEES perspective in 49% of patients in the intervention group, compared with only 9% of patients in the control group. 29 In line with this, PES also increased the decannulation rate in another randomized controlled monocentric trial. 30 Considering these results, PES could potentially help save costs for health insurers by improving severe dysphagia and subsequently shortening intensive care stay and ventilation times. Contrary to this hypothesis, however, in one of the studies, despite higher rates of decannulation after PES, length of stay in the intensive care unit was not shorter compared to standard treatment. 30
A synopsis of previous studies that have performed cost analyses of oropharyngeal dysphagia in stroke patients (published up to and including 2019) is available in a systematic review. 22 However, the most recent and comprehensive study to date was released after the review was published. 20 Here, 359 patients were divided into a dysphagic and non-dysphagic cohort based on a clinical swallowing examination (Volume-Viscosity-Test). Fixed costs for hospitalization were quantified with fixed amounts allocated for each day of stay. In addition, the costs for medications and enteral nutrition or nutritional supplementation were determined. The average cost of acute hospitalization was 5358€ for patients with dysphagia and thus 1.3 times the amount of the 3976€ in the patients without swallowing impairment. Consistent with the results of our study, regression analysis showed that dysphagia was a predictor of higher acute hospital costs, independent of age, concomitant disease, baseline functional status, and stroke severity. Besides country-specific factors, the difference in absolute numbers of costs compared to our study may be explained by different stroke severity in the cohorts. The mean NIHSS in the patient cohort was 5.1 in comparison to 11.2 in our study. Further, the prevalence of dysphagia in the study was lower, approximately 45%, than in our own cohort, approximately 80%. This may be attributed to the lower diagnostic sensitivity of the clinical diagnostic procedure compared to instrumental assessment. On the other hand, a particularly high prevalence of dysphagia may have occurred in our study cohort due to a patient preselection. However, only severe dysphagia showed an association with higher costs in our cohort, which in turn occurred in only approximately 15% of patients. A further study in a Taiwanese cohort of 311 ischemic stroke patients in a rehabilitation facility concluded that dysphagia was not an independent predictor of higher hospital costs in the regression analysis. 18 The cost analysis included amounts submitted by the hospital to the insurance company for reimbursement and amounts reimbursed by private parties. Dysphagia was determined based on a clinical examination, although no detailed protocol or timing of the examination is provided. One reason for the discrepancy to the own study results may be that dysphagia was probably not investigated during the acute phase of the disease due to the rehabilitation setting. This is important, since complications of dysphagia frequently occur early after patient admission. 31 In another study by the same group of investigators with similar study procedure, 237 patients with hemorrhagic stroke were investigated. 21 Here, dysphagia was shown to be a significant predictor of increased costs in a multivariate regression analysis (regression coefficient of 1025 with mean costs of 5940 + 3579$). However, due to the lack of information on stroke severity in both Taiwanese studies cohorts, it is difficult to compare the results. In two further studies, dysphagia rates were determined based on diagnostic coding. In an American study, the costs billed via Medicare (public insurance in the USA) in 2004 were considered within 1 year after stroke in a total of 3200 patients. 17 Hospital costs were 6637$ for patients with dysphagia, only 1.01 times higher than 6539$ for patients without dysphagia. After adjusting for cofactors such as age, comorbidities, ethnicity, and survival time and accounting for all costs within 1 year, the additional costs attributed to dysphagia were 4510$. In another study, 62,297 patients in a French cohort and 6037 patients in a Swiss cohort were analyzed and costs were determined from the hospitals’ perspective. 19 The cost for patients with dysphagia in the French cohort was 8770€, which was 1.5 times higher than the average cost of 5844€ in the patients without dysphagia. In the Swiss cohort, however, the costs of 27,801 Swiss francs (CHF) were approx. twice as high as in patients without dysphagia, with costs of 13,842 CHF. In all study cohorts with dysphagia assignment via diagnostic codes, the prevalence of dysphagia was around or below 10%. This is clearly lower compared to usually reported prevalence rates. 8 It can therefore be assumed that in these studies a significant proportion of patients with swallowing impairment were classified as non-dysphagic. Another prospective study from the USA compared the costs for a total of 48 patients with and without tube feeding. 16 The cost for patients with tube feeding was 12,538$, more than double the cost for patients without tube feeding (5949$). It should be noted, however, that the study is more than 20 years old and does not describe the determination of cost in detail. Furthermore, according to the results of our study, it is not so much the subdivision into dysphagic and non-dysphagic subjects that might be relevant, but rather the subdivision into severe dysphagia with impaired secretion management, which has not been decisively investigated in any of the previous studies.
Several limitations must be considered when interpreting the study. The retrospective and cross-sectional design and possible selection bias in the patient cohort is a major limitation. Thus, future prospective studies that assess the actual costs incurred by dysphagia are necessary to confirm the findings established here. Approximately 83% of the variance in costs is explained by the predictor variables in the regression analysis. Therefore, other relevant factors accounting for the not explained variance were likely not included in the regression analysis. Due to the DRG reimbursement system, costs for longer stays in normal wards without artificial ventilation are only relevant for reimbursement if there is a major deviation from the average length of stay. Thus, additional hospital costs incurred by a longer length of stay are not entirely reflected in the results. Conversely, ventilation times and intensive care are of great importance for reimbursement. This could explain why cost increases were attributed with high sensitivity to the severe forms of dysphagia, which typically affect these very parameters. Thus, there could also be health care costs for less severe dysphagia that were not reflected in our analysis due to the study design. The revenue structure via the DRG system does not consider additional charges (e.g. for hemodialysis procedures) and payments for new examination and treatment methods (“Neue Untersuchungs- und Behandlungsmethoden,” NUB). However, these payments play an extremely minor role in the overall hospital costs in general and for stroke patients in particular. The cost data showed heteroscedasticity and non-normally distributed residuals. In the regression analysis, this was countered with wild-bootstrapping but no correction was possible in the initial backward elimination regression.
In summary, our study suggests that post-stroke dysphagia is associated with significant additional costs for health insurers during acute hospitalization. However, the main cost driver is severe dysphagia with impaired secretion management. Rehabilitation methods that treat severe dysphagia and improve secretion management may therefore bear the potential to save costs.
Supplemental Material
sj-docx-1-eso-10.1177_23969873221147740 – Supplemental material for Costs of post-stroke dysphagia during acute hospitalization from a health-insurance perspective
Supplemental material, sj-docx-1-eso-10.1177_23969873221147740 for Costs of post-stroke dysphagia during acute hospitalization from a health-insurance perspective by Bendix Labeit, Almut Kremer, Paul Muhle, Inga Claus, Tobias Warnecke, Rainer Dziewas and Sonja Suntrup-Krueger in European Stroke Journal
Supplemental Material
sj-docx-2-eso-10.1177_23969873221147740 – Supplemental material for Costs of post-stroke dysphagia during acute hospitalization from a health-insurance perspective
Supplemental material, sj-docx-2-eso-10.1177_23969873221147740 for Costs of post-stroke dysphagia during acute hospitalization from a health-insurance perspective by Bendix Labeit, Almut Kremer, Paul Muhle, Inga Claus, Tobias Warnecke, Rainer Dziewas and Sonja Suntrup-Krueger in European Stroke Journal
Supplemental Material
sj-docx-3-eso-10.1177_23969873221147740 – Supplemental material for Costs of post-stroke dysphagia during acute hospitalization from a health-insurance perspective
Supplemental material, sj-docx-3-eso-10.1177_23969873221147740 for Costs of post-stroke dysphagia during acute hospitalization from a health-insurance perspective by Bendix Labeit, Almut Kremer, Paul Muhle, Inga Claus, Tobias Warnecke, Rainer Dziewas and Sonja Suntrup-Krueger in European Stroke Journal
Footnotes
Acknowledgements
This work is based on the master thesis by Bendix Labeit in the Master of Business Administration in Medical Management (MBA) at the University of Muenster.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: BL has received a grant from the medical faculty Münster and was involved in a study funded by Clexio Biosciences Ltd; AK has nothing to disclose; PM has nothing to disclose; IC has previously received honoraria from Abbvie, BIAL, STADAPHARM, Georg Thieme Verlag KG, and consultancies from STADAPHARM and was involved in a study funded by Clexio Biosciences Ltd.; TW reports advisory board memberships for AbbVie, UCB, Archimedes, Phagenesis, Zambon, Bial, Kyowa, honoraria for lectures from Bial, AbbVie, STADA, UCB, Biogen, Licher, Desitin, Pfizer, Zambon, Teva, Bayer, Grants (investigator-initiated) from UCB, Licher, Abbvie and Clexio Biosciences Ltd., academic grants from G-BA Innovation Fund, Deutsche Parkinson-Vereinigung (dPV), IZKF, Neuro NRW; RD reports non-financial support from Phagenesis, personal fees from Nestle Healthcare, grants from Olympus, personal fees from Sanofi, personal fees from Pfizer, personal fees from Daiichi Sankyo, outside the submitted work, SSK was supported by the Else Kröner-Fresenius-Stiftung with a clinician scientist endowed professorship.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a grant for BL from the medical faculty Münster and by a clinician scientist endowed professorship for SSK from the ElseKröner-Fresenius-foundation.
Informed consent
Informed consent was not sought for the present study because the ethics committee waived the need for informed consent due to the retrospective design.
Ethical approval
The nature of the study was approved by the local ethics committee at the University of Muenster.
Guarantor
BL.
Author contributions
BL was involved in the study design, data analysis and writing of the manuscript, AK was involved in collecting the cost-data from the DRG department, PM was involved in editing the manuscript, IC was involved in editing the manuscript, TW was involved in editing the manuscript, RD was involved in editing the manuscript, SSK was involved in the study design, the collection of data and in editing the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
