Abstract
Background:
Early identification of patients developing symptomatic intracranial hemorrhage and symptomatic brain edema after acute ischemic stroke is essential for clinical decision-making. Astroglial protein S-100B is a marker of blood-brain barrier disruption, which plays an important role in the formation of intracranial hemorrhage and brain edema. In this study, we assessed the prognostic value of serum S-100B for the development of these complications.
Methods:
Serum S-100B levels were measured within 24 h from symptom onset in 1749 consecutive acute ischemic stroke patients from the prospective, observational, multicenter BIOSIGNAL cohort study (mean age 72.0 years, 58.3% male). To determine symptomatic intracranial hemorrhage or symptomatic brain edema, follow-up neuroimaging was performed in all patients receiving reperfusion therapy or experiencing clinical worsening with an NIHSS increase of ⩾4.
Results:
Forty six patients (2.6%) developed symptomatic intracranial hemorrhage and 90 patients (5.2%) developed symptomatic brain edema. After adjustment for established risk factors, log10S-100B levels remained independently associated with both symptomatic intracranial hemorrhage (OR 3.41, 95% CI 1.7–6.9, p = 0.001) and symptomatic brain edema (OR 4.08, 95% CI 2.3–7.1, p < 0.001) in multivariable logistic regression models. Adding S-100B to the clinical prediction model increased the AUC from 0.72 to 0.75 (p = 0.001) for symptomatic intracranial hemorrhage and from 0.78 to 0.81 (p < 0.0001) for symptomatic brain edema.
Conclusions:
Serum S-100B levels measured within 24 h after symptom onset are independently associated with the development of symptomatic intracranial hemorrhage and symptomatic brain edema in acute ischemic stroke patients. Thus, S-100B may be useful for early risk-stratification regarding stroke complications.
Introduction
In patients with acute ischemic stroke (AIS), secondary neuronal damage can occur in subsequent hours to days, caused by complications such as symptomatic intracranial hemorrhage (sICH) or symptomatic brain edema (sBE). AIS followed by these complications is associated with poor functional outcome and higher mortality.1,2 Therefore, early risk-stratification is crucial to guide patient management, such as the decision over intensive care unit placement or intensified monitoring. Moreover, such information may facilitate time-sensitive therapy, such as decompressive hemicraniectomy. Inversely, identifying low-risk patients may help to improve health care allocation.
Patient history, clinical assessment and neuroimaging on admission provide some prognostic information, but their predictive value for complications is limited.3,4 Serum biomarkers of blood-brain barrier (BBB) disruption could be an important adjunct to predict complications, as BBB disruption has previously been associated with sBE and sICH. 5 S-100B, a calcium-binding protein produced predominantly by astroglial cells, is an established biomarker of BBB dysfunction.6,7 Its association with stroke severity, infarction volume and functional outcome in AIS patients is well-researched.8–11 Pilot studies have demonstrated an association of S-100B with sICH and sBE.12–14 In this study, we aimed to externally validate these associations and evaluate the incremental prognostic value of serum S-100B levels for the development of sICH and sBE during hospitalization when measured within 24 h after symptom onset in a large multicentric AIS cohort.
Methods
Study design and approval
The Biomarker Signature of Stroke Etiology (BIOSIGNAL) study is a prospective, observational, multicenter, inception cohort study to evaluate selected blood biomarkers in patients with confirmed AIS (NCT02274727). The study protocol was approved by each local Ethics Committee and conducted according to the principles expressed in the Declaration of Helsinki. Written informed consent was obtained from all patients or their welfare guardians for the collection blood samples and clinical data for further analysis.
Study population
The design of the BIOSIGNAL study has been described in detail elsewhere. 15 In brief, patients older than 18 years who were admitted with AIS and gave consent were recruited within 24 h from symptom onset between October 2014 to October 2017. Patients who were diagnosed with primarily hemorrhagic stroke, transient ischemic attack or stroke mimics (e.g. migraine, epilepsy) at hospital discharge, or who refused informed consent were excluded from the study. Recruitment was stopped after 150 recurring cerebrovascular events occurred, as intended for a different research question within the BIOSIGNAL study.
Data collection
Demographic variables, vital signs, vascular risk factors, National Institute of Health Stroke Scale (NIHSS) score, and modified Charlson Comorbidity Index were documented on admission. All patients received brain imaging with CT or MRI on admission. Standard etiological work up included 12-lead ECG, >24 h continuous ECG monitoring, transthoracic and/or transoesophageal echocardiography as well as cerebrovascular ultrasound and/or CT or MR angiography. Per protocol, in all patients who received any form of reperfusion therapy (i.e. intravenous or intraarterial thrombolysis (IVT/IAT) and/or mechanical thrombectomy), control neuroimaging was performed after 24 h. In addition, all patients showing clinical worsening during hospital stay, defined as an increase in NIHSS by ⩾4 points, received control neuroimaging. Patients who deceased, who were treated palliatively or who were transferred to a different hospital before control neuroimaging was performed were excluded from the final analysis (n = 10) (Figure 1).
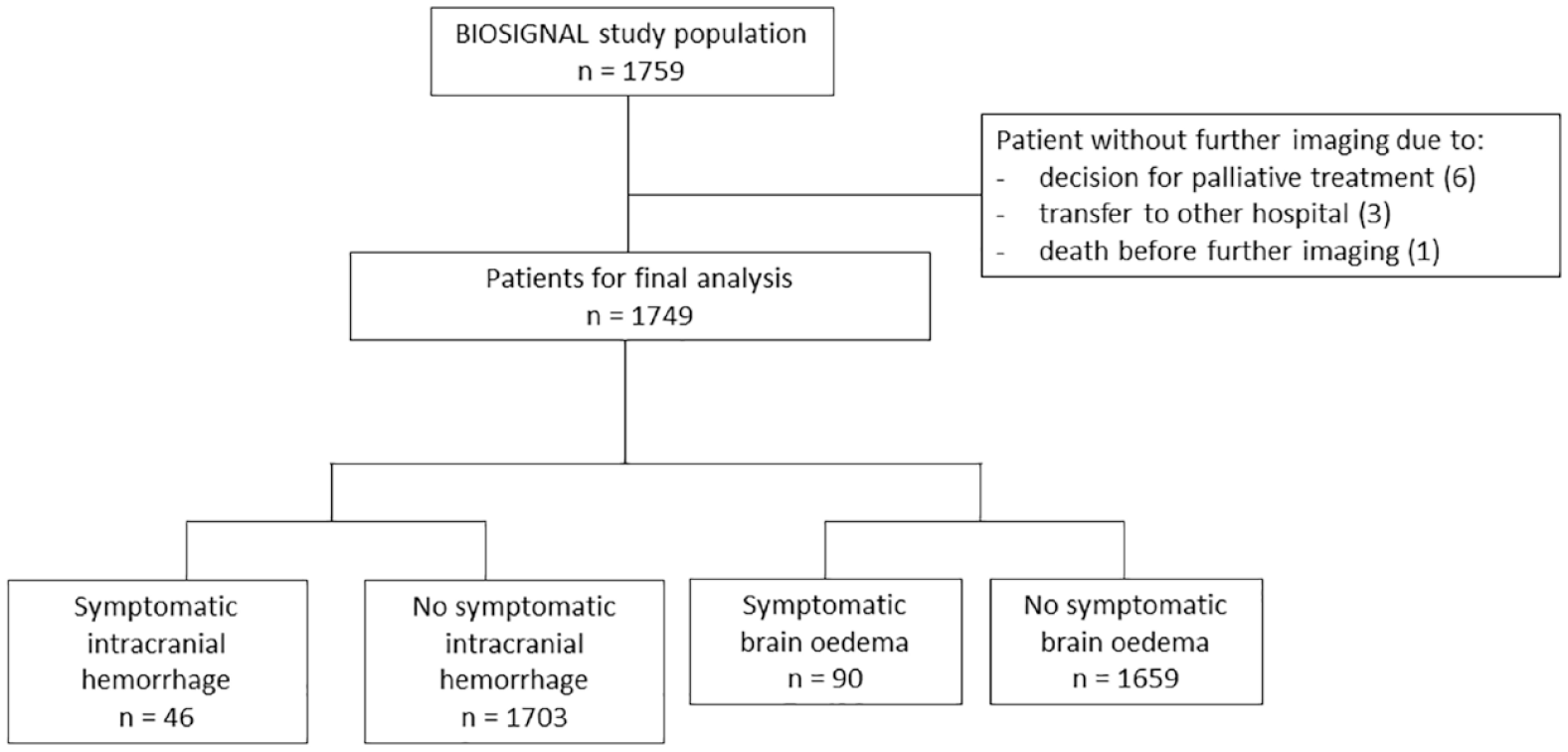
Final study population selection process.
Blood sampling
Venous blood was sampled in EDTA-containing plastic tubes within 24 h after symptom onset. Samples were immediately centrifuged at 3000g and 4°C for 20 min. Plasma was alliquoted and stored at −80°C until time of analysis. S-100B levels (µg/ml) were measured from plasma in a blinded batch analysis with the Roche Elecsys S100 assay running on the e801 unit of the Cobas 8000 platform (Roche Diagnostics, Mannheim, Germany) with a lower limit of detection of 0005 µg/l and a lower limit of quantification of 0.015 µg/l. Inter- and intraassay imprecision of the assay as expressed by coefficient of variance was less than 2.6%. No values below LoQ were observed. Analysis can be performed on two different immunoassay analyzing machines, is completed within 18 min and is commercially available.
Outcome variables
Primary outcome measures included the occurrence of sICH and sBE. Based on the ECASS-II study, we defined sICH as apparent extravascular blood in the brain or within the cranium on imaging in temporal correlation with clinical worsening (increase in NIHSS by ⩾4 points) or death due to neurological deterioration. 16 sBE was defined as apparent mass effect of the brain on control neuroimaging as the predominant cause of clinical deterioration (increase in NIHSS by ⩾4 points) or death as well as by the indication for decompressive hemicraniectomy. 17 The neuroradiologist on duty, who was blinded to S-100B levels, evaluated the presence of intracranial hemorrhage or brain edema at each site separately.
Statistical analysis
Discrete variables are expressed as counts (percentages), continuous variables as medians (interquartile range (IQR)). Logarithmic transformation (log10) was used to normalize skewed distributions, including serum levels of S-100B. For group comparisons, Fisher’s exact test or Mann-Whitney U test was conducted where appropriate.
To evaluate the association of S-100B levels with primary outcomes, multivariable logistic regression models were fitted to calculate odds ratios (OR) and 95% confidence interval (95% CI). Models for sICH and sBE were adjusted for pre-defined covariates based on previous studies and pathophysiological considerations. The pre-defined model for sICH included age,18,19 sex, 19 previous use of antithrombotic drugs (oral anticoagulants and/or antiplatelets), 18 NIHSS on admission,18,19 reperfusion therapy (IVT/IAT and/or mechanical thrombectomy), 16 as well as log10S-100B levels. The pre-defined model for sBE included age, 17 sex, 20 NIHSS on admission,17,20,21 CRP levels,22,23 reperfusion therapy, 24 affection of middle cerebral artery (MCA), 25 and log10S-100B levels. Interaction analyses were performed to assess effect modification of log10S-100B by demographic variables and established risk factors. Finally, we assessed the influence of time from symptom onset to blood draw on S-100B levels as well as both outcomes with separate logistic regressions. Patients with wake-up stroke and unknown symptom onset were excluded from this analysis, as the time interval could not reliably be determined.
For both outcomes we used additional stepwise model building strategies for a best out-of-sample predictive model. Variables were selected based on univariate logistic regression with a cut-off level of p < 0.15 to achieve retention of possibly meaningful covariates. 26 Backward selection was performed with a cut-off level of p < 0.05. Receiver Operating Characteristic (ROC) curves were plotted and Areas under the curve (AUC) were calculated as an overall discriminatory measure. Models with and without S-100B were compared using the likelihood-ratio test. To assess improvement in classification after adding S-100B to the model, continuous net reclassification improvement (NRI) was computed. Subgroup analyses were performed based on stroke severity as well as treatment with IVT.
Different S-100B cut-off values were evaluated for inclusion and exclusion of patients at increased risk for sICH, respectively sBE. We chose cut-off values with respective specificity values above 90% and calculated the corresponding sensitivity, negative and positive predictive values.
In addition, we applied six existing sICH prognostication scores for AIS patients after IVT to our cohort: SITS score, 27 SEDAN score, 28 HAT score, 29 SPAN-100, 30 MSS, 31 and GRASPS score. 19 Where necessary for calculation of the score, analysis was only performed in patients receiving CT on admission. Patients without IVT were excluded from this analysis. All statistical analysis was performed using Stata (Version 15.1, StataCorp LLC, College Station, Texas).
Results
Patient baseline characteristics
Of 1759 AIS patients enrolled in the study, 10 were excluded as pre-defined control neuroimaging could not be performed because of a switch to palliative care treatment (n = 6), transfer to a different hospital (n = 3), or death without prior follow up neuroimaging (n = 1). In the remaining 1749 patients, blood samples were available in 98.4% with a median S-100B concentration of 0.08 µg/l (IQR 0.05–0.15). The median S-100B concentration in patients who were excluded was 0.09 µg/l (IQR 0.08–0.17), which was not significantly different from that of included cases (p = 0.23). Mean age was 72.0 years, 41.7% were female. All patients who suffered apparent clinical worsening and all patients receiving re-canalization therapy received control neuroimaging. Participating centers were free to extend the indication for follow up neuroimaging, for example following clinical worsening with an increase in NIHSS of less than four points or as a standard of care. In total, 1584 patients (90.6%) received follow up neuroimaging. The complication sICH occurred in 46 patients (2.6%), while 90 patients (5.2%) developed sBE. Patients undergoing reperfusion therapy showed higher rates of stroke complications (sICH: 3.9% [n = 32/817] vs 1.5% [n = 14/932], sBE 6.7% [n = 55/817] vs 3.8% [n = 33/932]). Among patients with sICH, 32.6% (n = 15) died during hospital stay and among patients with sBE, 23.3% (n = 21) received decompressive hemicraniectomy while 46.7% (n = 42) died during the hospitalization. Further details on baseline characteristics are listed separately (Table 1).
Baseline characteristics categorized by sICH and sBE.
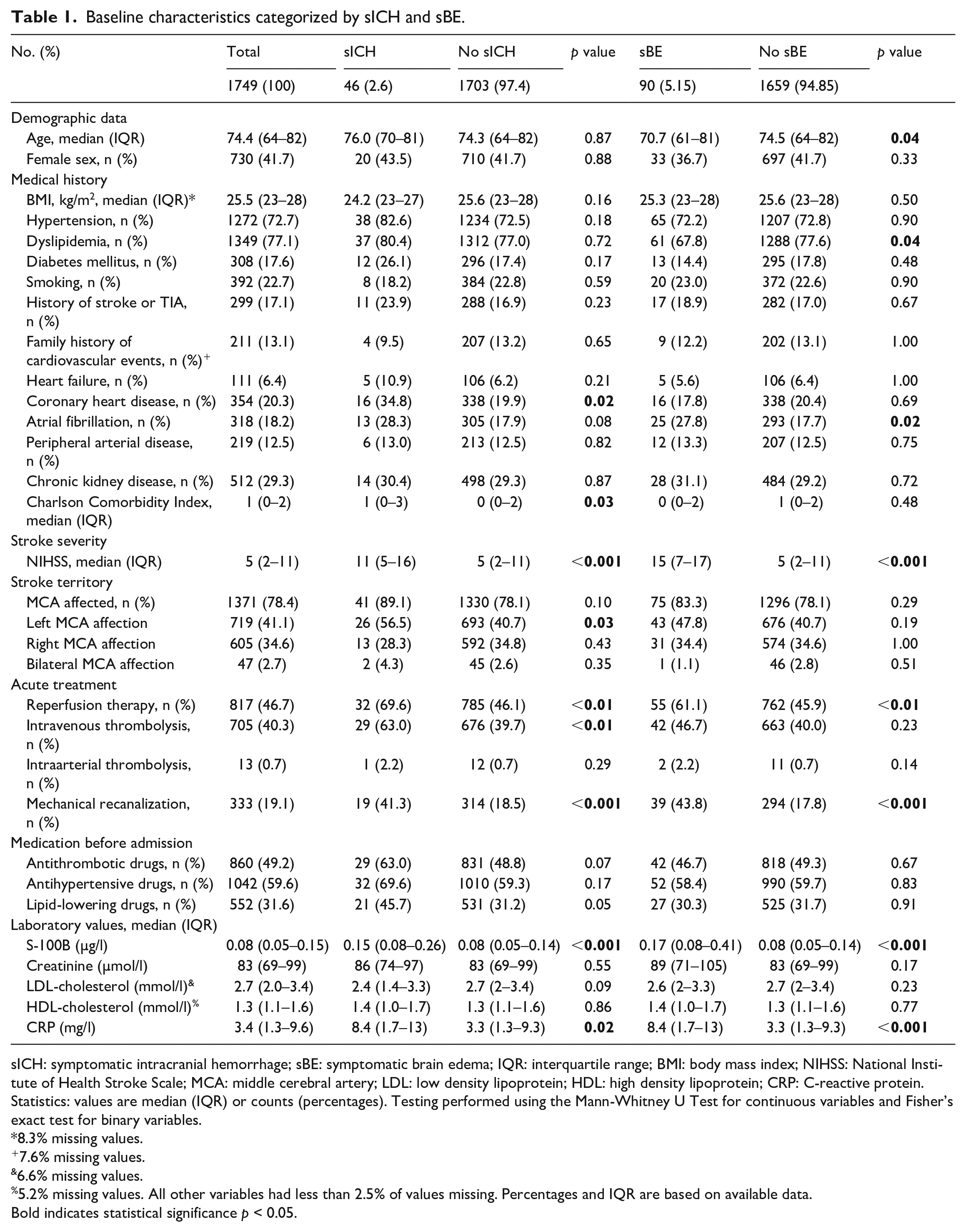
sICH: symptomatic intracranial hemorrhage; sBE: symptomatic brain edema; IQR: interquartile range; BMI: body mass index; NIHSS: National Institute of Health Stroke Scale; MCA: middle cerebral artery; LDL: low density lipoprotein; HDL: high density lipoprotein; CRP: C-reactive protein.
Statistics: values are median (IQR) or counts (percentages). Testing performed using the Mann-Whitney U Test for continuous variables and Fisher’s exact test for binary variables.
8.3% missing values.
7.6% missing values.
6.6% missing values.
5.2% missing values. All other variables had less than 2.5% of values missing. Percentages and IQR are based on available data.
Bold indicates statistical significance p < 0.05.
Predictive value of S-100B for sICH
Serum S-100B levels were significantly higher in patients who developed sICH (0.15 µg/l [IQR 0.08–0.26] vs 0.08 µg/l [0.05–0.14], p < 0.0001). In the pre-defined multivariable regression model, only log10S-100B (OR 3.40, 95% CI 1.69–6.87 per unit log10S-100B increase, p = 0.001) and reperfusion therapy (OR 2.18, 95% CI 1.09–4.36, p < 0.05) remained independently associated with sICH during hospitalization (Table 2). Adding log10S-100B to the model increased its discriminatory accuracy from an AUC of 0.72 (95% CI 0.65–0.78) to 0.75 (95% CI 0.68–0.82, p = 0.001) (Supplemental Figure 1). Overall continuous NRI was 0.36 (p = 0.02) with event NRI being 0.02 and non-event NRI 0.34, meaning that additional 2% of cases received higher probabilities to be correctly classified into the event group, and additional 34% of cases received higher probabilities to be correctly classified into the non-event group.
Multivariable logistic regression and ROC analyses for sICH with and without log10S-100B.
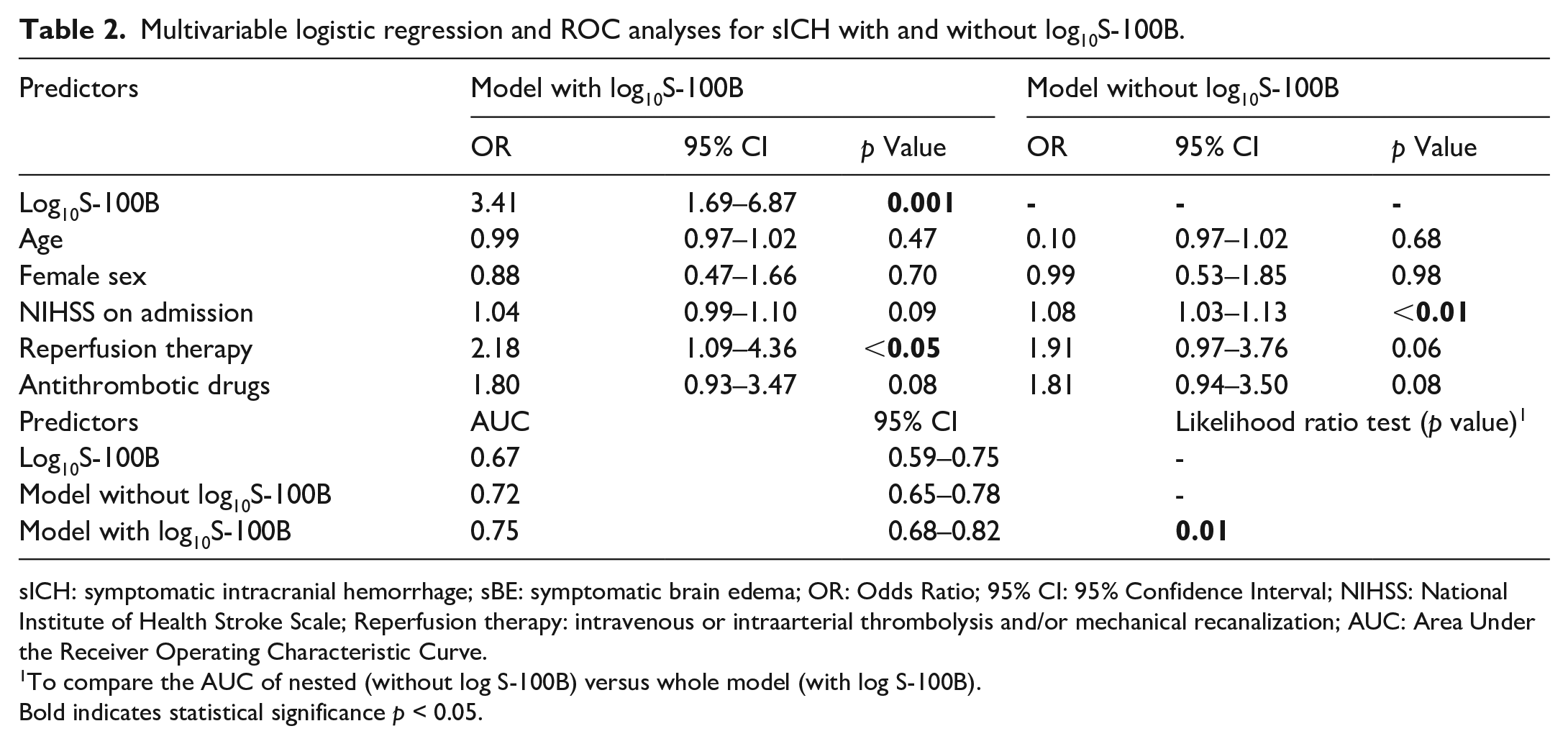
sICH: symptomatic intracranial hemorrhage; sBE: symptomatic brain edema; OR: Odds Ratio; 95% CI: 95% Confidence Interval; NIHSS: National Institute of Health Stroke Scale; Reperfusion therapy: intravenous or intraarterial thrombolysis and/or mechanical recanalization; AUC: Area Under the Receiver Operating Characteristic Curve.
To compare the AUC of nested (without log S-100B) versus whole model (with log S-100B).
Bold indicates statistical significance p < 0.05.
In the best out-of-sample predictive model based on stepwise model building, log10S-100B remained as an independent predictor (OR 4.17, 95% CI 2.2–7.8, p < 0.0001 per unit log10S-100B increase; improvement of AUC from 0.65 [95% CI 0.57–0.73] to 0.73 [95% CI 0.64–0.81], p < 0.0001). The final model included log10S-100B, reperfusion therapy and coronary heart disease.
Application of six existing sICH prognostication scores after IVT to our cohort revealed AUC values ranging from 0.48 to 0.63 versus 0.68 to 0.77 in the original studies.19,27–31 The addition of log10S-100B to the scores led to an increase of the AUC, ranging from 0.71 to 0.73. In all scores, this increase was significant (p < 0.01). Adjustment for other score items revealed that log10S-100B remained independently associated with sICH in every score (Table 3).
AUC of prediction scores for sICH after IVT in the original studies, in a comparison of the scores and applied to the BIOSIGNAL study cohort. (a) AUC of sICH prediction scores reported in previous studies. (b) AUC of sICH prediction scores calculated in the BIOSIGNAL study cohort.
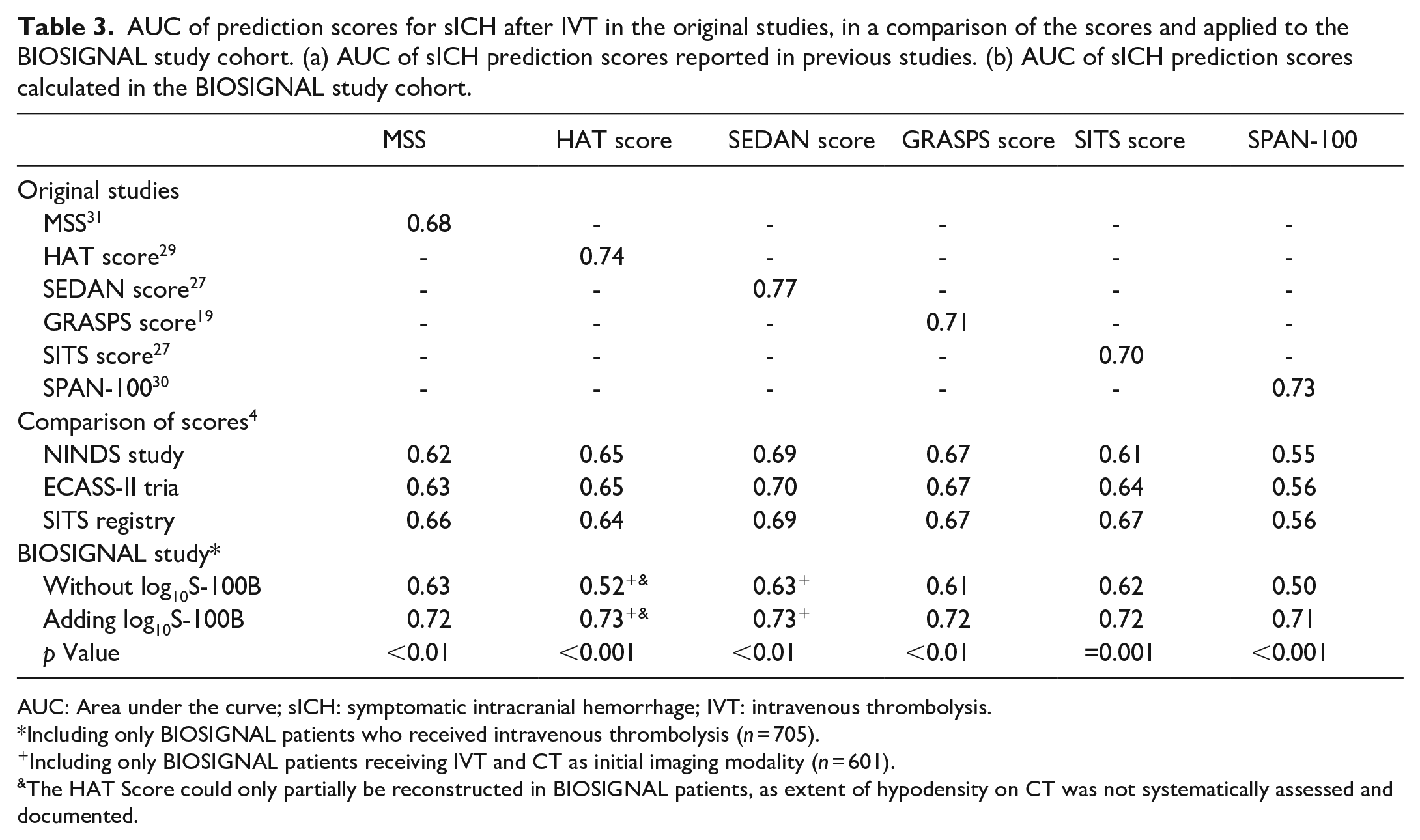
AUC: Area under the curve; sICH: symptomatic intracranial hemorrhage; IVT: intravenous thrombolysis.
Including only BIOSIGNAL patients who received intravenous thrombolysis (n = 705).
Including only BIOSIGNAL patients receiving IVT and CT as initial imaging modality (n = 601).
The HAT Score could only partially be reconstructed in BIOSIGNAL patients, as extent of hypodensity on CT was not systematically assessed and documented.
Predictive value of S-100B for sBE
Serum S-100B levels were significantly higher in patients with sBE (0.17 µg/l [IQR 0.08–0.41] vs 0.08 µg/l [0.05–0.14], p < 0.0001). In the predefined multivariable regression model, only log10S-100B (OR 4.08, 95% CI 2.3–7.1, p < 0.001 per unit log10S-100B increase), age (OR 0.97, 95% CI 0.95–0.98, p < 0.0001) and NIHSS on admission (OR 1.11, 95% CI 1.1–1.2, p < 0.001) remained independently associated with sBE (Table 4). Adding log10S-100B to the model improved its discriminatory accuracy from an AUC of 0.78 (95% CI 0.73–0.83) to 0.81 (95% CI 0.76–0.86, p < 0.001) (Supplemental Figure 2). Overall continuous NRI was 0.44 (p < 0.001) with event NRI being 0.13 and non-event NRI 0.31. Interaction analysis revealed no significant interaction between log10S-100B and the independent variables.
Multivariate logistic regression and ROC analyses for sBE with and without log10S-100B.
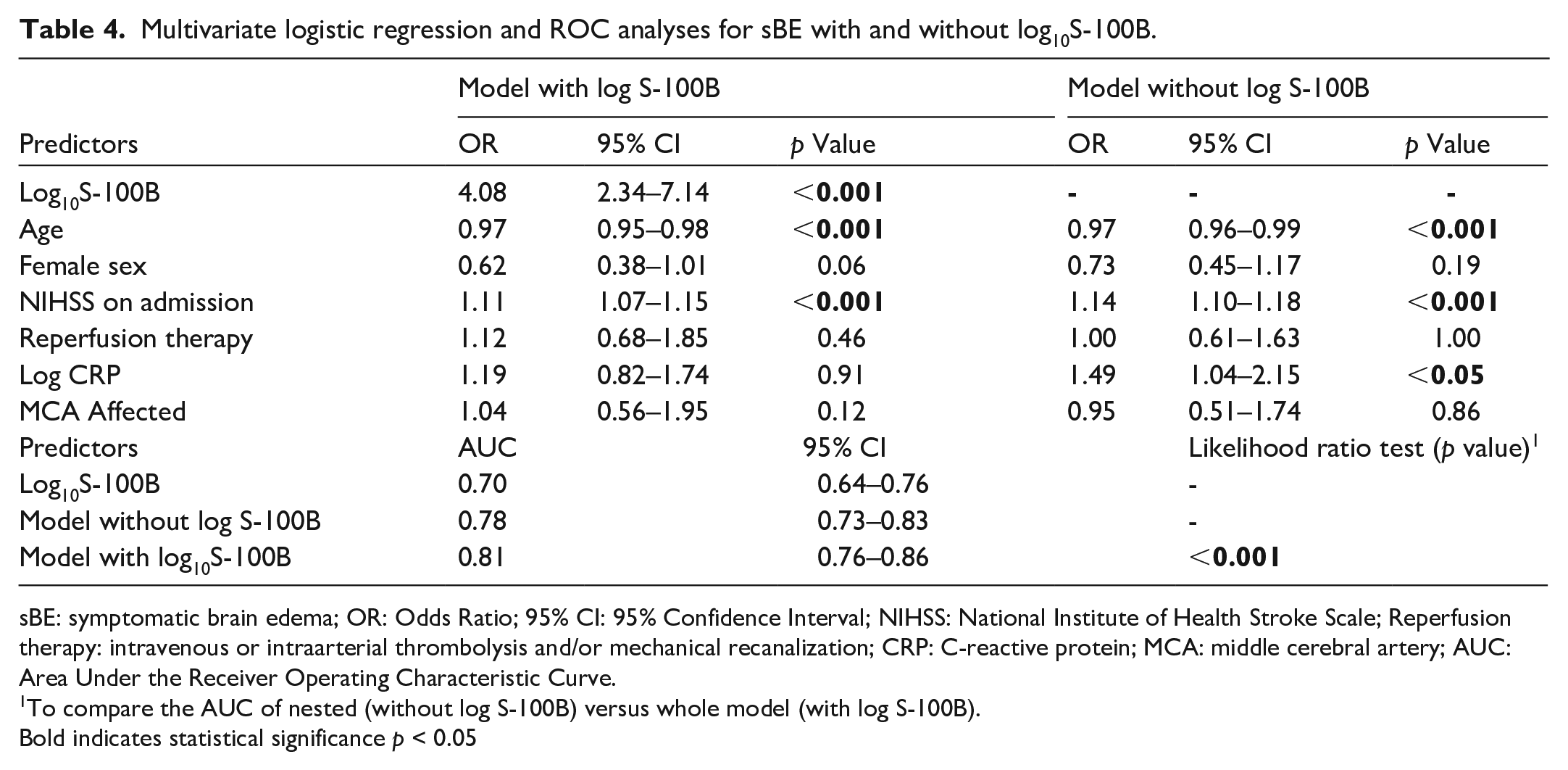
sBE: symptomatic brain edema; OR: Odds Ratio; 95% CI: 95% Confidence Interval; NIHSS: National Institute of Health Stroke Scale; Reperfusion therapy: intravenous or intraarterial thrombolysis and/or mechanical recanalization; CRP: C-reactive protein; MCA: middle cerebral artery; AUC: Area Under the Receiver Operating Characteristic Curve.
To compare the AUC of nested (without log S-100B) versus whole model (with log S-100B).
Bold indicates statistical significance p < 0.05
Using stepwise model building, log10S-100B remained as an independent predictor in the best out-of-sample model (OR 4.02, 95% CI 2.4–6.8 p < 0.001 per unit log10S-100B increase; improvement of AUC from 0.77 [95% CI 0.72–0.82] to 0.81 [95% CI 0.76–0.86], p < 0.001). The resulting model included log10S-100B, age, NIHSS, and log10creatinine.
Subgroup analyses
For sICH, the interaction analysis showed a significant interaction between log10S-100B and NIHSS on admission in the multivariable model (p = 0.04). Therefore, we performed a subgroup analysis for patients with minor stroke (NIHSS ⩽ 5) and non-minor stroke (NIHSS ⩾ 6). Log10S-100B remained as an independent predictor of sICH in patients with minor stroke (OR 18.8 [5.4–65.0], p < 0.001), while the association was attenuated in the non-minor stroke group (OR 1.9 [0.81–4.32], p = 0.14). In the bivariate model predicting sICH, both log10S-100B and NIHSS on admission remained independently associated (p < 0.01 respectively).
Further, we evaluated cut-off values for the inclusion and exclusion of patients at elevated risk for sICH in patients with minor stroke. A cut off-value of S-100B below 0.04 µg/l showed a specificity of 90.9% for not developing sICH (13th percentile of the cohort) (Table 5). Therefore, it could be used for exclusion of severe clinical courses after AIS. For inclusion of patients at high risk for the development of sICH, a cut off-value of S-100B above 0.31 µg/l showed a specificity of 90.2% (89th percentile).
Cut-off sensitivities and specificities for sICH in patients with minor stroke (NIHSS ⩽ 5).

sICH: symptomatic intracranial hemorrhage; sBE: symptomatic brain edema; NPV: negative predictive value; PPV: positive predictive value.
For the outcome variable sBE, log10S-100B remained significantly associated both in cases with minor and non-minor stroke (p < 0.001 respectively).
Moreover, subgroup analysis was performed based on whether patients received IVT. These patients did not show higher S-100B levels and did not develop sBE more often than patients receiving conservative treatment. However, patients developed sICH significantly more often after IVT than after conservative treatment (4.1% [n = 29] vs 1.6% [n = 17], p < 0.01). Lastly, we assessed the influence of time to blood draw on S-100B levels. S-100B was not significantly lower when blood was drawn before the median time of 270 min (IQR 110–900 min) compared to later blood draw (0.07 µg/l [0.05–0.13] vs 0.08 µg/l [0.05–0.14], p = 0.18). Inclusion of time from symptom onset to blood draw to the models for sBE and sICH did not alter the existing association of S-100B with both outcomes (OR 4.66 [2.3–9.3] and 4.63 [2.0–10.6] respectively, p < 0.001 for both). Time to blood draw showed no significant association with both outcomes (OR 1.00 [1.0–1.0] for both, p = 0.47 and 0.15 respectively).
Discussion
This study confirmed the predictive value of serum S-100B levels measured within 24 h after symptom onset for the occurrence of sICH and sBE in an unselected, prospective, large cohort of AIS patients. Adding log10S-100B to the pre-defined prediction models led to an increase in the diagnostic accuracy of both models measured by comparison of AUC and reclassification indices, showing its incremental value beyond previously established risk factors.
Of note, interaction analysis revealed only significant effect modification of S-100B levels by clinical stroke severity concerning the outcome of sICH. Subgroup analysis showed an attenuated association in patients with NIHSS ⩾ 6. We hypothesized that the explanatory power of BBB disruption measured by S-100B is strong especially in patients with smaller infarcts at generally lower risk of sICH, whereas the additional explanatory power of S-100B diminishes in patients with more severe infarcts (according to NIHSS levels) who inherently are at higher risk of sICH. This is particularly interesting as the assessment of the risk for development of sICH is especially challenging in patients with mild stroke. Our data suggest that serum S-100B levels over 0.7 µg/l in patients with minor strokes (NIHSS ⩽ 5) are highly specific for sICH and might indicate a need for intensified and prolonged monitoring.
Our findings regarding the association of S-100B and sICH are in line with those of pilot studies. In a prospective study of 458 AIS patients without IVT or anticoagulation therapy, S-100B was independently associated with sICH in multivariable models adjusted for age, NIHSS, platelet count, hypertension, diabetes, and antiplatelet therapy. 12 Another study including 275 AIS patients found S-100B levels in the highest quintile compared to the lowest quintile to be independently associated with hemorrhagic transformation 24 h after IVT. 13
Evidence regarding the association between S-100B levels and sBE is scarcer. A pilot study including 51 patients with proximal MCA occlusion showed significantly higher S-100B levels in patients with cerebral herniation or treatment with decompressive hemicraniectomy. S-100B levels 24 h after symptom onset >1.03 µg/l predicted a malignant infarct with a specificity of 83%. 14 While pre-selection of patients with proximal MCA occlusion and different definitions limit a direct comparison with this study, our findings confirm the previously hypothesized association between S-100B and brain edema.
Aside from S-100B, investigators have strived to predict both intracranial hemorrhage 12 and brain edema 14 using other blood biomarkers associated with BBB disruption including matrix metalloprotease-9, 32 glial fibrillary acidic protein, 33 neuron-specific enolase, 12 and vascular endothelial growth factor. 12 However, external validation in large studies including unselected AIS patients is lacking, thus it remains unclear which biomarkers may be most useful in clinical practice.
The extent of BBB permeability can also be estimated using novel modalities in neuroimaging. In a study including 106 patients with M1 or ICA occlusion treated with IAT, the contrast volume transfer coefficient (Ktrans) at ideal cut-off showed a sensitivity of 55% and a specificity of 68.2% for the prediction of hemorrhagic transformation. 34 Another study retrospectively assessed an automatically computed BBB permeability parameter from admission CT scans and aimed to predict either sBE or sICH in 32 AIS patients regardless of therapeutic interventions. 35 It showed a sensitivity of 100% and a specificity of 79% for the development of either outcome. Despite these promising results, an inherent disadvantage of advanced radiological examinations is the need for specialized neuroradiologists usually only available in hospitals involved in tertiary care. Blood-based biomarkers, on the other hand, are easily measurable and their interpretation does not require special training.
In attempt to predict the development of sICH in the early stages of stroke, numerous scores (HAT Score, SPAN-100 Score, SEDAN Score, SITS Score, GRASPS Score, Multicenter Stroke Survey) were evaluated specifically for the risk of sICH in the subgroup of patients treated with IVT. A previous comparison these sICH scores reported AUC values from 0.55 to 0.70 versus 0.68 to 0.77 in the original studies. 4 After application of score items to our cohort, we found AUC values from 0.50 to 0.63, which might be lower due to a stricter definition of sICH – four of the scores did consider any neurological deterioration as sufficient for sICH diagnosis, whereas in our study an increase of NIHSS of ⩾4 points was required. However, despite various pathophysiological considerations behind the scores, log10S-100B led to significant increase of the AUC in every score (Table 3). Also, log10S-100B remained independently associated with sICH after adjustment for the other variables in every score. This underlines the validity of S-100B as a useful prognostic adjunct regarding the development of sICH.
Scores have also been developed for the assessment of malignant MCA infarction. The EDEMA Score was developed based on data from 222 AIS patients with severe stroke and showed a sensitivity of 0.73 and a specificity of 0.70 at a score of 3 for death with midline shift >5 mm or need for decompressive hemicraniectomy. 36 However, as radiological features of brain edema are items of the score, it is not predictive in nature. The DASH Score was developed in a study with 119 AIS patients with ICA- or M1-occlusion receiving MRI on admission and showed an AUC of 0.88 for the detection of malignant MCA infarction within 48 hours. 3 This high diagnostic accuracy likely resulted from the pre-selection of stroke patients at risk for stroke complications. Furthermore, the potentially missing availability of MRI within the first hours of stroke onset would probably be a limitation. Alberta Stroke Program Early CT score (ASPECTS) of 7 was shown to have a sensitivity of 50% and a specificity of 86% for malignant infarction in 226 patients with MCA infarction. 37
In summary, while several clinical and neuroradiological approaches exist to predict sICH and sBE, none of these scores included specific serum markers of BBB permeability. According to our findings, such markers may be a helpful addition in the early assessment of sICH and sBE risk.
This study has limitations. We only analyzed single measurements of S-100B, while concentrations are known to gradually increase 8–10 h after symptom onset and reaching maximum plasma concentrations after 2–4 days.10,14,38 Biomarker trajectories created from repeated measurements may contain additional information regarding the development and extent of BBB dysfunction and therefore the risk of subsequent sBE or sICH. However, risk-stratification regarding time-sensitive therapy needs to be available in the hyperacute phase of stroke, therefore measurements beyond 24 h are unlikely to be useful for clinical application.
Second, control neuroimaging was only performed in in 90.3% of all cases. Per protocol, control imaging was systematically performed if patients showed clinical deterioration or if they had received any kind of reperfusion therapy. This approach was generally agreed to be feasible and sensible, as ICH or BE in patients who are clinically stable would not lead to any intervention.
Both brain edema and intracranial hemorrhage are more likely to occur with increased infarction size39,40 and higher S-100B levels were shown to correlate with final infarction volumes.9,10 In clinical practice, infarct size often cannot be correctly determined in the early phase of stroke as this would require initial MRI imaging in all patients, which may not routinely be available. Thus, lesion size may not be a feasible marker for very early risk-stratification in most hospitals. Further, blood draw was performed within 24 h of symptom onset – in some cases after initial diagnostic and therapeutic measures. Therefore, this study did not evaluate S-100B as a pre-treatment risk assessment tool. Lastly, 97.3% of the included patients were Caucasian, limiting the generalizability of our results to other ethnic groups.
In conclusion, we found that S-100B levels are significantly and independently associated with clinically important complications after acute ischemic stroke. Our data add to the preliminary evidence that S-100B levels may assist in improving risk prediction and in selecting either vulnerable individuals for intensified monitoring or stable patients for earlier discharge to a normal ward or rehabilitation facility. Further studies are required to elaborate effectiveness of measuring S-100B concerning improved clinical decision-making.
Supplemental Material
sj-docx-1-eso-10.1177_23969873221145391 – Supplemental material for Serum S-100B adds incremental value for the prediction of symptomatic intracranial hemorrhage and brain edema after acute ischemic stroke
Supplemental material, sj-docx-1-eso-10.1177_23969873221145391 for Serum S-100B adds incremental value for the prediction of symptomatic intracranial hemorrhage and brain edema after acute ischemic stroke by Tim Honegger, Juliane Schweizer, Antonela Bicvic, Laura P Westphal, Valerie Schütz, Corinne Inauen, Thomas Pokorny, Katja Bracher, Marcel Arnold, Urs Fischer, Leo H Bonati, Gian Marco De Marchis, Krassen Nedeltchev, Timo Kahles, Carlo Cereda, Georg Kägi, Joan Montaner, Alejandro Bustamante, Elena Palà, George Ntaios, Christian Foerch, Andreas Luft, Katharina Spanaus, Lanja Saleh, Arnold von Eckardstein, Markus Arnold and Mira Katan in European Stroke Journal
Supplemental Material
sj-docx-2-eso-10.1177_23969873221145391 – Supplemental material for Serum S-100B adds incremental value for the prediction of symptomatic intracranial hemorrhage and brain edema after acute ischemic stroke
Supplemental material, sj-docx-2-eso-10.1177_23969873221145391 for Serum S-100B adds incremental value for the prediction of symptomatic intracranial hemorrhage and brain edema after acute ischemic stroke by Tim Honegger, Juliane Schweizer, Antonela Bicvic, Laura P Westphal, Valerie Schütz, Corinne Inauen, Thomas Pokorny, Katja Bracher, Marcel Arnold, Urs Fischer, Leo H Bonati, Gian Marco De Marchis, Krassen Nedeltchev, Timo Kahles, Carlo Cereda, Georg Kägi, Joan Montaner, Alejandro Bustamante, Elena Palà, George Ntaios, Christian Foerch, Andreas Luft, Katharina Spanaus, Lanja Saleh, Arnold von Eckardstein, Markus Arnold and Mira Katan in European Stroke Journal
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article:
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported with research grants from the Swiss National Science Foundation (142422), the Swiss Heart Foundation, the Göhner Foundation and the Swiss Seaside Foundation.
Informed consent
Written informed consent was obtained from all patients or their welfare guardians for the collection blood samples and clinical data for further analysis.
Ethical approval
Ethical approval for this study was obtained from the ethics committees of the Canton of Zurich, Switzerland (BASEC No. PB_2016-00672, KEK-ZH-No. 2014-0001), of the Larissa University Hospital, Larissa, Greece (No. 46748), of the University Hospital Vall d’Hebron in Barcelona, Spain (No. 46228895E) and of the Goethe University Frankfurt am Main, Germany (No. 478/14).
Guarantor
MK
Contributorship
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
