Abstract
Introduction:
Migraine with aura (MA) accounts for up to 10% of “stroke mimics” and can present cerebral perfusion abnormalities. We aimed to compare perfusion-CT (PCT) findings in acute-onset MA mimicking an ischemic stroke with those observed in transient ischemic attack (TIA).
Methods:
We retrospectively studied patients admitted to our hospital between 2002 and 2014 with suspicion of acute ischemic stroke, having PCT and receiving a final diagnosis of MA. We visually assessed PCT for the presence and extent of focal hypoperfusion (FHP). MA patients with FHP were compared with consecutive TIA patients showing FHP. We performed both qualitative and quantitative analysis of PCT.
Results:
Of 47 patients with MA (median age = 33 years, 55% females), 16 (34%) displayed FHP. Compared to MA patients without FHP, MA patients with FHP had similar headaches and aura features, but a less frequent history of MA (p = 0.010). Compared to 74 TIA patients with FHP (median age = 69 years, 43% females), MA patients with FHP showed hypoperfusion that more frequently involved adjacent vascular territories or a whole hemisphere (p < 0.001). In addition, hypoperfusion in MA patients had a less pronounced increase in rMTT (1.2 vs 1.8, p < 0.001) and rTTP (1.1 vs 1.2, p < 0.001), and a lesser decrease in rCBF (0.8 vs 0.6, p < 0.001) compared to hypoperfusion in TIA. rMTT displayed the best discriminative ability to differentiate MA from TIA.
Conclusion:
Focal perfusion abnormalities in acute MA often involve adjacent vascular territories and hypoperfusion is less pronounced than in TIA. MA can be best differentiated from TIA by a smaller rMTT increase.
Introduction
Between 14% and 45% of patients with suspected acute ischemic stroke are affected by a non-stroke condition having a similar clinical presentation (stroke-mimics).1,2 After seizures and psychiatric disorders, migraine with aura (MA) is the third most common cause of such a condition, accounting for up to 10% of stroke mimics.1,2 Indeed, MA is responsible for about 18% of all erroneous thrombolytic treatments. 2
MA is defined as visual, auditory, somatosensory, or motor symptoms associated with migraine attacks, characterized by their gradual evolution (usually over 5–20 min) and a maximum duration of 60 min for each individual symptom. 3 The cortical spreading depression characteristic of MA is usually accompanied by a short-lasting cerebral hyperperfusion of about 1–2 min and a subsequent mild hypoperfusion of about 1–2 h. 4
In the emergency room (ER), differential diagnosis between MA and acute ischemic stroke (AIS) or transient ischemic attack (TIA) can be challenging. Improving the diagnostic accuracy is important, in order to avoid inappropriate therapies such as IV thrombolysis or unnecessary work-up and tertiary prevention for cerebrovascular diseases.
In the acute assessment of AIS and TIA patients, perfusion-CT (PCT) imaging is adopted widely and becoming a validated tool for identification of patients with good potential for acute recanalization therapies.5,6 However, the role of PCT in the acute evaluation of patients with MA has only been explored in single patient reports or small case series.7,8
The aim of our study is to assess the frequency of focal hypoperfusion (FHP) on PCT and the variables associated in a consecutive series of patients presenting with a clinical suspicion of AIS or TIA who underwent PCT and who received a final diagnosis of MA. In addition, we aimed to compare the characteristics of FHP in MA patients and in a control group of TIA patients with FHP.
Methods
Patient selection
We did a retrospective study including all patients admitted to the ER of our university hospital between 2002 and 2014 with clinical suspicion of AIS or TIA, who underwent acute PCT and who received a final diagnosis of event compatible with migraine attack with aura (MA). Included patients either fulfilled the International Classification of Headache Disorders (ICHD) criteria for migraine with aura, 3 or missed the criterion A only, that is presented with a first episode fulfilling all other criteria of migraine with aura, which was considered the most likely pathophysiological process according to a board-certified neurologist. In all cases the final diagnosis of MA was based on repeat history taking by board certified neurologists and follow-up MRI performed after the initial presentation, beyond the initial 24 h after symptom onset.
The inclusion criteria of the present study were: MA with hemispheric/lateralized symptoms and without symptoms suggestive of a migraine with brainstem aura (such as severe vertigo or diplopia); and the availability of a good quality PCT performed within 24 h of symptoms onset.
For each included patient we collected: demographics, vascular risk factors, personal history of headache, family history of migraine, neurological symptoms/signs, duration of neurological deficits and characteristics of migraine. We distinguished the modality of symptoms onset into (1) acute; (2) progressive topographical spreading of the initial symptoms, for example sensory deficit extending from the face to the arm; (3) sequential: sequence of different symptoms appearing one after the other (e.g. initial visual symptoms followed by the onset of aphasia). We recorded if symptoms were present during neuroimaging and, if not, the time between the end of the symptoms and PCT.
As control group, we identified all consecutive patients admitted to the ER of our hospital for hemispheric TIA in the same study period who underwent PCT within 24 h of symptoms onset, and who showed focal hypoperfusion (FHP) on PCT.
No informed consent nor local ethical committee approval was required for retrospective analysis of anonymized data according to the legislation of the Canton de Vaud, Switzerland.
Neuroimaging protocol and processing
In our institution, cerebral PCT following non-contrast CT (NCCT) and CT angiography of the head and neck is performed as part of the emergency diagnostic workup for patients with a clinical suspicion of AIS or TIA. We carried out cerebral CT on a 16-multidetector CT scanner (LightSpeed, GE Healthcare, Milwaukee, WI, USA) until November 2005 and on a 64-multidetector CT scanner (LightSpeed VCT, GE Healthcare, Milwaukee, WI, USA) thereafter. We acquired all PCT series in axial scan mode with 80 kV peak tube voltage, 240 mA tube current, 32 cm SFOV and 512 × 512 matrix. PCT series were positioned at the level of the basal ganglia and the third ventricle, above the orbits in order to protect the lens. We used 18 groups of 4 slices of 10 mm (40 mm z-axis coverage) until November 2005, and 18 groups of 16 slices of 5 mm (80 mm z-axis coverage) thereafter.
PCT raw data were reconstructed using FBP until February 2009 and using a blend of 40% ASiR with 60% FBP thereafter. We then analyzed PCT data using the Brilliance Workspace Portal® (Philips Medical Systems, Cleveland, OH, USA), based on the central volume principle. We used deconvolution to create parametric maps of time to peak (TTP), mean transit time (MTT); we calculated cerebral blood volume (CBV) maps from the area under the time-enhancement curves and cerebral blood flow (CBF) was derived from the formula CBF = CBV/MTT.
We defined FHP as any area that presented MTT increase or CBF decrease compared to contralateral mirror area on visual assessments, visible on ⩾2 consecutive slices and not attributable to an underlying chronic tissue lesion (as shown in the two exemplary cases in Figure 1). A vascular neurologist and a neuroradiologist blinded to the clinical symptoms independently analyzed PCT for qualitative analysis, scoring FHP as absent, focal, single vascular territory, adjacent territories or hemispheric. On a subsample of 40 patients, we calculated inter-rater agreement, which was substantial according to the Prevalence and Bias Adjusted Kappa of 0.69, similar to previous studies. 9 For patients showing FHP, we measured the ratio of PCT parameters (TTP, MTT, CBV, and CBF) between the visibly hypoperfused region and the contralateral side. A neuroradiologist with 12 years of experience and blinded to the clinical diagnosis manually delineated apparent FHP areas on all consecutive slices. The region-of-interest was automatically copied and pasted onto all parameter maps, then mirrored relative to the sagittal plane to calculate the ratio. Two exemplary cases of migraine and TIA, respectively are displayed in Figure 1.
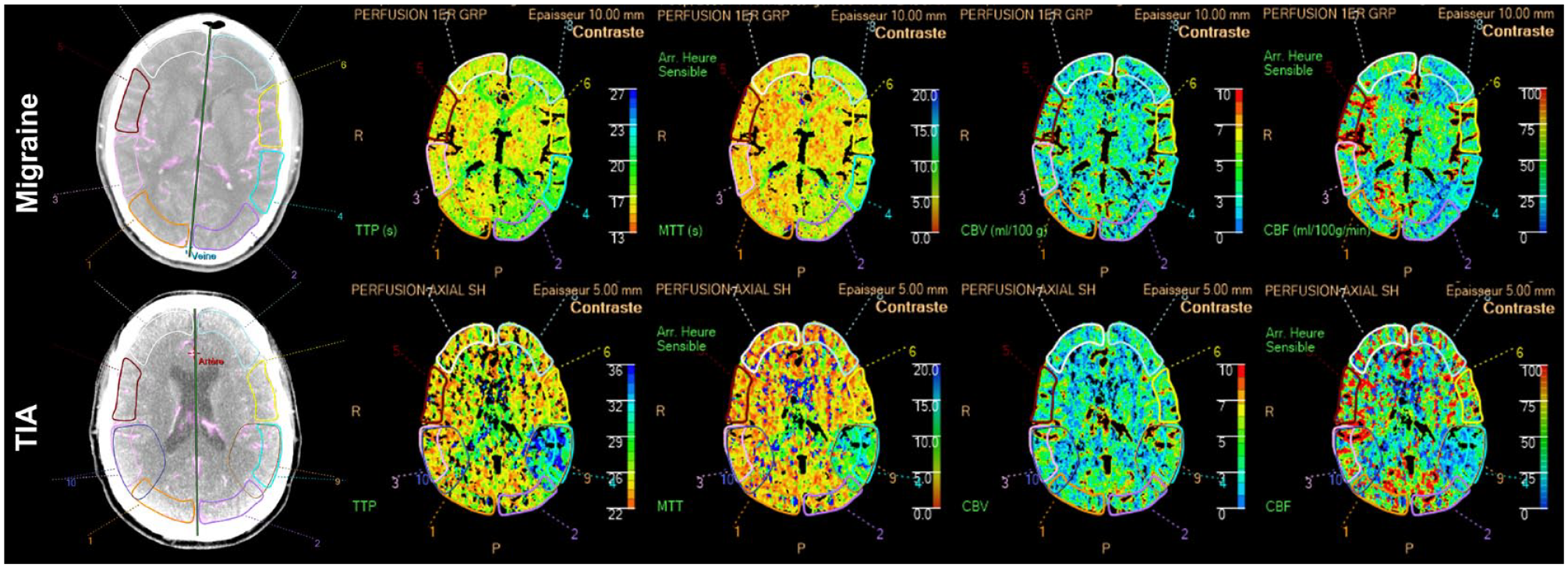
Two exemplary cases of perfusion CT analysis in a patient with migraine (upper panels) and in a patient with a TIA (lower panels). Raw images, time to peak, mean transit time, cerebral blood volume, and cerebral blood flow maps are displayed.
Statistical analysis
Continuous data are presented as median value and interquartile range (IQR), categorical data as absolute numbers and percentages and quantitative data from PCT as means and standard deviation.
First, we compared MA patients with and without FHP on acute PCT, using descriptive statistics and univariate analysis. Second, we compared the subgroup of MA patients with FHP with the control group of patients with hemispheric TIA and FHP. Group comparisons were performed with Fisher’s exact test (for categorical variables) and Kruskal-Wallis test (for numerical variables), considering significant p-values of <0.05. For PCT parameters showing significant differences between MA and TIA patients with FHP, we performed ROC curve analyses with computation of respective AUCs (95% CI). We used Liu method to identify the PCT values with highest sensitivity and specificity to correctly diagnose MA.
We performed statistical analysis with R statistical software (version 3.3.2, R Core Team [2016], R Foundation for Statistical Computing, Vienna, Austria).
This observational study is reported in accordance with the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines and with the Standards for Reporting Diagnostic accuracy studies (STARD) guidelines.
Data availability statement
Anonymized participant data are available upon reasonable request that includes a data analysis, authorship, and publication plan.
Results
Characteristics of MA patients with and without FHP
During the study period, we included 47 patients with MA having a median age of 33 years and 55% of whom were females. Sixteen (34%) patients displayed FHP on PCT. Compared to MA patients without FHP, MA patients with FHP had similar clinical features of headache and aura, similar onset modality (acute, progressive, or sequential), but less frequently presented a past history of MA (1/16[6.2%] vs 14/31[45.2%], p = 0.010), with a similar frequency of previous episodes of migraine without aura, tension type headache or aura without headache (Table 1). Interestingly, not all patients where hypoperfusion involved the posterior cerebral artery (PCA) territory had visual symptoms during the aura, which could indicate asymptomatic aura phenomena (Among 14 patients with hypoperfusion involving the PCA territory, 5 [35%] had no visual symptoms).
Clinical features of patients with migraine with aura (MA) with and without focal hypoperfusion (FHP) on perfusion-CT (PCT).
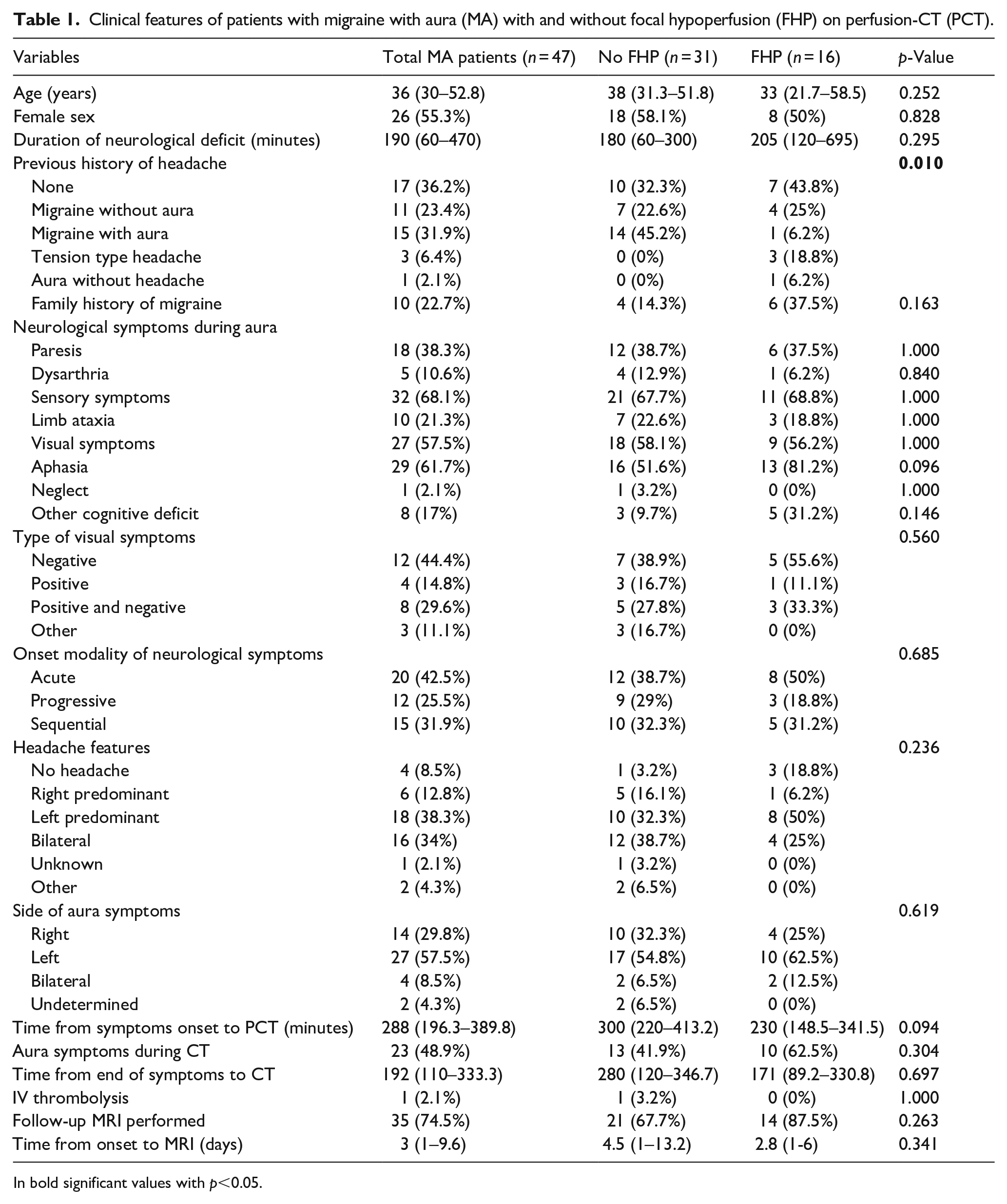
In bold significant values with p<0.05.
Comparison of MA and TIA patients with FHP
As a control group, we analyzed 74 TIA patients with FHP (median age = 69 years, 43% females). Compared with TIA patients, MA patients with FHP were younger and had a lower level of vascular risk factors (Table 2). MA patients presented a shorter duration of neurological deficits (mean 120 vs 205 min, p = 0.049), had more often a clinical presentation involving sensibility alteration (69% vs 34%, p = 0.021) or visual symptoms (56% vs 16%, p = 0.002) and less frequently dysarthria (6% vs 41%, p = 0.020). Neglect was rare in both conditions.
Clinical and radiological features of patients with migraine with aura (MA) and focal hypoperfusion (FHP) on perfusion CT and of patients with transient ischemic attack (TIA) and FHP.
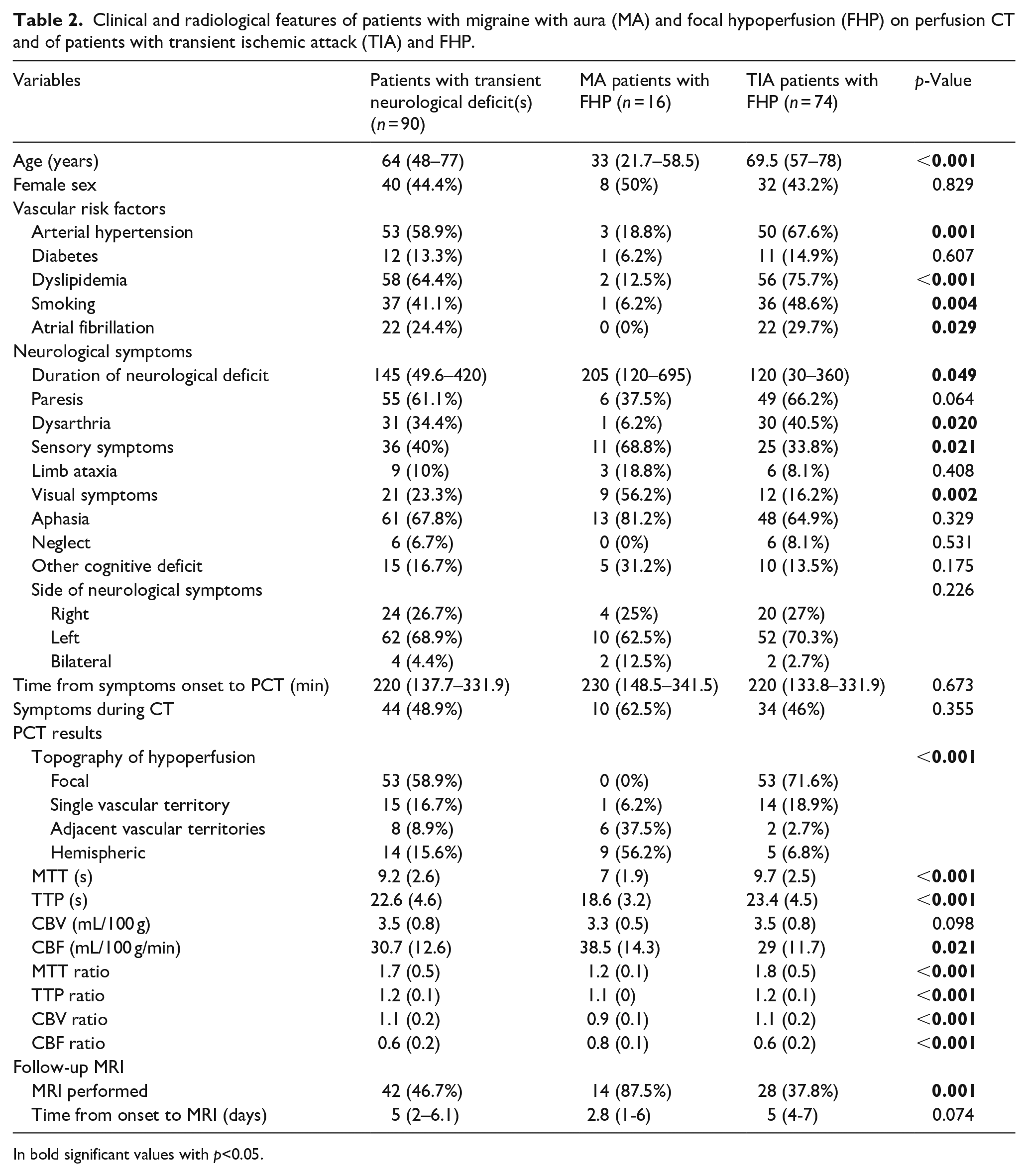
In bold significant values with p<0.05.
PCT was performed at a similar time point in MA and TIA patients. Focal hypoperfusion in MA patients more frequently involved adjacent arterial territories (38% vs 3%) or a whole hemisphere (56% vs 7%) (p < 0.001). The qualitative analysis of PCT capable of distinguishing focal or single-territory hypoperfusion versus adjacent-territory or hemispheric hypoperfusion showed an AUC of 0.92 (95% CI 0.85–0.99), with 90% sensitivity and 94% specificity.
On investigation of single PCT parameters, MA patients had a less pronounced increase in rMTT (1.2 vs 1.8, p < 0.001) and rTTP (1.1 vs 1.2, p < 0.001), and a lesser decrease in rCBF (0.8 vs 0.6, p < 0.001) compared with TIA patients. rCBV was slightly reduced in MA and slightly augmented in TIA (0.9 vs 1.1 respectively, p < 0.001) (Table 2).
From ROC curve analyses of PCT parameters, we found that rMTT displayed the best discriminative ability for differentiating MA from TIA (AUC 0.88, 95% CI 0.81–0.95) (Figure 2). We identified an optimal rMTT cut-off of <1.32, which had a sensitivity of 93% and a specificity of 82% for distinguishing FHP in MA from that of TIA.
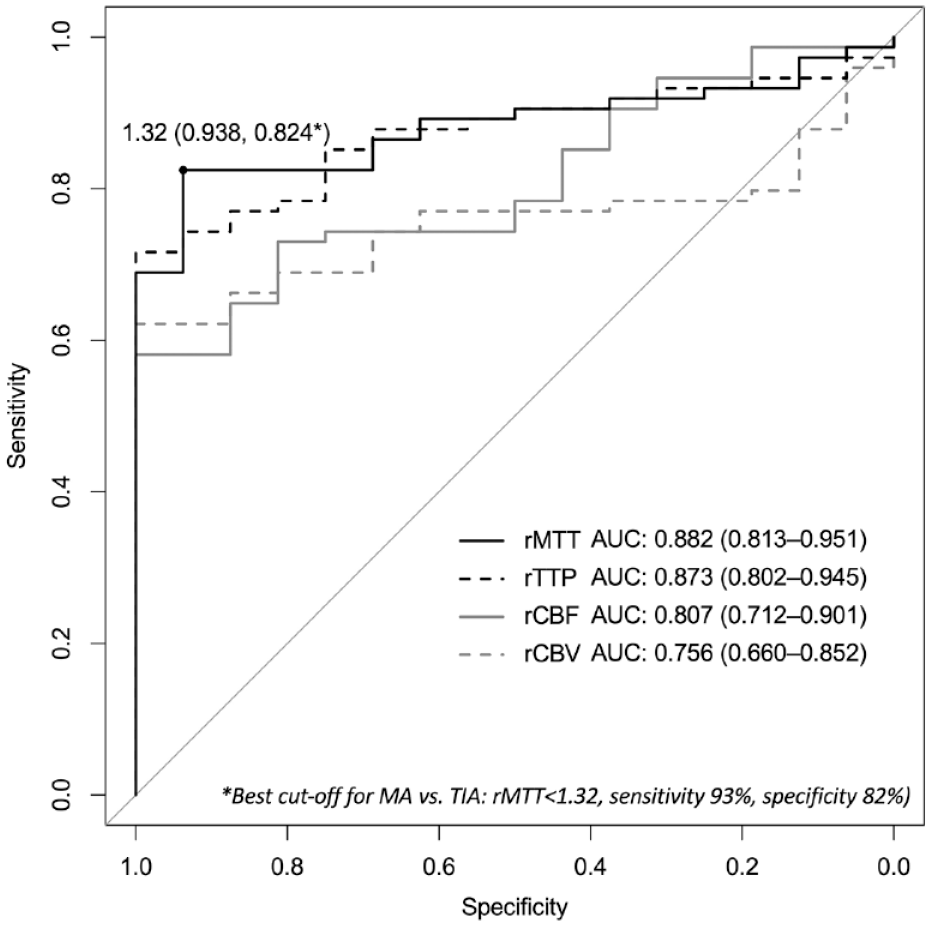
Receiver operating characteristic (ROC) curves for different PCT parameters to differentiate migraine aura from TIA.
Combining both qualitative and quantitative PCT data led to an AUC = 0.97, 95% CI 0.93–0.99), not significantly different from the AUC of the quantitative or qualitative data alone (De-Long tests, p > 0.05). After the addition of quantitative data, two patients were correctly reclassified as TIA from migraine and one patient incorrectly reclassified as migraine from TIA.
Discussion
In our retrospective study of MA patients with acute-onset hemispheric/lateralized neurological deficits who underwent PCT, we found focal hypoperfusion in one-third of patients. Compared with PCT findings in patients with TIA, focal hypoperfusion in MA involved adjacent arterial territories more frequently and was less severe. The analysis of single PCT parameters showed that the smaller increase in rMTT best differentiated MA from TIA.
The occurrence of cerebral hypoperfusion during MA is a known phenomenon, already described in SPECT and MRI studies. 10 On the other hand, the ability of PCT to detect such perfusion abnormality has mainly been described in single-case reports,11,12 and only in one case series. 8 In the latter, a quantitative assessment of PCT maps was adopted, showing perfusion abnormalities in 13/14 patients with MA, with the most frequently altered parameter being MTT. The observed perfusion changes were above the CBV threshold for ischemic core and below the MTT threshold for penumbra. This is in line with our finding of less marked perfusion changes in MA than in TIA patients. Of note, the time from aura symptoms onset and resolution and the presence of these symptoms at the time of imaging execution might affect the chance of detecting perfusion abnormality. Accordingly, in patients with FHP we observed a shorter delay from aura symptoms onset and resolutions to imaging and a higher frequency of symptom at the time of imaging. However, these differences were not statistically significant, possibly for the limited sample size, not allowing us to draw definite conclusion on the relationship between aura temporal evolution and prevalence of FHP.
Our results are also consistent with previous MRI-based studies showing that hypoperfusion in almost all cases of MA involved adjacent vascular territories,13,14 and was associated with lower TTP and MTT ratios compared to patients with AIS. 14 In addition, we demonstrated that the CBV ratio was significantly lower and the CBF ratio significantly higher in comparison to patients with TIA. These findings suggest that in migraine with aura, hypoperfusion is generally less severe and does not fall below a critical limit defining ischemic lesion.
Our research group already studied hypoperfusion characteristics in patients with TIA who underwent PCT, 15 and we previously demonstrated that they might be useful in differential diagnosis from other conditions, including transient global amnesia or posterior reversible encephalopathy syndrome.16,17 Similarly, in the present study, we suggest that a CTP-quantitative analysis might detect a moderate hypoperfusion pattern in the cerebral regions during MA, compatible with aura symptoms, which could help in distinguishing from TIA.
The clinical implications of our study are related to the potential role of PCT in the management of patients presenting with acute-onset focal neurological deficits. The differential diagnosis between MA and AIS can be clinically challenging, even more so with TIA given that TIA may be characterized by transient symptoms and absence of ischemic lesion on neuroimaging. Furthermore, focal hypoperfusion on PCT or PWI in MA patients could also be treated as ischemia, in particular if the above-described MA-specific features are not considered. This challenging diagnosis in the emergency setting is further complicated by the fact that migraine patients may themselves experience a cerebrovascular accident, which could be misinterpreted as an MA attack (i.e. stroke chameleons).
Even if the hemorrhagic risk for rtPA administered to patients with stroke mimic is low, and there is no report of post-rtPA intracerebral hemorrhage in patients with MA, the misdiagnosis of MA as a neurovascular event remains a non-negligeable matter. 2 Awareness of PCT characteristics can help to distinguish properly migraine with aura from cerebrovascular diseases and to avoid unnecessary procedures and treatments in patients showing perfusion deficit involving adjacent vascular territories and with only a moderate increase in MTT. On the contrary, hypoperfusion restricted to a single vascular territory in combination with a marked increase in MTT should be regarded as atypical for migraine aura and suggestive rather of TIA. This information may lead to a correct diagnosis based on clinical evaluation and CTP data, without the need of additional imaging such as brain-MRI.
Limitations of our study include the retrospective design and its moderate sample size; however, to our knowledge, this is the largest series of patients investigating perfusion abnormalities in MA compared with transient ischemic attack. Still, the limited number of included patients did not allow us to perform adjusted statistical analyses. Some patients in the MA group did not fulfill all ICHD criteria of migraine with aura as they had no previous attacks of MA. In these cases, the diagnosis was based on the evaluation by a board-certified neurologist who considered MA the most likely pathophysiological process to explain the event. Also, as perfusion imaging was carried out as part of clinical practice, no additional perfusion imaging was obtained that would have allowed studying the natural course of FHP. Of note, the decrease of brain transit time with aging should be considered as a potential confounder when comparing perfusion parameters between two population with large age difference such as MA and TIA (36 vs 64 years respectively in our study). We likely minimized this effect since we used the ratio of PCT parameters between the visibly hypoperfused region and the contralateral side.
In conclusion, in our collection of patients with migraine with aura assessed by PCT, we were able to identify a characteristic perfusion pattern with areas of hypoperfusion often affecting adjacent vascular territories and showing a lower increase in MTT ratio compared to transient ischemic attack. Knowledge of specific PCT findings in migraine with aura could be useful in the acute management and correct identification of these patients. Further prospective studies examining the role of emergency PCT in reducing the misdiagnosis of stroke mimics, including migraine with aura, are strongly welcomed.
Footnotes
Acknowledgements
We thank Melanie Price Hirt for English language correction and editing.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: D. Strambo: Research grants from the Swiss Heart Foundation and the University of Lausanne (not related to this work). V. Dunet: Research grant from the Swiss National Science Foundation (not related to this work). P. Michel: Research grants from the Swiss National Science Foundation and Swiss Heart Foundation; consulting fees from Medtronic (all used for research and education). The other authors declare no conflict of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Swiss Heart Foundation.
Ethical approval and informed consent
No informed consent nor local ethical committee approval was required for retrospective analysis of anonymized data according to the legislation of the Canton de Vaud, Switzerland.
Guarantor
DS
Contributorship
DS: clinical data acquisition, statistical analysis and interpretation and draft of the manuscript. SN: study conception and design, clinical data acquisition, and draft of the manuscript. LR: clinical data acquisition, critical revision of the manuscript. VD: radiological data acquisition, critical revision of the manuscript. PM: study conception and design, data acquisition, statistical analysis and interpretation and draft of the manuscript.
