Abstract
Background:
Data on the safety and effectiveness of once-daily (QD) versus twice-daily (BID) direct oral anticoagulants (DOAC) in comparison to vitamin K antagonists (VKA) and to one another in patients with atrial fibrillation (AF) and recent stroke are scarce.
Patients and methods:
Based on prospectively obtained data from the observational registry Novel-Oral-Anticoagulants-in-Ischemic-Stroke-Patients(NOACISP)-LONGTERM (NCT03826927) from Basel, Switzerland, we compared the occurrence of the primary outcome – the composite of recurrent ischemic stroke, major bleeding, and all-cause death – among consecutive AF patients treated with either VKA, QD DOAC, or BID DOAC following a recent stroke using Cox proportional hazards regression including adjustment for potential confounders.
Results:
We analyzed 956 patients (median age 80 years, 46% female), of whom 128 received VKA (13.4%), 264 QD DOAC (27.6%), and 564 BID DOAC (59%). Over a total follow-up of 1596 patient-years, both QD DOAC and BID DOAC showed a lower hazard for the composite outcome compared to VKA (adjusted HR [95% CI] 0.69 [0.48, 1.01] and 0.66 [0.47, 0.91], respectively). Upon direct comparison, the hazard for the composite outcome did not differ between patients treated with QD versus BID DOAC (adjusted HR [95% CI] 0.94 [0.70, 1.26]). Secondary analyses focusing on the individual components of the composite outcome revealed no clear differences in the risk-benefit profile of QD versus BID DOAC.
Discussion and conclusion:
The overall benefit of DOAC over VKA seems to apply to both QD and BID DOAC in AF patients with a recent stroke, without clear evidence that one DOAC dosing regimen is more advantageous than the other.
Introduction
For decades, vitamin K antagonists (VKA) were the mainstay of ischemic stroke prevention in patients with atrial fibrillation (AF). 1 This has changed with the introduction of the direct oral anticoagulants (DOAC) rivaroxaban, dabigatran, apixaban, and edoxaban, which were shown in large randomized clinical trials to be at least as effective as VKA while bearing a lower risk for intracranial hemorrhage (ICH). 2 Thus, DOAC are recommended in preference over VKA in the current guidelines,3,4 supported by additional observational evidence in their favor.5–7 However, since no direct comparisons between DOAC have been conducted, no randomized evidence exists on which the choice of a specific DOAC over another can be based. In this situation, systematically ascertained, standardized observational data – that is, “real-world” data – may be useful. 8
One differentiating factor among the DOAC is the dosing regimen, that is, once-daily (QD; rivaroxaban and edoxaban) and twice-daily dosing (BID; dabigatran and apixaban). 2 According to model-based simulations applied from other medications and based on the DOAC’ pharmacokinetic properties, BID DOAC might be expected to maintain a better continuity of drug plasma levels with smaller peak-to-trough variability than QD DOAC. It has been postulated that this might translate to a lower risk of bleeding and thrombotic events, and that BID DOAC might be “more forgiving” in the presence of suboptimal drug adherence. 9 On the other hand, several observational studies demonstrated a lower adherence to BID than QD DOAC,10–13 but others reported contradictory results. 14 For AF patients with a recent stroke, who are at high risk for both recurrence 15 and ICH,16,17 as well as non-adherence due to neurological and cognitive deficits, 18 any such differences influencing the outcomes of treatment with QD versus BID DOAC could be crucial.
With these considerations in mind, we investigated the safety and effectiveness of BID and QD DOAC compared to VKA and to one another in patients with AF and a recent ischemic stroke. In the absence of randomized data, we used prospectively collected data from a registry about the use of oral anticoagulants (OAC) in patients with AF following ischemic stroke from a certified Stroke Center.
Patients and methods
Study design, patient population, and data collection
We used prospectively collected individual patient data from the observational registry “Novel Oral Anticoagulants in Ischemic Stroke Patients (NOACISP)-LONGTERM” (NCT03826927). From April 2013 to December 2020, NOACISP-LONGTERM enrolled consecutive AF patients with an index acute ischemic stroke (defined as acute focal neurological deficits with a corresponding finding on neuroimaging) or transient ischemic attack (TIA; defined as acute focal neurological deficits of presumed ischemic origin lasting <24 h and without a corresponding finding on neuroimaging), as described previously.5–7,19–21 The choice of the OAC type in individual patients was at the discretion of the treating physicians and the patients. VKA therapy was defined as treatment with phenprocoumon, QD DOAC therapy as treatment with either rivaroxaban (15/20 mg) or edoxaban (30/60 mg), and BID DOAC therapy as treatment with apixaban (2.5/5 mg) or dabigatran (110/150 mg). Follow-up data were collected up to August 2021 at 3, 6, 12, and 24 months after the index event via telephone call, out-patient visit and/or from general practitioners’ and hospital records by trained study personnel using standardized electronic collection forms with predefined variables.5–7,19–21
In this study we included all consecutive NOACISP-LONGTERM patients with (i) an index recent (i.e., <3 months) ischemic stroke or TIA; (ii) nonvalvular AF (either known before index event or first diagnosed thereafter); (iii) treatment with VKA, QD, or BID DOAC, initiated within 3 months after the index event; and (iv) prospectively ascertained follow-up data for at least 3 months after the index event for the outcomes recurrent ischemic stroke, major bleeding, and all-cause death, occurring after OAC initiation and defined as reported previously.5–7 We excluded patients with valvular AF or mechanical heart valves, those without OAC treatment following the index event and patients missing follow-up.
We used the following variables from NOACISP-LONGTERM in line with prior research 5–7,19–21: Age, sex, type of index event (ischemic stroke or TIA), baseline National Institutes of Health Stroke Scale (NIHSS) score, AF known before index event, modified Rankin Scale (mRS) score before index event, time to OAC initiation after index event, concomitant antiplatelet use after index event, estimated glomerular filtration rate (eGFR; using the Cockcroft-Gault equation), history of ischemic stroke or TIA (prior to index event), history of ICH, history of major bleeding (including ICH), dyslipidemia, the CHA2DS2-VASc score (congestive heart failure, hypertension, age (65–74 or ⩾75 years), diabetes mellitus, stroke/TIA, vascular (coronary or peripheral artery) disease, sex) 15 and each of the score’s components separately. Information on medication administration (self-administration vs assisted by family or professional caregivers) was included post hoc.
Follow-up data included length of follow-up and absence or occurrence and timing of any of the following outcome events, which were defined in line with prior research5–7: (i) recurrent ischemic stroke (defined as new neurological deficits with a corresponding finding on neuroimaging); (ii) major bleeding (either major extracranial bleeding according to the ISTH criteria, 22 or ICH defined as new neurological deficits with detection of intracranial bleeding on neuroimaging); and (iii) all-cause death, defined as every death irrespectively of the cause and regardless of whether the cause was known or not.
Outcomes
The primary outcome was the time to occurrence of the composite of recurrent ischemic stroke, major bleeding, and all-cause death, in accordance with prior research.5–7 Secondary outcomes were the time to occurrence of each of these outcomes separately.
Statistical analysis
We categorized study patients in those with VKA, QD DOAC, or BID DOAC treatment. We presented their characteristics according to OAC type using frequencies and percentages for categorical data and the median and interquartile range (IQR) for continuous data. We compared categorical and continuous variables using the Chi-squared and Mann-Whitney U test, respectively. We calculated the annualized rate of outcome events as the total of observed events divided by patient-years of follow-up for each outcome.
As the main analysis, we modeled time to primary (composite) outcome using Cox proportional hazards regression in order to investigate the effect of OAC type on the primary outcome. For this, we analyzed time to first event without considering further events, and fitted both unadjusted models (with OAC type as the sole independent variable) and adjusted ones including not only OAC type, but also the following outcome predictors as independent variables in line with prior research,6,7,23 to account for imbalances in their distribution in the OAC subgroups: type of index event (ischemic stroke or TIA), NIHSS score, age, sex, CHA2DS2-VASc score (excluding the age and sex components, as in previously published research6,7), eGFR, history of major bleeding, time to OAC initiation after index event, AF known before index event, 24 and concomitant antiplatelet use. There were no missing values in any of the covariables used in the adjusted models except for NIHSS in 11 patients, eGFR in 22 patients, and time to OAC initiation in 1 patient, which we imputed with simple single imputation (using the respective median values of 4 points, 58.3 ml/min, and 4 days). In a post hoc analysis, we additionally adjusted our models for medication administration (missing in 95 patients, not imputed).
We fitted all models twice, (i) with OAC type as a categorical variable with three levels (QD DOAC, BID DOAC, and VKA, the latter as the reference group), and (ii) with OAC type as a dichotomous variable (BID DOAC and QD DOAC, the latter as the reference group) after excluding patients with VKA, in order to directly compare BID with QD DOAC.
For all models we report the model-based hazard ratio (HR) estimates along with the 95% confidence intervals (CI) and two-sided p-values. We present the composite outcome data in a Kaplan-Meier curve according to type of OAC.
As secondary analyses, we fitted the unadjusted and adjusted Cox models (i) and (ii) described above separately for the individual outcomes recurrent ischemic stroke, major bleeding, and all-cause death. As a sensitivity analysis, for the secondary outcomes recurrent ischemic stroke and major bleeding we additionally fitted unadjusted and adjusted Fine-Gray models 25 to account for death as a competing risk.
Statistical analyses were performed using Stata version 17.0 (StataCorp LLC, College Station, TX, USA).
We conducted this study in accordance with the STROBE Statement for observational studies. 26
Results
In total, 956 out of 1023 (93%) patients were eligible for analysis. Information on OAC type was complete. Only 26 patients were excluded for missing follow-up, amounting to a follow-up rate of 97% (study flowchart in Supplemental Figure 1).
Baseline characteristics
The index event was ischemic stroke in 853 patients (89%) and TIA in 103 (11%). The median (IQR) age was 80 (74–86) years and 442 patients (46%) were female. OAC was initiated at a median (IQR) of 4 (1–8) days after the index event with VKA in 128 patients (13%), QD DOAC in 264 (28%), and BID DOAC in 564 (59%).
Baseline characteristics of all study patients according to OAC type are displayed in Table 1. VKA-treated patients had more often concomitant antiplatelet use, coronary artery disease, heart failure, history of major bleeding and assisted medication administration, as well as lower eGFR and shorter time to OAC initiation than patients treated with QD and BID DOAC. Baseline characteristics were well balanced among patients with QD and BID DOAC, with the exception of known AF before stroke (more common in patients treated with BID DOAC and VKA) and ischemic stroke (as opposed to TIA) as the index event (more common in patients treated with BID DOAC).
Patient characteristics according to type of oral anticoagulant.
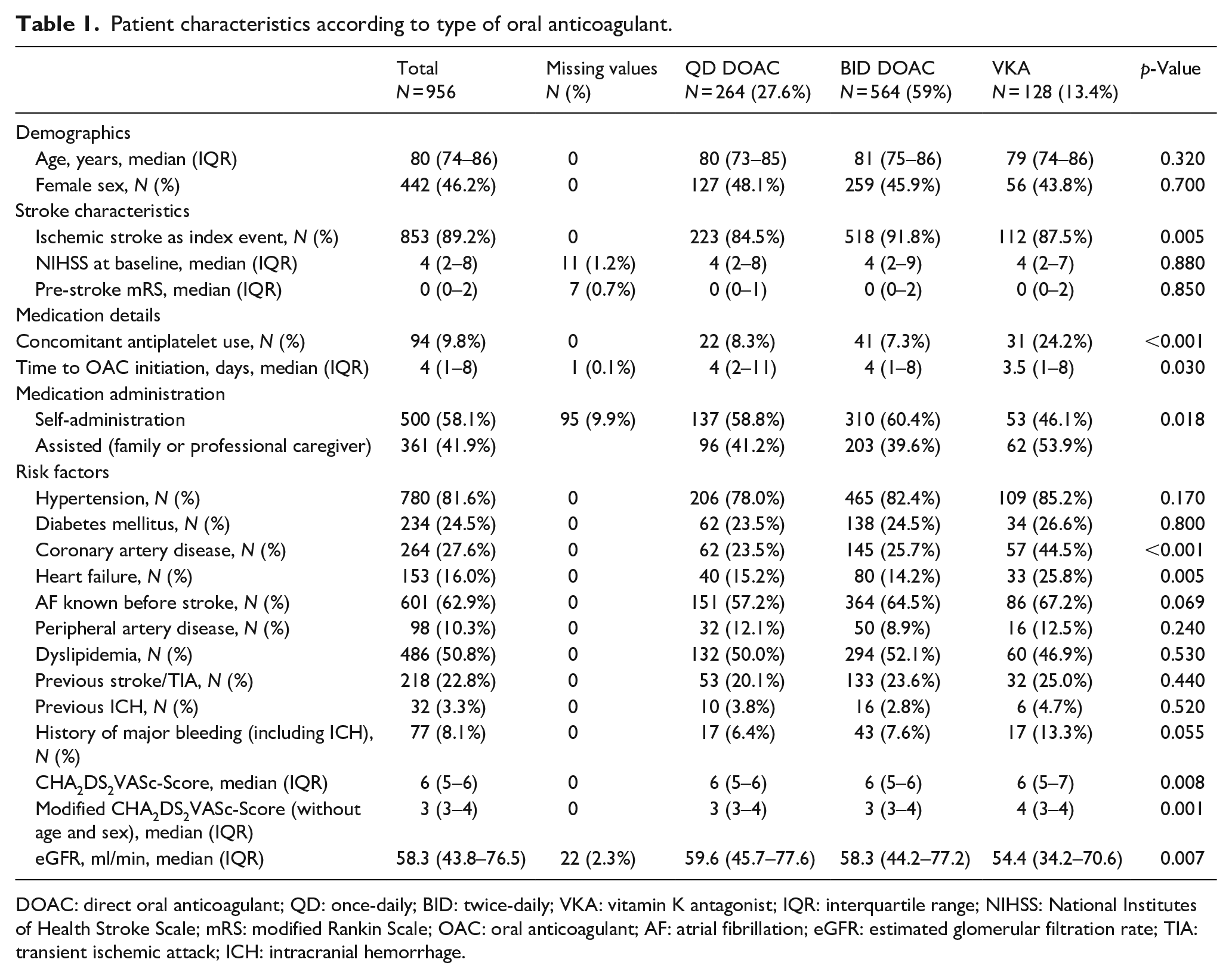
DOAC: direct oral anticoagulant; QD: once-daily; BID: twice-daily; VKA: vitamin K antagonist; IQR: interquartile range; NIHSS: National Institutes of Health Stroke Scale; mRS: modified Rankin Scale; OAC: oral anticoagulant; AF: atrial fibrillation; eGFR: estimated glomerular filtration rate; TIA: transient ischemic attack; ICH: intracranial hemorrhage.
Main analysis – primary composite outcome
During a total follow-up of 1,596 patient-years, 73 patients suffered recurrent ischemic stroke, 52 patients suffered major bleeding (36 major extracranial bleeding events and 18 ICH; two patients had both extracranial bleeding and ICH), and 173 patients died. This amounted to 265 patients suffering the primary (composite) outcome, a primary outcome event rate of 17.8%/year. The follow-up time, number, and crude rate of events for the primary outcome according to OAC type are given in Table 2. The Kaplan-Meier estimates for the composite outcome according to OAC type are presented in Figure 1.
Follow-up time, number, and crude rate of events for the primary and secondary outcomes.
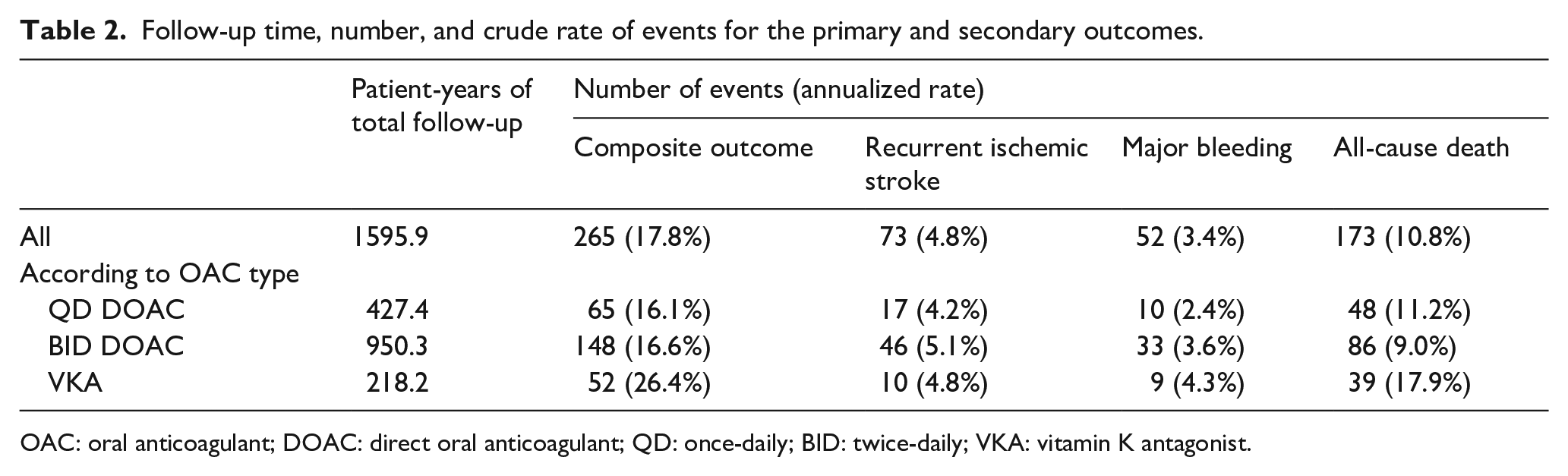
OAC: oral anticoagulant; DOAC: direct oral anticoagulant; QD: once-daily; BID: twice-daily; VKA: vitamin K antagonist.
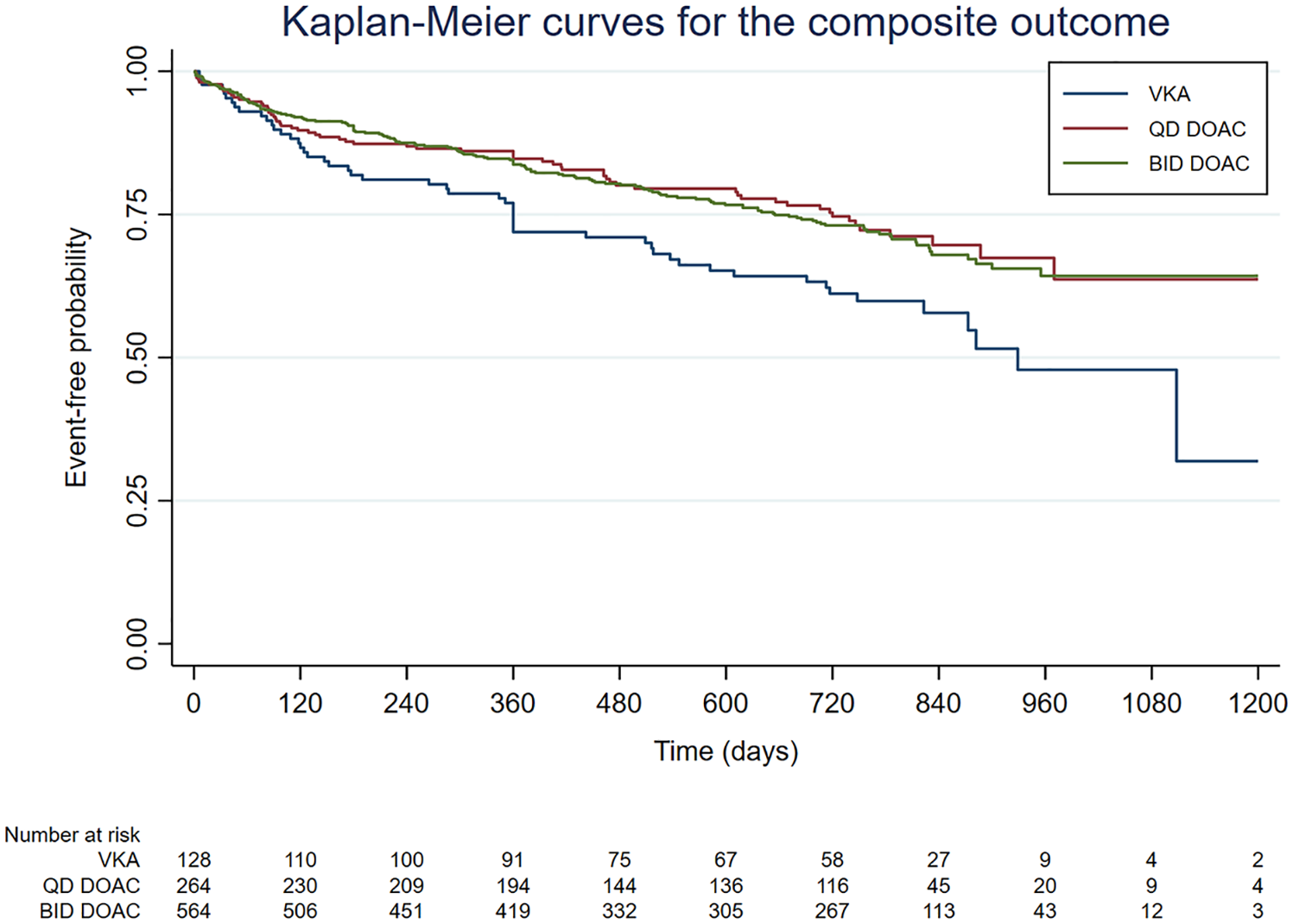
Kaplan-Meier curves for the primary (composite) outcome according to type of oral anticoagulant.
Both QD DOAC and BID DOAC were strongly associated with a lower hazard for the primary outcome compared to VKA in unadjusted Cox analysis. In the adjusted Cox model, BID DOAC retained their association with a lower hazard for the primary outcome compared to VKA. For QD DOAC, the association was weakened in the adjusted model, still showing a clear trend for a lower hazard for the primary outcome, but missing conventional statistical significance. Upon direct comparison, the hazard for the composite outcome did not differ between patients treated with QD versus BID DOAC. The detailed results of the main analysis for the composite outcome are presented in Table 3 and Figure 2. A post hoc analysis additionally adjusting for medication administration yielded consistent results (Supplemental Table 1). A post hoc power analysis indicated that the minimum effect of BID versus QD DOAC on the primary outcome that our unadjusted analyses could detect, would be a hazard ratio no larger than 0.66 and no smaller than 1.51 with a power of 80% at an alpha of 0.05.
Cox models for time to composite outcome.
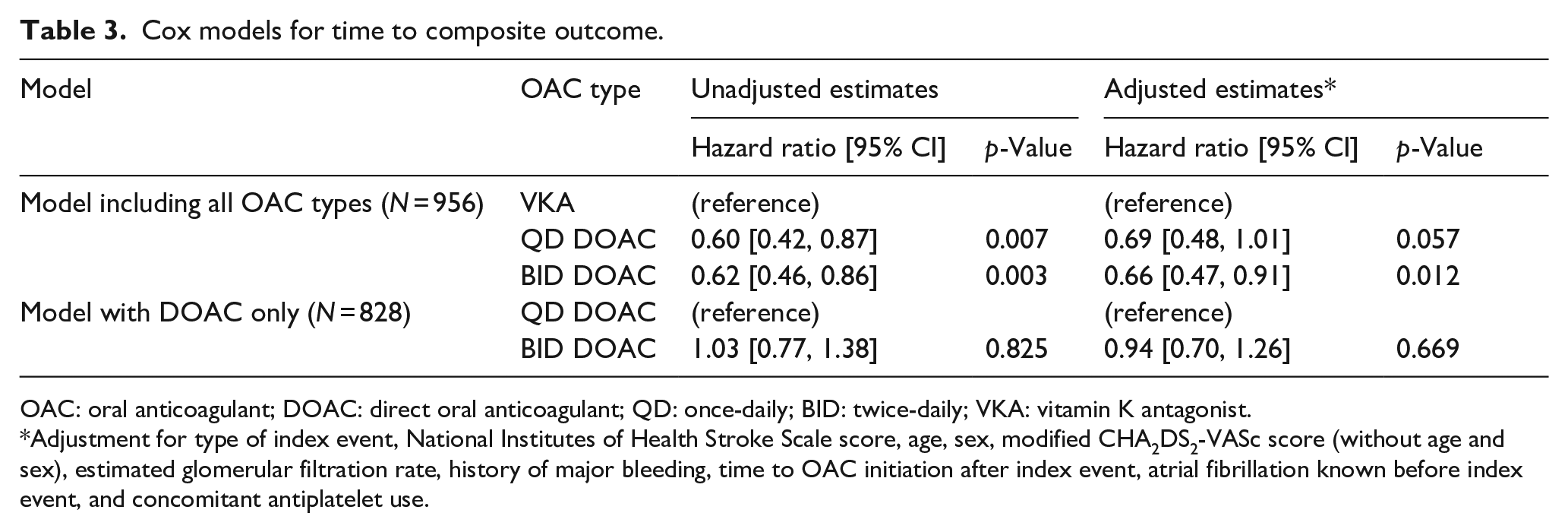
OAC: oral anticoagulant; DOAC: direct oral anticoagulant; QD: once-daily; BID: twice-daily; VKA: vitamin K antagonist.
Adjustment for type of index event, National Institutes of Health Stroke Scale score, age, sex, modified CHA2DS2-VASc score (without age and sex), estimated glomerular filtration rate, history of major bleeding, time to OAC initiation after index event, atrial fibrillation known before index event, and concomitant antiplatelet use.
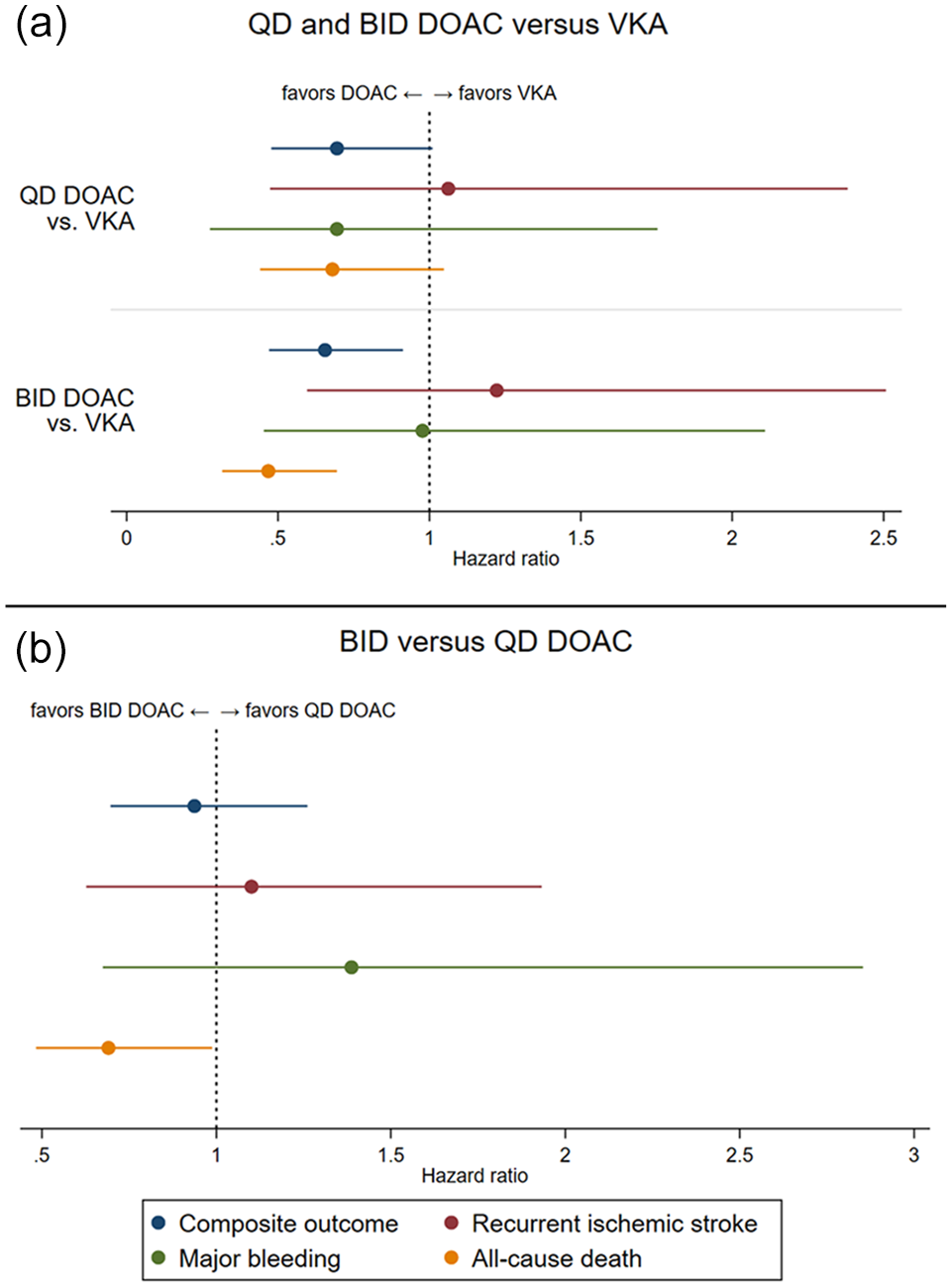
Adjusted hazard ratio estimates for the effect of oral anticoagulant type on the primary composite outcome and all its individual components, (a) from models including both DOAC types and VKA as reference and (b) from models including only DOAC (with QD DOAC as reference).
Secondary analysis – individual outcomes recurrent ischemic stroke, ICH, and death
Table 2 shows the follow-up time, number, and crude rate of events for the secondary (individual) outcomes according to OAC type.
The hazard for stroke recurrence did not differ between the OAC types, with similar point estimates close to 1 both for QD and BID DOAC compared to VKA, as well as for BID compared to QD DOAC, which was consistent in unadjusted and adjusted Cox models.
With regards to major bleeding, QD DOAC showed a trend for a lower hazard compared to both VKA and BID DOAC in unadjusted and adjusted analyses, with wide confidence intervals crossing 1.
Regarding all-cause death, both QD DOAC and BID DOAC were associated with a lower hazard compared to VKA in unadjusted and adjusted Cox models, an association which was more pronounced for BID DOAC. Upon direct comparison, BID DOAC were associated with a lower hazard for death compared to QD DOAC only in the adjusted Cox model.
The detailed results of the Cox models for the secondary outcomes are presented in Table 4 and Figure 2. The sensitivity analysis on recurrent ischemic stroke and major bleeding using Fine-Gray models to account for death as a competing risk yielded largely consistent results (Supplemental Table 2).
Cox models for time to recurrent ischemic stroke, major bleeding and death.
Adjustment for type of index event, National Institutes of Health Stroke Scale score, age, sex, modified CHA2DS2-VASc score (without age and sex), estimated glomerular filtration rate, history of major bleeding, time to OAC initiation after index event, atrial fibrillation known before index event, and concomitant antiplatelet use.
OAC: oral anticoagulant; DOAC: direct oral anticoagulant; QD: once-daily; BID: twice-daily; VKA: vitamin K antagonist.
Discussion
This explorative study focused on the safety and effectiveness of QD and BID DOAC following a recent ischemic stroke or TIA in patients with AF in a real-world setting. The two main findings were: (i) The overall beneficial effect of DOAC over VKA treatment was maintained both for the QD and BID dosing regimens, even when potential confounders were accounted for. (ii) There was no clear evidence to support the notion that the risk-benefit profile of QD versus BID DOAC might differ.
The first key finding of our study was that both QD and BID DOAC were associated with a lower hazard for the composite of recurrent ischemic stroke, major bleeding, and death compared to VKA among AF patients with a recent stroke. This remained true even after accounting for the less favorable baseline risk profile of VKA-treated patients in our observational cohort, although the association for the smaller group of patients treated with QD DOAC was weakened after adjustment, missing conventional statistical significance. Following the large randomized DOAC trials, 2 several observational studies confirmed the overall benefit of DOAC over VKA treatment in the general population of AF patients. 27 While observational studies later additionally showed that the trials’ findings also apply to patients with recent stroke,5–7,28 who had been excluded from the trials, 29 data on the “real-world” performance of QD and BID DOAC compared to VKA in patients with recent stroke were scarce. This is important, because patients with a recent stroke are at high risk for recurrence 15 and bleeding complications,16,17 and might be prone to non-adherence due to neurological and cognitive deficits.18,30 It has been previously postulated that the DOAC dosing regimen might have a differential impact on these factors.9,31 To our knowledge, only one previous study separately compared QD and BID DOAC to VKA in patients with a recent stroke and found both to be associated with a lower hazard for a combined outcome similar to ours. 5 Although this was only one among several post hoc analyses in this publication, it seems to support our finding that the overall benefits of DOAC over VKA are maintained regardless of QD or BID dosing regimen. Of note, in our study the lower hazard for the composite outcome with both DOAC regimens as opposed to VKA seemed to be mainly driven by death, consistent with prior observational studies comparing DOAC versus VKA in AF patients with recent stroke.6,7,28
More importantly and as a novelty, our study showed that the performance of QD versus BID DOAC with regards to the composite outcome did not differ upon direct comparison among AF patients with a recent stroke. While a number of observational studies previously compared DOAC directly to one another yielding somewhat inconsistent results,32–37 none focused on patients with recent stroke nor conducted comparisons of the QD versus BID DOAC regimen. Our finding is in contrast to a meta-analysis of the randomized DOAC trials that rendered indirect comparisons between QD and BID DOAC and reported that BID DOAC appeared to offer a more balanced risk-benefit profile with respect to stroke prevention and ICH. 31 We cannot rule out that this is attributable to lower power of our study, with a sample size of close to 1000 patients, which may have been insufficient to detect smaller differences in the effect of QD versus BID DOAC, as indicated by our post hoc power calculation. Alternatively, differences in the populations studied may matter as our population is characterized by advanced age and a high rate of multimorbid and severely affected patients. In addition, differences in adherence to medication might also explain this discrepancy. Patients’ adherence to DOAC was generally high in the well-controlled trial environment, 38 but several “real-world” studies have since shown a lower adherence to BID than QD DOAC.10–13 Although BID DOAC have been postulated to be “more forgiving” than QD DOAC with regard to stroke risk in the presence of non-adherence,9,31 this hypothesis is not supported by observational evidence from large datasets. 10 Regardless of any potential differences in adherence and its impact on the outcomes of treatment with QD versus BID DOAC, our “real-world” study on a large sample of AF patients with a recent stroke shows no clear evidence to support the notion that the risk-benefit profile of QD versus BID DOAC differs in a relevant manner.
Strengths and limitations
Our study has the following strengths: (i) We used prospectively collected, well-curated data from an established observational registry on a homogeneous cohort of consecutive stroke patients; (ii) the high data completeness, which reduces the risk of spurious findings; and (iii) the consistency regarding our results both in unadjusted and in adjusted analyses accounting for potential confounders, as well in the post hoc analysis accounting for medication administration, which underlines the robustness of our key findings.
We are aware of the following limitations: (i) As our data are observational rather than randomized, baseline imbalances in the allocation to the type of OAC that were unaccounted for might have introduced bias or confounding; (ii) The size of the OAC subgroups differed, which might have introduced imprecision in our model-based estimates; (iii) Our sample size may have been insufficient to detect smaller differences in the performance of QD versus BID DOAC; (iv) Our sample size disallowed analyses of each component of the composite outcome separately with confidence. Thus, the findings of the secondary analyses should be interpreted cautiously; (v) We did not account for changes in OAC treatment during follow-up, which is a potential source of bias.
Conclusion
In conclusion, our study showed that the overall benefit of DOAC over VKA seems to apply to both QD and BID DOAC in AF patients with a recent stroke, without clear evidence that one DOAC dosing regimen might be more advantageous than the other. Further larger studies are needed to more precisely elucidate potential differences in the risk-benefit profile of QD versus BID DOAC.
Supplemental Material
sj-docx-1-eso-10.1177_23969873221099477 – Supplemental material for Once versus twice daily direct oral anticoagulants in patients with recent stroke and atrial fibrillation
Supplemental material, sj-docx-1-eso-10.1177_23969873221099477 for Once versus twice daily direct oral anticoagulants in patients with recent stroke and atrial fibrillation by Alexandros A Polymeris, Annaelle Zietz, Fabian Schaub, Louisa Meya, Christopher Traenka, Sebastian Thilemann, Benjamin Wagner, Lisa Hert, Valerian L Altersberger, David J Seiffge, Flurina Lyrer, Tolga Dittrich, Ines Piot, Josefin Kaufmann, Lea Barone, Ludvig Dahlheim, Sophie Flammer, Nikolaos S Avramiotis, Nils Peters, Gian Marco De Marchis, Leo H Bonati, Henrik Gensicke, Stefan T Engelter and Philippe A Lyrer in European Stroke Journal
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: AP: research support from the Swiss Academy of Medical Sciences/Bangerter-Rhyner Foundation and the Swiss Heart Foundation; CT: travel honoraria from Bayer; research support from the Swiss Heart Foundation, Freiwillige Akademische Gesellschaft Basel, Bangerter-Rhyner Foundation, University of Basel, Novartis Foundation for Medical and Biological Research; ST: travel grants from BMS/Pfizer; DJS: research support from the Science Funds and Stroke Funds University Hospital Basel, Swiss National Science Foundation, Swiss Society of Neurology, Bangerter-Rhyner Foundation, Daiichi-Sankyo; Thrombosis research prize from Bayer Foundation; compensation for educational efforts from Stago; advisory board fees from Portola/Alexion, Bayer, Pfizer; NP: research support from the Swiss National Science Foundation, Swiss Heart Foundation; advisory boards for AstraZeneca and Daiichi-Sankyo; speaker honoraria from Vifor and OMPharma Suisse; GMDM: support from the Swiss National Science Foundation; Spezialprogramm Nachwuchsförderung Klinische Forschung, University of Basel; Science Funds (Wissenschaftspool) University Hospital Basel; Swiss Heart Foundation; ProPatient Foundation Basel; Bangerter-Rhyner-Stiftung; Swisslife Jubiläumsstiftung for Medical Research; Swiss Neurological Society; Fondazione Dr Ettore Balli; De Quervain research grant; Thermo Fisher GmbH; travel honoraria by Bayer and BMS/Pfizer; speaker honoraria by Bayer and Medtronic; consultant honoraria by Bayer and Novartis; member of the Steering Committee of PACIFIC Stroke; Industry payments are made to the research fund of the University Hospital Basel; LHB: research support from the Swiss Heart Foundation, Swiss National Science Foundation, Stiftung zur Förderung der gastroenterologischen und allgemeinen klinischen Forschung sowie der medizinischen Bildauswertung; unrestricted research grant from AstraZeneca, personal fees from Amgen, Claret Medical, InnovHeart; HG: research support from the Swiss National Science Foundation; advisory board honoraria from Daiichi-Sankyo; funding for travel from BMS/Pfizer and AbbVie; STE: research support from the Swiss National Science Foundation, Swiss Heart Foundation, Freiwillige Akademische Gesellschaft Basel, Science Fund Rehabilitation, Felix Platter Basel, the Stroke Fund and Scientific Fund of the University Hospital Basel, Pfizer, Daiichi-Sankyo; compensation from Stago for educational material; travel or speaker honoraria from Bayer, Boehringer-Ingelheim, BMS, Daiichi-Sankyo; advisory boards for Bayer, Boehringer-Ingelheim, BMS; PAL: research support from the Swiss National Science Foundation, Swiss Heart Foundation, Research Funds Neurology University Hospital Basel; advisory board compensation from Boehringer-Ingelheim, Bayer, Recordati SA, Daiichi-Sankyo; travel support from Pfizer. The remaining authors declare no relevant conflicts.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Bayer AG (Switzerland). NOACISP-LONGTERM was supported by the Science Fund Rehabilitation of the University Department of Geriatric Medicine Felix Platter Basel, the Stroke Fund and Scientific Fund of the University Hospital Basel, and grants from the Swiss Heart Foundation, Daiichi-Sankyo AG (Switzerland) and Bayer AG (Switzerland).
Informed consent
Written informed consent was obtained from the patients for their anonymized information to be published in this article.
Ethical approval
The NOACISP-LONGTERM registry including this study has been approved by the local ethics committee (BASEC PB_2016_00662).
Guarantors
AP, PAL.
Contributorship
All authors contributed to study design, data acquisition and analysis, and critically revised the manuscript. AAP performed statistical analyses and prepared the figures. AAP, STE, and PAL rendered the first manuscript draft.
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
