Abstract
Introduction
Micro-embolic signals (MESs) detected using transcranial Doppler (TCD) help in risk stratification in stroke patients. A systematic review and meta-analysis were performed to estimate the prevalence of MES and its association with stroke recurrence, functional outcome, and mortality in different stroke subtypes.
Methods
A comprehensive literature search was conducted before 26th January 2021 in PubMed, Embase, Google Scholar, Cochrane Library, and ClinicalTrials.gov. Studies were identified that used TCD to detect MES in stroke/TIA patients. Pooled prevalence and odds ratio (OR) along with 95% confidence interval (95% CI) were calculated for different outcome measures. The entire statistical analysis was conducted in R version 3.6.2.
Findings
Fifty-eight studies involving 5123 patients (1329 MES+, 3794 MES−) were included in our meta-analysis. The pooled prevalence of MES among all acute stroke/TIA patients was 30% (95% CI 25-34%). The pooled prevalence adjusted after the trim-and-fill analysis among all acute stroke/TIA patients was 18% (95% CI 14-23%). The prevalence of MES was high among all stroke subtypes except in patients with small vessel disease (SVD). In patients with new-onset stroke/TIA, the presence of MES was associated with a high risk of recurrence of cerebral ischemia (OR 4.03; 95% CI 2.38-6.82). Although no significant association was observed for the presence of MES with increased mortality (OR 2.37; 95% CI 0.75-7.50) and poor functional outcome (OR 2.11; 95% CI 0.20-22.50) among patients with new-onset stroke/TIA, this could only be determined in a smaller sample size of 477 patients.
Conclusions
Our meta-analysis showed a 30% prevalence of MES following acute stroke/TIA. The presence of MES increased the chance of recurrence of cerebral ischemia but was not associated with poor functional outcomes and mortality in the studied subgroup.
PROSPERO Registration Number: CRD42020200916
Introduction
Cerebral ischemia is one of the leading causes of morbidity and mortality across the world. Ever since the first description by Spencer et al. in 1990, 1 several studies evaluated the significance of micro-embolic signals (MESs) using transcranial Doppler (TCD) sonography in patients with cerebral ischemia. MES correspond to the micro-embolic particles present within the cerebral arteries. 2 They imply an unstable or vulnerable carotid plaque that may result in stroke/transient ischemic attack (TIA). Many studies have evaluated the prevalence of MES in stroke/TIA patients especially in large artery atherosclerosis (LAA) and cerebral embolism.3–5 Studies have also investigated the significance of MES with risk of future cerebral ischemic events and their outcome.6,7 Majority of these are small, underpowered studies and still substantial uncertainty exists about the results of these studies and their impact on clinical practice. Larger prospective studies on MES in stroke patients are hampered owing to the time consumption and difficulties involved in performing the procedure. Detection of MES could play an important role in the management of patients with stroke/TIA especially when the etiology is uncertain. This systematic review and meta-analysis aimed to summarize the available data and to obtain precise estimates on MES in newly diagnosed patients with stroke/TIA. This was primarily to determine the prevalence of MES among stroke subtypes. Additionally, we aimed to determine its association with stroke recurrence, functional outcome, and mortality.
Methods
Search strategy
This systematic review and meta-analysis were conducted in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) guidelines. 8 A checklist for the PRISMA guidelines is given in the Supplemental Material. A comprehensive literature search was performed using multiple electronic databases—PubMed, Embase, Google Scholar, Cochrane library, and ClinicalTrials.gov from 1996 to 26th January 2021.
The detailed search strategy is given in Supplemental material. The protocol for this systematic review and meta-analysis was registered in PROSPERO (ID: CRD42020200916), and there were no major deviations from the published protocol.
Population
Patients diagnosed with acute ischemic stroke (AIS) or TIA and underwent transcranial Doppler monitoring within seven days of the ischemic event.
Intervention
Transcranial Doppler monitoring for detecting micro-embolic signals.
Comparator
None.
Primary outcome
The pooled prevalence of micro-embolic signals among stroke subtypes.
Secondary outcomes
1) Recurrence of the cerebrovascular ischemic event. 2) Poor functional outcome (modified Rankin Scale (mRS) score ≥ 3). 3) Mortality at the last available follow-up.
Inclusion and exclusion criteria
Studies were included in the systematic review if they met all of the following inclusion criteria: 1) all prospective or retrospective studies including consecutive subjects diagnosed with AIS or TIA and underwent transcranial Doppler monitoring within seven days of the ischemic event; 2) studies stratifying etiology of stroke according to Trial of Org 10172 in Acute Stroke Treatment (TOAST) classification into cardioembolism, large artery atherosclerosis, cerebral small vessel disease, other, and unknown; 3) published studies in all languages were included and for those available in other languages, English translated version and/or English version results were sought by contacting the corresponding author twice via email.
Studies were excluded from the systematic review if they met any of the following exclusion criteria: 1) studies conducted on animal subjects; 2) unpublished or preprint studies; 3) case reports having individual cases and case series with a non-consecutive enrollment of cases; and 4) studies with full text not available or not translated to the English language.
Data synthesis
Two independent review authors (PS and SM) performed the literature search using the above-mentioned search terms. Full-text articles were selected if the abstract suggested the presence of relevant data. Inclusion or exclusion of studies was based on the uniform decision. Any divergence was resolved through discussion among all authors. The following information was extracted from each eligible study: journal name, first author, year of publication, study design, sample size, demographic characteristics including age, sex, MES positive/negative, and outcome measures. Data were grouped in each stroke subtype into MES positive and MES negative. The absolute number of MES was not considered. Patients were grouped into MES positive and MES negative according to outcome variables (recurrence of cerebrovascular ischemia, functional outcome, and mortality).
Risk of bias (quality) assessment
The risk of bias or quality assessment was done for all the studies included in our systematic review by two independent authors (PS and MN) using the new Cochrane risk of bias tool for randomized controlled trials (ROB-2), Newcastle Ottawa Scale (NOS) for cohort and case–control studies and Joanna Briggs Institute scale (JBI) for case-series and cross-sectional studies.9–11 Any disagreement was resolved by consulting with the remaining authors of the review. The risk of publication bias was assessed using funnel plots, and the asymmetry of the funnel plot was investigated using Egger’s regression test. 12
Statistical analysis
Dichotomous variables were represented by a number (percentage) and the continuous variables were represented by the mean and standard deviation (SD). A meta-analysis was performed only if data from more than two studies were available. The pooled prevalence of MES among stroke subtypes was calculated along with its corresponding 95% confidence interval (95% CI). The prevalence estimates were transformed using the Freeman–Tukey double arcsine transformation method. 13 The association of MES with stroke recurrence, functional outcome, and mortality was determined using odds ratio (OR) and 95% CI. We investigated the presence of heterogeneity among the included studies using the Cochran’s Q statistic and I-square, wherein heterogeneity was categorized as low: I2<25%, moderate: I2 25-75%, and high: I2>75%. 14 Results were combined using a random-effect model, and the DerSimonian–Laird estimator was used. Knapp–Hartung adjustments were applied to control for the uncertainty in our estimates of the between-study heterogeneity. The source of heterogeneity was further assessed by conducting the outlier and sensitivity analyses. A sensitivity analysis using the generalized linear mixed model (GLMM) method was done to account for the within-study uncertainties. Another sensitivity analysis was conducted using Hartung–Knapp–Sidik–Jonkman and Paule-Mandel estimators for random-effect models. A meta-regression analysis was conducted only if data from 10 or more studies could be pooled, to determine whether the predictor variables, namely, “risk of bias” and “study design,” were associated with the overall effect size difference for the respective outcome variables. Further, a subgroup analysis was conducted based on “risk of bias” and “study design.” A p-value of <0.05 was considered statistically significant, and all the statistical analyses were conducted using R version 3.6.2.
Findings
Study characteristics
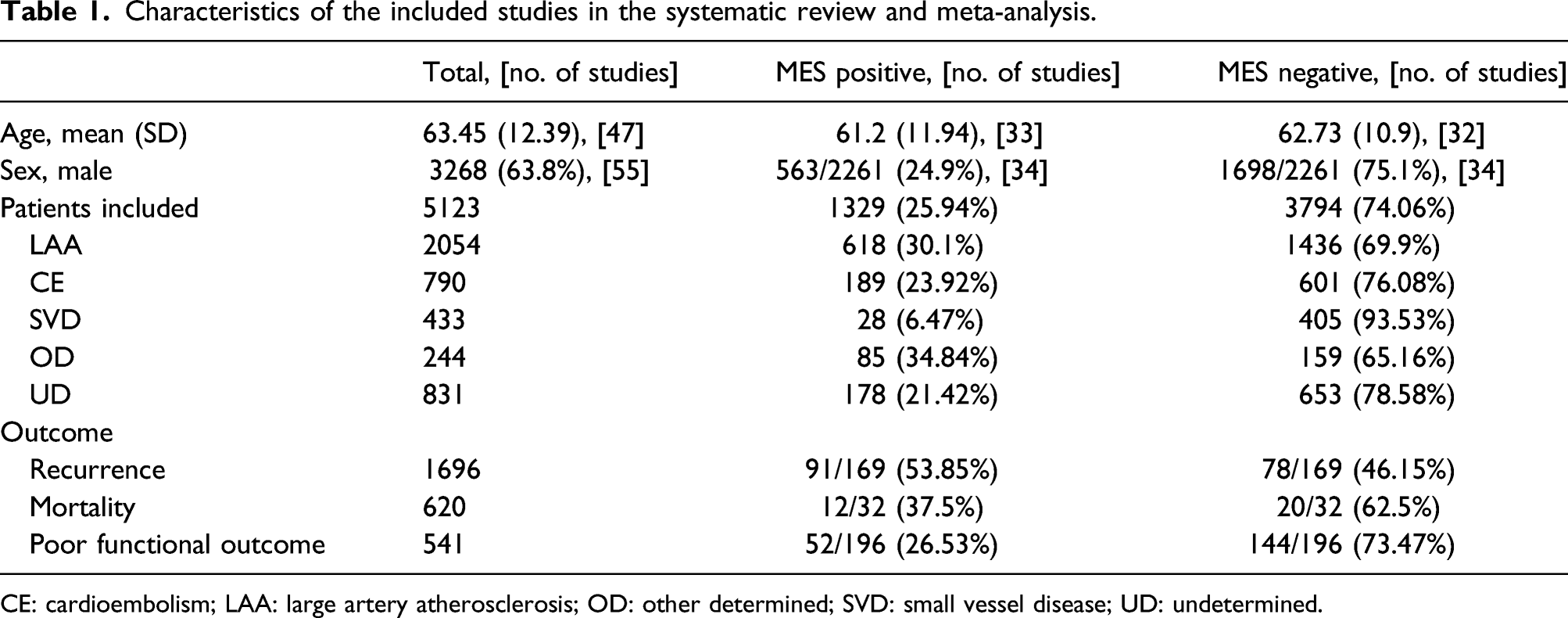
The systematic literature yielded 669 abstracts. After reading the full text of 239 articles, 58 studies finally met our inclusion criteria and were included in the systematic review and meta-analysis (Figure 1). A total of 5123 patients (1329 MES+, 3794 MES−) were included in these studies. Among the included studies, forty-two studies were prospective/retrospective cohort, six were cross-sectional, four were case–control, and three each was RCTs and case series studies. The studies were included from 22 distinct countries (details given in Supplemental material). The publication year of the included studies ranged from 1994 to 2020. The baseline characteristics of all the studies included in this systematic review and meta-analysis are given in Supplemental material (Table VI). The summarized characteristics of the included studies are shown in Table 1. The PRISMA study flow diagram. Characteristics of the included studies in the systematic review and meta-analysis. CE: cardioembolism; LAA: large artery atherosclerosis; OD: other determined; SVD: small vessel disease; UD: undetermined.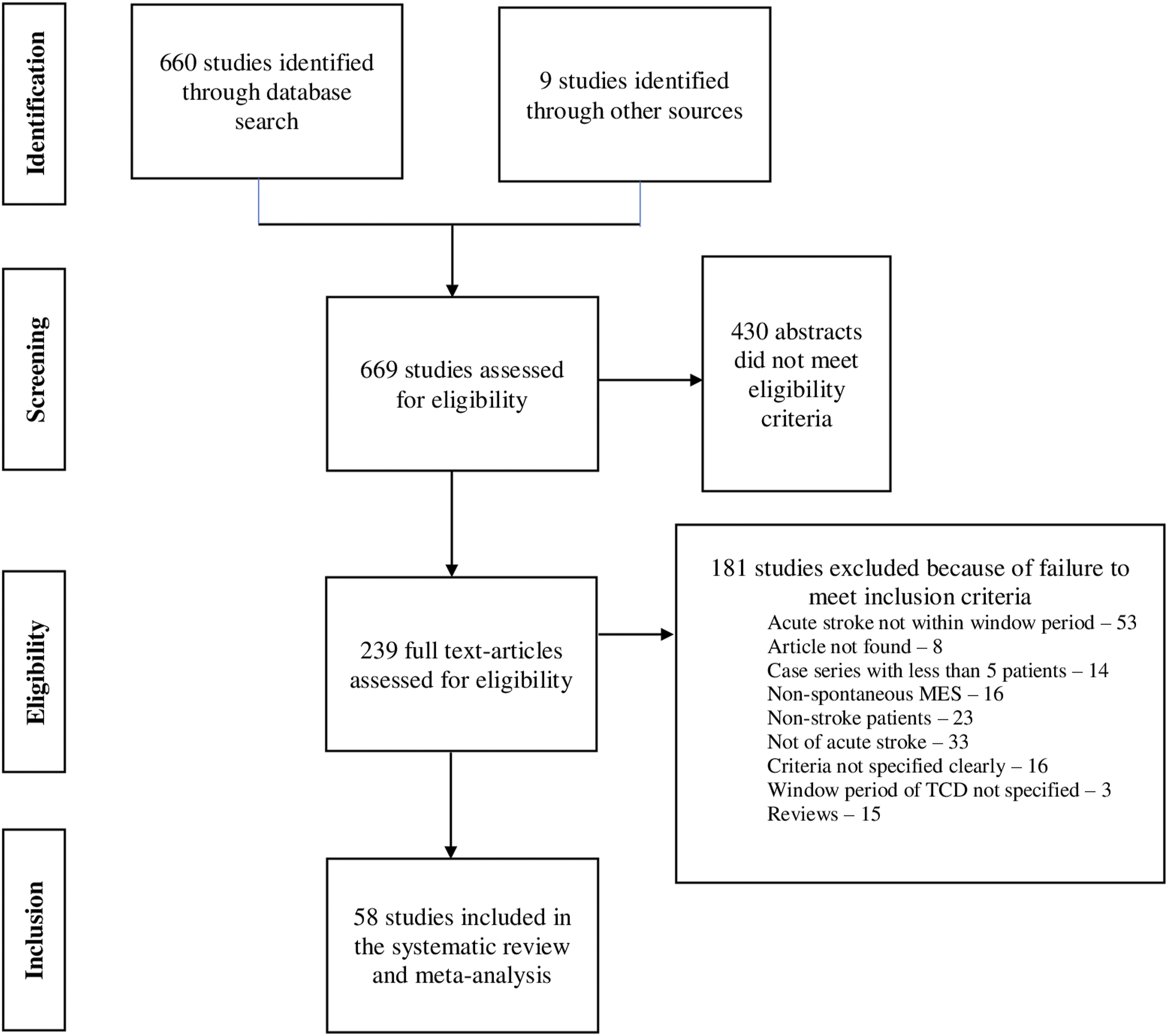
Primary outcome: Prevalence of MES among stroke subtypes
Among 58 studies included in the systematic review and meta-analysis, 5123 acute onset stroke/TIA patients underwent transcranial Doppler for MES monitoring within seven days of the event. The pooled prevalence of MES among all acute stroke/TIA patients was 30% (95% CI 25-34%). Data for MES concerning stroke subtypes according to TOAST classification was available as follows: Large artery atherosclerosis (LAA)—37 studies (2054 patients), cardio-embolism (CE)—23 studies (790 patients), small vessel disease (SVD)—18 studies (433 patients), other determined (OD)—17 studies (244 patients), and undetermined (UD)—25 studies (831 patients). The pooled prevalence of MES was high among all stroke subtypes [(LAA 32%; 95% CI 28-37%), (CE 24%; 95% CI 15-34%), (OD 35%; 95% CI 19-53%), (UD 24%; 95% CI 14-35%) except in patients with SVD (3%; 95% CI 0-10%)]. The forest plot for the pooled prevalence of MES among patients with stroke/TIA is represented in Figure 2. The tabulated results (Table I) and remaining forest plots (Figure I(a-e)) for the pooled prevalence of MES among different stroke subtypes are given in the Supplemental material. Pooled prevalence of MES positive in total stroke/TIA patients.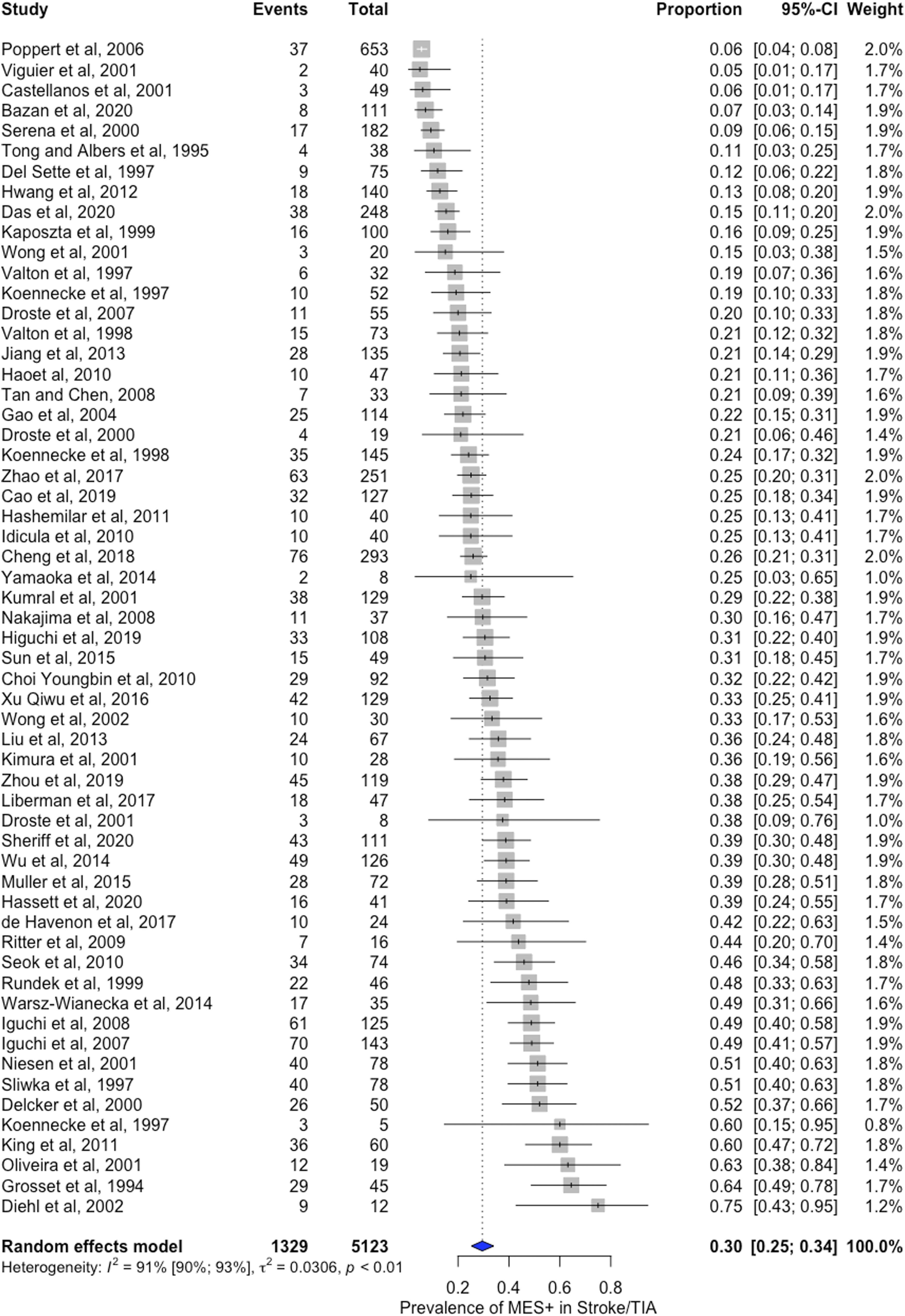
Secondary outcomes
The secondary outcomes of our study were to detect the association between the presence of MES and recurrence of cerebrovascular ischemia, mortality, and poor functional outcome in patients with acute onset stroke/TIA. A total of 19 studies having 1243 stroke/TIA patients reporting outcomes on recurrence of cerebrovascular ischemia were included in the systematic review and meta-analysis. The presence of MES was found to be associated with a high risk of recurrence of cerebral ischemia (OR 4.03; 95% CI 2.38-6.82). Data on the poor functional outcome (mRS score≥3) was available from three studies, including 443 patients. There was no significant association between MES and poor functional outcome (OR 2.11; 95% CI 0.20-22.50) in patients with acute stroke/TIA. Seven studies having 477 strokes/TIA patients reported outcomes on mortality and were included in the systematic review and meta-analysis. There was a trend of the near doubling of mortality (Labbé plot given in Figure II in the Supplemental material) toward stroke/TIA patients with MES positive compared to MES negative (OR 2.37; 95% CI 0.75-7.50); however, this was not statistically significant (Table II in Supplemental material). Figure 3(a-c) displays the forest plots on the outcomes of stroke/TIA patients with MES. (a-c) Forest plots of meta-analysis assessing the presence of MES with recurrence of cerebrovascular ischemia, poor functional outcome, and mortality.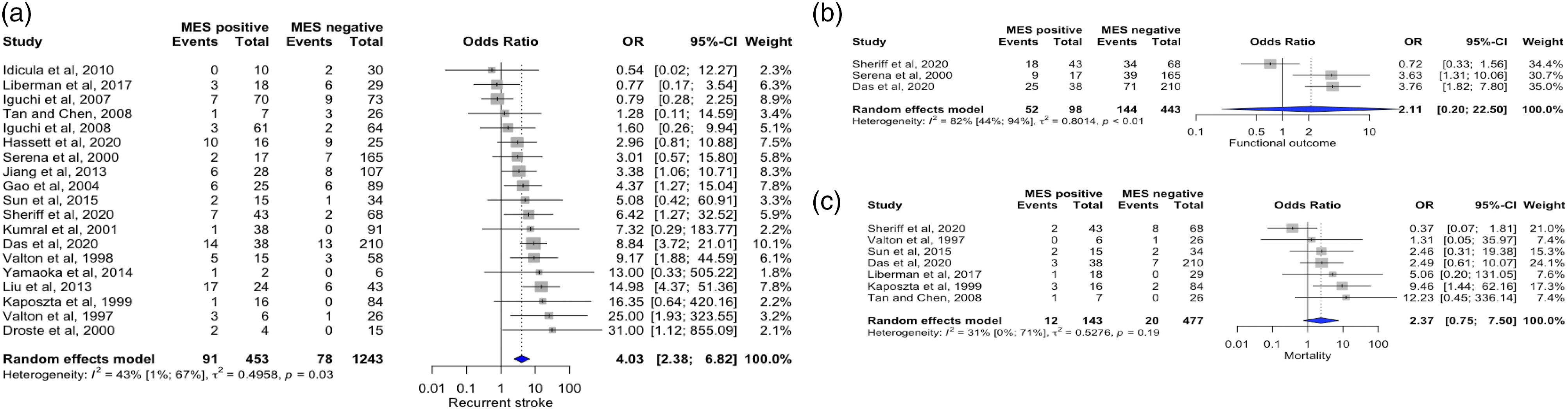
Risk of bias (quality) assessment
The risk of bias (quality) was assessed using the ROB-2 tool for RCTs, NOS for cohort and case–control studies, and JBI for case-series and cross-sectional studies. For NOS, the total score was divided into three categories: (a) 1-3 (high risk of bias); (b) 4-6 (some concerns); and (c) 7-9 (low risk of bias). For JBI, the total score was divided into three categories: (a) 1-4 (high risk of bias); (b) 5-7 (some concerns); and (c) 8-10 (low risk of bias). Overall, 18 (31.03%) studies included in our review had an overall low risk of bias, 39 (67.24%) studies had some concerns related to the risk of bias, while 1 (1.72%) study had a high risk of bias. One study which had a high risk of bias belonged to the RCT subgroup. Tables VIII to XII in the Supplemental material represent the quality assessment of each study included in the systematic review and meta-analysis.
Publication bias
The publication bias was assessed using funnel plots for all outcome variables wherein data from more than 10 studies could be pooled together. The shape of the funnel plots was found to be asymmetrical when assessing the prevalence of MES among all stroke/TIA patients (p-value: 0.0002), patients with LAA (p-value: 0.0004), and patients with strokes of undetermined etiology (p-value: 0.02). This was also confirmed by the significant p-values obtained after conducting Egger’s regression test. If the p-value in Egger’s regression test was <0.05, then the Duval and Tweedie trim-and-fill procedure was used to impute the missing studies into the funnel plots till symmetry was achieved. We observed that studies with a lower prevalence were missing in all stroke/TIA patients, patients with LAA, and patients with strokes of undetermined etiology. The pooled prevalence adjusted after the trim-and-fill analysis was as follows: Acute stroke/TIA (18%; 95% CI 14-23%), LAA (27%; 95% CI 21-32%), and UD (5%; 95% CI 0-16%). The shape of the remaining funnel plots as well as the p-value of Egger’s regression test was not significant for the presence of any publication bias for the rest of the outcome measures (Figures III & IV(a-c) in the Supplemental material).
Outlier, sensitivity, meta-regression, and subgroup analysis
Given in Supplemental material.
Discussion
Our meta-analysis provides a systematic summary of pooled prevalence of MES across different subtypes of stroke as well as its association with stroke recurrence, functional outcome, and mortality. In this systematic review and meta-analysis, we observed a high prevalence of MES during the acute phase of stroke/TIA across all subtypes except in patients with SVD. The majority of the studies included were prospective/retrospective cohort and had some concerns related to the risk of bias. However, meta-regression and subgroup analysis showed no association between risk of bias and overall effect size difference for any of the outcome measures.
MES is the “real time” imaging correlate of cerebral micro-emboli detected using transcranial Doppler sonography. They are composed of clots, platelet aggregates, and atherosclerotic plaque particles. The presence of micro-emboli indicates an unstable or vulnerable carotid plaque that may lead to artery-to-artery embolism. In this study, we found a high prevalence of MES in different stroke subtypes based on TOAST classification which signifies the pathogenic role of cerebral embolism in many stroke subtypes. This is in line with the previous meta-analysis by King et al. in 2009 where the prevalence of MES ranged between 9.3% and 71% in the acute phase after TIA and stroke. 15 However, they excluded retrospective studies and studies that did not provide data on future stroke/TIA events which may have underestimated the prevalence of MES. By including these studies in our meta-analysis, we might have estimated the true prevalence of MES following stroke/TIA. We could not evaluate the prognostic significance of MES in individual stroke subtypes in our study because of the limited number of studies reporting them. However, a previous meta-analysis published by King et al. found different clinical significance across few stroke subtypes such as large artery atherosclerosis and cardioembolism, although this could only be evaluated in a very small sample size. 15 In view of the very high number of studies included in our systematic review and meta-analysis, considerable heterogeneity was expected to be present while assessing the prevalence of MES among different stroke subtypes. However, after removing the outlier studies, the heterogeneity was significantly reduced for total stroke/TIA and LAA and SVD subtypes of stroke. A marginal decrease in the revised pooled prevalence was observed after the removal of outlier studies in all the stroke subtypes except the other determined subtype of the stroke where the pooled prevalence increased from 35% to 40% after removing the outlier studies. Another subgroup analysis based on study design observed statistically significant differences between the subgroups (p-value: 0.0004) with reduced heterogeneity when analyzing the pooled prevalence of MES in the LAA subtype. To test the robustness of the prevalence estimates obtained in our review, the analysis was rerun by using the GLMM method and different estimators for the random-effect model. Our prevalence estimates were found to be fairly similar across different sensitivity analyses; however, reduced heterogeneity was observed for all prevalence measures using the GLMM method. For SVD and OD subtypes, the heterogeneity reduced significantly from 78% to 0% and 75% to 47%, respectively, after using the GLMM method.
We found a significant association between the presence of MES and the recurrence of cerebrovascular ischemia. This is in line with the previous studies. A meta-analysis by King et al. in 2009 found that the presence of MES increased the risk of stroke/TIA (OR, 3.71; 95% CI, 1.64-8.38; P = 0.002). 15 In a meta-analysis of twenty-two studies by Best et al. in 2016, the positive likelihood ratio of having a future stroke/TIA was 2.43 (95% CI 1.85-3.18; p < 0.05) in the presence of MES. 16 In another meta-analysis of six studies by Safouris et al. in 2018, statin pretreatment in patients with cerebral ischemia was associated with a reduced risk of MES detection during TCD monitoring (risk ratio 0.67; 95% CI, 0.45-0.98) which may be related to lower risk of recurrent stroke in these patients. 17 This association was further strengthened by a multicentric RCT, CARESS trial published in 2005 which depicted an association between baseline MES and seven-day risk of cerebrovascular ischemia. 18 In this study, compared to monotherapy, the use of dual antiplatelet therapy following stroke/TIA was associated with a significant reduction in the prevalence of MES at 7 days (relative risk reduction 39.8%; 95% CI 13.8-58.0%; p = 0.0046). We could not include this study in our meta-analysis as TCD was done multiple times within seven days and patients were only included in the outcome analysis if they had MES at baseline. Therefore, our meta-analysis adds further evidence to the available literature that there is considerable evidence between MES and future risk of stroke/TIA. However, owing to the lack of studies on stroke subtypes other than large artery atherosclerosis, we could not perform a specific subtype prognosis in our meta-analysis. This signifies the importance of future large prospective, multicentric trials which were undermined currently due to the technical difficulties associated with TCD.
Data are sparse regarding the association of MES with functional outcomes which we addressed in our meta-analysis. Though we did not find a significant association between the presence of MES and functional outcome, we did find a trend toward poor functional outcome and increased mortality in patients with recent-onset stroke/TIA (OR 2.37; 95% CI 0.75-7.50). Lack of association may be explained by the effect of antithrombotic treatment in these patients which might have reduced the rate and prevalence of MES over time. The effect of antiplatelet and anticoagulation on the prevalence of MES has been evaluated before in various RCTs. In the CARESS trial, the use of dual antiplatelet medication was more effective in reducing asymptomatic embolization as compared to single antiplatelet (relative risk reduction 39.8%; 95% CI 13.8-58.0%; p = 0.0046). 18 Other trials showed no difference between aspirin and clopidogrel, 19 aspirin and low molecular weight heparin, 20 and clopidogrel and dipyridamole in reducing asymptomatic embolization. 21
Limitations
Although we ensured that our systematic review and meta-analysis were conducted very comprehensively, there were several limitations in our study. Firstly, there was considerable heterogeneity in the outcomes analyzed in our meta-analysis leading to a large variation in prevalence. This may be explained due to the high number of studies included, quality of studies, and technical factors related to TCD or maybe owing to real differences in the underlying prevalence of MES between patients. However, we conducted sensitivity, outlier, subgroup, and meta-regression analyses to explain the source of heterogeneity and used a random-effect model. The sensitivity analysis also confirmed that our prevalence estimates were robust after different methods and estimators were used to rerun the analysis. This suggests that the variation in prevalence in our meta-analysis might be due to real differences in the underlying prevalence of MES between patients or the accuracy and completeness of TCD recording rather than differences in the studies included. We also found a reduction in the overall prevalence after the trim and fill analysis. This indicates the presence of missing studies. Secondly, a large proportion of studies on MES were performed in patients with symptomatic carotid stenosis, and hence, the presence of publication bias cannot be completely ruled out. Thirdly, the presence of MES in different stroke subtypes can have different prognostic significance. We could not assess outcome measures among stroke subtypes due to the scarcity of literature. Absolute numbers of MES were also not considered. Fourthly, although we found a high prevalence of MES across several stroke subtypes, taking the intensity of MES into account may have helped in differentiating the stroke subtypes especially when the stroke workup is negative. Also, clinical outcomes may vary within each stroke subtype depending on the intensity of MES. Lastly, the effect of antiplatelet and/or anticoagulation on the presence and intensity of MES during the follow-up was not considered in this study. This can help in deciding the duration of antithrombotic treatment especially in those patients who were started on dual antiplatelets and combination therapy. We found a decrease in overall prevalence after the trim and fill analysis confirming the presence of missing studies.
Future implications
About a third of the ischemic strokes are cryptogenic. The majority of these cases have an unidentified etiology in the form of cardio-embolism, vasculopathy, and coagulopathy. There is also a lack of clarity regarding the recurrence rates and prognosis of cryptogenic strokes. We suggest MES monitoring might help in identifying the stroke subtype and risk stratification in these patients. Future studies should be performed that may help in differentiating stroke subtypes based on the prevalence and intensity of MES signals. The pooled prevalence estimates from our meta-analysis could be used to estimate sample size for conducting prospective cohort studies and for better risk stratification of different subtypes of stroke patients.
Conclusions
In summary, this meta-analysis provides evidence regarding the prevalence of MES across ischemic stroke subtypes during the acute phase. It also signifies the association of MES with a high risk of recurrence of cerebral ischemia and a trend toward increased risk of mortality and poor functional outcomes following stroke/TIA. The results of our meta-analysis might serve as a platform for future studies with a larger sample size and longer follow-up time in differentiating stroke subtypes based on MES and in evaluating the effect of antithrombotic treatment on the prevalence of MES and outcomes following acute ischemic stroke/TIA. It also provides evidence for including TCD in all patients of AIS for etiological evaluation and risk stratification.
Supplemental Material
sj-pdf-1-eso-10.1177_23969873211060819 – Supplemental Material for Micro-embolic signal monitoring in stroke subtypes: A systematic review and meta-analysis of 58 studies
Supplemental Material, sj-pdf-1-eso-10.1177_23969873211060819 for Micro-embolic signal monitoring in stroke subtypes: A systematic review and meta-analysis of 58 studies by Pachipala Sudheer, Shubham Misra, Manabesh Nath, Pradeep Kumar, Deepti Vibha, M.V.Padma Srivatsava, Manjari Tripathia, Rohit Bhatia, Awadh Kishor Pandit and Rajesh K Singh in European Stroke Journal
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
