Abstract
Introduction
Cerebral small vessel disease (SVD) predicts all-cause mortality in Eastern Asian and Caucasian populations. However, little is known about SVD impact in individuals of different races/ethnic groups. In this study, we sought to estimate the mortality risk according to the total SVD (tSVD) score in older adults of Amerindian ancestry.
Methods
Participants aged ≥60 years from the prospective population-based Atahualpa Project cohort underwent brain MRI between June 2012 and June 2017. The tSVD score was calculated based on the presence of moderate-to-severe white matter hyperintensities, enlarged perivascular spaces, one or more lacune, and one or more cerebral microbleed. We ascertained all-cause mortality during post-MRI follow-up. Poisson regression and Cox-proportional hazards models adjusted for demographics and cardiovascular risk were obtained to estimate mortality risk according to the tSVD score.
Results
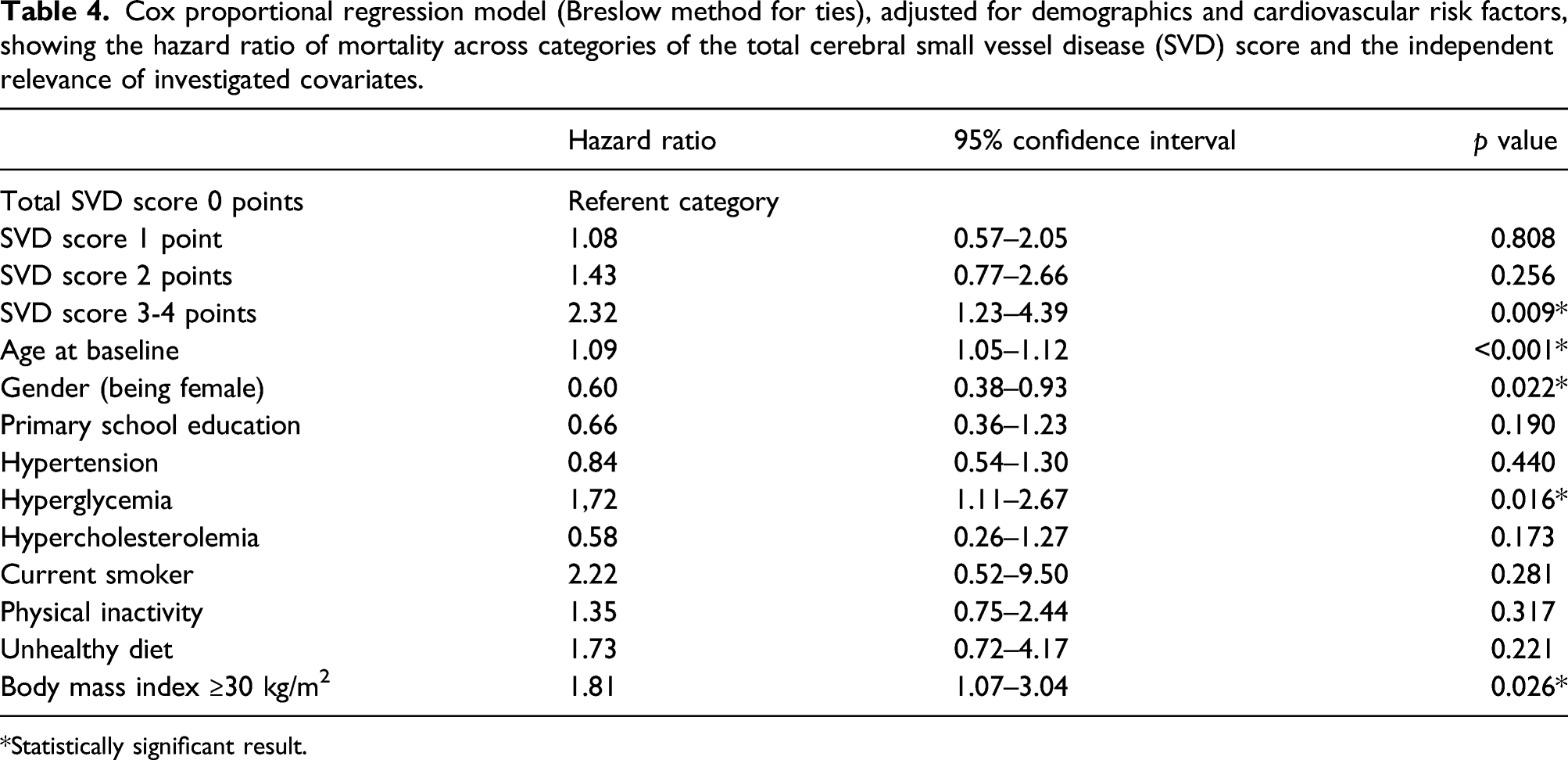
Analysis included 375 participants with available brain MRI and clinical data (mean age 69.0 ± 8.3 years, 56.3% women). The tSVD score was 0 point in 216 individuals (57.6%), 1 point in 71 (18.9%), 2 points in 53 (14.1%), and 3–4 points in 35 (9.3%). Increasing tSVD score was associated with advancing age, hypertension, low level of education, and physical inactivity. Using tSVD score of 0 as reference, a multivariate Poisson regression model showed an increased mortality for individuals with a tSVD score 3–4 points (IRR: 2.27; 95% CI: 1.20–4.28). Likewise, in the Cox-proportional model adjusted for demographics and cardiovascular risk, participants with a tSVD score 3–4 maintained a greater than two-fold mortality risk when compared to those with tSVD score of 0 points (HR: 2.32; 95% CI: 1.23–4.39).
Conclusions
High-burden SVD as determined by the tSVD score predicts mortality in community-dwelling older adults of Amerindian ancestry. Incidental diagnosis of covert SVD should prompt aggressive control of cardiovascular health.
Keywords
Introduction
Cerebral small vessel disease (SVD) is a highly prevalent disorder of the cerebral microvasculature characterized by endothelial dysfunction, vessel stiffening, dysfunctional blood flow, white matter rarefaction, ischemia, inflammation, and secondary neurodegeneration. 1 Clinically, SVD is responsible for one-fourth of all ischemic strokes, most hemorrhagic strokes in older adults, and contributes to almost half of dementia cases.2,3 Brain parenchymal lesions associated to SVD include white matter lesions, lacunes, cerebral microbleeds (CMB), and enlarged perivascular spaces (EPVS). 4 SVD-related parenchymal lesions can be detected on brain MRI, which has become the mainstay for SVD diagnosis and grading.4,5
Beyond the morbidity associated with stroke and dementia, individual SVD biomarkers have shown to be associated to all-cause mortality in hospital-based and population-based studies.6,7 However, there is limited information on whether SVD biomarkers accumulation is a better predictor for unfavorable outcomes than each biomarker alone. The total SVD (tSVD) score is a simple and reliable method to stratify SVD burden, 7 yet its association with mortality has not been widely studied. A Korean study among patients with ischemic stroke found that only high-burden SVD as determined by a maximal tSVD score independently predicted all-cause mortality. 8 A more recent study found that all-cause mortality increased with advancing tSVD score in a heterogeneous population with and without established cerebrovascular disease. 9
Most studies on the association between SVD and mortality risk focused in either Eastern Asian or Caucasian populations.6–10 Formerly, our group reported that SVD prevalence in Amerindians living in rural Latin America is comparable to those reported from the developing world,11,12 despite a better cardiovascular profile in the former. 13 In order to expand our understanding on the association between SVD burden and mortality risk, as well as the impact that SVD has on the health of native inhabitants of Latin America (Amerindians), we sought to determine the association between the tSVD score and all-cause mortality in a population-based longitudinal study conducted in a rural Ecuador.
Methods
Study Population
Subjects were participants of the Atahualpa Project, a population-based prospective study focused on risk factors for stroke and cognitive decline in rural coastal Ecuador. Participants have been identified since June 2012 by means of yearly door-to-door surveys and other overlapping sources, and were considered eligible if aged ≥40 years, were established inhabitants of the village, and provided informed consent. Population characteristics have previously been described. 14 Atahualpa is a relatively isolated and underserved community, more than 95% of inhabitants share ethnicity (Amerindian ancestry), and most villagers have primary school education only and low socioeconomic status.
Study design
All participants aged ≥60 years were invited from June 2012 to June 2017 to undergo brain MRI if they were not disabled, had no contraindications for MRI, and provided signed consent. Poisson regression models were fitted to estimate the mortality incidence rate ratio (IRR), and Cox-proportional models were fitted to calculate the hazard ratio (HR) in order to estimate the risk for all-cause mortality according to the presence of individual SVD biomarkers and the tSVD score. The study was approved by the Institutional Review Board of Hospital-Clínica Kennedy, Guayaquil (FWA: 00030727).
Brain MRI analysis
Brain MRIs were performed in a 1.5T MRI system (Phillips Medical Systems, Eindhoven, the Netherlands) and included multi-slice turbo spin echo T1-weighted, fluid attenuated inversion recovery (FLAIR), T2-weighted, and gradient-echo sequences in the axial plane. Slice thickness was 5 mm with 1 mm gap between slices. White matter hyperintensities (WMH) were defined as hyperintense lesions on T2-weighted images that remained bright on FLAIR (without cavitation) and graded according to the modified Fazekas scale. 15 WMH were considered moderate-to-severe when periventricular WMH extended into the deep white matter (Fazekas 3) or deep WMH became confluent or semi-confluent (Fazekas 2–3). Lacunes were defined as fluid-filled cavities measuring 3–15 mm in diameter located in the territory of perforating arterioles. 4 CMB were identified as small (≤10 mm) round areas of signal void with blooming seen on gradient-echo sequences and rated according to the microbleed anatomical rating scale. 16 EPVS were defined as small sharply delineated spaces of CSF intensity on T2-weighted images that followed the course of perforating arterioles. EPVS were graded at the level of the basal ganglia and considered moderate-to-severe if >10 lesions were seen in one side of the brain. 17 MRIs were independently read by two investigators blinded to clinical data and disagreements were resolved by consensus. Kappa coefficients for inter-reader agreement were 0.93 for moderate-to-severe WMH, 0.84 for EPVS, 0.88 for lacunes presence, and 0.82 for CMB presence. The tSVD score was estimated as an ordinal scale ranging from 0 to 4 points, where 1 point was awarded for each of the following: moderate-to-severe WMH, moderate-to-severe EPVS, one or more lacunes, and one or more CMB. 5
Outcome measure
Participants were screened yearly to determine changes in vital status. In addition, field investigators continuously reminded participants and their proxies to notify us in case of a new major health problem or in the event of death. Positive screens were further evaluated by proxy-delivered medical history and medical records review. Death certificates were revised to confirm the date and cause of death. For the current analysis, the last censoring date was set as June 2021.
Clinical covariates
Demographics and cardiovascular risk factors were selected as potential confounders. Age was recorded at the time of MRI. The American Heart Association (AHA) criteria were used to assess cardiovascular risk factors (health metrics). 18 A poor smoking status was designated if the subject was a current smoker, a poor body mass index if ≥30 kg/m2, a poor physical activity if the subject engaged in no moderate or vigorous physical activity, a poor diet if the individual had none or only one component of the AHA healthy diet, a high blood pressure if ≥140/90 mm Hg, a poor fasting glucose if ≥126 mg/dL, and a poor total cholesterol levels if ≥240 mg/dL.
Statistical analysis
Baseline characteristics of the study population were compared according to the tSVD score using linear models for continuous variables and the chi-squared test for categorical variables. We calculated the crude mortality IRR overall and stratified it by the tSVD score using Poisson regression models. To compute the person-years of follow-up, we considered the time from MRI to the last censoring date, study drop-out, or time of death. Cox-proportional models (Breslow method for ties) adjusted for demographics and cardiovascular risk were fitted to calculate the HR with its 95% confidence interval (CI) in order to estimate the risk for all-cause mortality according to the presence of individual SVD biomarkers and the tSVD score. Scores of 3 and 4 were merged as the high-burden SVD group to reduce variability. All data analyses were carried out by using STATA version 17 (College Station, TX, USA).
Results
Characteristics of Atahualpa residents aged ≥60 years identified by means of door-to-door surveys that were included and not included in this study.
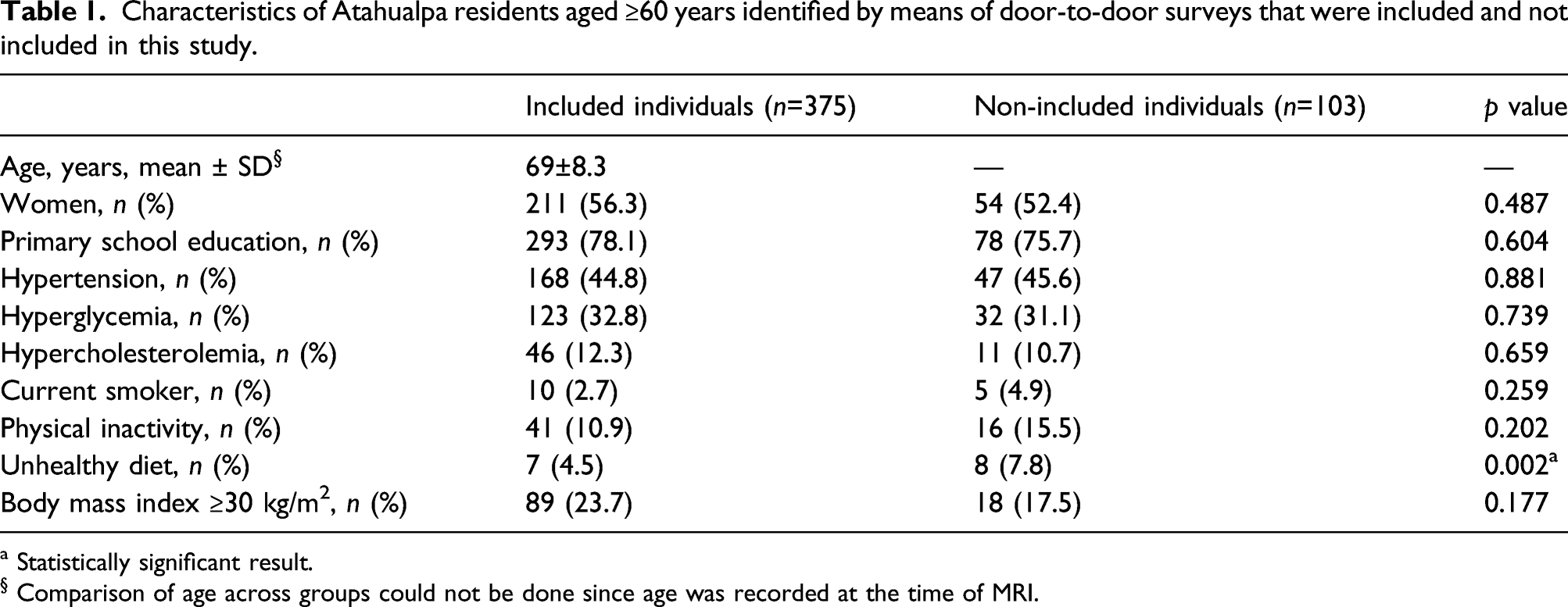
a Statistically significant result.
§ Comparison of age across groups could not be done since age was recorded at the time of MRI.
The mean age of the studied population at the time of MRI was 69 ± 8.3 years, 56.3% were women, and 78.1% had primary school education only. Cardiovascular risks factors were distributed as follows: 44.8% had hypertension, 32.8% had hyperglycemia, 12.3% had hypercholesterolemia, 23.7% were obese, 10.9% were physically inactive, 4.5% had an unhealthy diet, and 2.7% were current smokers.
Study population characteristics according to the total small vessel disease score.
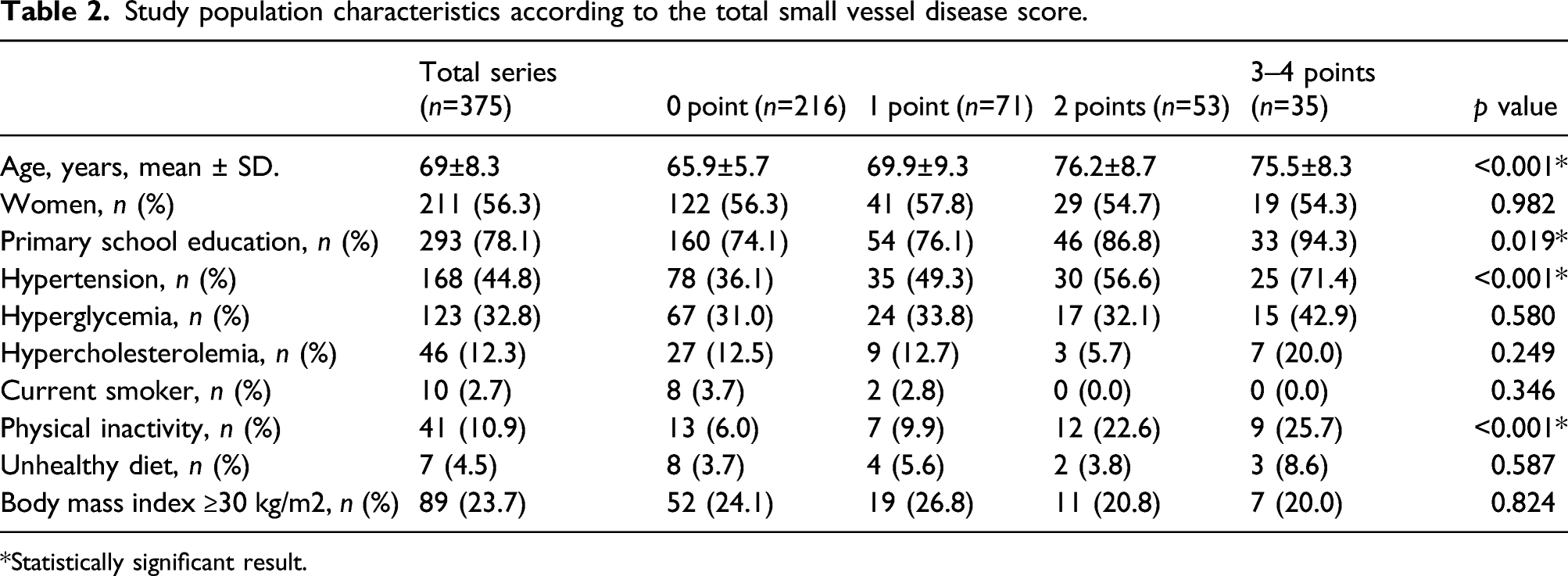
Statistically significant result.
Crude mortality incidence rate according to the total small vessel disease (SVD) score.
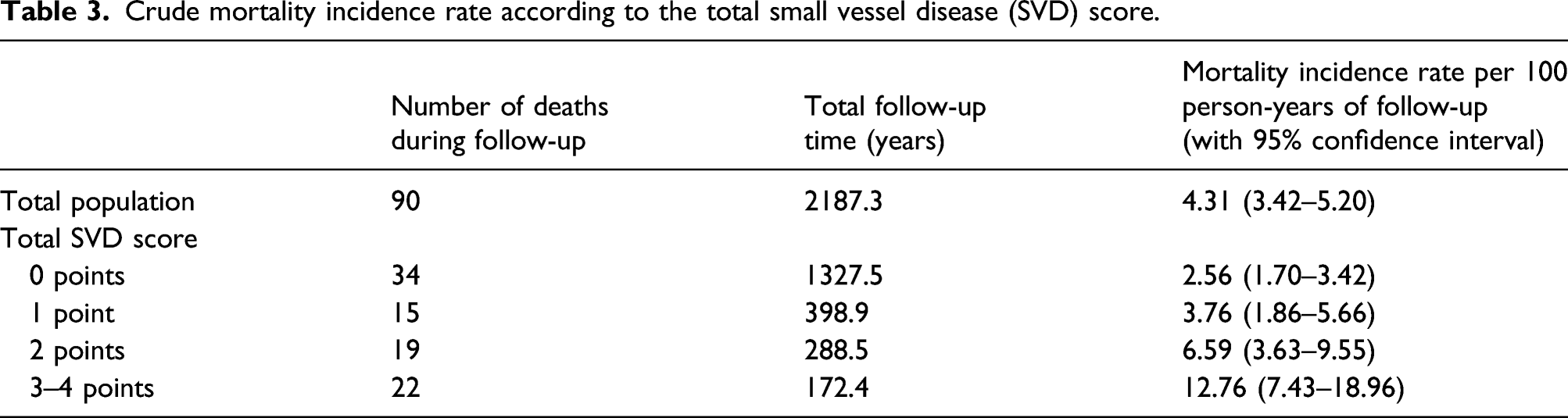
Using Cox-proportional models, moderate-to-severe WMH (HR: 3.24; 95% CI: 2.14–4.91), EPVS (HR: 2.36; 95% CI: 1.56–3.57), lacunes (HR: 3.00; 95% CI: 1.89–4.76), and CMB (HR: 2.57; 95% CI: 1.56–4.23) were associated with increased mortality in unadjusted analyses. After adjusting for demographics and cardiovascular risk factors, only moderate-to-severe WMH and lacunes remained associated to mortality risk (Figure 1). When compared to tSVD score of 0, unadjusted all-cause mortality HR was greater with tSVD score 2 points (HR: 2.72; 95% CI: 1.55–4.78), and score of 3–4 points (HR: 5.12; 95% CI: 2.98–8.78), but not with score of 1 point (HR: 1.51; 95% CI: 0.82–2.78). In the Cox-proportional model adjusted for demographics and cardiovascular risk, participants with high-burden SVD (tSVD score 3–4) maintained a greater than two-fold mortality risk when compared to those with tSVD score of 0 point (Figure 2). Covariates remaining independently significant in this model were age at baseline, being male, a poor BMI, and high fasting glucose levels (Table 4). Kaplan–Meier survival curves and hazard ratios with 95% confidence intervals (adjusted for demographics and cardiovascular risk) for all-cause mortality according to the presence of individual small vessel disease biomarkers: Moderate-to-severe white matter hyperintensities (upper left), one or more lacunes (upper right), one or more cerebral microbleeds (lower left), and enlarged basal ganglia perivascular spaces (lower right). Numbers in the x-axes correspond to months of follow-up. Kaplan–Meier survival curves and hazard ratios with 95% confidence intervals for all-cause mortality according to the total small vessel disease score, adjusted for demographics and cardiovascular risk. Numbers in the x-axis correspond to months of follow-up. Cox proportional regression model (Breslow method for ties), adjusted for demographics and cardiovascular risk factors, showing the hazard ratio of mortality across categories of the total cerebral small vessel disease (SVD) score and the independent relevance of investigated covariates. Statistically significant result.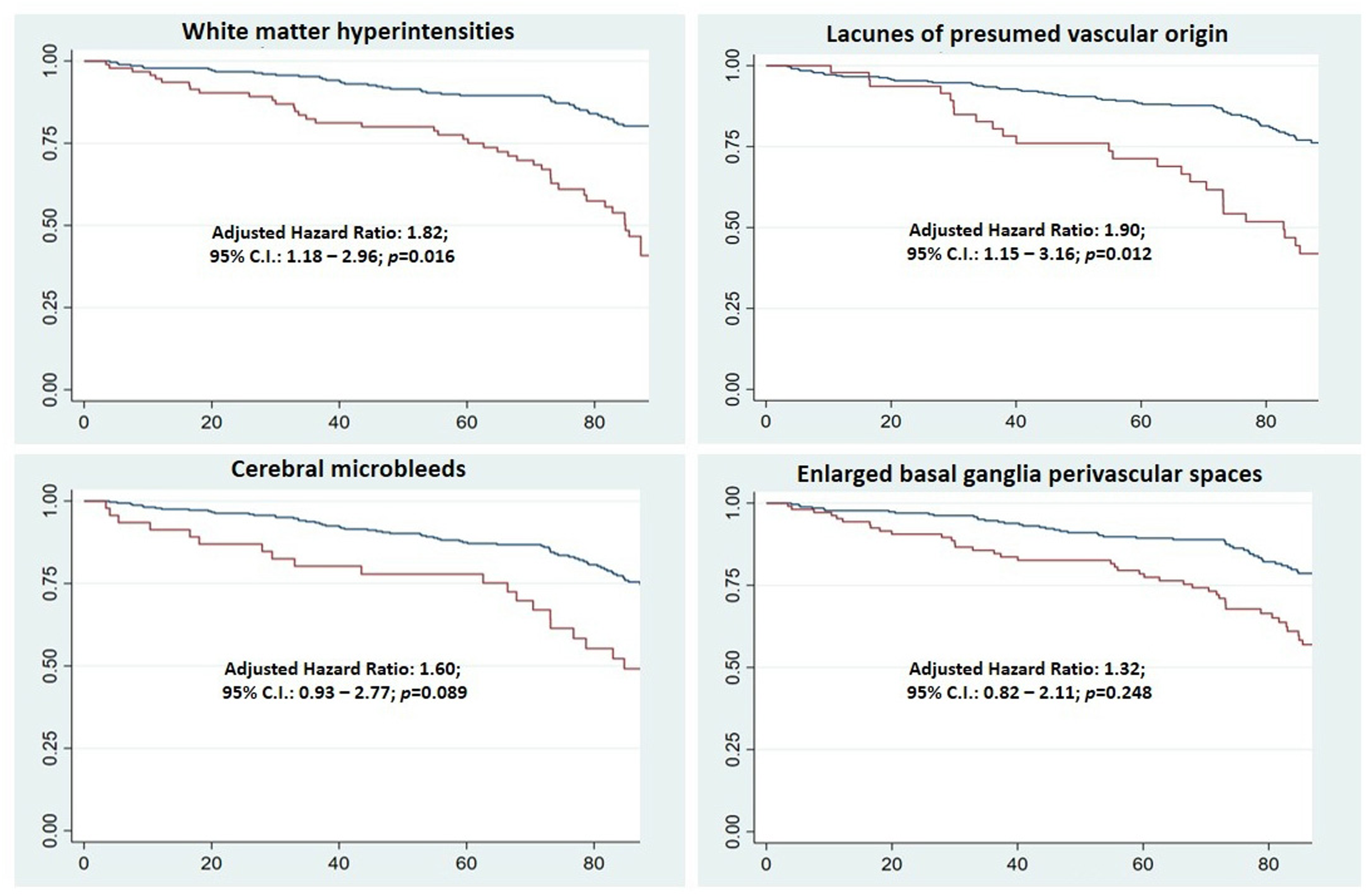
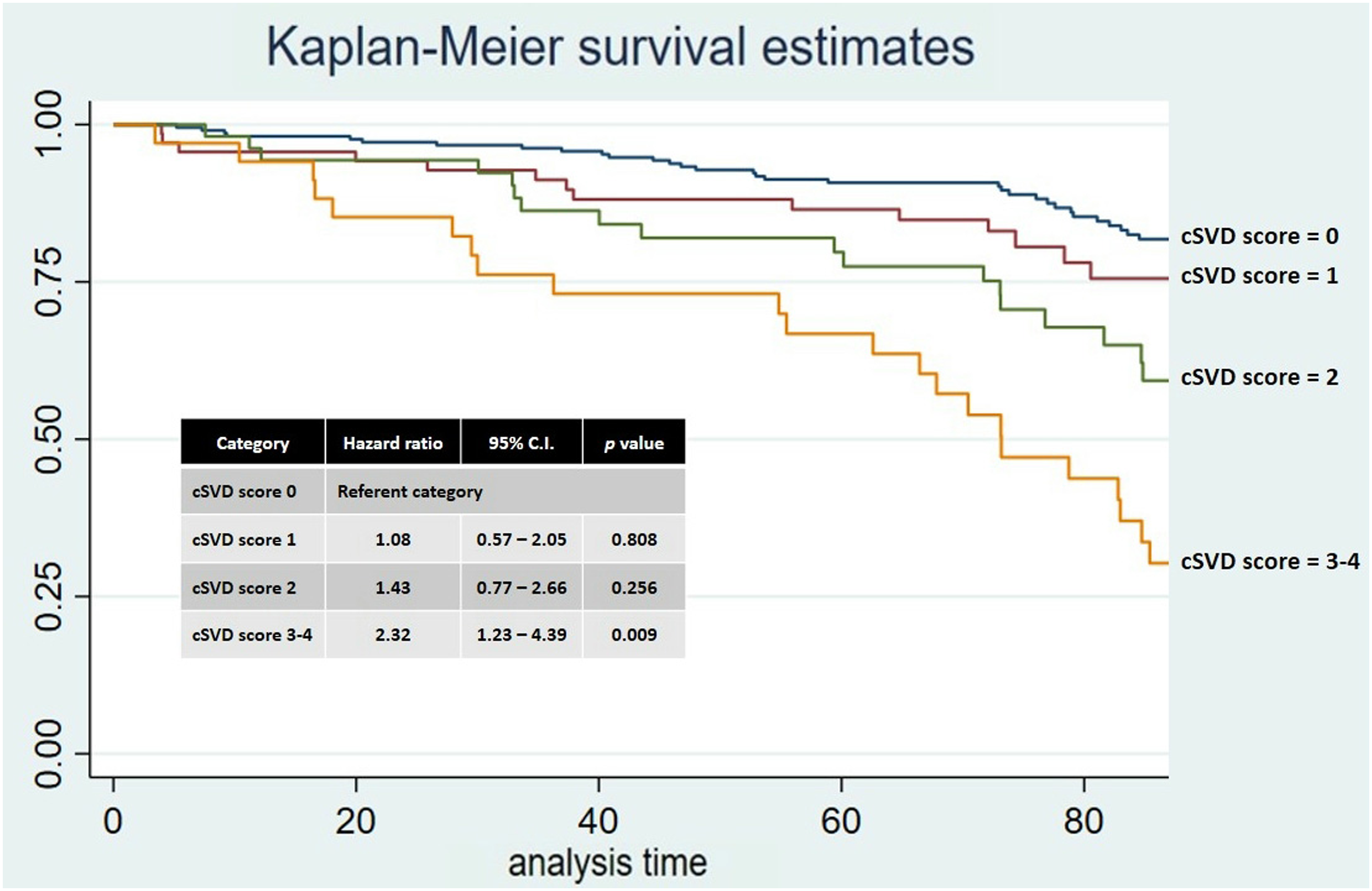
Separate univariate Cox proportional models for each one of the individual SVD biomarkers and the total SVD score showed that the latter model had the highest log likelihood and LR test (chi2). Furthermore, when the best predictor of mortality among individual components of the SVD score (WMH) was added to a model where tSVD score was already present, the latter remained significant (HR: 1.49; 95% CI: 1.13–1.96; p = 0.004) while the former became non-significant (HR: 1.56; 95% C.I.: 0.81–2.99; p=0.181).
Discussion
In a population-based cohort of non-disabled older adults of Amerindian ancestry living in a rural setting, high-burden SVD as determined by a tSVD score of 3–4 points predicted all-cause mortality independently of age, gender, level of education, and cardiovascular health. SVD biomarkers accumulation ascertained by the tSVD score showed to be a better mortality predictor than individual biomarkers alone, thus supporting the value of the tSVD score as a reliable visual scale to estimate SVD-related health consequences. Although several studies have addressed the risk of stroke and cognitive decline with the tSVD score,12,19–21 only few studies have reported its association with all-cause mortality.8–10
The reason why the accumulation of SVD biomarkers is a better predictor of mortality than individual biomarkers could be explained beyond the accumulation of vascular risk factors. The tSVD score denotes a more comprehensive view of the overall impact of SVD on the brain. 5 Although each of the SVD biomarkers denotes cerebral microvascular injury, distinct imaging biomarkers represent different pathogenic mechanisms which may increase the risk of stroke and cognitive decline individually.
Similar to our findings, the Rotterdam Study found that WMH and lacunes, but not EPVS or CMB, predicted mortality in community-dwelling older adults. 10 In the same study, the sum of SVD biomarkers was a better predictor of vascular events and mortality compared to single SVD biomarkers. However, mortality HR for a tSVD score of 3–4 tempered from 1.65 (95% CI: 1.04–2.06) to 1.39 (95% CI: 0.87–2.21) after adjusting for vascular risk. 10 Interestingly, our population of Amerindians showed a higher relative mortality risk when compared to the Rotterdam’s population despite comparable mean age and SVD prevalence, thus suggesting that the health impact of SVD may vary according to the underlying characteristics of the population studied. Other studies on the association between tSVD score and mortality focused in hospital-based populations8,9; therefore, results are not comparable to the present study.
In our cohort, higher tSVD scores were associated with hypertension and physical inactivity, both potential intervention targets to reduce SVD-related morbidity and mortality. Hypertension is known to be the most important contributor for developing SVD, and guidelines advocate strict blood pressure control to prevent SVD progression.22,23 However, a much-debated question is whether targeting blood pressure levels lower than standard targets is beneficial in subjects with SVD. 24 Previous research has established that physical activity relates to a lower incidence of stroke, cognitive decline, and mortality in individuals with established SVD.25,26 Although broadly recommended, other lifestyle interventions such as cognitive training, dietary supplements, weight loss, and smoking cessation have limited evidence showing benefit in individuals with SVD.22,23 On the other hand, deliberate use of antiplatelets, hypoglycemic, or anti-dementia medications are not recommended in individuals with SVD who otherwise have no indications for these therapies. 22 Therefore, despite the absence of high-quality evidence, cardiovascular risk control may result in cost-effective interventions in underserved populations to reduce adverse outcomes related to SVD.
Another factor that has been associated with a high tSVD burden in developed societies is illiteracy. 27 In the present study, low levels of education were associated with higher tSVD scores in unadjusted analysis, but educational disparities did not independently influence mortality rate in multivariate Cox-proportional models. The lack of effect could probably be related to the fact that almost 80% of the study population had primary school education only.
Study strengths include the population-based design, imaging acquisition uniform protocol, and the detailed study population characterization. Our SVD burden assessment might be limited by shortcomings related to the tSVD score, which weights each SVD biomarkers equally and ignores severity grading for each biomarker. Nevertheless, our study aligns with others in that the tSVD score is a reliable and practical tool to predict adverse outcomes, even in remote populations with limited access to healthcare. Other potential study limitations include low generalizability to other populations, potential selection bias related to declined consent and disability, and the absence of cause of death sub-analysis due to suspected inaccuracies in death certificate reports and limited number of participants.
Conclusion
In summary, the tSVD score is associated with all-cause mortality in community-dwelling Amerindians living in a rural setting, similar to that previously reported in Eastern Asian and Caucasian populations. Incidental diagnosis of covert SVD should prompt cardiovascular risk control as stated in current guidelines.22,23 Further trials on risk factors management are needed to identify SVD disease-modifying therapies with the potential to be applied in underserved populations, where cerebrovascular disease burden is on the rise. 11
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Study supported by Universidad Espíritu Santo—Ecuador. The sponsor had no role in the design of the study, nor in data collection or analyses, or in the decision to submit this work for publication.
Informed consent
All individuals enrolled in the Atahualpa Project signed comprehensive informed consent and were informed about the use of their anonymized data for publication.
Ethical approval
Guarantor
The corresponding author (OHD) takes full responsibility for the data, the analyses and interpretation, and the conduct of the research, and has full access to all of the data and the right to publish any and all data.
Contributorship
V.J.D.: Study design, imaging readings, and manuscript drafting; R.M.M.: statistical analysis of data; B.Y.R.: study coordinator; D.A.R.: data collection and interpretation; A.F.C.: data collection and interpretation; O.H.D.: study design, imaging readings, and significant intellectual contribution to manuscript content. All authors read and approved the final version of the submitted manuscript.
