Abstract
Introduction
Hyperintense acute reperfusion marker (HARM) is an indicator of early disruption of the blood-brain-barrier. Our aim was to investigate the incidence of HARM in patients with a diffusion weighted imaging (DWI) - fluid attenuated inversion recovery (FLAIR) mismatch and determine the association between this marker and hemorrhagic complications as well as clinical outcome.
Patients and Methods
We included patients from the Efficacy and Safety of MRI-Based Thrombolysis in Wake-Up Stroke (WAKE-UP) trial who underwent baseline perfusion weighted imaging (PWI). HARM was defined as a hyperintense signal in the cerebrospinal fluid space on FLAIR imaging at 24 h after baseline imaging. We compared baseline characteristics in patients with and without HARM and investigated the association between HARM and any hemorrhagic transformation (HT) and parenchymal hematoma (PH) in a multivariate logistic regression. We also explored HARM as an independent predictor of poor outcome, defined as a modified Rankin Scale of 3–6 at 90 days.
Results
HARM was present in 14 of 223 (6%) patients with a DWI-FLAIR mismatch and baseline characteristics were similar in patients with vs without HARM. HARM showed an independent relationship with any HT (OR 6.67; 95%CI 1.72–26.58) and any PH (OR 6.92; 95%CI 1.34–29.49). The rate of HARM was similar in patients with good and poor outcome (5%, p = 0.90).
Conclusion
In the WAKE-UP trial, the incidence of HARM was only 6% at 24 h. An association was present between HARM and hemorrhagic complications, but no relationship with functional outcome was observed.
Introduction
A paradigm shift towards tissue-based treatment strategies instead of relying on rigid time-windows has reshaped the current landscape of acute stroke care. The interest in imaging markers to guide treatment decisions and to predict tissue fate is increasing. Magnetic resonance imaging (MRI) can visualize both parenchymal and hemodynamic changes in ischemic brain tissue. Evaluating a mismatch between the DWI lesion vs the penumbral tissue, as visualized by perfusion weighted imaging (PWI-DWI mismatch),1–3 or in relation to the presence of vasogenic edema on FLAIR (the DWI-FLAIR mismatch) 4 are selection strategies to increase the amount of acutely treated stroke patients.
Another less frequently reported imaging characteristic is the hyperintense acute reperfusion marker (HARM) defined as a delayed sign of early blood-brain-barrier (BBB) disruption. Leakage of gadolinium contrast results in enhancement of the cerebrospinal fluid (CSF) compartment on postgadolinium FLAIR images; an altered function of the BBB is thought to be responsible for this phenomenon.5–8 The reported incidence of HARM in ischemic stroke patients is highly variable and ranges from 5.5% to 40.4%.5,6,9–12 No consensus exists on the association of HARM with hemorrhagic transformation (HT) and clinical outcome.5,6,10,11
We investigated the presence of HARM in patients with a DWI-FLAIR mismatch from the randomized Efficacy and Safety of MRI-Based Thrombolysis in Wake-Up Stroke (WAKE-UP) trial. 4 Furthermore, we explored the association of HARM with hemorrhagic transformation and functional outcome at 90 days.
Methods
Study design and patients
We analyzed imaging data from patients included in the WAKE-UP trial who underwent PWI at baseline. The WAKE-UP trial was an investigator-initiated, multicenter, randomized, double blind, placebo-controlled clinical trial. 4 Acute stroke patients with unknown stroke onset were randomized to alteplase (IV tPA) or placebo after assessing the presence of a diffusion restrictive lesion on DWI in the absence of a FLAIR hyperintense lesion (i.e. DWI-FLAIR mismatch). The standard imaging protocol at baseline consisted of MRI with at least DWI, FLAIR, time-of-flight magnetic-resonance angiography (TOF-MRA) and gradient echo (GE) or susceptibility weighted imaging (SWI). At the discretion of the local investigator, PWI using echoplanar T2* weighted sequence with gadolinium contrast bolus could be added to the MRI protocol. This was done subsequent to performing DWI and FLAIR imaging. Randomized patients, i.e. those with a DWI-FLAIR mismatch, received follow-up imaging similar to the baseline protocol at 22–36 h after randomization. PWI could be added to this protocol, after performing DWI and FLAIR imaging. National or local ethics committees or institutional review boards approved the trial. Informed consent was signed by patients or their legal representatives, according to national and local regulations.
Image analysis
In randomized patients receiving PWI and thus gadolinium contrast at baseline, we visually inspected FLAIR images at follow-up for the presence of HARM. HARM was defined as a hyperintense signal in the CSF space (sulci or ventricles) on more than two consecutive slices and without signs of subarachnoid hemorrhage on SWI or GE (Figure 1). Baseline DWI and PWI images were analyzed using RAPID software (IschemaView, Menlo Park, version 4.9 and 5.0), which calculates the ischemic core lesion based on an apparent diffusion coefficient (ADC) value <620*10−6 mm2/s and the area of critical hypoperfusion based on a time to maximum of the residue function (Tmax) with a threshold of > 6 s. A visual DWI lesion was mandatory for inclusion in the WAKE-up trial, but lesions were small and could therefore not always be detected by quantitative analysis resulting in DWI- and PWI-lesion volumes of 0 ml in some patients. For randomized subjects with available PWI at both baseline and follow-up and with a PWI deficit >0 ml at baseline, reperfusion was defined as a reduction of the baseline area with a Tmax >6 s of >50% at follow-up.
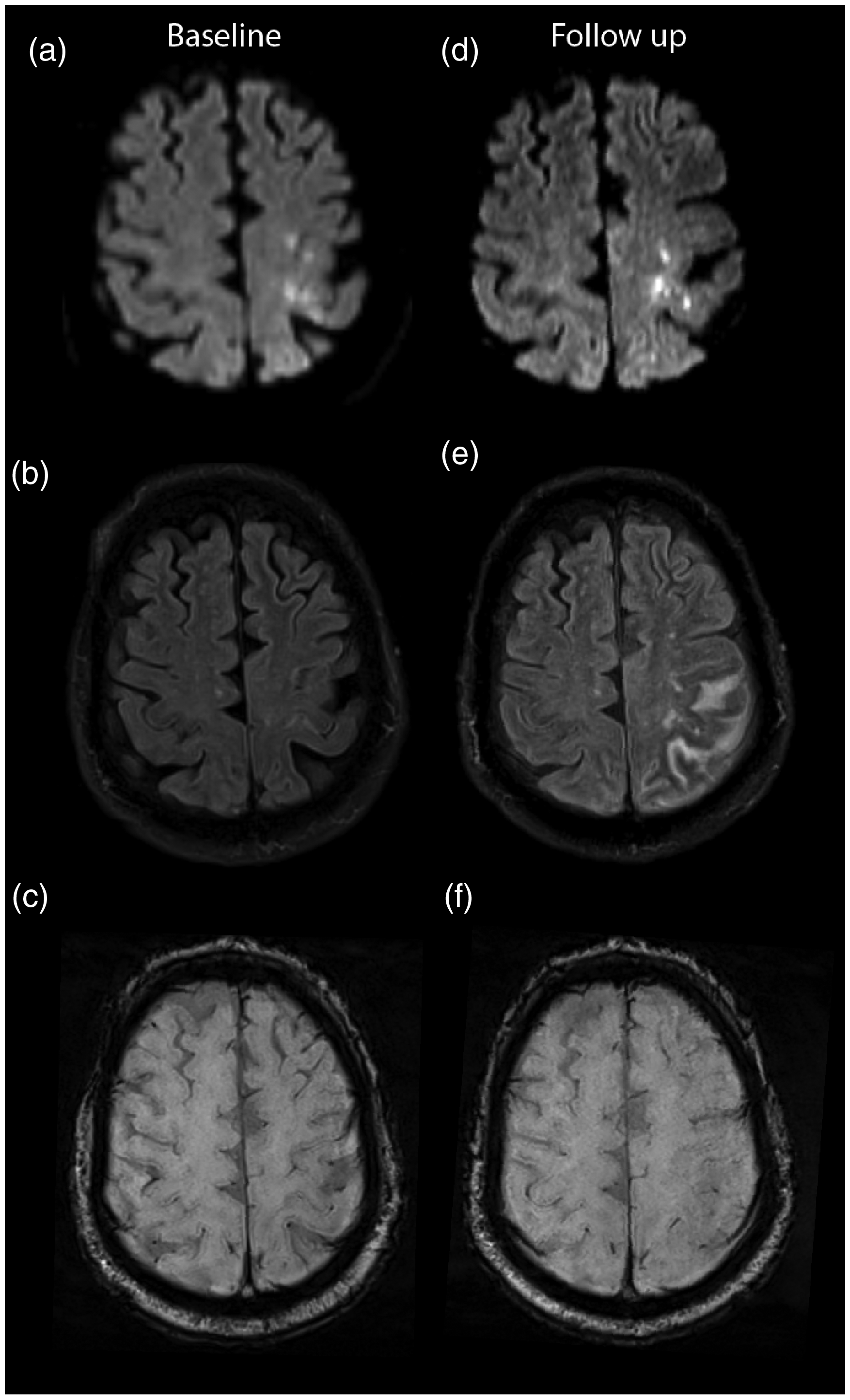
Baseline (a) and follow-up (d) diffusion-weighted imaging shows multiple cortical ischemic lesions left fronto-parietal, without (b) a correlating hyperintense lesion on fluid attenuated inversion recovery imaging (FLAIR) (i.e. a DWI-FLAIR mismatch). At follow-up imaging, (e) HARM can be seen on FLAIR as diffuse sulcal enhancement. Susceptibility weighted imaging at baseline (c) and follow-up (e) excludes subarachnoid hemorrhage.
Outcome measures
Any hemorrhagic transformation (HT) was defined according to the Heidelberg criteria as hemorrhagic transformation of infarcted brain tissue: hemorrhagic infarction (HI) type 1 and 2 (Class 1a and 1 b), parenchymal hematoma (PH) type 1 (Class 1c) or type 2 (Class 2). 13 Functional outcome was defined based on scores on the modified Rankin Scale (mRS) at 90 days: poor outcome as mRS 3–6 and good functional outcome as mRS 0–2.
Statistical analysis
We compared baseline characteristics with the Chi-Square test for categorical and the Mann–Whitney-U test for continuous variables. We constructed multivariate models for any HT, PH and poor outcome with backward logistic regression based on the Akaike information criterium (AIC). Age, the baseline National Institute of Health Stroke Scale (NIHSS), large vessel occlusion, HARM, DWI-volume, PWI-volume and treatment with IV tPA were introduced as explanatory variables. We used R for all statistical analyses.
Results
Baseline characteristics
We analyzed 223 patients of the 503 randomized patients (44%) in the WAKE-UP trial who underwent baseline perfusion imaging before randomization and in whom follow-up FLAIR and GE/SWI was available. The baseline characteristics did not differ between patients included in this analysis vs those excluded (Supplementary table 1). HARM was detected in 14 patients (6%). One of these patients had an additional FLAIR scan performed at the time of randomization, 5 min after the end of the PWI. At that moment, HARM was already present for this patient, but less pronounced compared to the FLAIR scan at 24 h (Supplementary figure 1). Relevant baseline characteristics did not differ between patients with vs without HARM (Table 1). Reperfusion based on follow-up PWI was present in 49 of 69 patients (71%). We could not identify an association between reperfusion and HARM (p = 0.33).
Baseline characteristics and HT in patients with vs without HARM.
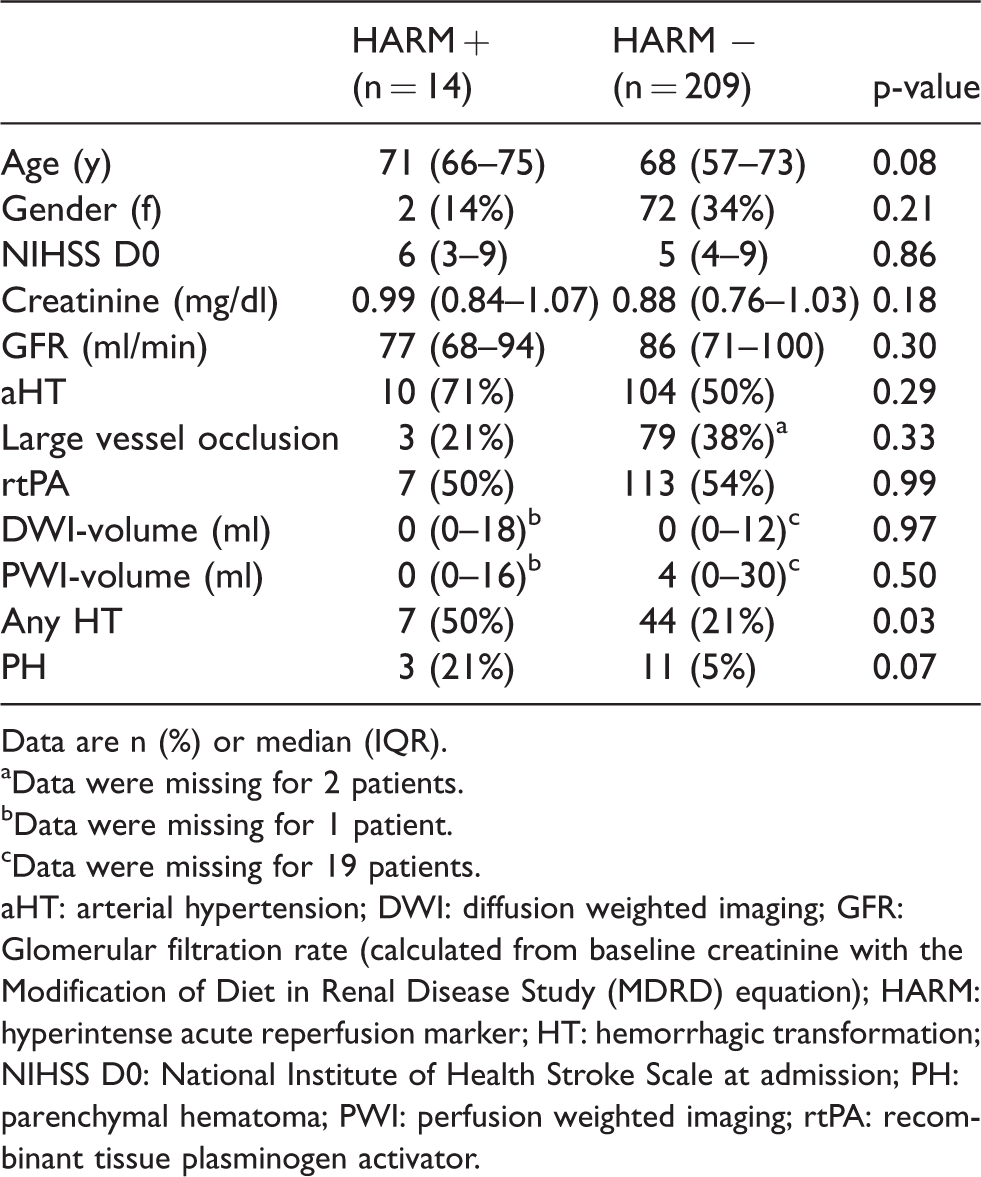
Data are n (%) or median (IQR).
aData were missing for 2 patients.
bData were missing for 1 patient.
cData were missing for 19 patients.
aHT: arterial hypertension; DWI: diffusion weighted imaging; GFR: Glomerular filtration rate (calculated from baseline creatinine with the Modification of Diet in Renal Disease Study (MDRD) equation); HARM: hyperintense acute reperfusion marker; HT: hemorrhagic transformation; NIHSS D0: National Institute of Health Stroke Scale at admission; PH: parenchymal hematoma; PWI: perfusion weighted imaging; rtPA: recombinant tissue plasminogen activator.
Association of HARM with hemorrhagic transformation, parenchymal hematoma and poor outcome
At follow-up imaging, any HT was revealed in 51 out of 223 patients (23%) (Figure 2). The presence of HARM was higher in patients with any HT (n = 7, 14%) compared to patients without any HT (n = 7, 4%; (p = 0.03)). This association remained present in multivariate analysis (OR 6.67, 95% CI 1.72–26.58). Other independent predictors of any HT were treatment with thrombolysis, DWI-volume, age, NIHSS and the presence of a large vessel occlusion (Table 2). PH occurred in 3 (21%) patients with HARM versus 11 (5%) patients without HARM (Figure 2). In multivariate analysis HARM and DWI-volume were independently associated with PH (Table 2).
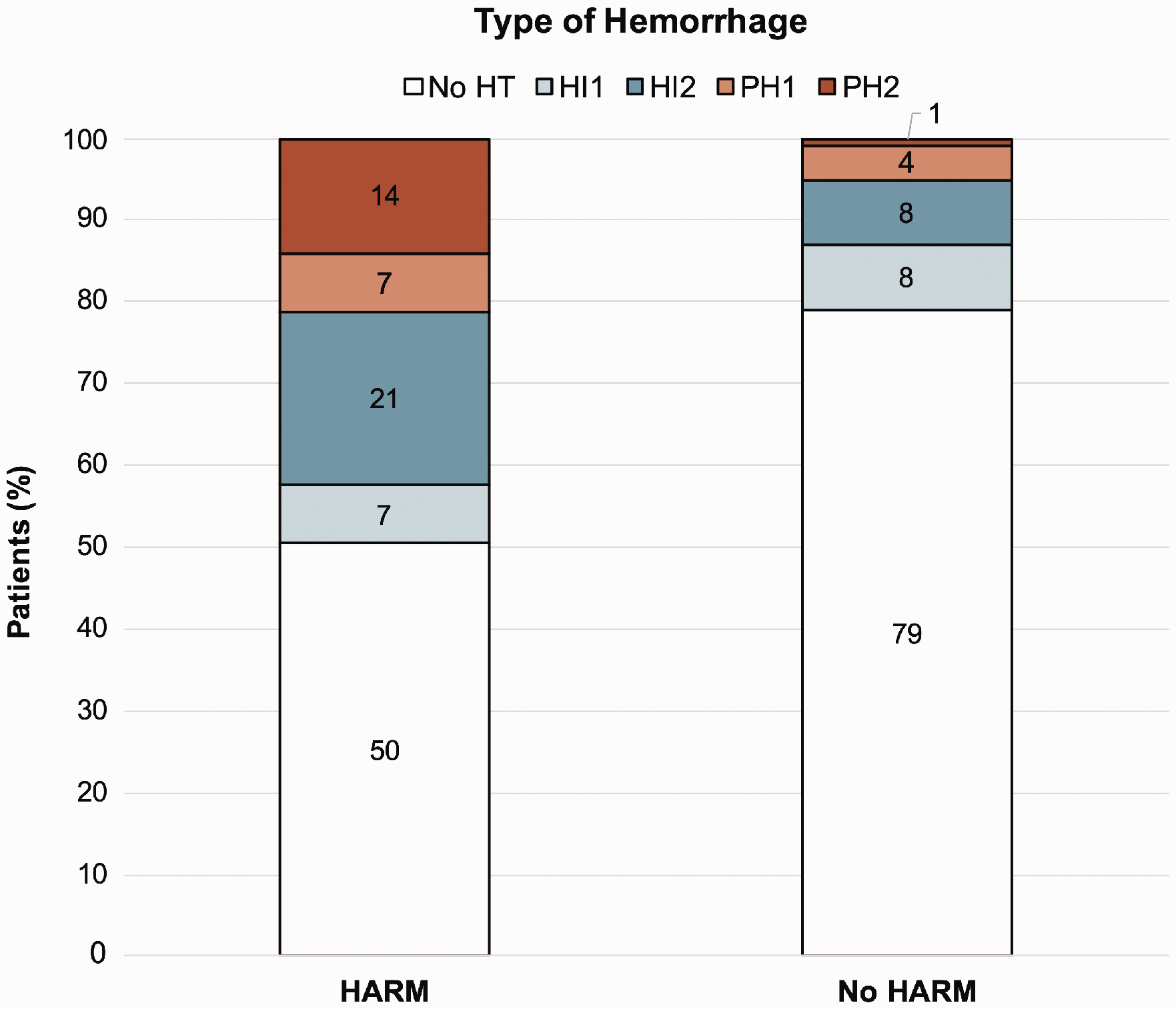
Distribution of hemorrhage types for HARM positive and HARM negative patients.
Results of backward logistic regression.
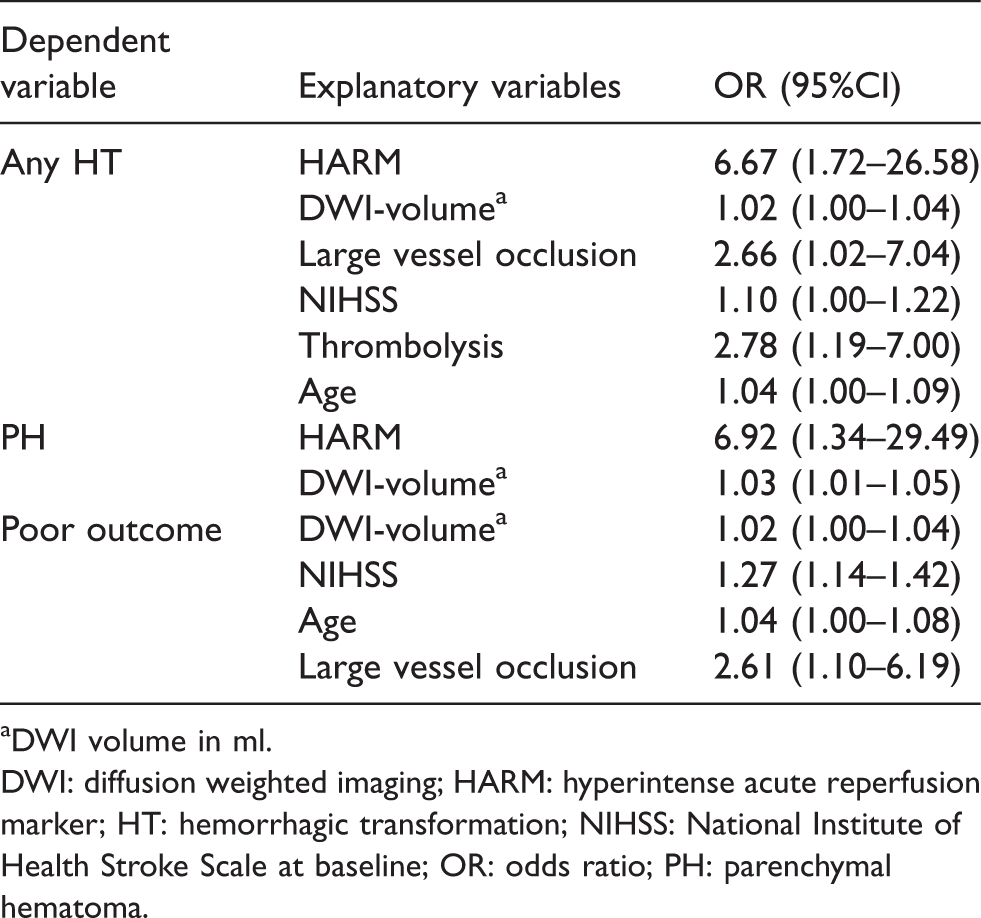
aDWI volume in ml.
DWI: diffusion weighted imaging; HARM: hyperintense acute reperfusion marker; HT: hemorrhagic transformation; NIHSS: National Institute of Health Stroke Scale at baseline; OR: odds ratio; PH: parenchymal hematoma.
At 90 days, poor outcome was observed in 59 patients (26%). The rates of HARM in patients with poor outcome (n = 3, 5%) vs good functional outcome (n = 11, 5%) were similar (p = 0.90). The association between HARM and outcome remained absent after adjusting for other predictors of poor outcome (large vessel occlusion, age, DWI volume and baseline NIHSS) (Table 2).
Discussion
In this descriptive study of the WAKE-UP trial, HARM occurred in 6% of ischemic stroke patients with a DWI-FLAIR mismatch. We identified an association with any hemorrhagic transformation and with parenchymal hematoma, but not with outcome at 90 days.
The incidence of HARM in our study was low compared to previous studies. However, our analysis was limited to randomized patients with a DWI-FLAIR mismatch, a marker of recent stroke onset. 14 In these subjects the absence of a FLAIR hyperintense signal within the DWI lesion is an indicator of cytotoxic edema without ionic and vasogenic edema. The combination of these pathological changes could eventually lead to BBB reversible leakage, irreversible breakdown and potentially even hemorrhage in some patients. 15 The incidence of HARM in acute stroke patients without a DWI-FLAIR mismatch who likely present later after onset, although of interest, could not be determined since follow-up imaging in WAKE-UP was only available for randomized patients (i.e. patients with a DWI-FLAIR mismatch).
Other variables such as time between administration and follow-up imaging, stroke lesion characteristics, the dosage of gadolinium contrast and reperfusion status might also explain the variability in the incidence rates of HARM.10–12 Early FLAIR imaging after gadolinium contrast administration could result in too low gadolinium contrast concentrations in the CSF. 9 In the WAKE-UP trial follow-up imaging occurred 22–36 h after baseline imaging which seems a sufficient enough time interval between PWI and follow-up FLAIR. Indeed, HARM may be less easily visible early after the administration of gadolinium as confirmed in one patient for whom early and late FLAIR was available. The rate of HARM is lower in patients with smaller infarcts and in general patients randomized in the WAKE-UP trial had minor to moderate infarct volumes which could partially explain the low rate of HARM.9,11,12 One study reported an increased percentage of HARM with higher dosages of gadolinium contrast, in patients who underwent both contrast enhanced imaging of the cervical arteries and PWI resulting in a double dosage of gadolinium contrast. 11 Because we did not collect gadolinium contrast dosages in WAKE-UP patients, we lack data to draw any conclusion on an association between dosage of gadolinium and HARM.
HARM was independently associated with HT and PH in multivariate analysis, although the small number of patients with any HT or PH was a limitation. The small numbers also prevented from studying possible associations of HARM with all the different radiological subtypes of HT. The early timing of follow-up imaging may have led to a lower incidence of HT since this can occur over a longer period than the 24 h in the WAKE-UP trial. Previous reports are contradictory on the association of HARM with hemorrhagic transformation.5,6,10,11 In contrast to these studies, WAKE-UP was a randomized study, which overcomes any selection bias in treatment allocation. In addition, in our study with blinded outcome assessment, HARM was not associated with functional outcome. Based on our data the presence of HARM should raise awareness for potential hemorrhagic transformation. However currently there is no evidence that identifying HARM can predict poor outcome.
Given the low incidence of HARM and hemorrhagic complications, the small sample size is a major limitation of our study and the results should be interpreted with caution. Another limitation is the lack of PWI in all randomized patients. Hence, a certain selection bias cannot be excluded.
In conclusion, for patients with a DWI-FLAIR mismatch, the incidence of HARM was lower compared to previously reported in other patient cohorts. Although HARM was associated with hemorrhagic complications, we found no association with outcome after ischemic stroke.
Supplemental Material
sj-pdf-1-eso-10.1177_23969873211007686 - Supplemental material for Hyperintense acute reperfusion marker associated with hemorrhagic transformation in the WAKE-UP trial
Supplemental material, sj-pdf-1-eso-10.1177_23969873211007686 for Hyperintense acute reperfusion marker associated with hemorrhagic transformation in the WAKE-UP trial by Anke Wouters, Lauranne Scheldeman, Patrick Dupont, Bastian Cheng, Martin Ebinger, Märit Jensen, Matthias Endres, Christian Gerloff, Keith W Muir, Norbert Nighoghossian, Salvador Pedraza, Claus Z Simonsen, Florent Boutitie, Vincent Thijs, Götz Thomalla, Jochen Fiebach and Robin Lemmens in European Stroke Journal
Footnotes
Acknowledgements
WAKE-UP Steering Committee: CG (Chair, Coordinator of WAKE-UP), GT (Coordinating Investigator of WAKE-UP), MEb, CS, SP, RL, NN, KM, IF, and MEn.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: GT received Consulting fees from Acandis, grant support and lecture fees from Bayer, lecture fees from Boehringer Ingelheim, Bristol-Myers Squibb/Pfizer, and Daiichi Sankyo, and consulting fees and lecture fees from Stryker.
CG received funding from German Research Council (DFG), European Union, Federal Ministry of Education and Research (BMBF), German Statutory Pension Insurance Scheme (RV Nord), National Innovation Fond, Wegener Foundation, and Schilling Foundation; he received honoraria as speaker or consultant from Abbott, Amgen, Bayer Vital, Bristol-Myers-Squibb, Boehringer Ingelheim, Sanofi Aventis, and Prediction Biosciences.
JF reports consulting and advisory board fees from BioClinica, Cerevast, Artemida, Brainomix, Biogen, BMS, and EISAI, outside the submitted work.
KM has participated in advisory boards for Bayer, Boehringer-Ingelheim and Daiichi-Sankyo; and receives research support from Boehringer-Ingelheim for the ATTEST-2 trial.
ME reports grant support from Bayer, and fees paid to the Charité from Bayer, Boehringer Ingelheim, Bristol-Myers Squibb, Pfizer, Daiichi Sankyo, Amgen, GlaxoSmithKline, Sanofi, Covidien, Novartis, all outside the submitted work.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: WAKE-UP received funding from the European Union Seventh Framework Program (FP7/2007–2013) under grant agreement 278276. LS is supported by a fund for Scientific Research Flanders (FWO, PhD fellowship fundamental research grant number1193620N). RL is a senior clinical investigator of FWO Flanders. CS was funded by a grant from Novo Nordisk Foundation.
Informed consent
Informed consent was signed by patients or their legal representatives, according to national and local regulations.
Ethical approval
National or local ethics committees or institutional review boards approved the trial.
Guarantor
AW.
Contributorship
All authors were responsible for acquisition of data. AW, LS, PD, JF, GT, RL, and MJ were responsible for analyzing the data. AW and LS wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
