Abstract
Introduction
The global incidence of ischemic stroke among patients with human immunodeficiency virus is increasing. The aim of this controlled case analysis was to study patient characteristics, stroke etiologies, and risk factors in human immunodeficiency virus-positive patients in a medical system with easy access to antiretroviral therapies.
Patients and methods
We conducted a retrospective, observational study of human immunodeficiency virus-positive patients treated in our stroke unit and outpatient clinic in Germany between 2012 and 2018. A control group of all patients treated for acute ischemic stroke in 2018 was used to elicit possible differences in stroke localization, etiology, and distribution of risk factors.
Results
Out of 3615 patients with an acute ischemic stroke, 24 (0.7%) were newly or previously diagnosed as human immunodeficiency virus-positive. Strokes in this cohort were caused by large-vessel disease (37.5%), small-vessel disease (20.8%), cryptogenic-embolism (20.8%), vasculitis (16.7%), and cardio-embolism (4.2%). Large-vessel disease-related strokes were more often located in the posterior circulation (77.8%). Compared to the control group, cardio-embolic strokes were less and vasculitis and large-vessel disease more frequent. Human immunodeficiency virus-positive patients were younger at stroke onset (53.7 ± 12.8 vs. 70.2 ± 14.6 years, p = 0.05).
Discussion
Although the prevalence of human immunodeficiency virus-infection among patients treated for acute ischemic strokes appears low, this collective merits special attention due to a high burden of potentially modifiable risk factors and vasculitis.
Conclusion
Compared to previous studies, age at stroke onset was one of the highest, supporting the hypothesis that as antiretroviral therapy increases the life expectancy of people living with HIV/AIDS, they will be increasingly affected by cardiovascular disease. Human immunodeficiency virus-related cerebral vasculitis was associated with high mortality (75%).
Introduction
Medical progress not only alleviates individual suffering, but can have profound effects on the population as a whole leading to demographic changes. For example, the emergence of anti-infective agents in the 20th century has improved overall life expectation. More recently, this has also been true for people living with HIV/AIDS (PLWHA) due to antiretroviral therapy (ART). These advances have caused a measurable change of morbidity with increasing prevalence of non-acquired immune deficiency syndrome (AIDS) complications such as cardiovascular disease and diabetes. 1 On the other hand, this might lead to an increasing prevalence of age-related diseases such as stroke. Moreover, the incidence of ischemic strokes in human immunodeficiency virus (HIV)-positive patients appears to be higher than in the general population, and the age at onset appears to be younger. 2 Chronic infections as well as antiretroviral medication itself seem to increase the risk of stroke by mechanisms that are not yet fully resolved. Pathophysiological explanations include atherosclerosis fostered through chronic inflammation or dyslipidemia due to antiretroviral medication. 3 The assumption of a specifically HIV-associated vasculopathy and its discrimination from common atherosclerosis and vasculitis has been discussed previously. 4
Our aim was to provide information about the characteristics of HIV-infected patients with stroke treated at a German stroke unit and to put this into the context of case series and observational studies published worldwide to describe differences that may be linked to socioeconomic standards in general and access to antiretroviral therapy in particular.
Methods
We performed a retrospective observational study on patients treated in the department of Neurology, University Hospital Frankfurt, between 2012 and 2018. Ischemic stroke was defined clinically by sudden onset of a new neurological deficit and an acute ischemic lesion found on cranial CT or MRI. Transitory ischemic attacks were excluded. HIV-infection was defined by positive serological test either for antibodies (HIV enzyme immunoassay) and confirmed Western blot or detection of HIV–RNA (polymerized chain reaction). Patients were identified by a hospital-wide databank search using an ICD-10 code combination I63 for ischemic stroke and B20/21/22/23/24 or Z21 for HIV-infection. For the classification of stroke etiology, we employed a previously described algorithm of HIV-related ischemic stroke for research studies. 4 All ischemic strokes treated in 2018 served as a control group. Except for exclusion of those patients with known HIV-positive status, no further selection was performed. The retrospective study design has been approved by the local ethics committee (file number 46/19) on 5 March 2019.
The manuscript was drafted in accordance with the guidelines of Strengthening the reporting of Observational Studies in Epidemiology (STROBE). 5 Data were analyzed using SPSS (IBM, Version 22). Descriptive statistics (mean, standard deviation (SD)) were used to describe the baseline data. Normally, distributed data were compared by Student’s t-test and non-parametric data by Mann–Whitney U or Kruskal–Wallis test. To compare categorical or proportional characteristics, χ2 tests were used. All tests were two-sided with P values < 0.05 considered significant.
Literature search: studies were identified performing a PUBMED search using the following term: ((HIV) OR (human immunodeficiency virus)) AND ((stroke) OR (cerebral ischemia)) AND ((case series) OR (cohort) OR (retrospective analysis) OR (prospective analysis)). No date limits were applied. Additional studies were chosen from the reference list of identified papers. Pediatric or post-mortem studies were excluded.
Results
Twenty-four patients were treated for acute ischemic stroke between 2012 and 2018 with a diagnosis of HIV prior to or during diagnostic work-up for the index event. There was a total of 3651 patients with an ICD-10-diagnosis of I63; hence, the estimated prevalence of positive HIV-status was 0.66%. In the reference year 2018, 47.4% of stroke patients were tested for HIV. The stroke etiologies were distributed as follows: large-vessel disease n = 9 (37.5%), small-vessel disease n = 5 (20.8), vasculitis n = 4 (16.7%), cardio-embolism n = 1 (4.2%), and cryptogenic-embolism n = 5 (20.8%). Two cases of cerebral vasculitis were deemed to be of infectious origin due to treponema pallidum in one and aspergillus fumigatus as well as nocardia cyriacigeorgigica in the other patient. One cardio-embolic stroke was caused by bacterial endocarditis. Despite comprehensive diagnostic-workup consisting of at least 24-h Holter–ECG monitoring, transesophageal echocardiography, vasculitis- and thrombophilia screening, and color-coded duplex sonography, the source of five cases with a presumably embolic stroke could not be determined and were therefore classified as cryptogenic embolism.
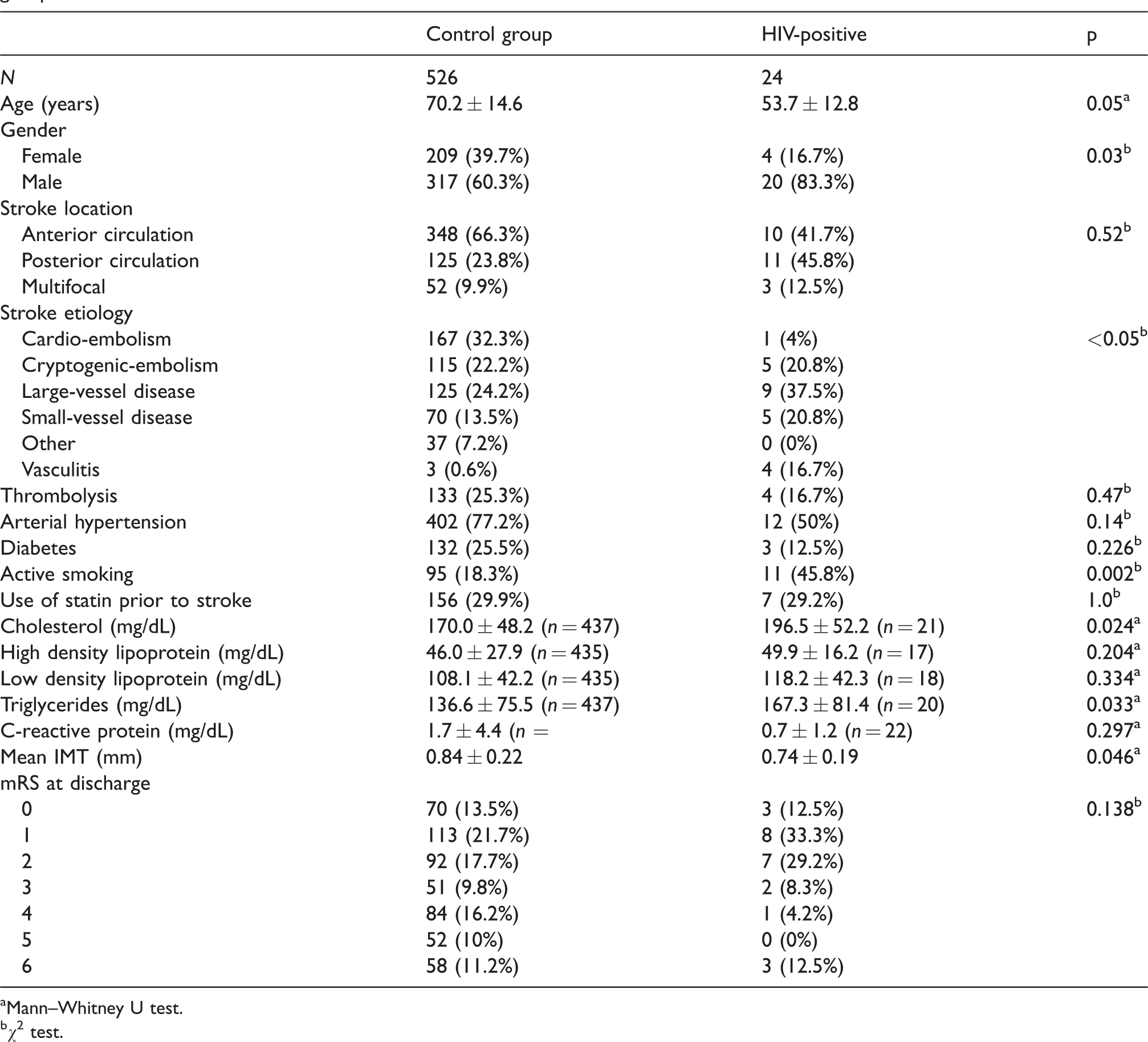
Mean age at stroke onset was substantially younger in patients with HIV compared to the pre-specified control group (53.7 ± 12.8 years vs. 70.2 ± 14.6 years; p = 0.05, Mann–Whitney U test) (Table 1). Other significant differences included a higher percentage of large-vessel disease in HIV-positive patients (37.5% vs. 24.2%; p < 0.05, χ2 test), and vasculitis was higher in HIV-positive patients, who in turn experienced less cardio-embolic strokes (Figure 1). The distribution of stroke locations—classified as anterior or posterior circulation or multifocal lesions in multiple territories—did not reveal any significant differences with only a numerically higher proportion in the posterior circulation for HIV-positive patients (Figure 2). Vasculitic stroke in particular occurred in multiple territories in half of cases (2/4) with the other half being localized in the anterior circulation. Overall, HIV-associated strokes were slightly more frequent in the posterior (n = 11, 45.8%) compared to the anterior circulation (n = 10, 41.7%) and multifocal strokes (n = 3, 12.5%). Notably, strokes attributed to large-vessel disease were more common in the posterior circulation (7/9 patients, 77.8%).
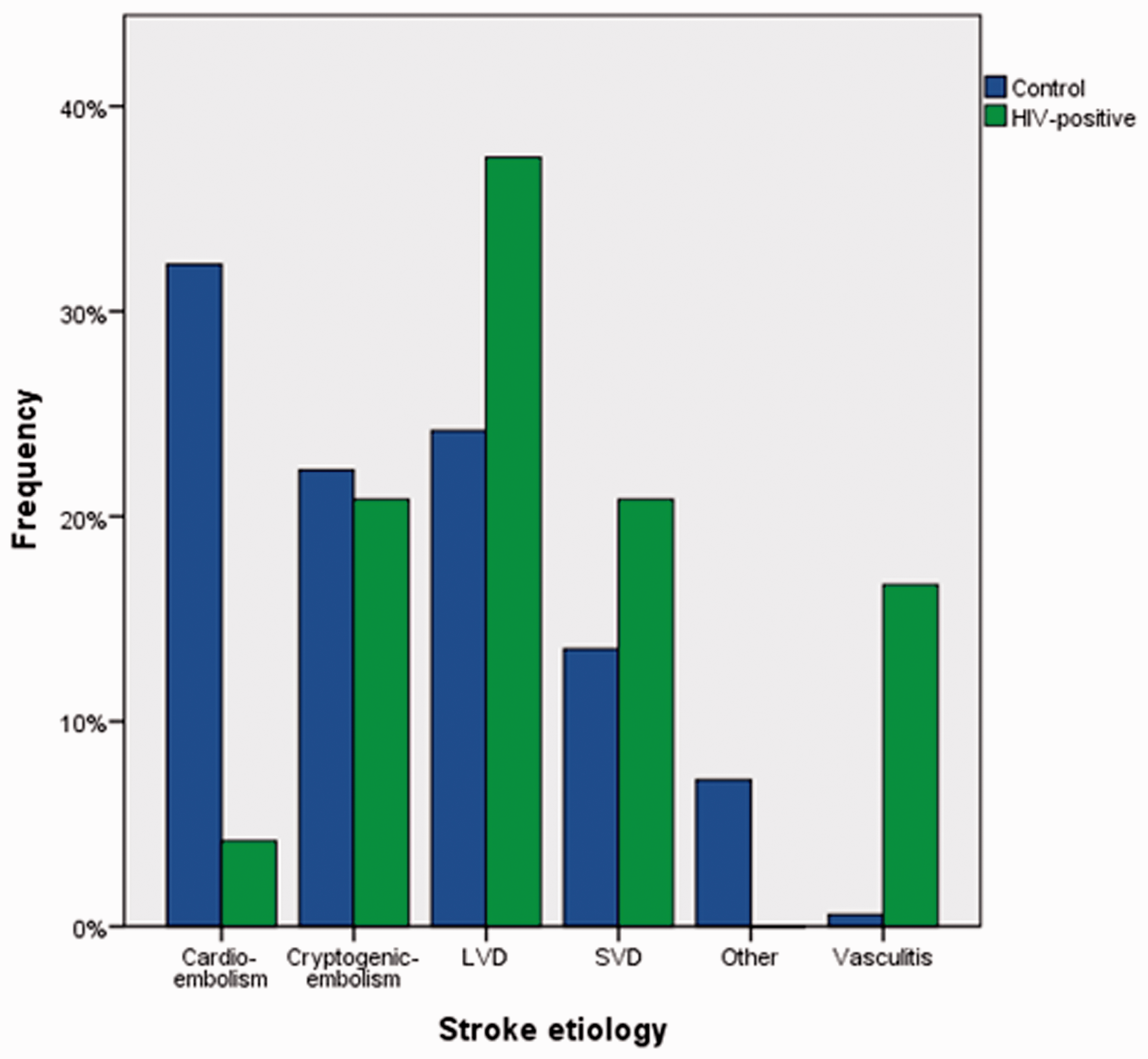
Distribution of stroke etiologies for the control group (acute strokes in 2018, n = 526) and HIV-positive stroke patients (n = 24).
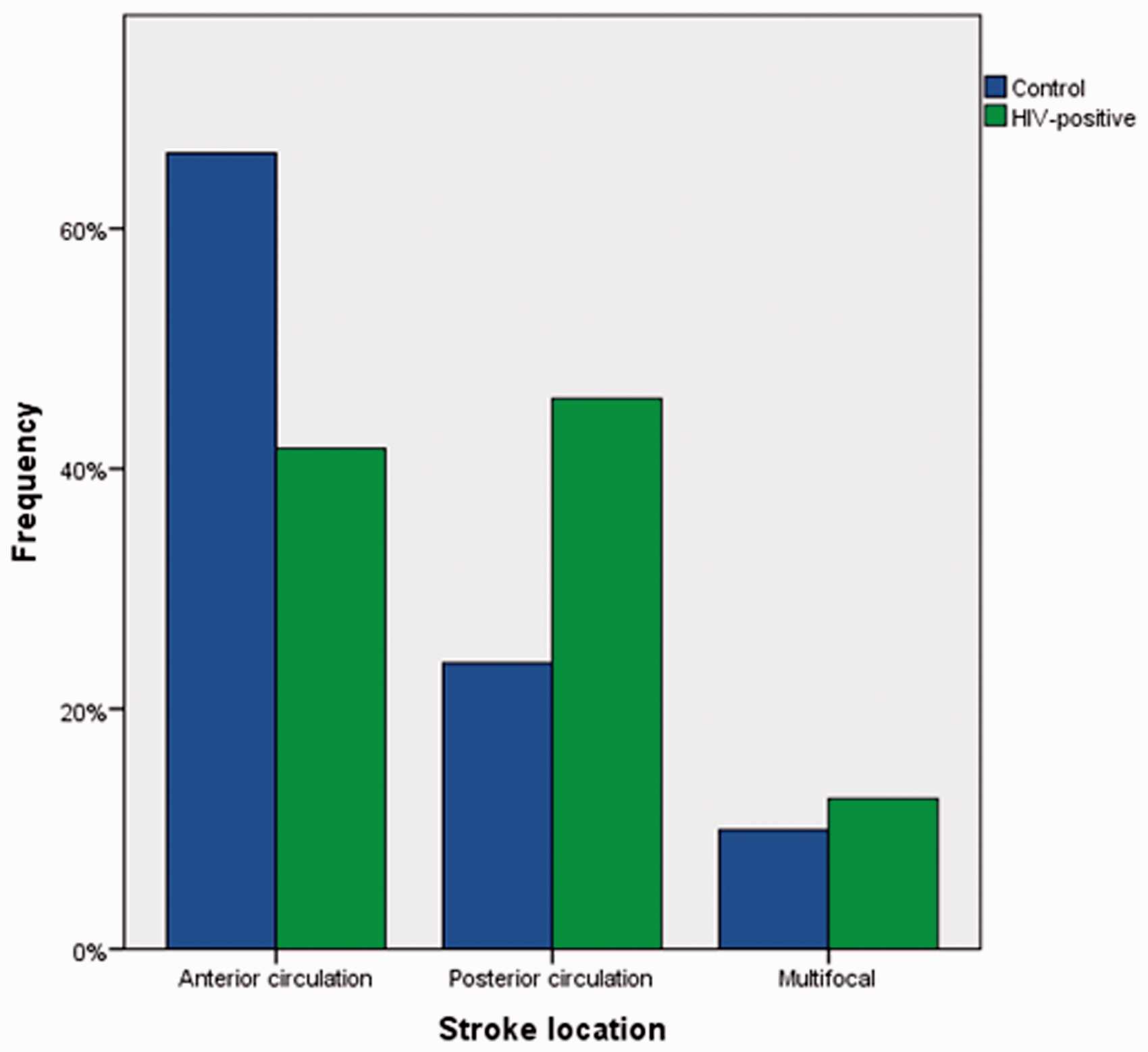
Distribution of stroke location for the control group (acute strokes in 2018, n = 526) and HIV-positive stroke patients (n = 24).
Regarding vascular risk factors, the prevalence of arterial hypertension in HIV patients was 50% with a higher proportion in small-vessel disease (80%, Table 2). Similarly, the rate of confirmed diabetes was numerically higher for small-vessel disease (40% vs. 13% in all patients), though not reaching statistical significance. Active smoking was more frequent in the HIV group compared to controls (45.8% vs. 18.3%; p = 0.002, χ2 test). Cholesterol levels (170.0 ± 48.2 mg/dL vs. 196.5 ± 52.2 mg/dL; p = 0.024, Mann–Whitney U test) and triglycerides (136.6 ± 75.5 mg/dL vs. 167.3 ± 81.4 mg/dL; p = 0.033, Mann–Whitney U test) were higher in HIV patients compared to the control group.
The intima media thickness (IMT) of both common carotid arteries was measured in 23 of 24 patients with a mean ± SD value of 0.74 ± 0.19, significantly lower as in the control group (0.84 ± 0.22; p = 0.046, Mann–Whitney U test). There was a significant correlation in both groups between the IMT and the number of comorbidities such as hypertension, diabetes, and smoking in HIV-positive patients: Spearman r = 0.496, p = 0.019, n = 22; controls: Spearman r = 0.255; p < 0.05; n = 451. After adjusting for age, the correlation was still significant in the HIV-negative cohort (standardized coefficient β = 0.22, p < 0.05), but age had a stronger effect on IMT (standardized coefficient β = 0.33, p < 0.05). In the HIV-positive cohort, the correlation was not significant when adjusted for age (standardized coefficient β = 0.75, p = 0.46).
No significant differences were found comparing the rate of thrombolysis, arterial hypertension, diabetes, use of statins prior to stroke, level of C-reactive protein at admission, and modified Rankin scale at discharge (p > 0.05, respectively).
Comparison of patient factors and stroke characteristics between confirmed HIV-positive patients and pre-specified control group.
aMann–Whitney U test.
bχ2 test.
Comparison of patient and stroke characteristics with confirmed HIV-infection according to stroke etiology.
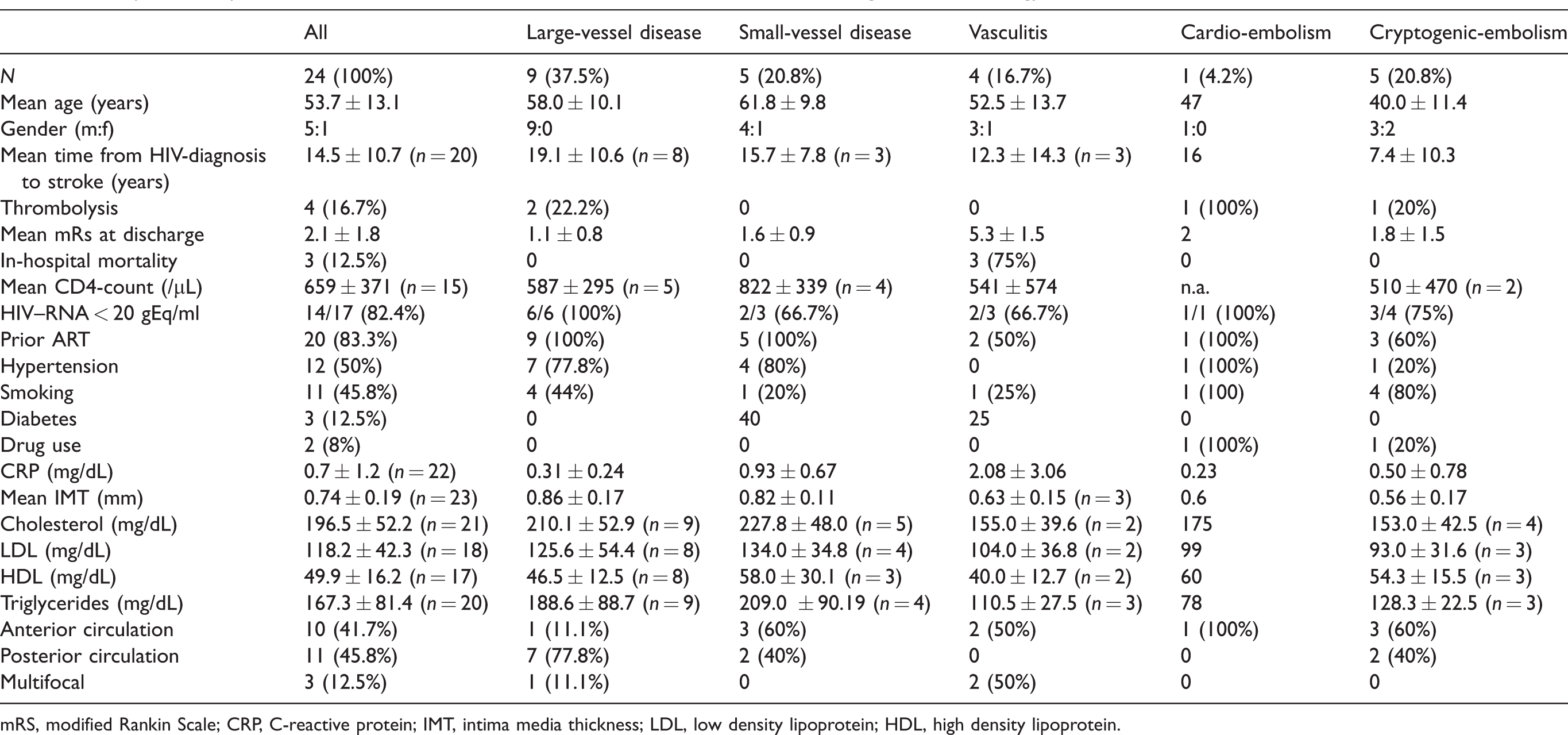
mRS, modified Rankin Scale; CRP, C-reactive protein; IMT, intima media thickness; LDL, low density lipoprotein; HDL, high density lipoprotein.
Mean CD4-counts were available in 14 patients and, on average, demonstrated a competent immune system (659 ± 371/µl). Prior to their stroke event, 83.3% of patients were regularly taking ART, which led to non-detectable HIV–RNA levels in 82.4% of patients (detection limit: 20 gEq/ml). These drugs were in order of frequency: two nucleoside reverse-transcriptase inhibitors (NRTI) and one integrase inhibitor (INI) in 6/20 cases (30%), two NRTIs and one non-nucleoside reverse-transcriptase inhibitor (NNRTI) in 5/20 cases (25%), two NRTIs and at least one protease inhibitor (PI) in 4/20 cases (20%), one INI and one PI in 3/20 cases (15%), and last, individual combinations of NNRTI + INI + CCR5-inhibitor (one patient) and PI + CCR5-inhibitor + Cobicistat (one patient).
Within 4.5 h after stroke onset, 16.7% of HIV-positive patients received recombinant tissue plasminogen activator (rtPA). The subsequent median score on the modified Ranking scale (mRS) at discharge did not differ significantly compared to patients who did not receive thrombolysis (2.0 vs. 1.5; p = 0.84, Kruskal–Wallis test). The mean mRS at discharge was 2.1 ± 1.8.
Mortality was significantly higher in patients with vasculitis as compared to other stroke etiologies (75% vs. 0%; p = 0.002, χ2 test, Figure 3). Other recorded baseline variables did not differ significantly between vasculitis- and non-vasculitis-associated strokes (percentage of prior antiretroviral therapy (ART), mean CD4-counts, mean serum HIV–RNA copies, leukocyte count in cerebrospinal fluid, and CRP; Table 3).
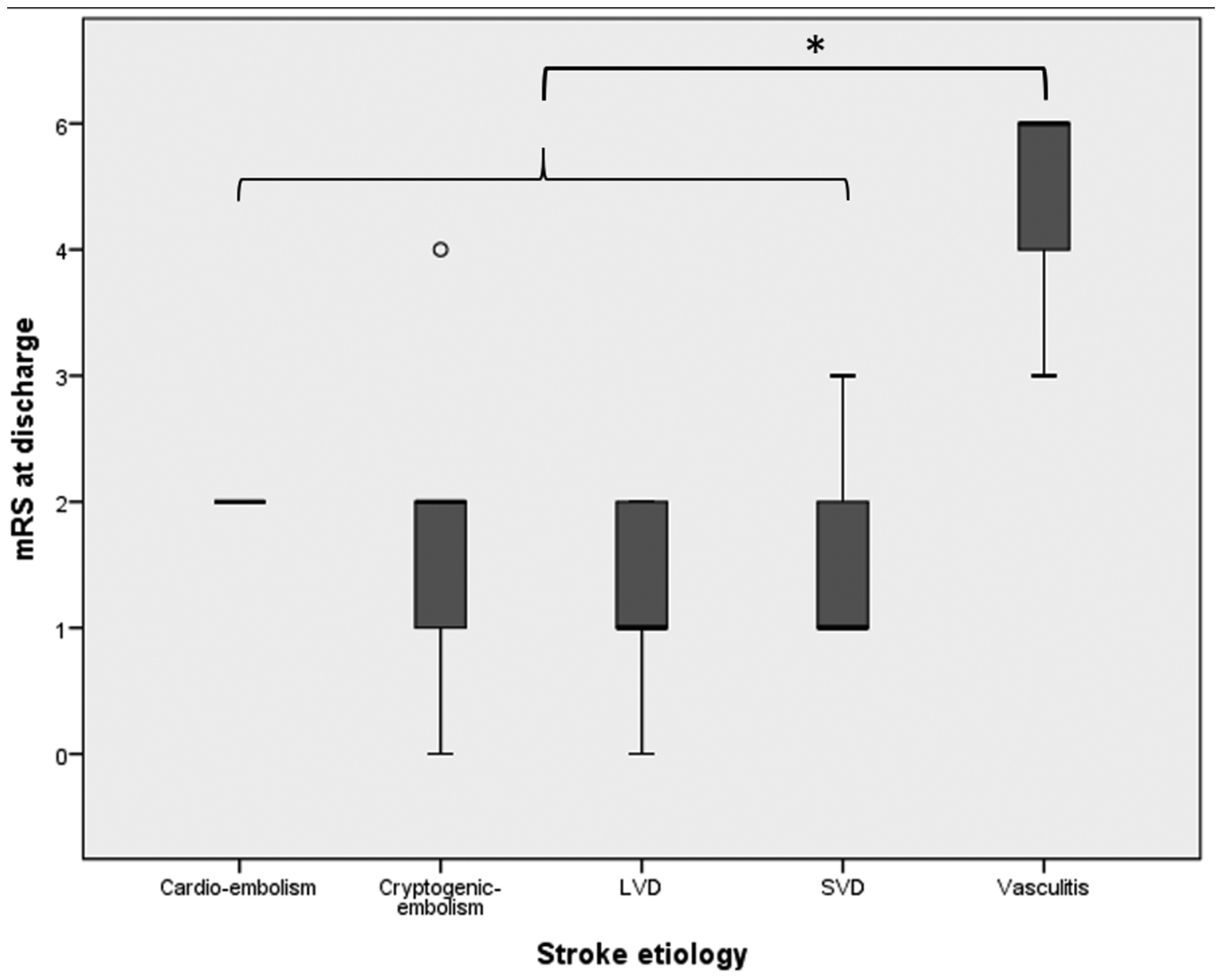
Clinical outcome according to the different stroke etiologies in HIV-positive patients. * = significant difference, p = 0.002.
Comparison of vasculitic- and non-vasculitic strokes.
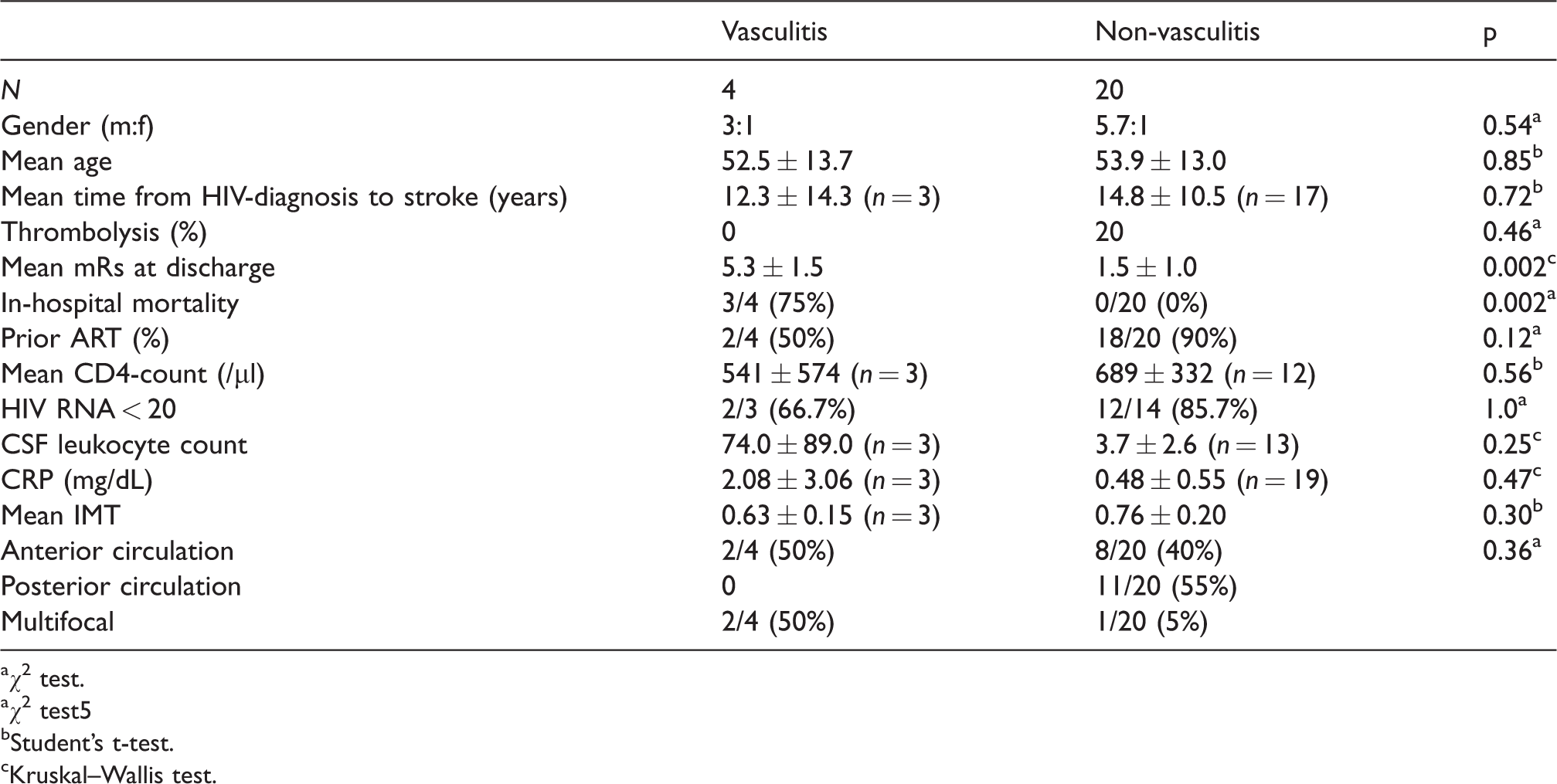
aχ2 test.
aχ2 test5
bStudent’s t-test.
cKruskal–Wallis test.
Discussion
We present a case series of 24 HIV-positive patients, who were treated at our department with ischemic strokes of different etiologies. The most common cause of stroke in this collective was large-vessel disease, which supports the hypothesis that accelerated atherosclerosis might be a hallmark of HIV-infection. The estimated prevalence of HIV-positive patients among all stroke patients from 2012 to 2018 was 0.66%. This is 6.6-fold higher than the general prevalence of HIV-infection in Germany. 6 However, the location of our hospital in a metropolitan area has to be taken into account. Males were even more frequently affected in the HIV+ group (83.3% vs. 60.3% in the control group, p = 0.3). This finding is in line with the general trend toward a higher number of men with HIV and closely reflects the German HIV statistics in 2018 of 80.3%. 6
Comparison of patient characteristics, stroke etiologies, and risk factors between the HIV-positive patients and the pre-specified control group yielded some interesting differences. Ischemic strokes tended to occur earlier in life for HIV patients (mean onset 53.7 ± 12.8 years vs. 70.2 ± 14.6 in the control group; p = 0.05). Our collective was slightly older than prior descriptions from other European countries (Germany: 477–51 years; 8 Spain 46 years; 9 Portugal 51 years; 10 Table 4). Across all reported studies, mean ages ranged from 28 to 75 years. Compared to European countries, the mean age of strokes patients in sub-Saharan African countries appears to be much younger (e.g. Malawi 3911–40 years 12 and South Africa 3213–33 years 14 ), though the reasons for this seem unclear and have to be correlated with general baseline characteristics of African stroke patients.
Previous case series or cohort studies describing ischemic strokes in HIV-positive patients.
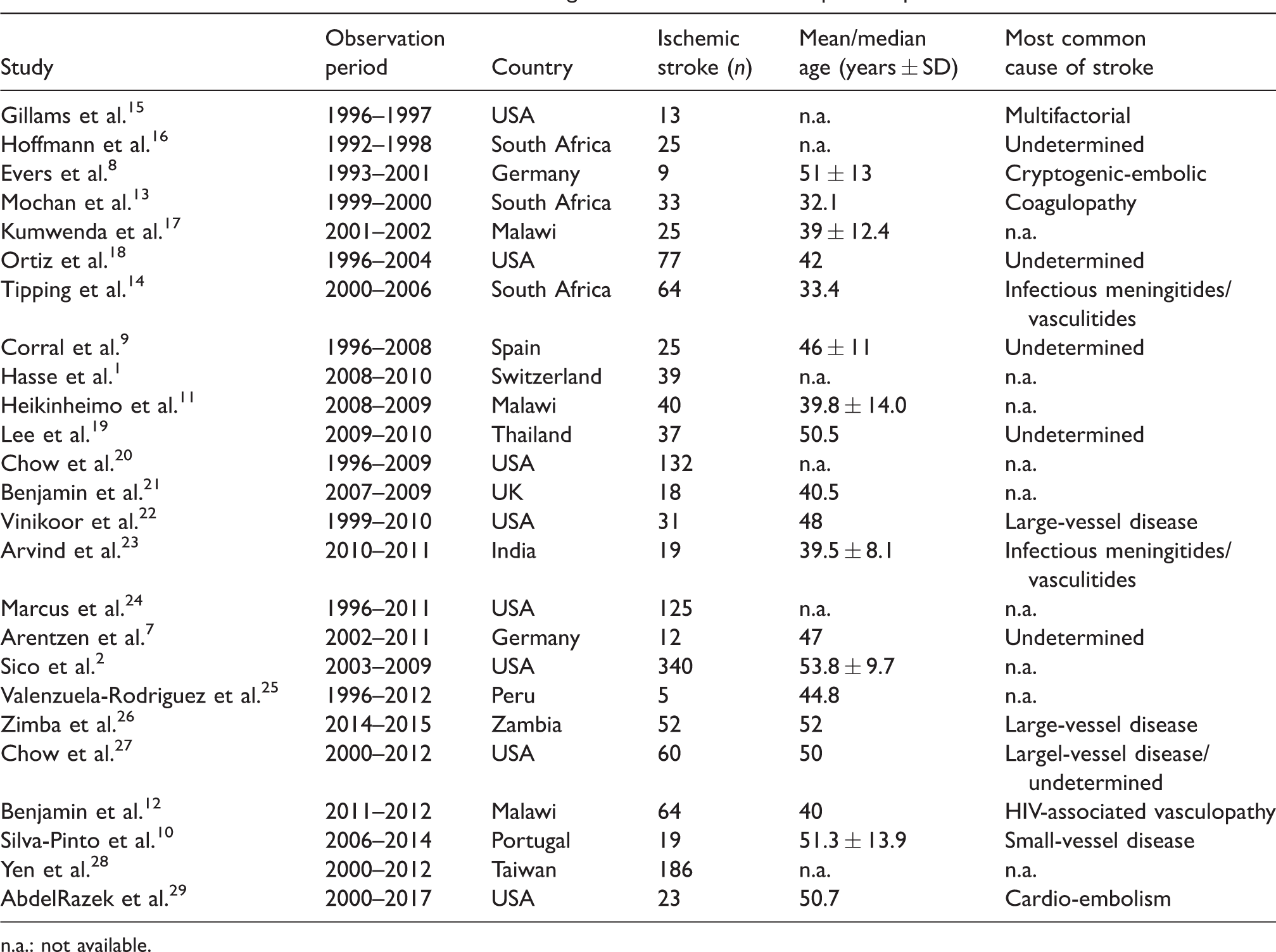
n.a.: not available.
Notably, the distribution of stroke etiology was significantly different in HIV-positive patients with a higher rate of large-vessel disease (37.5% vs. 24.2%), small-vessel disease (20.8% vs. 13.5%), and vasculitis (16.7% vs. 0.6%). Conversely, cardio-embolisms were much less frequent (4% vs. 32.3%). Risk factors included high rates of active smoking (45.8%) and arterial hypertension (50%). Especially smoking was more frequent compared to controls (18.3%; p = 0.002). Diabetes, on the other hand, was not as prevalent (12.5% vs. 25.5%, p = 0.23). HIV-positive patients had significantly higher cholesterol (196.5 ± 52.2 vs. 170.0 ± 48.2, p = 0.024) and triglyceride levels (136.6 ± 75.5 vs. 167.3 ± 81.4, p = 0.033), whereas high density lipoprotein (HDL) and low density lipoprotein (LDL) were similar.
There was no evidence of non-atherosclerotic vascular lesions such as intimal hyperplasia or aneursyms, which have been described as HIV-associated.4,30 A significantly lower IMT in HIV-positive patients (0.74 ± 0.19 vs. 0.84 ± 0.22, p = 0.046) could very well be explained by the younger mean age compared to the general stroke patients. By contrast, a large prospective study has shown that HIV-infection is an independent risk factor for early onset carotid atherosclerosis as measured by IMT. 31 In another large case study, the IMT correlated more with traditional vascular risk factors than with HIV. 32 Nonetheless, if chronic vascular inflammation accelerated atherosclerosis, IMT could be a suitable parameter to detect increased atherosclerotic burden in HIV-positive patients.
Interestingly, there was a predilection toward ischemic strokes of the vertebro-basilar circulation in HIV-positive patients, which has been observed in other inflammatory diseases such as giant cell arteriitis. 33 A recently published study has described similar abnormalities for systemic lupus erythematodes. 34 Whether this reflects increased inflammation in the posterior circulation or a difference in vessel anatomy and hence increased susceptibility remains unclear. One hypothesis is involvement of elastic tissue in inflammation, which is thinning once the vertebral arteries reach the skull. 35
Intravenous thrombolysis was administered in 16.7% of cases without a significant effect on the mRS at discharge, explained by multiple confounders, in particular that patients without relevant neurological deficits do not routinely receive rtPA and have a better outcome per se. No major complications such as symptomatic intracerebral hemorrhage or death were recorded, which is in accordance with reports that thrombolysis is safe in HIV-associated strokes. 29 Nevertheless, caution should be advised in cases of suspected vasculitis which may provoke hemorrhages.
Contrary to some reports, 36 the outcome of HIV-related stroke was mostly favorable with mRS ≤3 at discharge. Furthermore, HIV did not appear to worsen the outcome in comparison to the control group (2.68 ± 1.9 vs. 2.08 ± 1.8, p = 0.14). In other case–control studies, a HIV-positive status in itself had no detrimental impact on the outcome after stroke. 11 However, cases with confirmed vasculitis had significantly worse outcome with increased in-hospital mortality rate compared to patients without vasculitis (75% vs. 0%; p = 0.002).
The CD4-cell count was not particularly suppressed (659 ± 371/µl). An association between a CD4-cell count <200/µl and increased risk of ischemic stroke has previously been described. 24 Nevertheless, it is unclear if immunosuppression is an independent risk factor for cardiovascular disease. 37 Opportunistic or HIV-related infections can, via para-infectious vasculitis, lead to ischemic strokes. Neurosyphilis is an essential differential diagnosis in HIV-positive stroke patients 38 and was found in one patient, who suffered a fatal stroke due to associated vasculitis. A second patient had a concomitant cerebral infection with aspergillus fumigatus and as nocardia cyriacigeorgigica, which were assumed to have caused vasculitis, again with a fatal outcome. Other known risk factors for vasculitis, such as hepatitis C, were not found in our cohort.
Whether HIV-infection is an independent risk factor for cerebrovascular ischemia is a subject of debate. Some authors concluded from large cohort studies that patients with well-controlled infection status do not exhibit an increased risk for ischemic strokes. 24 By contrast, other authors found a risk increase for HIV-infection independent of current immune status.22,39 Due to the inherently low incidence of cerebrovascular disease in younger patients (<50 years), statistically, there seems to be a more robust risk increase for HIV-positive compared to HIV-negative patients when stratified for age. 20
Even after thorough diagnostics, a definite source could not be established in five patients with presumably embolic strokes. A recent study proposed that there might be an augmented risk of stroke of undetermined etiology in HIV-positive patients, highlighting the possibility of multifactorial etiologies with underlying HIV vasculopathy. 27 Furthermore, even when atrial fibrillation could be detected, treatment with warfarin did not consistently prevent new episodes of stroke, suggesting additional underlying pathomechanisms. 40
Contrary to other reports, our cohort did not contain any strokes within the first six months after starting ART, which has been attributed to an immune reconstitution inflammatory syndrome. 41 Through various mechanisms, the intake of ART appears to pose an independent vascular risk. 42 Interestingly, increased vasculopathy attributed to antiretroviral medication has previously been observed. 43 Conversely, there have been reports of a protective effect of ART as evidenced by a reduction of stroke risk with longer duration of drug therapy, suggesting that any vascular risks stemming from ART such as dyslipidemia might be offset by antiretroviral effects.9,20 Our present cohort was too small to validly analyze the vascular risk of ART. Larger prospective studies with longitudinal analyses of suitable surrogate parameters for endothelial pathology (e.g. IMT) and chronic inflammation/immunosuppression would provide further insights.
Another limitation of our study was the absence of routine HIV testing in stroke patients (in 2018 only 47.4% of patients were screened for HIV); therefore, we cannot completely rule out further (asymptomatic) cases in the control group and hence an underestimation of the true prevalence. Furthermore, not all cases of HIV-related stroke were completely screened for CD4-count and serum HIV–RNA-count, and there were some missing values of serum lipids, CRP, and IMT in the control group, which are indicated in the respective tables. Lastly, there was inadequate data on follow-up examinations. Therefore, we cannot draw any conclusions about the risk of recurring strokes and the effectiveness of secondary prevention.
Summary
To our knowledge, this is the largest description of a HIV-positive cohort with ischemic strokes in Germany as an example of a health care system with easy access to ART to date. Similar to several other published case studies, we found a younger age at stroke onset as well as a high incidence of large-vessel occlusions as the causative mechanism. Particularly high was the proportion of vessel-occlusion in the posterior circulation, a finding that requires further research. In a global context, the age of our cohort is one of the oldest described so far, supporting the hypothesis that as ART increases the life expectancy of PLWHA, they will be increasingly affected by cardiovascular disease. Although prevalence of HIV-infection among patients treated for acute ischemic strokes appears to be low (0.7%), this collective merits special attention due to several potentially modifiable vascular risk factors and a higher incidence of vasculitis, which was associated with increased mortality.
Footnotes
Contributorship
JHS gathered data; CS collected literature; CF and WP were involved in protocol development; JHS, CS, CF and WP were involved in data analysis. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: JHS has received non-financial travel grants from Biogen and Boehringer Ingelheim. CS reports personal honorary fees from scientific advisory board activities for pharmaceutical companies (MSD, Gilead, ViiV, Janssen Cilag) and travel grants for participation in medical conferences (BMS, Gilead, Janssen). CF reports personal fees from Boehringer Ingelheim and Prediction Bioscience. WP reports no conflicts in regard to the manuscript.
Ethical approval
The ethics committee of the University of Frankfurt has approved the study design (file number 46/19) on 5 March 2019.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
No informed consent was required due to the retrospective study design
Guarantor
JHS.
