Abstract
Background
Small vessel disease causes a quarter of ischaemic strokes (lacunar subtype), up to 45% of dementia either as vascular or mixed types, cognitive impairment and physical frailty. However, there is no specific treatment to prevent progression of small vessel disease.
Aim
We designed the LACunar Intervention Trial-2 (LACI-2) to test feasibility of a large trial testing cilostazol and/or isosorbide mononitrate (ISMN) by demonstrating adequate participant recruitment and retention in follow-up, drug tolerability, safety and confirm outcome event rates required to power a phase 3 trial.
Methods and design
LACI-2 is an investigator-initiated, prospective randomised open label blinded endpoint (PROBE) trial aiming to recruit 400 patients with prior lacunar syndrome due to a small subcortical infarct. We randomise participants to cilostazol v no cilostazol and ISMN or no ISMN, minimising on key prognostic factors. All patients receive guideline-based best medical therapy. Patients commence trial drug at low dose, increment to full dose over 2–4 weeks, continuing on full dose for a year. We follow-up participants to one year for symptoms, tablet compliance, safety, recurrent vascular events, cognition and functional outcomes, Trails B and brain MRI. LACI-2 is registered ISRCTN 14911850, EudraCT 2016–002277-35.
Summary
LACI-2 will determine feasibility, tolerability and provide outcome rates to power a large phase 3 trial to prevent progression of cerebral small vessel disease.
Introduction
Stroke and vascular dementia are increasing in prevalence, 1 have enormous economic and societal costs, share many risk factors, and are major Government targets for health improvement.2,3 Cerebral small vessel disease (SVD) is a central link between stroke and dementia. 4 It accounts for 20–25% of clinical strokes as either recent small subcortical or ‘lacunar’ infarcts 5 (hereafter referred to as ‘lacunar ischaemic stroke’) and most intracerebral haemorrhages in older people, 6 and up to 45% of all dementias, alone or combined with Alzheimer’s disease. 7 Patients presenting with a lacunar ischaemic stroke are often younger than with other stroke subtypes, 6 and about a third experience post-stroke cognitive impairment or dementia,8,9 which may restrict their independence, including ability to return to work. 9
The causes of lacunar ischaemic stroke are still poorly understood. 4 Most appear due to an intrinsic cerebral perforating arteriolar disease. This includes impaired vasoreactivity, increased blood-brain barrier permeability and perivascular inflammation, all causing brain damage 4 visible on magnetic resonance imaging (MRI) including white matter hyperintensities (WMH), lacunes, prominent perivascular spaces and microbleeds. Additionally, the damaged endothelium may precipitate local thrombosis and ischaemia, which worsens the damage. 4
Despite the profound health impacts, few RCTs specifically targeted either SVD lesion progression or lacunar stroke. 10 The largest trial in lacunar ischaemic stroke, the Secondary Prevention of Small Subcortical Stroke (SPS3) trial, tested 3.4 years of aspirin + clopidogrel versus aspirin 11 and 3.7 years of target versus guideline blood pressure (BP) reduction 12 in 3020 lacunar ischaemic stroke patients, to prevent recurrent stroke and cognitive decline. The aspirin + clopidogrel versus aspirin arm stopped early due to increased death; 11 target (versus guideline) BP lowering was consistent with a small reduction in recurrent stroke, but neither it nor aspirin + clopidogrel versus aspirin reduced cognitive decline. 13
Given the long timeline to develop new drugs, available licensed drugs with relevant actions should be evaluated. 10 Prostacyclin (PGI2)-cyclic AMP-phosphodiesterase 3 pathway or nitric oxide (NO)-cyclic GMP-phosphodiesterase 5 modulators may improve vasodilatation, reduce inflammation and smooth muscle hypertrophy and improve cerebral endothelial integrity. 10 Amongst several drugs licensed in Europe, cilostazol and isosorbide mononitrate (ISMN) show promise; two drugs with complementary actions could have synergistic effects.
Cilostazol, a phosphodiesterase 3-inhibitor (PDE3-inhibitor), enhances the PGI2-cAMP pathway, with weak antiplatelet effects (so low bleeding risk).
14
Routine cilostazol use reduced dementia incidence in the Taiwanese registry,
15
reduced infarct size
16
and improved myelin repair in animal models,
17
and has been trialled for secondary stroke prevention in more than 6400 patients.18–22 Trials where >50% of participants had lacunar stroke (
ISMN is an NO-donating organic nitrate that enhances vasodilation, is widely used in ischaemic heart disease with a known safety profile and has no antiplatelet activity. 23 NO maintained cerebral perfusion short term after subacute stroke, 24 but drugs that increase NO availability are rarely used in stroke. 25
The LACI-1 trial 26 tested short-term dose escalation protocols for cilostazol and ISMN individually and together, tolerability and early markers of safety and efficacy in patients with lacunar stroke. 27 LACI-1 recruited 56 patients in two centres, showed that headache, nausea, palpitations were common pre-randomisation, increased on starting either trial drug, but by incrementing the dose slowly, symptoms returned to background levels within two weeks. 27 Most patients achieved the target dose 27 without safety concerns. 28 The LACI-2 trial aims to determine if cilostazol and ISMN, alone or together, are tolerated at the target dose for a year, provide feasibility, safety and efficacy data, prior to proceeding to a large phase III trial.
Methods
LACI-2 is a phase IIb randomised, partial factorial, open label, blinded end-point trial, aiming to recruit 400 patients from UK Stroke Network Centres, with follow-up to one year. In addition to experience from LACI-1, LACI-2 trial design benefitted from:
a National Institutes of Health Research (NIHR) Stroke Research Network (SRN) Portfolio Development Expert Writing Group (members indicated*), which discussed trial design options in detail; the USA NIH-funded SPS3 trial11,12 (3020 patients with lacunar stroke): Chief Investigator (CI) O Benavente*, which informed on recruitment and outcome event rates; the CSPS, CSPS-2 (3740 patients)18,20 and CSPS-3 trials
22
testing cilostazol to prevent stroke in Japan: CI K Toyoda, informed on cilostazol dose and escalation to increase tolerability; monogenic SVDs:
29
CI H Chabriat*, informed on timing and practicality of potential outcome assessments, and the relative merits and rates of functional, cognitive and other outcomes to optimise sensitive outcomes against the burden of assessments on the participants; the PRESERVE trial:
30
CI H Markus*, informed on approaches to patient recruitment, cognitive assessments, and MRI rates; the PODCAST trial:
31
CI P Bath*, Co-CI LACI-2, informed on minimisation algorithms, data collection, likely availability of informants, approaches to loss of mental capacity during the trial, cognitive assessments and frequency of outcome assessments; CROMIS-2 and the Microbleeds International Collaborative Network,32,33 informed on clinical and imaging assessments, frequency and practicalities of potential outcome assessments.
The UK SRN Prevention Clinical Studies Group strongly supported testing cilostazol and ISMN in SVD prevention and many UK Clinical Research Network Centres expressed interest in joining LACI-2.
Patients undergo guideline-based investigations as per usual stroke prevention practices in the UK including carotid and cardiac investigations and for other rarer causes in young strokes as appropriate.
Patients are eligible if they are independent in activities of daily living, have capacity to give consent, are aged over 30 years, have suffered a minor stroke with clinical features compatible with a lacunar syndrome (most frequently, but not limited to, a pure motor hemiparesis, pure sensory stroke, ataxic hemiparesis, sensorimotor stroke, or dysarthria-clumsy hand syndrome; designated as the ‘index stroke’) and have, on contemporaneous CT or MRI brain imaging, either a visible relevant recent small subcortical (lacunar) infarct or no alternative pathology to explain symptoms (Table 1). There is no maximum time limit for recruitment because (a) lacunar stroke indicates the presence of SVD, which is a longstanding condition, (b) recurrent stroke after lacunar stroke and signs of SVD progression are thought to occur slowly and inexorably (in contrast to atherothromboembolic stroke where the risk of recurrence is immediate then declines), (c) to avoid guideline dual antiplatelets and minimise the burden of participation on the patient soon after the stroke, (d) since both drugs are likely to affect long-term chronic vascular dysfunctions, (e) to increase recruitment by avoiding clashes with high periods of recruitment to acute treatment and secondary prevention trials and (f) since any time limit would be very arbitrary after considering all the above. However, time after stroke is accounted for in minimisation and will be accounted for in the statistical analysis.
Inclusion and exclusion criteria.

CTIMP: Clinical Trial of Investigational Medicinal Product; FLAIR: fluid attenuated inversion recovery; MR: magnetic resonance.
Exclusion criteria include other significant active neurological illness, alternative pathologies visible on imaging (cortical infarct, subcortical infarct exceeding 2 cm diameter likely to be large artery, haemorrhage, tumour, etc), athero- or cardio-embolic source requiring specific treatment, renal or hepatic impairment, unlikely to complete follow-up due to illness or location, current enrolment in another Clinical Trial of Investigational Medicinal Product (CTIMP), and other general and specific reasons listed in Table 1. Co-enrolment is allowed in non-CTIMPs.
Consent
Participants must have capacity and consent is taken from them for the trial and for secondary data uses; consent is taken from a relative or other informant to provide outcome data.
Intervention
ISMN 40–60 mg (or equivalent formulation) daily only; cilostazol 200 mg daily only; or both ISMN 40–60 mg and cilostazol 200 mg daily. The range of ISMN doses is allowed to account for different formulations provided in hospital pharmacies. The trial is open-label since placebo tablets were not available in the UK and masking by encapsulation was impractical and expensive due to dose escalation and factorial trial design. However, outcomes including structured symptom questionnaires, ascertainment of clinical outcomes and MRI analysis are collected by individuals blinded to treatment allocation. Participants with indications for or contraindications to one drug can be randomised to the other drug only. The partial factorial design allows testing of both drugs when given alone and together.
Comparator
Best medical guideline stroke prevention therapy as per UK guidelines, which include neither cilostazol nor ISMN. Throughout the trial, participants continue to take their normal prescribed medication, information on which is collected at randomisation.
Randomisation, minimisation
Randomisation is by central computer-generated allocation at the University of Nottingham after recording key patient variables and minimisation data. Minimisation uses key prognostic factors including: age, sex, stroke severity (National Institute of Health Stroke Scale), dependency resulting from the stroke, systolic BP ≤/>140, smoker status, time after stroke, and years of education. Years of education give an estimate of pre-morbid cognitive ability and predicts post-stroke cognitive impairment; 9 BP and smoking predict recurrent stroke; delay since stroke reflects disease activity; age, sex and stroke severity are standard minimisation variables. 27 This approach ensures concealment of allocation, minimises differences in key baseline prognostic variables, and improves statistical power. Randomisation is not minimised by Centre because this may result in high rates of allocation prediction, but a pre-specified post-hoc analysis by centre will be performed to investigate and adjust for heterogeneity of treatment effect by centre.
Dispensing
Participants are allocated in a 1:1 ratio to cilostazol versus no cilostazol and to ISMN versus no ISMN, resulting in a partial factorial comparison of cilostazol versus ISMN versus both drugs versus neither drug. The algorithm generates a unique trial ID and number corresponding to a prescription of allocated trial treatment, which is prepared and dispensed by the local Pharmacy (Figure 1).
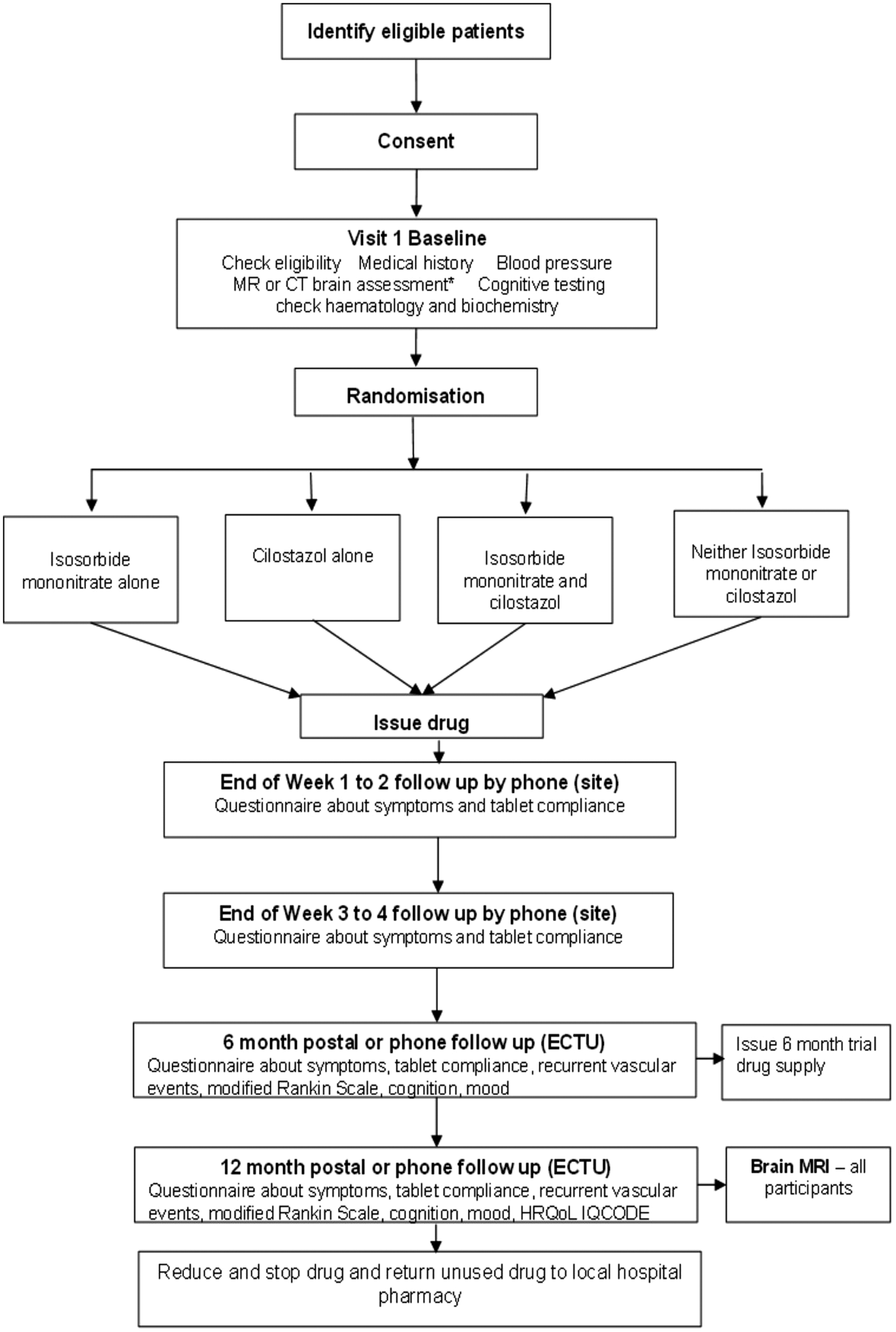
CONSORT diagram. Diagnostic MR or CT brain scan assessment (Baseline) refers to a visual assessment of the scan that has already been performed as part of the patient’s routine stroke clinical assessment and diagnosis. Features present on the scan are scored for their presence and severity to create a total sum of SVD score. This is used in the minimisation algorithm. Dispensing may be at 3-monthly intervals if preferred in particular centres.
Drug initiation and maintenance
After randomisation, patients are given instructions to start on a low dose and escalate to target dose over two-to-four weeks. For ISMN, 25 mg once daily in the first week, 40–60 mg daily in the second week and thereafter. For Cilostazol, 50 mg once daily for the first week, twice daily in the second week rising to 100 mg twice daily over the next two weeks until participants are on their full dose by one month, remaining on full dose for the rest of the year. Escalation can proceed more slowly if necessary. Participants can remain on less than full dose if they are unable to tolerate the full dose, since some drug may be better than no drug and dose is recorded.
Assessments
At baseline, the study researcher records details of the presenting stroke, past medical history, medications, socioeconomic status, education, lifestyle factors, neurological findings, brain imaging findings (index stroke, background WMH, lacunes, microbleeds), BP, cognitive testing (Montreal Cognitive Assessment, Trails B), haematology and biochemistry (Table 2).
Study assessments.
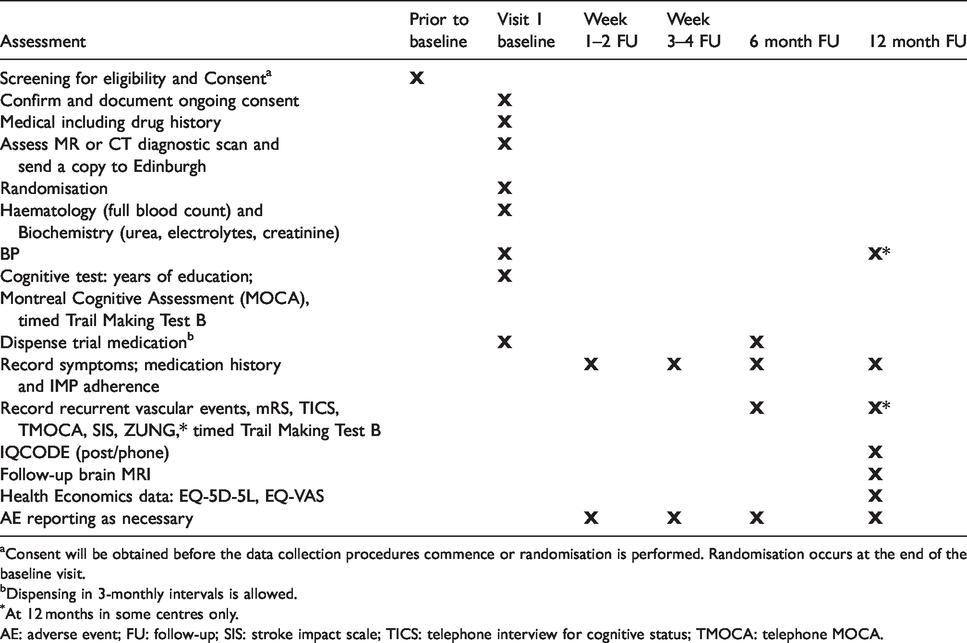
Consent will be obtained before the data collection procedures commence or randomisation is performed. Randomisation occurs at the end of the baseline visit.
Dispensing in 3-monthly intervals is allowed.
At 12 months in some centres only.
AE: adverse event; FU: follow-up; SIS: stroke impact scale; TICS: telephone interview for cognitive status; TMOCA: telephone MOCA.
After commencing trial treatment, local researchers telephone the participants at 1–2 and 3–4 weeks post randomisation, with intermediary phone calls as necessary, to advise on dose escalation, symptoms, events, check they have achieved full dose or are established on a lower tolerated dose by four weeks.
At 6 and 12 months, central trial staff (Edinburgh, Nottingham), masked to treatment allocation, perform follow up by postal questionnaire and phone. Additionally, at 12 months, the patient attends their local trial centre for Trails B and brain MRI (to assess SVD lesion burden, new stroke lesions). Information recorded at 6 and 12 months follow-up includes symptoms, recurrent ischaemic or haemorrhagic stroke or transient ischaemic attack (TIA), cardiac events, smoking status, current medications, modified Rankin scale (mRS), stroke impact scale 34 and separately the amount of trial medication (Table 2). Date and cause of death are recorded. Participants are encouraged to contact their local Trial Centre to record vascular events occurring between follow-up points. Unused drug is returned to pharmacy at the participating hospital for accountability and destruction at the end of the trial.
Brain imaging
All MR or CT scans performed for stroke diagnosis, to assess new neurological events during the trial, and at one-year follow-up, are collected centrally, anonymised, for blinded assessment. MRI at one year must include diffusion-weighted imaging (DWI), T2-weighted, fluid attenuated inversion recovery (FLAIR), susceptibility-weighted (blood sensitive), and T1-weighted sequences (exemplar sequences are provided). Scans are rated, blind to patient characteristics and treatment allocation, to assess the index infarct location, size, WMH, lacunes, old cortical infarcts, old haemorrhages, microbleeds, siderosis, perivascular spaces and brain atrophy, with validated scores using a web-based system. 5
Adverse events are recorded as serious (SAEs), reported to the Sponsor, if they result in hospitalisation, persistent disability, death or are life threatening. Recurrent vascular events (stroke, myocardial infarction (MI), TIA) and bruising, bleeding or other types of haemorrhage are collected as outcomes, not SAEs. Serious adverse reactions (SARs) and suspected unexpected SARs (SUSARS) are also reported. Adverse events (AEs) that are common in patients with stroke and do not meet criteria for seriousness or drug related are recorded.
Outcomes
The primary outcome is feasibility, i.e. that eligible participants can be identified correctly, in sufficient numbers, enrolled and >95% retained in follow-up at one year, to achieve target sample of 400 participants in 24 months in the UK. The secondary outcomes are tolerability (can patients tolerate trial medication at least in half dose for up to a year?), safety (systemic or intracranial bleeding, vascular and non-vascular causes of death) and efficacy (stroke, TIA, myocardial ischaemia, cognitive impairment and dementia).
Sample size
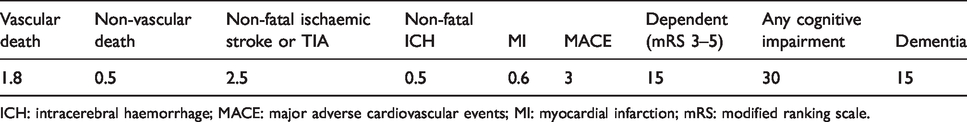
Data to estimate sample size were limited. Based on LACI-1, we estimated that 75% of participants would take trial medications, in at least half dose, up to one year. We calculated mean annual event rates (Table 3) from trials (SPS3,
11
lacunar participants in ENOS,
25
IST-3,
35
of cilostazol18,20,36) and observational data (LADIS;
37
our9,38,39 and other
40
studies). We expect deaths including vascular deaths of median 2.0% pa, with 4% being the upper 95% CI of 2% in 400 participants.
11
We propose that the absolute risk of death, including fatal haemorrhage, will not exceed 4% per year on trial drugs versus no trial drugs, given in addition to guideline drugs, and will not increase bleeding or ischaemic SVD lesions significantly (at the
Annual absolute risks (%) of outcome events after lacunar stroke – see text for data sources.
ICH: intracerebral haemorrhage; MACE: major adverse cardiovascular events; MI: myocardial infarction; mRS: modified ranking scale.
Sample size for composite outcome in main trial, estimated event rates – see text for data sources.
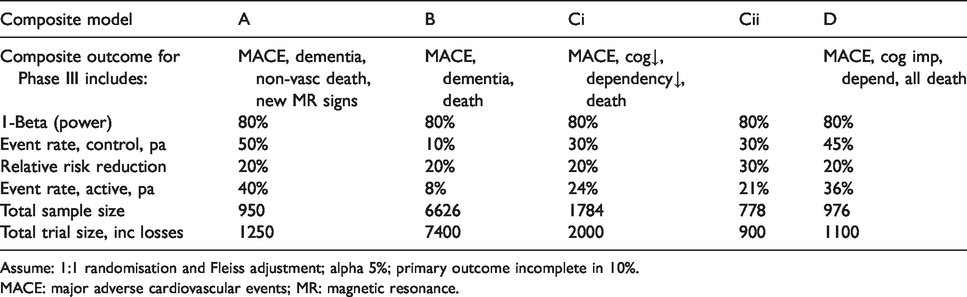
Assume: 1:1 randomisation and Fleiss adjustment; alpha 5%; primary outcome incomplete in 10%.
MACE: major adverse cardiovascular events; MR: magnetic resonance.
Statistical analysis
A statistical analysis plan will be published prior to recruitment ending, blinded to data. A brief summary of planned analyses are as follows. We will compare cilostazol versus no cilostazol, ISMN versus no ISMN, cilostazol and ISMN v neither, by intention to treat (ITT) analysis. The proportion of patients achieving and sustaining target dose and adverse symptoms will be assessed using odds ratios. ITT analysis will use logistic regression adjusted for minimisation variables and other baseline prognostic variables (to maximise power 44 ) including SVD score, age and cilostazol+/–ISMN versus control on efficacy and safety outcomes, with secondary tests of subgroup interactions. Ordinal logistic regression will be used for ordinal categorical outcomes (e.g. mRS) to increase power. 45 To analyse safety, we will use Kaplan-Meier and Cox proportional regression for time-to-event outcomes of death, recurrent stroke, MI; binary logistic regression for SAEs, recurrent stroke, MI, and in those completing 12 month MRI, microbleeds, siderosis, new infarcts, and WMH burden.
Trial and data management
LACI-2 has an operational management committee meeting alternate weeks, a Trial Steering Committee (TSC) with an independent chair, two scientific advisors, participant, funder and sponsor representatives meeting six monthly, a Data Monitoring Committee (DMC) with four independent members meeting yearly, and an International Advisory Board that provides informal advice. The Sponsor, the University of Edinburgh/NHS Lothian joint Academic and Clinical Central Office for Research and Development (ACCORD) approves all trial procedures and provides monitoring. The trial is managed from the Edinburgh Clinical Trials Unit, registered with UKCRC. The secure, password-protected, electronic database and case record form is held at the University of Nottingham; statistical support is provided by the Nottingham Stroke Trials Unit and UKCRC-registered Nottingham Clinical Trials Unit; image data management and analysis is at the University of Edinburgh. Only the DMC members see unblinded data during the trial. Members of each group are listed in Appendix 1.
Discussion
LACI-2 is testing drugs that aim to improve the endothelial dysfunction seen in lacunar ischaemic stroke, given for one year. LACI-2 builds on experience in LACI-1 including inclusion and exclusion criteria, data collection, randomisation and statistical analysis, has a similar TSC, the same DMC and International Advisory Boards, to enhance and retain knowledge and experience. LACI-2 will provide methods for balancing randomisation, streamlining of follow-up and reliable data on key event rates for lacunar stroke/SVD trials, preparatory to a Phase III trial of these drugs for definitive efficacy and safety data. LACI-2 aims to promote more personalised approaches to brain vascular disease by recognising and addressing key mechanistic and susceptibility differences between ischaemic stroke subtypes.
LACI-2 is pragmatic; we faced several practical issues in designing the trial and recognise that some of our decisions were, of necessity, empirical since there have been few trials in lacunar stroke to guide design. In LACI-1, target sample 60 patients in two centres, recruitment time was reduced by 8 months and the total cost by £85,000 (equivalent to a third of total available funding) using enrolment based on CT or MRI compared with only allowing enrolment based on MRI. 27 Although an MRI-based trial could be more specific for lacunar stroke, we showed that it would delay trial completion by at least 33% thus inflating staff costs significantly, it would exclude patients unable to tolerate MRI, and delay scientific progress since the time to finding a treatment for lacunar ischaemic stroke would take much longer. Since no imaging modality is perfect, and not all acute ischaemic lesions are visible on MRI even with diffusion imaging (e.g. 30% of patients with lacunar or mild cortical stroke do not have an acute lesion on DWI yet have similar rates of recurrent stroke, functional and cognitive impairments 46 ) appropriate use of CT and MRI can exclude alternative causes of symptoms, and CT can detect moderate to severe WMH, lacunes, atrophy, and old stroke lesions including differentiating some old haemorrhages from old infarcts.47,48 LACI-2 also does not set a maximum time from stroke to recruitment after consideration of pathological (lacunar stroke due to intrinsic cerebral arteriolar pathology indicates the presence of a chronic condition, SVD, and disease progression is thought to occur steadily and slowly), pharmacological (putative effects of cilostazol and ISMN are on chronic vascular dysfunctions), and practical reasons (avoid subacute post-stroke period when guideline dual antiplatelet drugs are used, and minimise clashes with other acute treatment and short term secondary prevention trials).
There is no placebo in LACI-2 since, after considerable searching, none was available, but central follow-up and MRI are blinded to allocation. LACI-2 encourages best medical management in all patients including good BP control using guideline medications. Good BP control may help reduce WMH progression 49 and prevents stroke. LACI-2 is therefore testing cilostazol or ISMN or both plus best medical management versus best medical management alone.
LACI-2 started recruiting in March 2018 in one site, had opened 26 sites and recruited 258 participants by February 2020. Apart from delays in opening sites due to the large administrative burden of clinical trials, it is on course to complete recruitment by Dec 2020, to complete follow-up by Dec 2021, and report in spring 2022. The DMC have met twice (August 2018 and 2019) and recruitment continues. To the best of our knowledge, LACI-2 appears to be the only ongoing trial specifically testing agents for long term prevention of recurrent lacunar stroke, MACE and worsening of SVD. It is one of only five trials listed in the Alzheimer’s Drug Discovery Foundation Report 2018 (
Trial registration
The trial is registered: EudraCT 2016–002277-35, ISRCTN 14911850.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is supported by British Heart Foundation (CS/15/5/31475), the Alzheimer’s Society (AS-PG-14–033), EU Horizon 2020 SVDs@Target (666881), MRC UK DRI, Fondation Leducq (16/05 CVD), NHS Research Scotland, The Stroke Association and Garfield-Weston Foundation, Chief Scientist Office (UC), and National Institute of Health Research. PB is Stroke Association Professor of Stroke Medicine and a NIHR Senior Investigator.
Ethical approval
The trial is conducted in accordance with the principles of the International Conference on Harmonisation Tripartite Guideline for Good Clinical Practice (ICH GCP). Ethics approval was granted by the East Midlands Nottingham 2 Research Ethics Committee of the Health Research Authority number 17/EM/0077 on 10/05/2017. NHS Research and Development Approval is given by each participating Centre. The Medicines and Healthcare Regulatory Authority approved the trial on 01/06/2017. LACI-2 was adopted by the UK Clinical Research Network and Scottish Stroke Research Network.
Informed consent
Participants must have capacity and consent is taken from them for the trial and for secondary data uses; consent is taken from a relative or other informant to provide outcome data.
Guarantor
JMW.
Contributorship
JMW, PMWB, FD, NS, VC, THE, AH, DW designed the trial and secured funding; JMW is the Chief Investigator, obtained ethics and regulatory approvals; GB, JPA helped design the assessments, and with JMW and PMWB, designed the case record form; AH is the trial manager, responsible for daily running of the trial including regulatory compliance; FD, THE, AH, DW are Principle Investigators; NS provides independent blinded event review; AM provides statistical expertise; LW is the trial statistician; JMW drafted the manuscript; all other authors commented and edited it; all authors approved the final version for submission.
Acknowledgements
We thank the patients and their relatives for their time and effort to participate in LACI-2, the Trial Steering Committee, Sponsor, Data Monitoring Committee, Edinburgh Clinical Trials Unit staff, UK Clinical Research Network, Scottish Stroke Research Network, International Advisory Panel, and all staff at participating sites, for their support.
Appendix 1. The LACI-2 Trial Investigator Group
Writing Committee: Joanna M Wardlaw, Philip MW Bath, Fergus Doubal, Anna Heye, Niki Sprigg, Lisa Woodhouse, Gordon Blair, Jason P Appleton, Vera Cvoro, Timothy J England, Ahamed Hassan, David Werring, Alan Montgomery.
Trial Steering Committee: John Bamford, Independent Chair; Christine Roffe, Stoke, John T. O’Brien, Cambridge, Independent Advisors; Euan Haig, Patient Representative; Joanna M Wardlaw, CI, Fergus Doubal, PI, Edinburgh; Philip MW Bath, Co CI, Nikola Sprigg, Safety Adjudication, Alan Montgomery, Statistical Oversight, Lisa Woodhouse, Statistician, Richard Dooley, Programmer, Nottingham; Timothy England, PI Derby, David Werring, PI UCL London, Ahamad Hassan, PI Leeds, Vera Cvoro, PI Fife; Anna Heye, Trial Manager, Edinburgh; F O’Mahoney, Sponsor Representative, Edinburgh; Shannon Amoils, British Heart Foundation, Funder Representative.
Independent Advisors: Oscar Benavente, Vancouver, Canada; Kasuo Toyoda, Kyoto, Japan; Hugues Chabriat, Paris, France.
Data Monitoring Committee: Colin Baigent, Oxford; Gary Ford, Oxford; Alison D Murray, Aberdeen; Jonathan Emberson, Oxford.
Trial Staff: Anna Heye, Carol Williams, Anna Foster, Kaye Ferguson, Guen Innes, Edinburgh; Di Havard, Sharon Ellender, Patricia Robinson, Lisa Woodhouse, Richard Dooley, Lee Hayward, Nottingham.
Image Management: Eleni Sakka, Image Data Manager, Jeb Palmer, Programmer, David Buchanan, Programmer, Edinburgh.
Independent blinded event adjudication: Nikki Sprigg, Nottingham.
Participating Sites:
Edinburgh, Royal Infirmary, C001: Fergus Doubal (PI)
Nottingham, University Hospitals, C002; Kailash Krishnan (PI)
Kircaldy, Victoria Hospital, C004: Vera Cvoro (PI)
Glasgow, Queen Elizabeth Hospital, C005: Jesse Dawson (PI)
Bradford, Royal Infirmary, C006: Christopher Patterson (PI)
Aberdeen Royal Infirmary, C007: German Gutierrez (PI)
Leeds General Infirmary, C008: Ahamed Hassan (PI)
Derby, Royal Derby Hospital, C009: Timothy England (PI)
Inverness Raigmore Hospital, C010: Stephen Makin (PI)
London, St George’s Hospital, C011: Usman Khan (PI)
London, Kings College Hospital, C012: Laszlo Sztriha (PI)
Essex, Broomfield Hospital, C013: Ramanathan Kirthivasan (PI)
Stockton-on-Tees, University Hospital of North Tees, C014: Anwar Ijaz (PI)
Sheffield, Royal Hallamshire Hospital, C015: Kirsty Harkness (PI)
Sandwell, General Hospital, West Bromich, C016: Sissi Ispoglou (PI)
Winchester, Royal Hampshire County Hospital, C017: Nigel Smith (PI)
London, University College, C018: David Werring (PI)
Harrow, Northwick Park Hospital, C019: Aravinth Sivagnanaratnam (PI), David Cohen,
Luton, Luton and Dunstable NHSFT University Hospital, C020: Lakshmanan Sekaran (PI)
Doncaster Royal Infirmary, C021: Dinesh Chadha (PI)
Wolverhampton, New Cross Hospital, C022: Kenneth Fotherby (PI)
Halifax, Calderdale Hospital, C023: Pratap Rana (PI)
Taunton, Musgrove Park Hospital, C025: Malik Hussain (PI)
Southampton General Hospital, C026: Nic Weir (PI)
London, Homerton University Hospital, C027: Thomas Harrison (PI)
Exeter, Royal Devon and Exeter Hospital, C028: Salim Elyas (PI)
