Abstract
Introduction
In patients with spontaneous intracerebral haemorrhage, it is uncertain if diagnostic and therapeutic measures are time-sensitive on their impact on the outcome. We sought to determine the influence of the time to admission to a comprehensive stroke centre on the outcome of patients with acute intracerebral haemorrhage.
Patients and methods
We studied a prospective database of consecutive patients with intracerebral haemorrhage attended at two comprehensive stroke centres (2005–2017). We excluded patients with an unwitnessed time of onset of the intracerebral haemorrhage, or previous modified Rankin Scale >3 or in those in whom withdrawal of life-sustaining interventions were decided <24 h from admission. We recorded the time from the intracerebral haemorrhage onset to admission, demographic, clinical, radiological data, the functional outcome (favourable when modified Rankin Scale ≤3) and mortality at 90 days. We conducted a propensity score-matching analysis to evaluate functional outcome and mortality.
Results
We included 487 patients (mean age 72.3 ± 13.9 years), and 53.2% were men. Compared to patients with an admission >110 min, patients who were admitted ≤110 min were significantly younger, and had higher National Institutes of Health Stroke Scale scores. Moreover, patients admitted ≤110 min were more likely to have basal ganglia intracerebral haemorrhage, and to show neurological deterioration. The propensity score groups were well matched. We did not find an association between time to admission and the favourable outcome (OR: 1.42 (95% CI: 0.93–2.16)) or mortality (OR: 0.64 (0.41–0.99)) at 90 days.
Conclusions
Our results suggest that in patients with intracerebral haemorrhage and known symptom onset who are admitted to a comprehensive stroke centre, an early admission (≤110 min) does not influence the outcome at 90 days.
Keywords
Introduction
Spontaneous intracerebral haemorrhage (ICH) is a significant cause of morbidity and mortality.1,2 Neurological deterioration of patients with ICH occurs within the first hours and days after symptoms onset and increases the risk of a poor outcome.1,2
The main objective of the pre-hospital management in patients with suspected stroke is to provide airway management, cardiovascular support and transport of the patient to the nearest hospital.1,3 The admission to a comprehensive stroke centre (CSC) can improve the outcome of patients with ICH due to an aggressive medical and surgical management and specialist care.1,2,4
In addition, compared to a less-organised service, the organised inpatient care in a stroke unit reduces the risk of death and dependency in patients with ICH. 5 Overall, the use of emergency medical services, the development of Stroke Code systems and the admission to CSC with organised inpatient care have improved the outcome of stroke patients. In ischaemic stroke, the efficacy of acute reperfusion therapies is time-dependent and, therefore, an early admission to a CSC increases the odds of favourable functional outcome.4,6 However, in patients with ICH, the association of an early compared to a delayed admission to the CSC and favourable outcome is uncertain. One possible explanation is that medical and surgical treatments for acute ICH have a limited efficacy.1,2 However, ICH is a dynamic process and recent studies reported that some early therapeutic measures may improve outcome.1,2,7 Therefore, patients with ICH who are admitted early may have a high risk of neurological deterioration but also an opportunity to receive beneficial treatments. It is more likely that a CSC can provide a protocolised care and all the possible therapeutic measures.
There is a lack of data about the influence of an early admission to a CSC on long-term outcome of patients with ICH. We hypothesised that, after an adjustment by factors associated with an unfavourable outcome, an early CSC admission may improve the outcome. We investigated the relationship between the time from the onset of the ICH to the CSC admission and the functional outcome and mortality at 90 days.
Patients and methods
This is a retrospective study of patients with acute spontaneous ICH who were consecutively included in a prospective registry from two CSCs (A and B). CSC A included patients from July 2013 to December 2017 and CSC B from January 2005 to December 2017. The Ethics Committees of both institutions approved the use of the ICH registries for clinical research. Most patients were transferred directly to the CSC and were treated according to the updated European guidelines of ICH.
We used the following inclusion criteria: a diagnosis of spontaneous ICH; age ≥18 years; witnessed symptom onset; and time from the onset of ICH symptoms to CSC admission ≤24 h. We excluded those patients with a baseline modified Rankin Scale (mRS) > 3; those in whom withdrawal of life-sustaining interventions were applied within the first 24 h from the CSC admission. In our ICH registries, we did not include patients with ICH-associated with trauma, thrombolysis, or subdural and epidural haematoma or significant subarachnoid haemorrhage.
The following variables were collected prospectively: admission unit (Stroke unit, Intensive Care Unit, others), demographics (age and sex); vascular risk factors (hypertension, dyslipidaemia, diabetes mellitus, atrial fibrillation, ischaemic stroke, ICH, ischaemic heart disease); previous medication (oral anticoagulants, antiplatelet agents); vital signs at admission (blood pressure and temperature); baseline capillary glucose; international normalised ratio (INR); time from the onset of symptoms to the CSC admission; severity of the neurological deficit rated with the National Institutes of Health Stroke Scale (NIHSS) or the Glasgow coma scale (GCS) score. Neurological deterioration was defined as an increase of four or more points in the NIHSS score or decrease of two or more points in the GCS score within the first 72 h from the admission. From the baseline cranial computed tomography (CT), we evaluated the ICH volume (ABC/2 formula), presence of intraventricular haemorrhage and ICH localisation (lobar, deep, infratentorial). We defined ICH growth as an absolute growth ≥6 mL or >33% from baseline to the follow-up imaging and was evaluated in patients who had a second cranial CT within the first 72 h from admission. In CSC A, we collected neurosurgical procedures and oral anticoagulants reversal. At 90 days, a stroke neurologist evaluated the functional outcome with the mRS (favourable when 0–3) and mortality.
Statistical analyses
To compare baseline characteristics, we used contingency tables with the χ2 or the Fisher Exact test for categorical variables. We used Student’s t-test for quantitative variables with a normal distribution, and Mann–Whitney U test when a nonparametric test was appropriated. The median time from the onset of symptoms to CSC admission was our reference to define an early (≤110 min) or late (>110 min) admission.
We used a propensity score (PS) stratification approach because non-random assignment of early or late admission to the CSC may have produced a selection bias. Time to admission was treated like scalar and dichotomised, from the median, by its skewed and multimodal distribution. The effect of this binary variable was estimated by stratification of the PS. The propensity for time to admission >110 min instead of ≤110 min was computed by means of a multiple logistic regression model, accounting for the following variables: age, admission unit, atrial fibrillation, use of antiplatelet agents, baseline capillary glucose, baseline NIHSS, baseline GCS score, ICH volume and intraventricular haemorrhage. Our criteria to include variables in the PS model were plausibility of a relationship between any of the selected variables and eligibility for the time to admission or with the outcome. Confounding factors were evaluated by contrasting Odds Ratios (ORs) among models with and without each potential variable, included factors were those that altered OR by 10% or more. 8 Then, patients were stratified in four blocks (quartiles), according to PS ascending values in the region of common support (four cases excluded because PS values outside the region). Figure 1 shows the box plots of the distribution of PS by time to admission within blocks. To calculate the PS estimate, we used the psmatch2 and the Kernel approach. 9 We estimated the average OR through blocks with the Mantel–Haenszel test statistics and we performed also a test for homogeneity between blocks with Breslow-Day test. All analyses were performed using IBM-SPSS version 25 and STATA version 15. A two-tailed p value of <0.05 was considered significant for all tests.
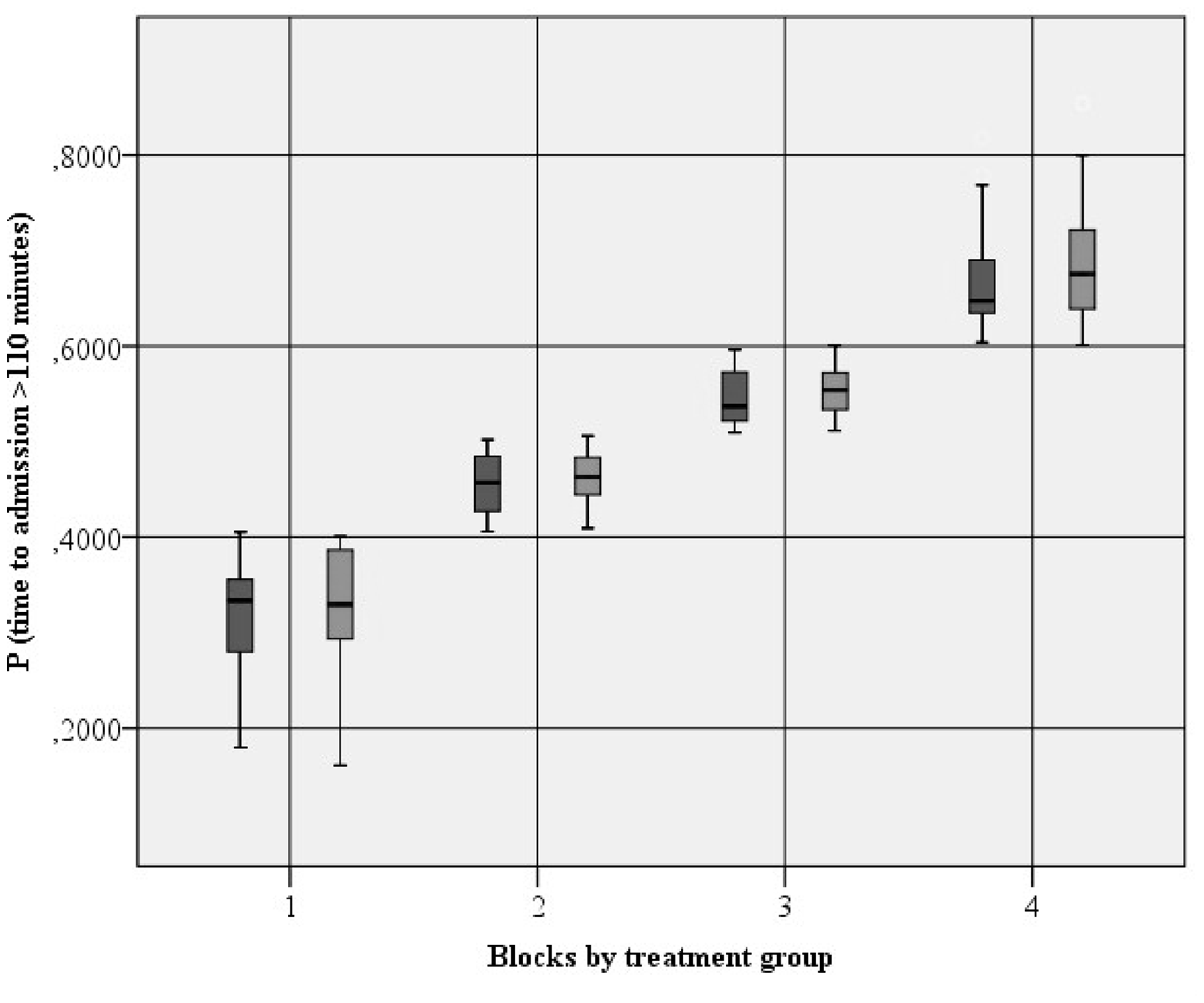
Distribution of propensity score blocks by time to admission. For each pair of blocks, the box on the left represents the group of patients with an admission ≤ 110 min and the one on the right represents the group who was admitted > 110 min.
Results
A total of 979 ICH patients were admitted to both CSCs within the study period. Four-hundred and ninety-two patients were excluded (Figure 2), leaving 487 patients for the analyses. We found baseline differences between CSC A and CSC B (Supplemental Table 1). Mean age was 72.3 years (standard deviation: 12.9), 259 (53.2%) patients were men, median NIHSS was 12 (interquartile range (IQR): 4–19), GCS was 15 (IQR: 11.7–15) and baseline median ICH volume was 18.4 mL (IQR: 4.1–49.7). Table 1 shows characteristics of patients stratified by early or late admission. Compared to patients with an admission >110 min, patients who were admitted ≤110 min were significantly younger, and had higher NIHSS scores, and were less likely to have a history of atrial fibrillation or use of oral anticoagulants. In addition, patients who were admitted ≤110 min were more likely to have a basal ganglia ICH and to show neurological deterioration.
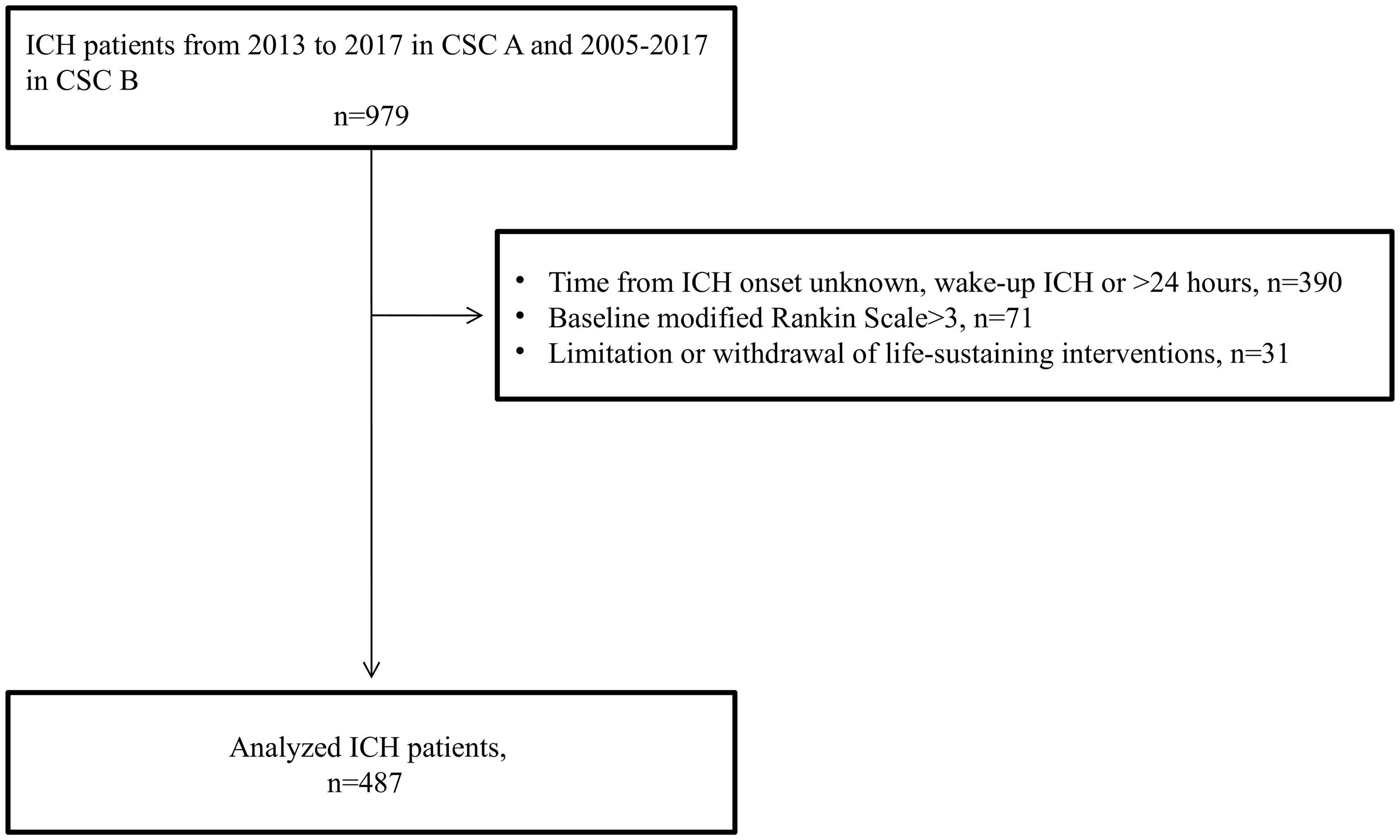
Study flow diagram.
Baseline characteristics of all patients according to time from onset to stroke centre admission.
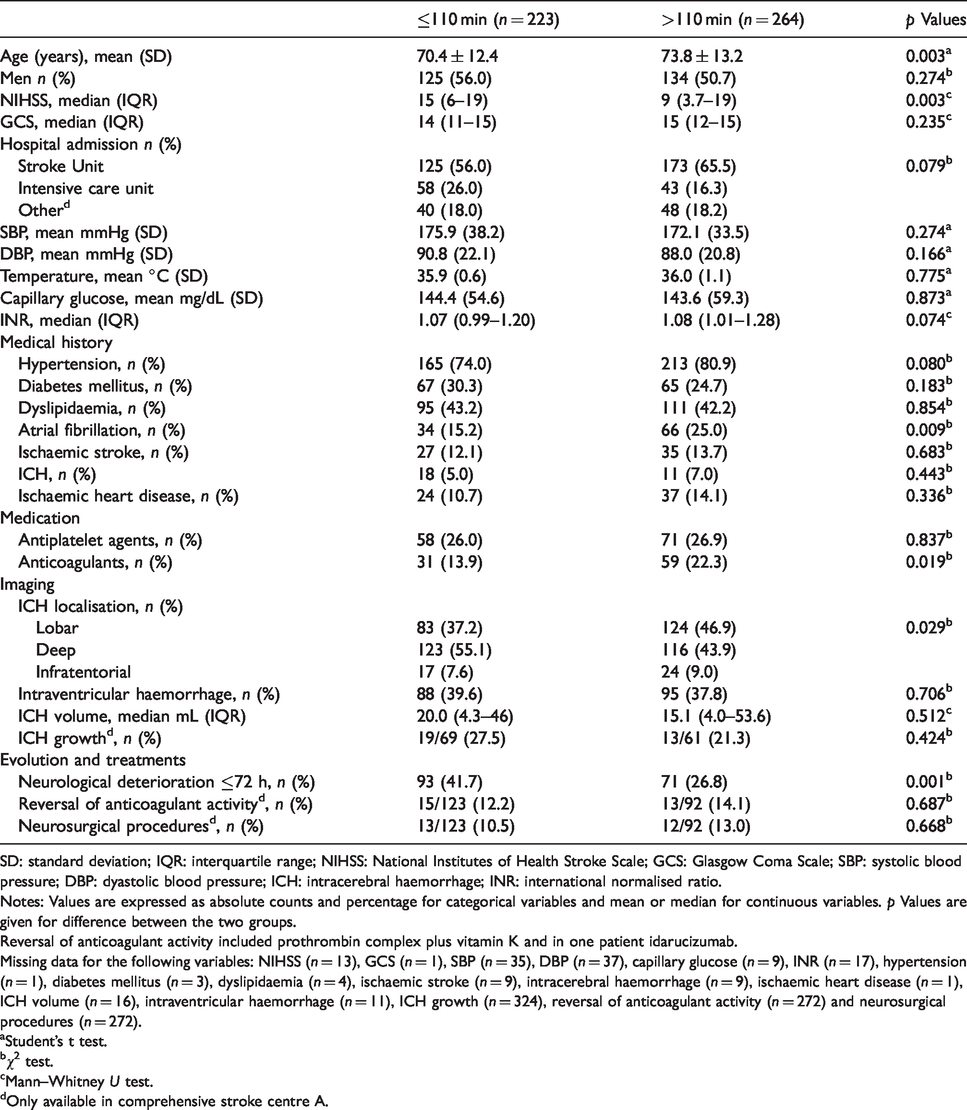
SD: standard deviation; IQR: interquartile range; NIHSS: National Institutes of Health Stroke Scale; GCS: Glasgow Coma Scale; SBP: systolic blood pressure; DBP: dyastolic blood pressure; ICH: intracerebral haemorrhage; INR: international normalised ratio.
Notes: Values are expressed as absolute counts and percentage for categorical variables and mean or median for continuous variables. p Values are given for difference between the two groups.
Reversal of anticoagulant activity included prothrombin complex plus vitamin K and in one patient idarucizumab.
Missing data for the following variables: NIHSS (n = 13), GCS (n = 1), SBP (n = 35), DBP (n = 37), capillary glucose (n = 9), INR (n = 17), hypertension (n = 1), diabetes mellitus (n = 3), dyslipidaemia (n = 4), ischaemic stroke (n = 9), intracerebral haemorrhage (n = 9), ischaemic heart disease (n = 1), ICH volume (n = 16), intraventricular haemorrhage (n = 11), ICH growth (n = 324), reversal of anticoagulant activity (n = 272) and neurosurgical procedures (n = 272).
aStudent’s t test.
bχ2 test.
cMann–Whitney U test.
dOnly available in comprehensive stroke centre A.
We obtained a balanced PS stratification between groups, except for age in block 4 (Supplemental Table 2). In block 1, those patients who were admitted ≤110 min showed higher mortality (OR: 0.39 (0.16–0.99)) at 90 days than those who were admitted >110 min (Table 2). However, in the rest of the blocks, the Mantel–Haenszel estimation showed no differences between groups in favourable functional outcome or mortality at 90 days (Table 2).
Association between time to comprehensive stroke centre admission and outcome at 90 days.
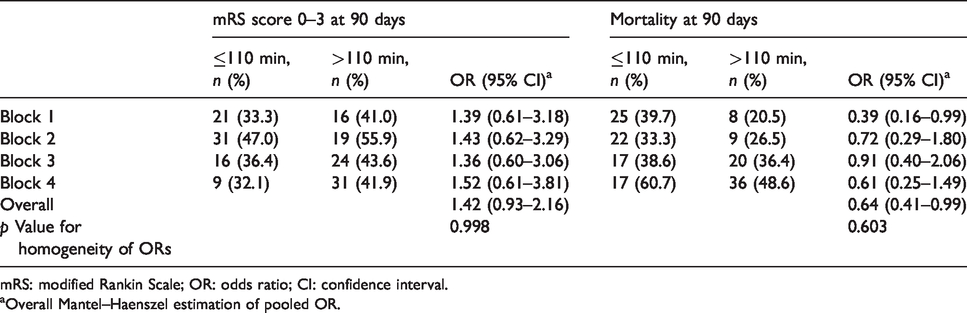
mRS: modified Rankin Scale; OR: odds ratio; CI: confidence interval.
aOverall Mantel–Haenszel estimation of pooled OR.
Discussion
We investigated the association of time to admission with outcome at 90 days in a sample of consecutive patients with ICH admitted to two different CSCs. An early CSC admission did not influence the outcome at 90 days. It is notable that, in spite of the PS stratification according to baseline characteristics, those patients who were admitted early had higher rates of neurological deterioration than patients who were admitted later. This is an expected finding since patients admitted early had more time to exhibit this neurological worsening compared to those admitted later. Therefore, we suggest that in patients with ICH who are admitted to a CSC and the symptom onset is known, a rapid time-to-admission (≤110 min) is not associated with favourable functional outcome or mortality at 90 days.
We believe that our results may be explained by the natural history of ICH and the fact that ICH treatments have a limited efficacy. ICH is a dynamic process over time in which the haematoma expands and induces injury.10,11 Within the first 24 h from the symptom onset, ICH growth and the oedema in the area surrounding the haematoma contribute to the secondary neuronal injury and neurological deterioration.10–12 The probability of ICH growth and neurological deterioration is greater in the first three hours from the symptom onset and the risk declines in the following hours. 12 In Table 1, we showed that patients who were admitted early had greater neurological deterioration than those who were admitted later. This was an expected result since these patients had a more severe clinical presentation (as evidenced by a higher NIHSS score) and this was the reason for applying the PS stratification. To evaluate neurological deterioration, we used scores such as the NIHSS or the GCS that do not completely substitute the comprehensive neurological examination, the information about the localisation of the ICH, the impact of subtle deficits or medical complications. Moreover, the PS stratification used variables that may change in the following hours after the CSC admission (NIHSS, GCS, ICH volume, and presence of intraventricular haemorrhage) and have an impact in the outcome. To date, the INTERACT 2 is the only clinical trial that showed that a rapid and intensive reduction of blood pressure (target systolic blood pressure <140 mmHg) within one hour of randomisation and continued for a duration of seven days was safe and increased the odds of lower mRS scores. 13 Nevertheless, a second trial (antihypertensive treatment of acute cerebral haemorrhage 2 (ATACH-2)), involving patients with similar demographic and clinical characteristics, did not show clinical benefits of an intensive blood pressure lowering regimen and reported an increase in renal adverse events of such treatment. 14 In our study, we did not record patients who reached an optimal blood pressure control and our sample is different than those who were included in these trials.13,14 It is notable that approximately 18% of our patients were not admitted to a Stroke Unit or the Intensive Care Unit, and we included patients with very low GCS, or high NIHSS baseline scores. We included also patients with massive ICH volumes, and we applied an aggressive management of blood pressure after the publication of the INTERACT2 trial in 2013.
Few studies have evaluated the implications of an early admission to a CSC in patients with ICH.15,16 Our findings are similar to a study of 157 patients with spontaneous ICH in whom the authors reported close associations between time to admission and clinical severity. Patients who were admitted early had a lower GCS, and larger ICH volume, as well as higher Graeb scores, compared to those who were admitted later. 15 Another multicentre study analysed 6564 patients with spontaneous ICH and reported that transferring patients with the Emergency Medical Service was associated with an early hospital admission, but the early admission did not improve survival or functional outcome. 16
We believe that a rapid management in some subgroups of patients with ICH may increase the odds of favourable outcome and survival and, therefore, our hypothesis might be supported in some patients.17–22 A meta-analysis of 40,138 patients showed that it is necessary to start tranexamic acid within the first three hours to obtain a clinical benefit. 17 A secondary analysis of the INTERACT2 trial showed that a greater systolic blood pressure reduction was associated with reduced ICH growth and the least mean ICH growth was found in patients who achieved target systolic blood pressure <1 h. 18 An exploratory analysis of the ATACH-2 trial showed that an intensive reduction of blood pressure decreases haematoma expansion specifically in haemorrhages in deep structures of the brain. 19 A pooled analysis on individual patient-level data derived from the INTERACT2 and ATACH-2 trials showed that achieving early and stable control of systolic blood pressure (as low as 120–130 mmHg) was safe and associated with favourable outcomes in adult patients who are admitted to hospital with mild-to-moderate ICH. 20 Finally, a multicentre retrospective study of oral anticoagulants associated ICH showed that reversal of INR < 1.3 within four hours and systolic blood pressure <160 mmHg at four hours were associated with lower rates of ICH enlargement. 21 Similarly, in the ‘INR Normalisation in Patients with Coumarinrelated intracranial Haemorrhages’ (INCH) trial, a fast INR normalisation (three hours after the start of treatment) was associated with reduced ICH growth. 22 These findings suggest that there are few therapeutic measures that have a modest effect in selected patients. Therefore, the improvement was not observed when we analysed the whole sample of patients.
Our study had limitations. For example, it was retrospective but we used only those variables which were collected prospectively. We excluded approximately 50% of ICH patients and, thus, the results are limited by a sample selection bias. To reduce the bias and to adjust significant differences in baseline characteristics, we performed a PS stratification. However, our PS stratification did not include other confounding factors such as transferal to the CSC effects (direct transfer or not, medical complications), volume of the surrounding oedema, degree of intraventricular haemorrhage or the ICH growth assessment. Another limitation is that the PS stratification has a considerable loss of subjects when data are missing. A considerable reduction of subjects may increase the risk of not finding differences that may actually exist (type II error). Therefore, we decided to avoid analyses in subgroups of patients from our sample such us those patients who were admitted in the CSC after the publication of the INTERACT2 trial or those patients who were admitted in the stroke unit. We analysed two CSC registries with different time frames and the spontaneous ICH management has changed over time. Notably, in 2014, the management of blood pressure and deep venous thromboembolism prevention changed in both centres. Moreover, we recommended strategies of neutralisation of the effect of direct oral anticoagulants since their commercialisation (Supplemental Table 3). We did not record neurosurgical procedures, anticoagulant reversal or ICH growth assessment in CSC B because these data were not collected prospectively and there was a large amount of missing information. In addition, we did not record if blood pressure control or anticoagulant reversal was achieved early or not, and all of these variables may an influence in the long-term outcome.
Conclusions
In our study, the early time to CSC admission (≤110 min) of patients with ICH and witnessed symptom onset was not associated with functional outcome or mortality at 90 days. Our data suggest that the current therapies for ICH and the set for measures in a CSC have a limited efficacy on the long-term functional outcome, although they may be effective in some patients. To confirm whether time is brain in ICH, we suggest that future studies should control the selection of patients; investigate which subgroups of ICH patients might benefit from a rapid CSC admission and the impact of a rapid implementation of different measures and therapies; and evaluate the optimal triage and transport strategy of these patients. In the meantime, we suggest that patients with ICH should be admitted as soon as possible to a stroke unit.
Supplemental Material
ESO901616 Supplemental material - Supplemental material for Influence of time to admission to a comprehensive stroke centre on the outcome of patients with intracerebral haemorrhage
Supplemental material, ESO901616 Supplemental material for Influence of time to admission to a comprehensive stroke centre on the outcome of patients with intracerebral haemorrhage by Luis Prats-Sánchez, Marina Guasch-Jiménez, Ignasi Gich, Elba Pascual-Goñi, Noelia Flores, Pol Camps-Renom, Daniel Guisado-Alonso, Alejandro Martínez-Domeño, Raquel Delgado-Mederos, Ana Rodríguez-Campello, Angel Ois, Alejandra Gómez-Gonzalez, Elisa Cuadrado-Godia, Jaume Roquer and Joan Martí-Fàbregas in European Stroke Journal
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Fondo de Investigaciones Sanitarias, Instituto de Salud Carlos III (FI12/00296; RETICS INVICTUS PLUS: RD16/0019/0010 and RD16/0019/0002; FEDER).
Ethical approval
The Ethics Committees of both institutions (Hospital de la Santa Creu I Sant Pau and Hospital del Mar) approved the use of these ICH registries for clinical research.
Informed consent
Written consent was obtained from all the subjects for their anonymised information to be published as clinical research articles.
Guarantor
JM-F
Contributorship
LP-S, JR, JM-F: Design of the work; draft the work and revising it critically; final approval of the version to be published; agreement to be accountable for all aspects of the work. MG-J, EP-G, NF, PC-R, DG-A, AM-D, RD-M, AR-C, AO, AG-G, EC-G: Acquisition and interpretation of data; revising it critically; final approval of the version to be published; agreement to be accountable for all aspects of the work. IG, LP-S: Analysis and interpretation of data.
Acknowledgements
We thank Professor William Stone for providing helpful comments.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
