Abstract
Abstract
Introduction
Recent data suggest that statin use after intracerebral haemorrhage might be beneficial. However, data on the effects of early in-hospital statin exposure are lacking. Therefore, we sought to assess whether (1) early statin exposure during the acute phase after intracerebral haemorrhage and (2) early continuation of prevalent statin use are associated with favourable functional outcome.
Patients and methods
Data were obtained from the Virtual International Stroke Trials Archive. Patients were categorised according to use patterns of statins during this early in-hospital phase (continuation, discontinuation or new initiation of statins). Univariate and multivariable analyses were conducted to explore the association between early statin exposure and functional outcome.
Results
A total of 919 patients were included in the analysis. Early in-hospital statin exposure (n = 89, 9.7%) was associated with better functional outcome (modified Rankin Scale ≤ 3) compared with 790 patients without statin exposure before or early after the event (66% versus 47%, adjusted OR 2.1, 95% confidence interval 1.3–3.6).
Compared with patients without exposure to statins before and early after the event, early continuation of statin therapy (n = 57) was associated with favourable functional outcome (adjusted odds ratio 2.6, 95% confidence interval 1.3–5.2). The association between early continuation of statins and outcome remained robust in sensitivity analyses restricted to patients able to take oral medication within 72 h and one-week survivors.
Discussion
It is possible that part of the observed associations are not due to a protective effect of statins but are confounded by indication bias.
Conclusion
Statin exposure and continuation of prevalent statin therapy early after intracerebral haemorrhage are associated with favourable functional outcome after 90 days.
Introduction
Treatment with HMG-CoA-reductase-inhibitors (statins) in patients with acute ischaemic stroke is associated with improved survival and better functional outcome.1,2 However, in intracerebral haemorrhage (ICH), which constitutes 10–15% of all strokes, 3 use of statins is considered more controversial.4,5
Treatment with statins in patients with recent stroke might be associated with a modestly increased risk for future ICH.4,6,7 On the other hand, there is evidence from observational and animal studies that exposure to statins at any time during hospitalisation for ICH is associated with better outcome and reduced mortality, while discontinuation of statins after ICH is associated with higher rates of poor clinical outcome and mortality.8–12 However, previous studies did not provide data regarding timing of statin use, specifically continuation of statin therapy during the first few days after ICH.8–10 This early in-hospital phase is relevant because of potential early rebound effects associated with statin discontinuation.13–16 Animal model studies in mice with cerebral ischaemia showed loss of protective statin effects as early as two days after their discontinuation. 17
Altogether, uncertainty remains on how to proceed with pre-existing statin therapy, especially in the early in-hospital phase after ICH. This is mirrored by the current guidelines from the American Heart Association and American Stroke Association on the management of spontaneous ICH, which have no clarity on statin use in patients with recent ICH. 18 There are, however, expert recommendations outside of guidelines suggesting that prevalent statin use should be continued during the early in-hospital phase.19–21
Using the Virtual International Stroke Trials Archive (VISTA) database we sought to study whether exposure to statins during the early in-hospital phase, and especially whether continuation of prevalent statin use is associated with a better functional outcome at 90 days.
Methods
Data source and patients
Data were obtained from the Virtual International Stroke Trials Archive – ICH database (VISTA-ICH, URL: http://www.virtualtrialsarchives.org/vista/). The VISTA database collects data from a variety of clinical trials for exploratory analyses; its design and goals have been published previously. 22 Prior to the data analysis, a project proposal with definitions of our outcome parameters was approved by the independent VISTA steering committee.
We included all patients with primary ICH and having available data on age, prior and concurrent medication, clinical severity as measured by Glasgow Coma Scale (GCS), haematoma volume, systolic blood pressure at baseline and functional outcome after 90 days. Figure 1 shows patient inclusion into our analysis as well as distribution of patients according to statin therapy. Functional outcome was measured using the modified Rankin Scale (mRS). Patients on oral anticoagulation before the event were excluded from the study, as there are differences in clinical outcome in these patients. 23
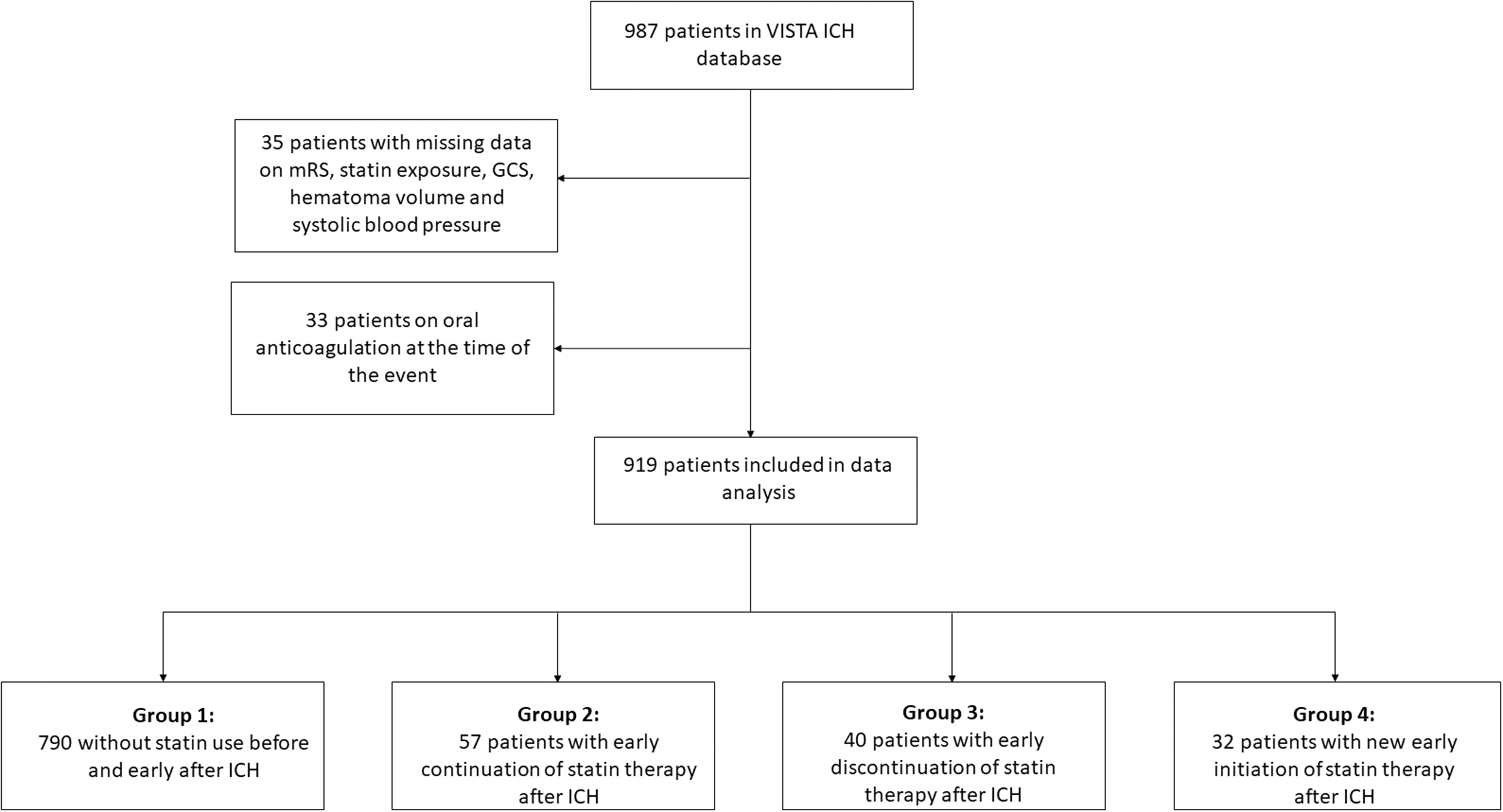
Data collection and group allocation. GCS: Glasgow Coma Scale; ICH: intracerebral haemorrhage; mRS: modified Rankin Scale; VISTA: Virtual International Stroke Trials Archive.
Definition of early statin use
We categorised patients into four groups according to early in-hospital statin exposure in the first 48 h after the event: (1) patients without statin exposure before and early after the event (‘no statin use’), (2) patients who continued prevalent statin treatment within 48 h and continued this therapy at least until day 7 after the event (‘early continuation’), (3) patients who discontinued statin use after ICH in the first 48 h after ICH (‘early discontinuation’) and (4) patients newly started on statins within 48 h after the event and continued this therapy at least until day 7 after the event (‘new early initiation’, see Figure 2). Early in-hospital statin exposure was defined as documented treatment with statins within the first 48 h after admission irrespective of prevalent statin therapy before ICH (combination of groups 2 and 4).
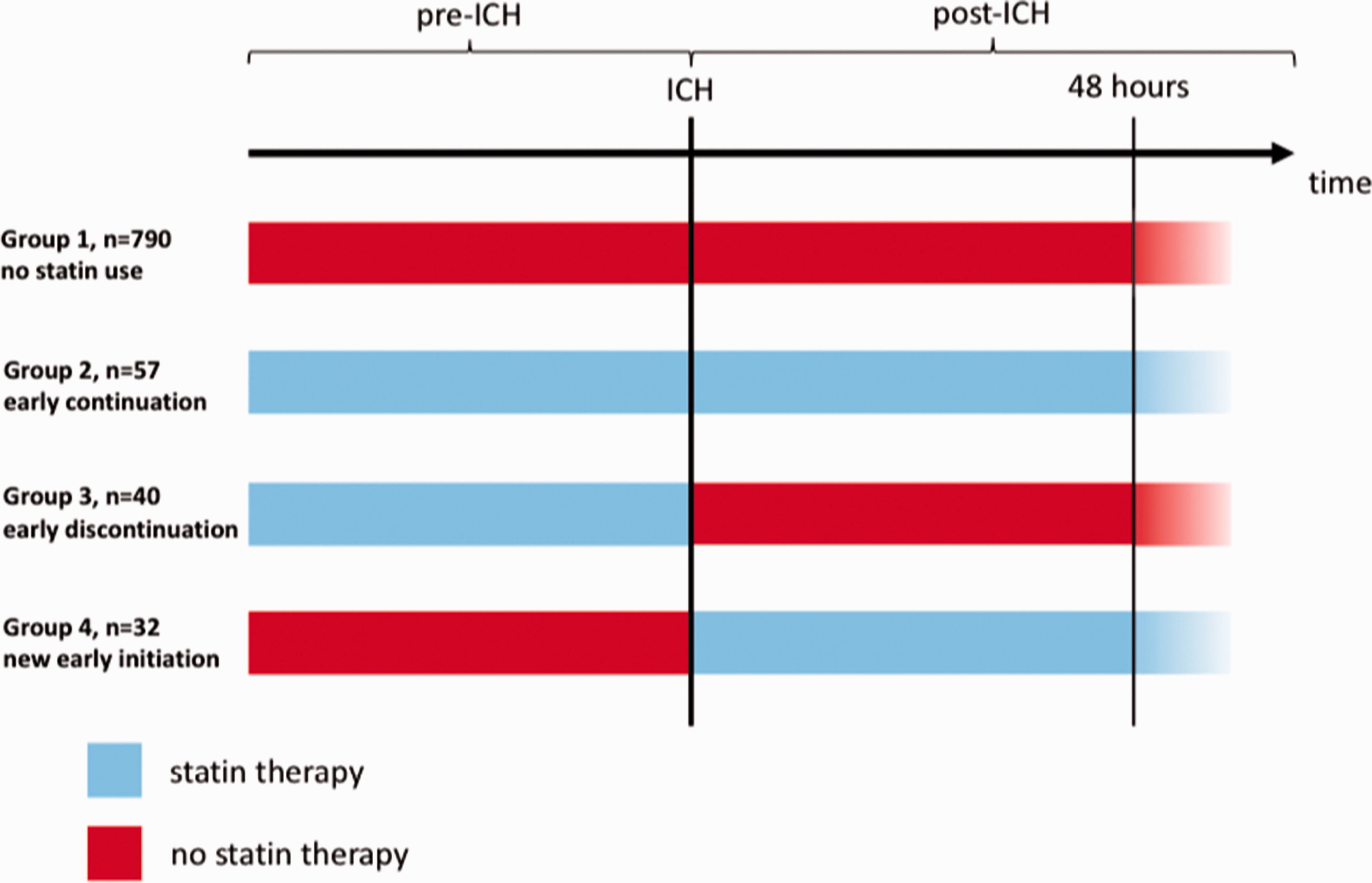
Patient groups according to statin exposure. ICH: intracerebral haemorrhage.
Group 1 included patients with late initiation of statins after 48 h but within the first week after ICH, and group 3 included patients with late continuation after 48 h but within the first week after the event.
Study outcomes
Outcome of interest was favourable functional outcome after 90 days, defined as mRS ≤ 3. This outcome measure was chosen to facilitate comparability with other studies on ICH.9,24 Further outcomes were distribution of 90-day mRS and mortality at 90 days.
Statistical analysis
For univariate analysis Pearson’s chi-squared test, Mann–Whitney U-test and Student’s t-test were used where appropriate. Unadjusted and adjusted multiple logistic regression analyses were conducted to explore the association between statin exposure and favourable functional outcome. Statin use was entered as a four-group categorical variable. Patients without statin exposure before and early after ICH (‘no statin use’) were used as reference. In order to explore the association of early statin therapy after ICH and favourable shift in 90-day mRS, we conducted an ordinal logistic regression analysis. The models were adjusted for age, sex, haematoma volume, history of prior antithrombotic use, GCS and systolic blood pressure on admission. These variables are established risk factors for unfavourable outcome after ICH and could be linked to clinical decisions regarding statin treatment (re)initiation.25–28 The variables had to be available for at least 90% of the cohort and were checked for relevant multicollinearity within the model, which was defined as the largest condition index ≥30. 29
We also compared the association between early continued statin exposure (‘early continuation’) and favourable outcome with patients who discontinued statins (‘early discontinuation’). Due to limited numbers, the multiple logistic regression analysis only included age and haematoma volume. Furthermore, we compared functional outcomes of patients who continued statin therapy early after ICH (‘early continuation’) with those who continued statins later (i.e. between days 3 and 7 after ICH, ‘late continuation’).
In addition, we performed two sensitivity analyses, restricting our multiple logistic regression to patients with documented intake of oral medication in the first 72 h in order to try to rule out patients unable to take statins because of dysphagia and to one-week survivors in an attempt to reduce selection bias. In two further sensitivity analyses we (1) replaced GCS with National Institute of Health Stroke Scale (NIHSS) into our original model and (2) added intraventricular haemorrhage into our original model.
Two-sided p-values < 0.05 were considered statistically significant. All data were analysed using SPSS version 25 (IBM, Chicago, USA).
Results
Of 987 ICH patients recorded in VISTA-ICH, 919 fulfilled inclusion criteria. Thirty-five patients were excluded because of missing data on either mRS at 90 days (19 patients), information on early statin exposure (1 patient), baseline GCS and/or haematoma volume (12 patients) and/or systolic blood pressure (3 patients). Another 33 patients were excluded because they were on oral anticoagulant treatment at the time of the event (Figure 1).
Of these 919 patients, mean age was 65.6 ± 12.4 years, 66% were male, median baseline GCS was 15 (interquartile range (IQR) 13–15), median baseline NIHSS was 13 (IQR 9–18), median ICH volume was 15 ml (IQR 7–30 ml) and median onset to randomisation time was 3.5 h (IQR 2.75–4.65).
Statin exposure
Prevalent statin use upon hospital admission was present in 97 patients (10.6%). Of these, 57 patients (6.2%) continued statin treatment within 48 h after ICH, while 40 patients (4.4%) discontinued statin use in the first 48 h after the event. The latter included seven patients (0.8%) who continued statins later than 48 h but within the first week after ICH. New early statin exposure (i.e. new initiation of treatment within 48 h) was observed in 32 patients (3.5%), while 790 (86.0%) patients were not on statins before and early after the event. The latter included 49 patients (5.3%) with late initiation of statin therapy later than 48 h but within the first week. Any early in-hospital statin exposure was documented in 89 patients (9.7%). Table 1 shows baseline characteristics according to early in-hospital statin exposure.
Baseline characteristics according to early in-hospital statin use.
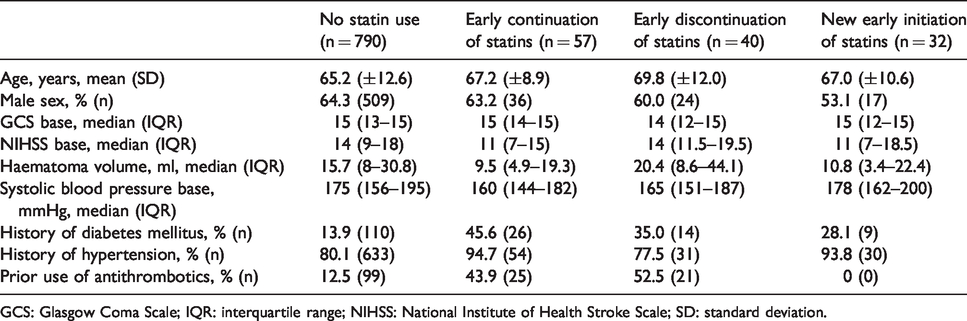
GCS: Glasgow Coma Scale; IQR: interquartile range; NIHSS: National Institute of Health Stroke Scale; SD: standard deviation.
Compared with patients not on statins before and early after the event, patients with continued statin use had lower haematoma volume (median 9.5 ml versus 15.7 ml, p = 0.002), lower systolic blood pressure at admission (median 160 mmHg versus 175 mmHg, p = 0.003) and were more often on antithrombotic drugs before the event (43.9% versus 12.5%, p <0.001).
Functional outcome at 90 days
Median mRS at day 90 was 4 (IQR 2–5), and frequency of unfavourable outcome (mRS 4–6) was 50.9% (n = 468). Frequency of favourable outcome according to early in-hospital statin exposure is shown in Table 2. Early in-hospital statin exposure (‘early continuation’ and ‘new early initiation’) was associated with better functional outcome at 90 days compared with no early statin exposure (66.3% versus 47.2%, unadjusted odds ratio (OR) 2.20, 95% confidence interval (95% CI) 1.39–3.48). These results were confirmed after adjusting for age, sex, haematoma volume, prior use of antithrombotics and GCS and systolic blood pressure at initial presentation (adjusted OR (adjOR) 2.13, 95% CI 1.26–3.60).
Association between early in-hospital statin use after ICH and functional outcome.
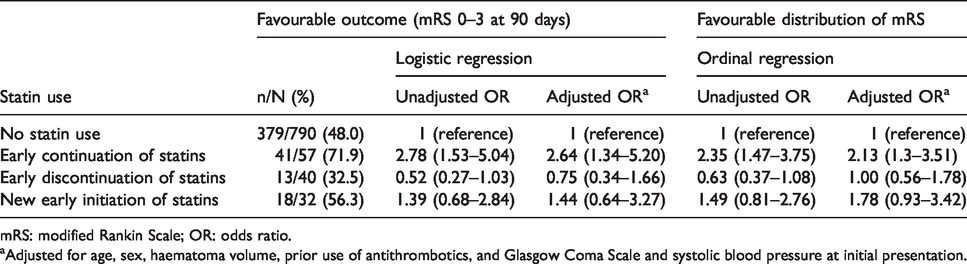
mRS: modified Rankin Scale; OR: odds ratio.
aAdjusted for age, sex, haematoma volume, prior use of antithrombotics, and Glasgow Coma Scale and systolic blood pressure at initial presentation.
When statin exposure was entered as a categorical variable into the model with patients without statin use (‘no statin use’) as reference, early continuation of pre-existing statin therapy was associated with higher probability of favourable functional outcome (unadjusted OR 2.78, 95% CI 1.53–5.04). After adjustment for age, sex, haematoma volume, prior use of antithrombotics and GCS and systolic blood pressure at initial presentation, early continuation of statins remained significantly associated with favourable functional outcome (adjOR 2.64, 95% CI 1.34–5.20), while early discontinuation of statins (adjOR 0.75, 95% CI 0.34–1.66) and new early initiation of statins (adjOR 1.44, 95% CI 0.64–3.27) were not (Table 2). To account for bias conveyed by lower clinical severity in patients with early statin continuation we, first, replaced GCS by NIHSS into our original model, and, second, included presence of intraventricular haemorrhage at baseline (n = 265) into our original model. The results regarding the association between early statin continuation and favourable outcome remained robust (adjOR 2.37, 95% CI 1.19–4.72 and adjOR 2.41, 95% CI 1.20–4.88, respectively).
Using ordinal regression analysis there also was an association between early statin continuation and a shift towards a favourable mRS score (adjusted common OR for early statin continuation 2.13, 95% CI 1.30–3.51; Table 2 and Figure 3).
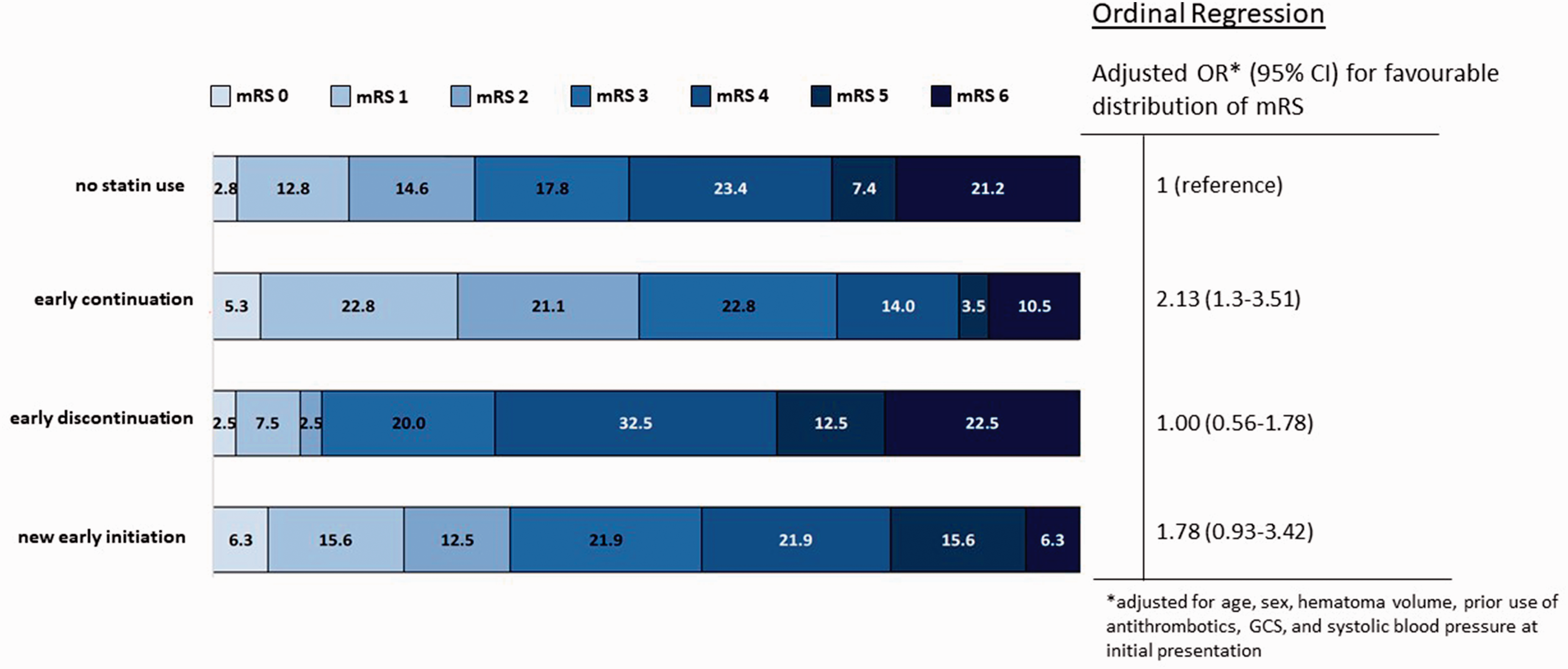
Distribution of mRS at 90 days according to statin exposure. mRS: modified Rankin Scale; 95% CI: 95% confidence interval; OR: odds ratio.
When directly compared with early discontinuation of statin therapy, early continuation of pre-existing statin therapy was also associated with higher probability of favourable functional outcome (72% versus 33%, p < 0.001, unadjusted OR 5.32, 95% CI 2.21–12.21, adjOR 3.87, 95% CI 1.46–10.27, adjusted for age and haematoma volume). Similarly, early statin continuation was associated with a shift towards favourable mRS score (adjusted common OR for continued statin use 2.75, 95% CI 1.24–6.08; versus early discontinuation of statins).
Finally, we compared early and late continuation of statins (n = 57 and n = 7). Frequency of favourable outcome was higher in the early continuation group (71.9% versus 42.9%, p = 0.117, unadjusted OR 3.4, 95% CI 0.7–17.0). Because of the small number of patients with late continuation of statin therapy, we did not perform a multiple logistic regression analysis.
Mortality at 90 days
Within the first 90 days after ICH, 183 patients (19.9%) died. Patients with early in-hospital statin exposure (‘early continuation’ and ‘new early initiation’) were less likely to die within 90 days than patients without statin exposure (9% versus 21%, p = 0.007, unadjusted OR 0.37, 95% CI 0.18–0.8, adjOR 0.4, 95% CI 0.2–0.97).
Ninety-day mortality was non-significantly lower in the early continuation group, when compared with the ‘no statin’ group (21% versus 11%, p = 0.074, unadjusted OR 0.4, 95% CI 0.2–1.1, adjOR 0.6, 95% CI 0.2–1.5, Table 3), and when compared with the early discontinuation group (11% versus 23%, p = 0.108, unadjusted OR 0.4, 95% CI 0.1–1.3, adjOR 0.7, 95% CI 0.2–2.5).
Association between early in-hospital statin use after ICH and mortality at 90 days.
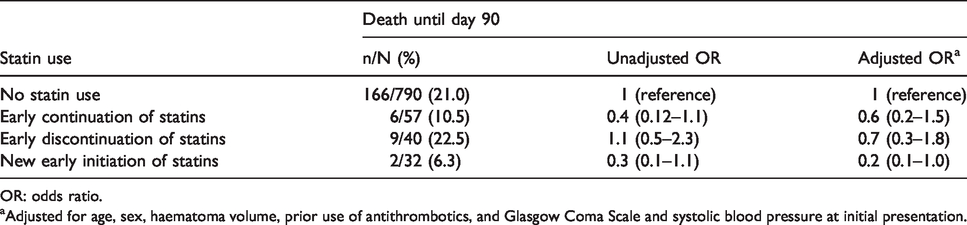
OR: odds ratio.
aAdjusted for age, sex, haematoma volume, prior use of antithrombotics, and Glasgow Coma Scale and systolic blood pressure at initial presentation.
Sensitivity analysis regarding functional outcome
In an attempt to reduce selection bias generated by patients unable to take statins because of dysphagia, we performed a sensitivity analysis regarding favourable 90-day outcome. First, we excluded 313 patients for whom no intake of any oral medication within the first 72 h was documented. Our results stayed robust (frequency of favourable outcome in patients with early continuation of statins compared with no statin exposure 75% versus 53%, unadjusted OR 2.65, 95% CI 1.27–5.54; adjOR 2.92, 95% CI 1.26–6.75). Similarly, another sensitivity analysis restricted to 839 patients, who survived the first week after ICH, showed similar associations in the multiple logistic regression analysis (frequency of favourable outcome in patients with early continuation of statins compared with no statin exposure 73% versus 53%, unadjusted OR 2.40, 95% CI 1.31–4.42; adjOR 2.55, 95% CI 1.28–5.09).
Discussion
In this study, early in-hospital exposure to statins after acute ICH was associated with better functional outcome compared with no statin exposure early after the event. Our data suggest that this association is particularly driven by continuation of pre-existing statin use within the first two days after the event. Thus, our findings provide clinical evidence to support current expert recommendations that prevalent statin use should be continued during the early in-hospital phase.19–21
Several hospital-based observational studies have explored the association between in-hospital statin exposure and functional outcome after ICH8,9,30: In a study of 3218 Chinese patients with ICH, in-hospital statin use (at any time during hospitalisation) was associated with a better functional outcome at three months and one year, in comparison to patients without in-hospital statin use. 30 Data from other studies show more conflicting results.8,9 In an analysis of data from the Ethnic/Racial Variations of Intracerebral Hemorrhage study, a prospective hospital-based multicentre study of 2457 patients with ICH, there was a signal towards better functional outcomes among patients with in-hospital statin use (combined group of patients with continued statin use and new statin use) compared with discontinuation of statins. This was, however, no longer statistically significant when a propensity score accounting for variables that could have influenced the decision to treat with statins was included in the analysis. 8 Similarly, an analysis of data from 2466 patients from the registry of the Canadian stroke network found an association between in-hospital statin discontinuation (at any time) and poor functional short-term outcome which became borderline statistically significant after excluding patients treated palliatively. 7
Importantly, these studies did not define the timing of in-hospital statin exposure. We hypothesise that any protective or detrimental effects of statins would occur early following index ICH. Clinically, the early phase after ICH is critical because of the risk for haematoma expansion, development of perihaematomal oedema and re-bleeding.31–34 Hence, continuation of statin therapy early after ICH is reasonable to avoid rebound effects associated with statin discontinuation.13–15 This argument is supported by animal models, where the protective effect of statins was lost within two days after their withdrawal. 17 Moreover, clinical data show that withdrawing statin therapy within the first 24 h after myocardial infarction is associated with increased in-hospital mortality, compared to patients without statin use. 35 These findings might explain that our observed associations remained stable even after adjusting for strong predictors of outcome after ICH.
It should be mentioned, that a direct comparison between early and late continuation of statins also is of high interest, when assessing the necessity to continue statin therapy early after ICH. However, late continuation of statins was rare in our cohort limiting the statistical power to detect statistically significant differences.
In our study, statin exposure early after ICH was significantly associated with a reduced odds of 90-day mortality compared with patients without statin exposure. Likewise, there was a signal towards lower mortality in patients who continued statins within 48 h after ICH, though this was not statistically significant. This is in line with most data derived from observational studies focusing on mortality as outcome parameter. In a recent meta-analysis by Lei et al. 36 comprising seven studies, in hospital-statin exposure was associated with an approximately 60% reduced odds of mortality. Interestingly, our observed associations fit well within these results and extend these observations to the early in-hospital phase.
Strengths and limitations
The strength of our study is the large sample size of the VISTA-ICH database which is based on multicentre randomised controlled trials (RCTs) on (hyper-)acute ICH. The high data quality required in clinical trials with rigorous documentation of time points of drug administration allowed us to define and measure early in-hospital statin use.
There are limitations to this analysis. First, our results could be confounded by indication bias. Patients with presumed good prognosis at index ICH might have received statins more likely compared to those with presumed poor prognosis. This also falls into weight as our database lacked information on the exact reason to start or continue statin treatment, such as prevalence of coronary disease, severe carotid artery disease or history of ischaemic stroke. Reassuringly, results from the sensitivity analysis, limited to one-week survivors, remained statistically significant with similar effect measures being detected. Moreover, as our database consists of patients that participated in RCTs, confounding by indication bias could be lower than in registry studies. Patients enrolled in RCTs usually differ from those registered in hospital-based cohorts in particular with regard to co-morbidities and prognosis as patients with a presumably bad prognosis are often not included in RCTs. This presumption is corroborated by the high median GCS in our patient population suggesting an overall mild-to-moderate ICH severity. Thus, our findings may not apply to a more severely affected ICH population.
Second, statins cannot be administered intravenously. Therefore, patients with dysphagia, which is a common complication of stroke, are unable to take statins. 37 To account for this, we performed yet another sensitivity analysis, excluding patients for whom no intake of oral medication was documented in the first 72 h. Again, our results stayed robust in this analysis. However, it has to be noted that the reason for not administering oral medication was not documented in our database. It is therefore possible that there were other reasons to withhold oral medication (and therefore statins) than dysphagia.
Third, the number of patients on statins was small, especially in the continuation and discontinuation groups. Therefore, we were not able to study potential effects of statin type or statin dose. This could be important as there is an association between higher dose of statin use and risk of symptomatic ICH after intravenous thrombolysis for ischaemic stroke. 38 Moreover, sample size did not allow us to provide reasonable comparison of early versus late continuation, as well as early versus late new treatment initiation.
Fourth, we have no data on the exact location of ICH, presence and location of cerebral microbleeds, haematoma expansion and recurrent ICH. This could be of significance as all these factors are associated with outcome and are potentially affected by statin exposure.39–43 Finally, our data lacked information on cholesterol levels of our patients. Therefore, we were unable to interpret the association between outcome and cholesterol levels. 44
Conclusion
We observed an association between continued statin use during the early in-hospital phase after ICH (<48 h) and a favourable functional outcome at 90 days. We are unable to differentiate if this association was due to a protective effect of early statin use in ICH patients or whether the use of statins was a surrogate parameter for an expected better prognosis. Still, our results provide clinical evidence supporting current expert recommendations to continue statins early after ICH. Well-designed randomised controlled trials are warranted to further answer questions about statin continuation in the setting of acute ICH.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: JID, AHA, BS and CHN report no disclosures. ME reports grants from Bayer and fees paid to the Charité from Bayer, Boehringer Ingelheim, BMS/Pfizer, Daiichi Sankyo, Amgen, GSK, Sanofi, Covidien, Novartis, all outside the submitted work. KRL reports no relevant disclosure, he has received Fees/expenses from Boehringer Ingelheim (for data monitoring committees); ACI Clinical, Parexel, Nestlé, American Heart Association, and EVER NeuroPharma. JFS has received fees for lectures from Stryker GmbH & Co. KG and Bristol-Myers Squibb, all outside the submitted work.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Not applicable.
Ethical approval
Not applicable.
Guarantor
JFS.
Contributorship
Study concept and design: JID and JFS. Acquisition of data: Vista collaboration, JFS. Analysis and interpretation of data: All authors. Drafting of the manuscript: JID and JFS. Critical revision of the manuscript for important intellectual content: All authors. Statistical analysis: JID, JFS.
Acknowledgements
VISTA-ICH Steering Committee: Daniel F. Hanley (Chair), Kenneth S. Butcher, Stephen Davis, Barbara Gregson, Kennedy R. Lees, Patrick Lyden, Stephan Mayer, Keith Muir, Else C. Sandset, and Thorsten Steiner.
