Abstract
Abstract
Introduction
We assessed whether modest systemic cooling started within 6 hours of symptom onset improves functional outcome at three months in awake patients with acute ischaemic stroke.
Patients and methods
In this European randomised open-label clinical trial with blinded outcome assessment, adult patients with acute ischaemic stroke were randomised to cooling to a target body temperature of 34.0–35.0°C, started within 6 h after stroke onset and maintained for 12 or 24 h , versus standard treatment. The primary outcome was the score on the modified Rankin Scale at 91 days, as analysed with ordinal logistic regression.
Results
The trial was stopped after inclusion of 98 of the originally intended 1500 patients because of slow recruitment and cessation of funding. Forty-nine patients were randomised to hypothermia versus 49 to standard treatment. Four patients were lost to follow-up. Of patients randomised to hypothermia, 15 (31%) achieved the predefined cooling targets. The primary outcome did not differ between the groups (odds ratio for good outcome, 1.01; 95% confidence interval, 0.48–2.13; p = 0.97). The number of patients with one or more serious adverse events did not differ between groups (relative risk, 1.22; 95% confidence interval, 0.65–1.94; p = 0.52).
Discussion
In this trial, cooling to a target of 34.0–35.0°C and maintaining this for 12 or 24 h was not feasible in the majority of patients. The final sample was underpowered to detect clinically relevant differences in outcomes.
Conclusion
Before new trials are launched, the feasibility of cooling needs to be improved.
Introduction
In selected patients with acute ischaemic stroke, intravenous thrombolysis and endovascular treatment improve functional outcome.1–3 However, among patients treated with thrombolysis within 3 hours of stroke onset in clinical trials, 59% were dead or dependent at the end of follow-up, 1 and in trials of endovascular treatment, 29–67% of the patients randomised to the intervention arm were dead or dependent at three months. 3 In addition, only a minority of patients with acute ischaemic stroke are eligible for revascularisation therapies: the Action Plan for Stroke in Europe 2018–2030 aims to achieve rates above 15% for intravenous thrombolysis and above 5% for endovascular therapy across Europe. 4 For this reason, there is an urgent need for additional treatment strategies that can be applied in a broad range of patients.
Therapeutic hypothermia appears a promising candidate treatment. Systematic review and meta-analysis of animal studies modelling ischaemic stroke have shown convincingly that therapeutic hypothermia reduces infarct size and improves neurological outcomes. The benefit of hypothermia appears inversely related to the temperature achieved, with a reduction in infarct size of about one third with cooling to 35°C and by around 45% with cooling to 34°C. 5 Most phase II clinical trials have reported that cooling awake patients with ischaemic stroke to target temperatures of 33–35°C for 12–24 h is feasible and safe, despite an increased risk of pneumonia in some studies.6–10 Both safety and feasibility were confirmed in a recent phase III clinical trial that was terminated prematurely after inclusion of 120 of the intended 1600 patients. 11 All published trials were underpowered to detect any effect of hypothermia on functional outcome.
In this randomised clinical trial, we aimed to assess whether systemic cooling to a target body temperature between 34.0°C and 35.0°C, started within 6 h of symptom onset and maintained for 12 or 24 h, improves functional outcome at three months in awake patients with acute ischaemic stroke.
Methods
Study design
EuroHYP-1 was an investigator-initiated, international, multicentre, randomised, open-label, blinded outcome (PROBE), parallel-group, clinical superiority trial, registered at ClinicalTrials.gov as NCT01833312. The trial was coordinated at the University Hospital Erlangen and funded by the European Union’s (EU) Seventh Framework Programme under grant agreement n° 278709. The final full study protocol is available as an online supplement to this paper; a summary of the original study protocol and the final statistical plan have been published.12,13 The trial was overseen by a steering committee and by an independent data and safety monitoring board. Coordination of other trial activities such as training, monitoring, data management, and data analysis were divided over eight institutions in seven European cities.
Participants could be enrolled by investigators from 36 hospital sites in nine countries: Belgium, Denmark, Finland, France, Germany, Italy, Lithuania, Spain and the UK. Investigators were certified by web-based training modules on cooling procedures; training provided by manufacturers of cooling systems on their devices; web-based certification on assessment of the National Institutes of Health Stroke Scale (NIHSS) 14 and modified Rankin Scale (mRS) 15 and provision of written instructions on trial-specific procedures. Experience with therapeutic hypothermia in awake patients with acute stroke was not a requirement. Ethics approval was obtained in each country and (where required) at each site before the start of the study in that country or site. Patients or their representatives provided written informed consent according to national and local regulations.
Patients
Patients were eligible if they had a diagnosis of acute ischaemic stroke; an age of 18 years or older; a score on the NIHSS of 6–18 inclusive; a score on the motor response sub-scale of the Glasgow Coma Scale of 5 or 6; a pre-stroke score on the mRS ≤ 2; an estimated body weight of 50–120 kg; a possibility to start therapeutic hypothermia within 6 h after onset of stroke; and a possibility to start therapeutic hypothermia within 90 min after start of alteplase administration in patients receiving thrombolysis or a possibility to start therapeutic hypothermia within 90 min after admission to the trial site if not receiving thrombolysis. All patients underwent CT or MRI brain imaging to exclude haemorrhage and other non-ischaemic causes of stroke; the presence of early acute ischaemic signs (e.g. subtle brain hypoattenuation, hyperattenuated artery), prior infarcts, leukoaraiosis or brain atrophy were not exclusion criteria as long as the acute changes were consistent with the known time of symptom onset. An overview of the reasons for exclusion from the trial is available in the full study protocol.
In an attempt to enhance recruitment after inclusion of the first 50 patients, and to accommodate the widespread use of endovascular treatment, the upper limit on the NIHSS was dropped and the time window to start of treatment was increased to 150 min after start of alteplase administration in patients receiving thrombolysis at the trial site, to 150 min after start of endovascular treatment, if this was later, and to 150 min after admission to the trial site in patients not receiving thrombolysis or in patients who had received thrombolysis at a different site, without a change in the maximum time to start of hypothermia of six hours after symptom onset.
Treatment
Patients were allocated to hypothermia plus standard care or to standard care alone. Randomisation was performed through a web-based allocation service and involved stratification by country and minimisation on: intention to give alteplase; intended method of cooling (surface or endovascular); sex; stroke severity (dichotomised between NIHSS 12 and 13); age (≤65 years or >65 years); visibility of a relevant ischaemic lesion on the first brain imaging (yes or no) and time since symptom onset (≤4 h or 4–6 h).
In patients randomised to hypothermia, cooling was started within 6 h after onset of symptoms and within 90 min of start of thrombolysis (or within 90 min of hospital admission in patients who were not treated with thrombolysis) with intravenous infusion of 20 mL/kg refrigerated normal saline (4°C) over 30–60 mins or a pre-specified surface cooling method, depending on local practice and preference. As mentioned above, the maximum delay to start of treatment after intravenous thrombolysis or study site admission was increased to 150 min after the first 50 patients. Cooling was maintained at 34.0–35.0°C for 24 h with a surface or endovascular technique. The choice of the cooling technique was at the discretion of the local investigator, as long as protocol-specified devices were used. Thereafter, patients were passively rewarmed at a rate of 0.2 ± 0.1°C per hour until the rectal or bladder temperature reached 36.0°C, after which the cooling device was disconnected. To increase the feasibility of cooling and thereby to increase recruitment, the duration of active cooling was reduced from 24 to 12 hours after inclusion of the first 50 patients.
In patients randomised to hypothermia, shivering and discomfort were prevented and treated with intravenous pethidine (max 500 mg/24 h) and oral buspirone (max 30 mg/24 h). Patients who had difficulties swallowing did not receive buspirone unless they had a nasogastric tube. For the prevention of opioid-induced nausea and vomiting, ondansetron or granisetron were recommended.
All patients were treated according to published guidelines for the treatment of acute ischaemic stroke and for secondary prevention.
Outcomes
The primary outcome was the score on the mRS at 91 days (±14 days), as analysed with ordinal logistic regression and expressed as a common odds ratio (OR). Secondary outcome included death or dependency, defined as a score on the mRS > 2; death; score on NIHSS; brain infarct size (on CT or MRI, volume, centrally and blindly assessed); World Health Organization Disability Assessment Schedule (WHODAS) 2.0 score and EuroQoL-5D-5L score,16–18 all at 91 days, except for brain infarct size that was assessed at 48 h. The primary safety variable was the occurrence of serious adverse events (SAEs) until day 91.
To allow blinded assessment of the primary outcome and to enhance statistical power, the mRS at 91 days was assessed by at least four independent adjudicators based on a video recording of the mRS interview, with masking to treatment allocation, and coordinated from the University of Glasgow. 19
Sample size
We originally estimated that a total of 1474 patients was required to detect an absolute reduction in the risk of death or disability (mRS > 2) of 7% (corresponding to an OR of 0·74) with 90% power at the 5% significance level (two sided) and allowing for 3% loss to follow-up. This target sample size was rounded to 1500 patients. When recruitment proved to be much slower than expected, we aimed for 80% power, allowing a reduction in the target sample size to 750 patients, which were rounded to 800.
Statistical analysis
Primary and secondary efficacy outcomes were assessed in the intention-to-treat population (all included patients). The primary efficacy variable, the score on the mRS at 91 days, was intended to be determined with ordinal logistic regression. However, because the proportional odds assumptions of the ordinal regression analyses were not fulfilled in any of the analyses of the primary outcome, the two groups were also compared using van Elteren’s test stratified by nationality, as defined in the statistical analysis plan. 13
For secondary and exploratory outcomes frequencies and percentages per group as well as risk ratios with 95% confidence intervals (CIs) were reported for binary outcomes. We used logistic regression for binary quantities, the general linear univariate model for continuous outcomes and the Poisson distribution or negative binomial distribution for rate outcomes. If the assumptions of the Poisson or negative binomial models were not fulfilled with reasonable approximation, a non-parametric method (van Elteren adjusted by nationality of centre) would be used.
Predefined per-protocol analyses were performed in all patients who had a body temperature ≤35.0°C for at least 6 hours during the active cooling period.
Results
Enrolment into the trial started in November 2013 and was stopped in March 2018 after inclusion of 98 patients at 23 study sites because of slow recruitment and the expiration of the funding period. Forty-nine patients were randomly assigned to the hypothermia group and 49 to the control group (Figure 1). At baseline, the mean age of the patients was 70 years (range, 34–92) and the median score on the NIHSS 11 (interquartile range (IQR), 7–17). Forty three (44%) were female. Baseline characteristics were balanced between the two groups (Table 1). One patient in the hypothermia group and three in the control group were lost to follow-up at 91 days.
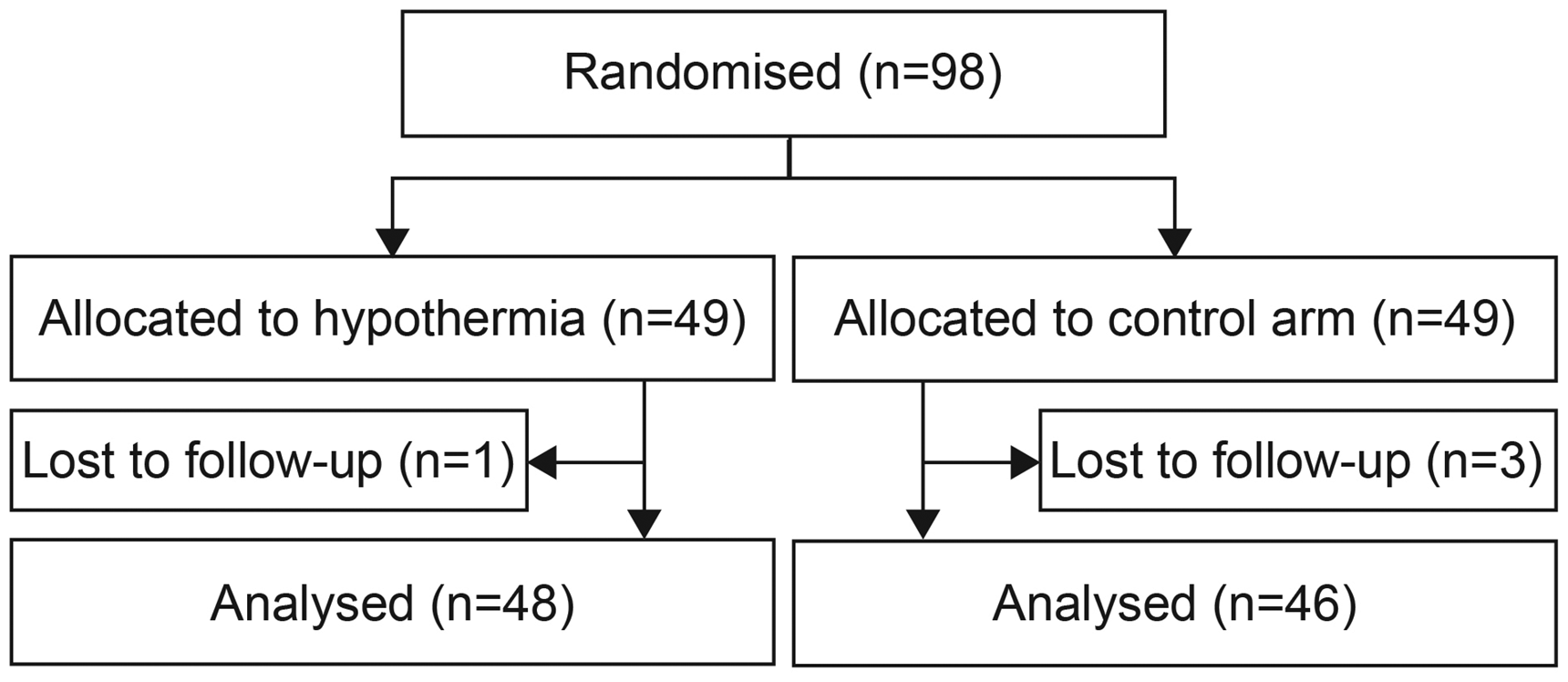
Flow of patients through the study.
Of the 49 patients randomised to hypothermia, 38 (77%) were cooled with surface cooling and 11 with an endovascular technique. Twenty five patients (51%) had a planned duration of active cooling of 24 h, and 24 (49%) of 12 h. Only 15 (31%) of the 49 patients randomised to hypothermia achieved cooling to the extent defined in the study protocol and had a body temperature ≤35.0°C for at least 6 h during the active cooling period. On the first day after randomisation, 18 (37%) patients randomised to hypothermia and 12 (24%) controls were treated on an intensive care unit or intermediate care unit/high dependency unit. The other patients were treated on a stroke unit or normal ward.
Body temperatures in the first 12 h after randomisation are shown in Figure 2.
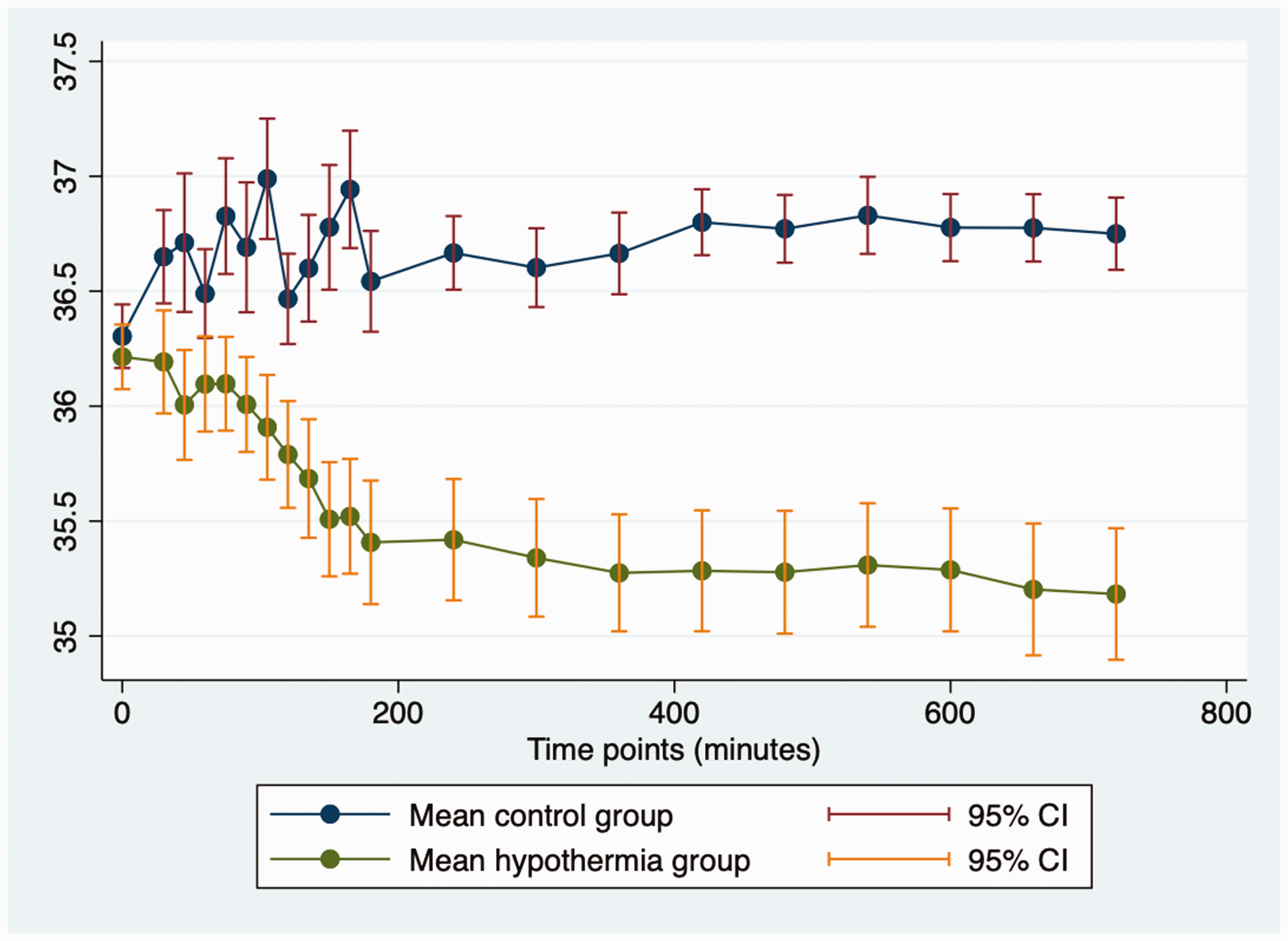
Body temperatures in the first 12 h after start of treatment in the overall trial population.
Efficacy outcomes
In the intention-to-treat analysis, there was no difference between the groups in the scores on the mRS at 91 days (OR for good outcome, 1.01; 95% CI, 0.48–2.13; p = 0.97; Figure 3). The same applied to the per-protocol analysis, including 15 patients randomised to hypothermia and 46 to standard treatment (OR, 1.07; 95% CI, 0.35–3.32; p = 0.91). There were also no differences in the risks of death or death or dependency at 91 days, the scores on the NIHSS or Visual Analogue Scale (VAS) of the EQ-5D-5 L at 91 days, or infarct volume at 48 h (Table 2). Per-protocol analyses yielded essentially the same results as the intention-to-treat analyses (data not shown).
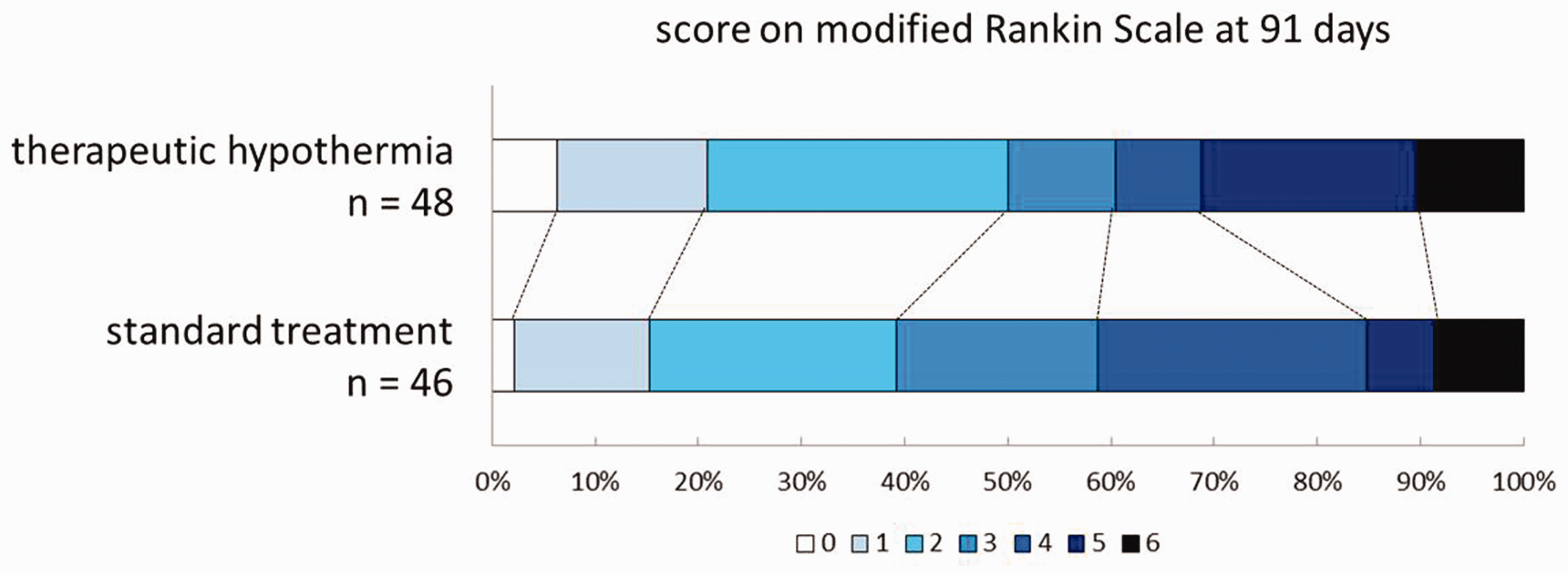
Distribution of scores on the modified Rankin Scale at 91 days.
Characteristics of patients at baseline.

mRS: modified Rankin Scale; NIHSS: National Institutes of Health Stroke Scale; IQR, interquartile range.
Secondary efficacy outcomes.
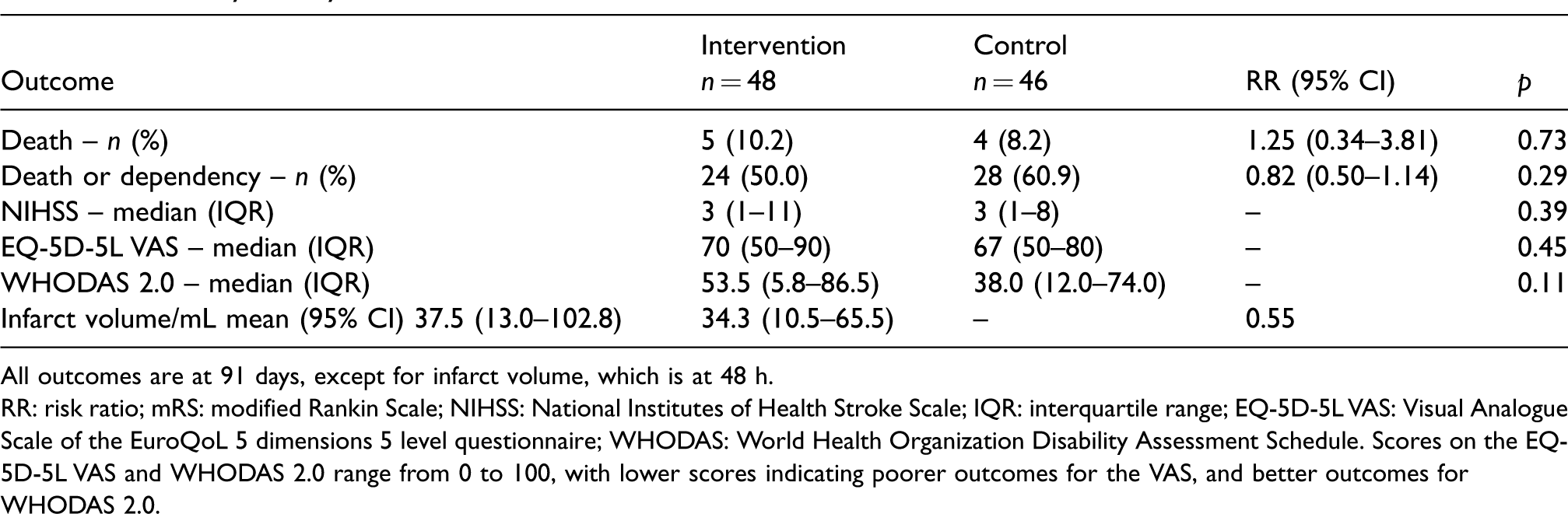
All outcomes are at 91 days, except for infarct volume, which is at 48 h.
RR: risk ratio; mRS: modified Rankin Scale; NIHSS: National Institutes of Health Stroke Scale; IQR: interquartile range; EQ-5D-5L VAS: Visual Analogue Scale of the EuroQoL 5 dimensions 5 level questionnaire; WHODAS: World Health Organization Disability Assessment Schedule. Scores on the EQ-5D-5L VAS and WHODAS 2.0 range from 0 to 100, with lower scores indicating poorer outcomes for the VAS, and better outcomes for WHODAS 2.0.
Safety outcomes
Seventeen patients (38%) in the hypothermia group and 14 controls (29%) had at least one serious adverse event (relative risk, 1.22; 95% CI, 0.65–1.94; p = 0.52; Table 3).
Serious adverse events.
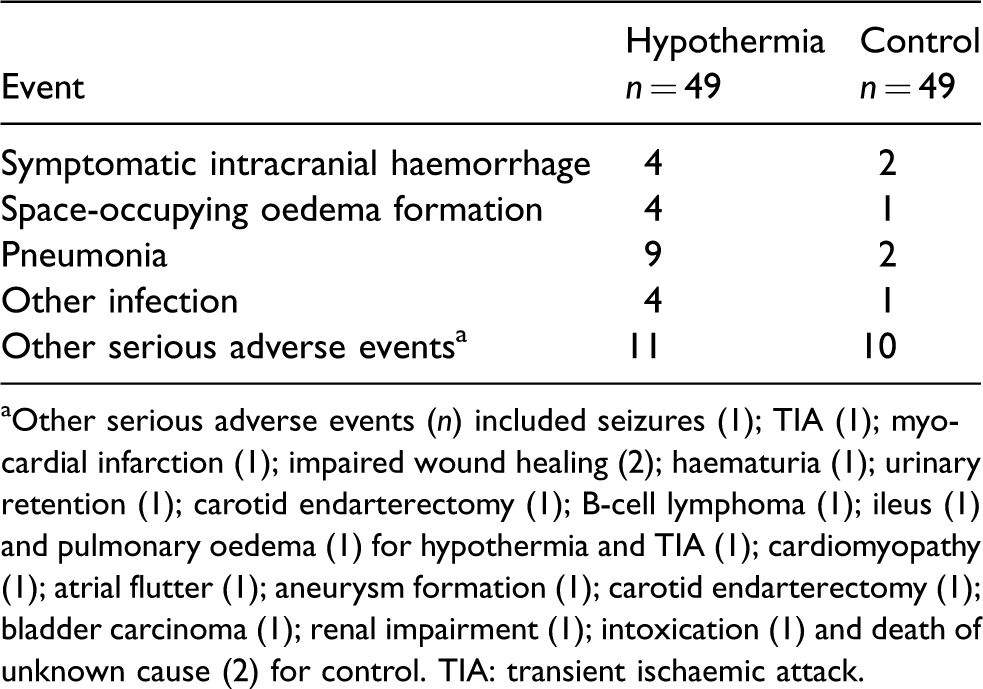
aOther serious adverse events (n) included seizures (1); TIA (1); myocardial infarction (1); impaired wound healing (2); haematuria (1); urinary retention (1); carotid endarterectomy (1); B-cell lymphoma (1); ileus (1) and pulmonary oedema (1) for hypothermia and TIA (1); cardiomyopathy (1); atrial flutter (1); aneurysm formation (1); carotid endarterectomy (1); bladder carcinoma (1); renal impairment (1); intoxication (1) and death of unknown cause (2) for control. TIA: transient ischaemic attack.
Discussion
The results of this trial provide no evidence that active cooling to a target of 34.0–35.0°C for 12–24 h started within 6 h after onset of ischaemic stroke has an impact on functional outcome at three months. However, with 98 patients included against an original target of 1500, the trial was substantially underpowered to detect any clinically relevant benefit or harm. In addition, only one third of the patients randomised to hypothermia reached the predefined cooling targets, which further limits the interpretation of the findings.
We were disappointed by the small number of patients included into the trial despite a project duration of six years, involvement of 36 trial sites and generous funding. We think this may be explained by several factors. First, cooling awake patients with ischaemic stroke is a complex intervention with which most study sites had no experience. While centres recognised the importance of the scientific question, there was some understandable caution in proceeding from trial approval to recruitment. Regardless of training and experience, inclusion of an eligible patient into the trial required a major investment in time of study staff during the full treatment period of 12 or 24 h, and this was often not feasible. Next, the trial design, regulatory and reporting requirements and the overall degree of complexity had more in common with industry-initiated trials than with academic-led so-called ‘pragmatic’ trials. This further increased the burden to local investigators. Earlier phase II trials of hypothermia for patients with acute ischaemic stroke in centres without previous experience with cooling have suggested that much higher recruitment rates are possible with more pragmatic trial designs.9,10 Third, we had chosen to distribute core trial activities across a range of universities and enterprises across Europe – on the view that such distribution was more likely to lead to funding success – rather than a more conventional centralised approach based in the institution of the study sponsor. In retrospect this was an error, and impeded effective trial coordination. We suggest that future EU-funded trials should have central provision of core trial functions unless the applicants can provide a compelling reason why this should not be the case. Finally, initiation of study sites was severely delayed due to very high and inconsistent bureaucratic hurdles across Europe and the trial being variously considered a device trial, a drug trial, both, or neither in different jurisdictions, each operating under the same EU Clinical Trials Directive.
Previous small randomised trials had suggested that cooling to targets ranging from 33°C to 35°C was feasible in the large majority of patients with acute ischaemic stroke.6–11 However, these studies were performed in a few centres, often with experience with hypothermia, whereas our phase III trial included patients in a total of 23 study sites of which just a few had previous experience with cooling awake patients with ischaemic stroke. Before being opened for recruitment, the staff at each study site were not only trained in the study protocol, but also in the technique of cooling. Unfortunately, our study showed that it was not possible to achieve cooling targets in the majority of non-sedated stroke patients, at the current time with typical limited staffing and equipment resources that are available even in expert centres. One small phase II trial in patients with ischaemic stroke has suggested that surface cooling is feasible to 35.0°C, but not to 34.5°C or 34.0°C. 10 Future trials of hypothermia in awake patients with acute ischaemic stroke could therefore target a modest temperature reduction to 35.0°C within the usual care environment. Larger reductions in temperature probably require not only more investigator experience, but also intensive nursing care on an intensive care unit or high-dependency unit.
Shivering occurred in all our cooled patients and has been reported as a common adverse effect in all previous cooling trials involving awake patients with acute ischaemic stroke, despite anti-shivering regimes. Shivering could be the most important reason for not reaching and maintaining target temperatures. In our study, the dose of pethidine as the main anti-shivering drug was limited to a maximum of 500 mg during the period of cooling, which is about half that administered in previous trials targeting a body temperature of 33°C for 24 h8,11 and also substantially less than that used in a trial testing surface cooling from 34.0 to 35.0°C. 10 The maximum pethidine dose of 500 mg in our trial was motivated by the maximum daily dose in the drug’s summary of product characteristics (SPC), to which other trials were apparently not bound. The pethidine dose in our trial was however higher than the dose of 2·3 mg/kg used in a recent surface cooling trial targeting a body temperature of 35°C for 12 h. 9 In that trial, pethidine was combined with intravenous dexmedetomidine, which we did not use because the SPC of dexmedetomidine reports acute cerebrovascular conditions as a contraindication to its use. We think that any new study on hypothermia in patients with acute ischaemic stroke should only commence after the development and validation of better methods to prevent shivering and discomfort.
In our trial, cooling was associated with an increased risk of pneumonia. This is in line with most previous studies of hypothermia in patients with acute ischaemic stroke, and with findings of a systematic review on infections in patients treated with hypothermia for any indication. 20 In patients with acute stroke, the occurrence of pneumonia has been associated with a greater risk of a poor outcome. 21 It is however uncertain whether pneumonia can be prevented with prophylactic treatment with an antibiotic, and whether this might have an impact on outcome. 22
In addition to evidence from animal studies that cooling improves functional and pathological outcomes in acute ischaemic stroke, 23 the concept of our trial was supported by randomised trials in adult patients with cardiac arrest in the previous two decades, demonstrating benefit of hypothermia. 24 However, in a more recent randomised trial in this patient population, cooling to a target of 33°C did not confer a benefit as compared with maintaining body temperature at 36°C. 25 This suggests that in patients with cardiac arrest outcomes may be improved by either cooling to 36°C, or by the prevention of sub-febrile temperatures and fever. In patients with acute stroke, elevated body temperatures have been associated with an increased risk of a poor outcome23,26,27 and systematic review and meta-analysis of animal studies modelling ischaemic stroke have suggested that this relation may in part be causal. 28 Rather than active cooling, the prevention of fever could therefore be a target to improve outcomes after stroke. A randomised trial on prevention of infections and fever in patients with moderately severe to severe stroke is ongoing. 29
Hypothermia is considered a powerful neuroprotective intervention in animal models of stroke, to the extent that inadvertent cooling was held to confound early laboratory studies. It might be argued that a failure of hypothermia to provide neuroprotection in human stroke is further evidence that animal studies do not model human pathophysiology with sufficient fidelity to be a useful guide to developing new treatments. However, differences in thermal mass mean that the process of cooling a rodent brain can proceed much more rapidly and with greater control than can be achieved in the awake human brain in a busy ward, and very few patients indeed achieved cooling to target, so we do not believe that our findings have any relevance to the question of the usefulness of animal models. Future studies might explore the effect of more direct approaches to cooling, for instance through the instillation of cooled fluids directly to the cerebral circulation during endovascular procedures.
In conclusion, the limited feasibility of the intervention under study at the current time and practical hurdles preventing sufficient recruitment into our trial have prevented final conclusions on the benefit of hypothermia in awake patients with acute ischaemic stroke to be made. Improvement of the feasibility of hypothermia in awake stroke patients and simplification of the study design are required before this treatment can be tested again in a randomised trial. For now, the low feasibility that we encountered militates against the premise of our study: with the current methods, hypothermia is unlikely to be a widely applicable treatment for patients with acute ischaemic stroke.
Supplemental Material
Supplemental material for Therapeutic hypothermia for acute ischaemic stroke. Results of a European multicentre, randomised, phase III clinical trial
Supplemental Material for Therapeutic hypothermia for acute ischaemic stroke. Results of a European multicentre, randomised, phase III clinical trial by H Bart van der Worp, Malcolm R Macleod, Philip MW Bath, Raj Bathula, Hanne Christensen, Bridget Colam, Charlotte Cordonnier, Jacques Demotes-Mainard, Isabelle Durand-Zaleski, Christian Gluud, Janus Christian Jakobsen, Bernd Kallmünzer, Rainer Kollmar, Derk W Krieger, Kennedy R Lees, Dominik Michalski, Carlos Molina, Joan Montaner, Risto O Roine, Jesper Petersson, Richard Perry, Nikola Sprigg, Dimitre Staykov, Istvan Szabo, Geert Vanhooren, Joanna M Wardlaw, Per Winkel, Stefan Schwab and the EuroHYP-1 investigators in European Stroke Journal
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: HBvdW has received speaker’s fees and serves as a consultant to Boehringer Ingelheim; PMB has received honoraria for advisory work from DiaMedica, Moleac, Nestle, Phagenesis, Sanofi and ReNeuron; HC has received speaker’s fees from MSD, Bayer and Medtronic and served as a consultant to Boehringer Ingelheim. IDZ has received speaker’s fees and/or has served as a consultant to Bayer Pharma, BMS Pfizer, Boehringer Ingelheim, Medtronic and Sanofi. BK has received speaker’s fees from Zoll Medical, Medtronic, Boehringer Ingelheim, Daiichi Sankyo and Pfizer. RK has received speaker’s fees from Boehringer Ingelheim, Bayer, Bard Medical and Pfizer and serves as consultant to Bard Medical. KRL has received honoraria from Boehringer Ingelheim for participation in Independent Data Monitoring Committees; JP has received speaker’s fees and/or has served as a consultant to Biogen, Braincool, MSD and QuickCool. ROR has received speaker’s fees and/or has served as a consultant to Bayer Pharma, BMS Pfizer, Boehringer Ingelheim, Orion Pharma, Biogen, Merck, Medtronic, Gore, Cardialysis and BrainCool; DS has received speaker’s fees from Boehringer Ingelheim, Bayer, Daiichi Sankyo, Zoll Medical, Bard Medical, Pfizer and serves as consultant to Zoll Medical and Boehringer Ingelheim. ISZ serves Becton Dickinson in a senior management role as of June 2017. The other authors report no conflicts of interest. Bard Medical and Zoll Medical have supported the EuroHYP-1 trial through the provision of temperature management systems at a preferential prize and through training of local study staff; Vacutest Kima has donated the EuroHYP-1 with materials for blood sampling.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The research leading to these results has received funding from the European Union’s Seventh Framework Programme (FP7/2007–2013) under grant agreement n° 278709. HBvdW was supported by the Dutch Heart Foundation (2010T075); PMB is Stroke Association Professor of Stroke Medicine and is a National Institute of Health Research Senior Investigator. The prefunding phase was supported by an unconditional grant from W. Hacke from his 2009 Max Jarecki and Mihara awards.
Ethical approval
Ethics approval was obtained in each country and (where required) at each site before the start of the study in that country or site.
Informed consent
Patients or their representatives provided written informed consent according to national and local regulations.
Guarantor
HBvdW
Contributorship
HBvdW, MRM, PMB, CG, RK, DWK, KRL, JP, IS, JMW and SS conceived the study and developed the protocol. All authors contributed to the conduct of the study, with the exception of CG, JCJ and PW who performed the blinded statistical analyses. HBvdW wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version.
Acknowledgements
Members of the EuroHYP-1 trial who included patients are (local principal investigator; number of patients included): Universitätsklinikum Erlangen, Erlangen, DE (S. Schwab; 13); Department of Neurology, University of Leipzig, Leipzig, DE (D. Michalski, C. Hobohm; 6); Universitätsklinikum Carl Gustav Carus, Dresden, DE (H. Schneider; 1); Martin-Luther-Universität Halle-Wittenberg, Halle, DE (K. Wartenberg; 3); Klinikum Altenburger Land, Altenburg, DE (J. Berrouschot; 1); Universitätsklinikum Frankfurt, Frankfurt, DE (M. Lorenz 3); Bispebjerg Hospital, Copenhagen, DK (H.C. Christensen; 7); University of Nottingham, Nottingham, UK (N. Sprigg; 7); Northwick Park Hospital, Northwick, UK (R. Bathula; 10); Royal Liverpool, Liverpool, UK (P. Fitzsimmons; 1); University College London Hospital, London, UK (R. Perry; 8); Royal London Hospital, London, UK (K. Saastamoinen; 1); Royal Surrex County Hospital, Surrey, UK (B.C. Creagh-Brown; 1); The Newcastle upon Tyne Hospitals, Newcastle upon Tyne, UK (A. Sweenie; 1); Sheffield Teaching Hospital NHS Foundation Trust, Sheffield, UK (R. Lindert; 1); Hospital Vall d'Hebron, Barcelona, ES (M. Rubiera; 4); Hospital Universitario Dr. Josep Trueta de Girona, Girona, ES (J. Serenal 2); AZ Sint Jan Brugge-Ostende, Brugge, BE (G. Vanhooren; 2); Centre Hospitalier Chrétien, Liège, BE (P. Desfontaines; 1); Centre Hospitalier Régional Universitaire de Lille, Lille, FR (C. Cordonnier; 21); Hopital Pellegrin CHU Bordeaux, Bordeaux, FR (I. Sibon; 1); Vilnius University Hospital Santariskiu klinikos, Vilnius, LT (D. Jatuzis; 2); Azienda Ospedaliera Sant'Andrea, Rome, IT (F. Orzi; 1).
We also thank the Data and Safety Monitoring Committee: Philip Bath (Stroke Trialist, Nottingham; Chair), David Erlinge (Cardiologist, Lund), Gordon Murray (Biostatistician, Edinburgh) and Polly Scutt (Statistician, Nottingham).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
