Abstract
Abstract
Aim
To investigate whether lesions on diffusion-weighted imaging (DWI+) after carotid artery stenting (CAS) or endarterectomy (CEA) might provide a surrogate outcome measure for procedural stroke.
Materials and Methods
Systematic MedLine® database search with selection of all studies published up to the end of 2016 in which DWI scans were obtained before and within seven days after CAS or CEA. The correlation between the underlying log odds of stroke and of DWI+ across all treatment groups (i.e. CAS or CEA groups) from included studies was estimated using a bivariate random effects logistic regression model. Relative risks of DWI+ and stroke in studies comparing CAS vs. CEA were estimated using fixed-effect Mantel-Haenszel models.
Results
We included data of 4871 CAS and 2099 CEA procedures (85 studies). Across all treatment groups (CAS and CEA), the log odds for DWI+ was significantly associated with the log odds for clinically manifest stroke (correlation coefficient 0.61 (95% CI 0.27 to 0.87), p = 0.0012). Across all carotid artery stenting groups, the correlation coefficient was 0.19 (p = 0.074). There were too few CEA groups to reliably estimate a correlation coefficient in this subset alone. In 19 studies comparing CAS vs. CEA, the relative risks (95% confidence intervals) of DWI+ and stroke were 3.83 (3.17–4.63, p < 0.00001) and 2.38 (1.44–3.94, p = 0.0007), respectively.
Discussion
This systematic meta-analysis demonstrates a correlation between the occurrence of silent brain infarcts on diffusion-weighted imaging and the risk of clinically manifest stroke in carotid revascularisation procedures.
Conclusion
Our findings strengthen the evidence base for the use of DWI as a surrogate outcome measure for procedural stroke in carotid revascularisation procedures. Further randomised studies comparing treatment effects on DWI lesions and clinical stroke are needed to fully establish surrogacy.
Introduction
Atherosclerotic stenosis of the internal carotid artery is a major cause of stroke. Early randomised trials demonstrated that carotid endarterectomy (CEA) reduces stroke risk in patients with symptomatic and asymptomatic carotid stenosis.1–4 In recent years, carotid angioplasty and carotid artery stenting (CAS) has emerged as an alternative to CEA, avoiding general surgical complications and morbidity associated with neck incision, as well as shortening hospital stay. In a systematic review of 11 randomised trials including 5778 patients with symptomatic carotid stenosis, CAS was associated with a higher risk of procedural stroke or death than CEA (8.2% vs. 5.0%), although the risk difference between CEA and CAS mainly concerned minor strokes which did not lead to disability or death and was seen in patients older than 70 years only.5,6 Conversely, CAS avoided cranial nerve palsy and was associated with lower risks of access site haematoma and myocardial infarction. Both treatments appear to be equally effective at preventing recurrent stroke after the procedural period.7–10
Further development of surgical and interventional techniques and optimisation of patient selection may lead to a reduction in the risk of procedural stroke associated with carotid revascularisation. However, phase III trials of new carotid interventions designed to show improvements in treatment safety will require many patients and take a long time to complete. For example, a clinical trial would require about 2000 patients to detect a true reduction in the risk of procedural stroke from 6% to 3% with a new intervention compared to an established intervention, at a significance level of 5% and 90% power. The use of a surrogate outcome measure, defined as ‘a variable that provides an indirect measurement of effect in situations where direct measurement of clinical effect is not feasible or practical’, 11 may therefore be justified to investigate new interventions in proof-of-concept or phase II studies and inform the decision whether a large phase III trial should be initiated. 12
A large number of small to medium sized studies have reported acute cerebral ischaemic lesions on diffusion-weighted magnetic resonance imaging (DWI) to occur in a substantial proportion of patients after treatment of carotid stenosis by CAS or CEA (Figure 1). Recent review articles highlighted the potential of DWI as a surrogate safety outcome measure in clinical trials of carotid interventions.13–15 Furthermore, the existing evidence has never been systematically reviewed in the context of validating ischaemia on DWI as a surrogate outcome measure for procedural ischaemic stroke.
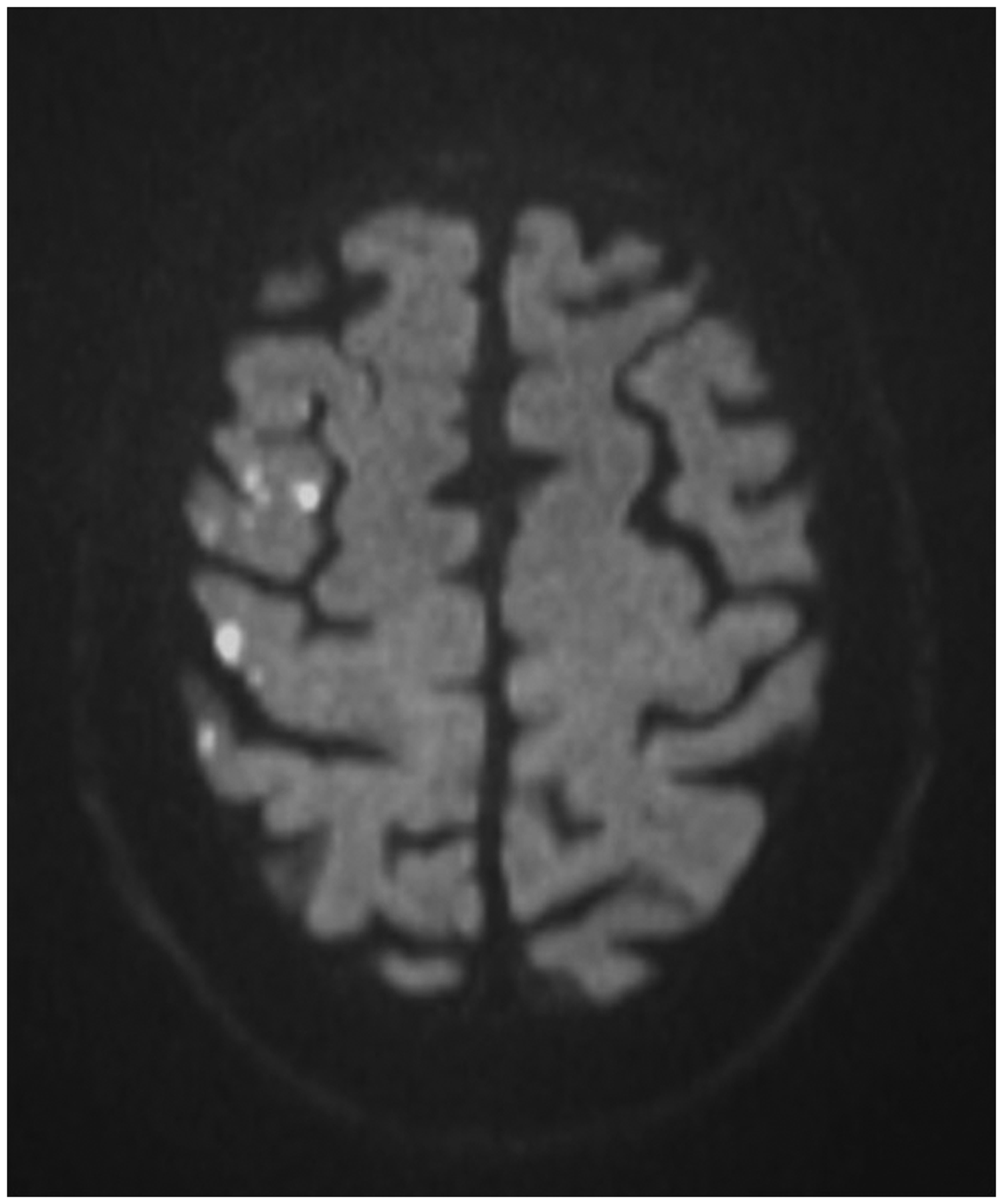
Diffusion-weighted magnetic resonance imaging (DWI) of a patient following stenting of the right carotid artery. Multiple hyperintense signals representing acute ischaemic lesions in the territory of the right middle cerebral artery are present. The patient did not experience any symptoms. Copyright Department of Radiology, University Hospital Basel.
In general, the strength of evidence for a validated surrogate outcome measure depends on the following criteria: Firstly, the biological plausibility of a relationship between the surrogate and the clinical outcome measure; secondly, the demonstration of a correlation between the occurrence of the surrogate and the clinical outcome measure and thirdly, evidence from clinical trials that the effect of treatment on the surrogate outcome measure (e.g. the relative risk of cerebral ischaemia on DWI in CAS vs. CEA) correlates with the treatment effect on the clinical outcome measure (e.g. the relative risk of procedural stroke in CAS vs. CEA). 11 Based on its specificity to image processes in the brain directly linked to cellular hypoxia, and its sensitivity in detecting acute cerebral ischaemia,16–18 the use of DWI as a surrogate outcome measure for procedural ischaemic stroke seems biologically plausible. In order to explore the second and third criterion for surrogacy, we performed a systematic literature review and meta-analysis. We investigated the association between the risk of procedural stroke and the risk of cerebral ischaemia on DWI in patients treated by CAS or CEA, and compared the effect of CAS vs. CEA on the risk of stroke and the risk of cerebral ischaemia on DWI.
Methods
Search strategy and study eligibility criteria
A systematic MedLine® search was conducted to identify all studies investigating patients undergoing treatment of carotid artery stenosis by CAS or CEA with DWI, published online with access to the full article or in-print version up to 31 December 2016. The search term (‘carotid endarterectomy’ OR ‘carotid stenting’ OR ‘carotid angioplasty’) AND (‘DWI’ OR ‘diffusion’ OR ‘MRI’ OR ‘embolism’ OR ‘ischemia’ OR ‘silent’ OR ‘ischemic lesion’ OR ‘emboli’) was entered on http://www.ncbi.nlm.nih.gov/PubMed. We also checked cited studies in retrieved articles and existing reviews on MRI in carotid interventions.13–15,19
Studies were included in the analysis if they fulfilled the following criteria: (1) Patients were treated for atherosclerotic carotid stenosis (any degree of stenosis, symptomatic or asymptomatic stenosis) with either CEA or CAS (with or without use of cerebral protection devices); (2) patients were examined with DWI both before and within seven days after treatment and (3) the numbers or percentages of patients with new lesions on DWI after treatment and procedural ischaemic stroke were reported. If several publications resulted from the same cohort of patients, we chose the report which included the highest number of patients. Studies assessing brain ischaemia on magnetic resonance imaging (MRI) sequences other than DWI were not included. Studies with prospective or retrospective design were included as long as they fulfilled the inclusion criteria above. Reports on single cases were not included. Likewise, studies reporting on emergency procedures (i.e. including intracranial thrombectomy in acute ischemic stroke) were not included. Two researchers (CT and LHB) reviewed the abstracts of all publications identified by the search, retrieved the full publications where abstract data were consistent with the inclusion criteria and selected the studies for inclusion based on the full published data.
Data extraction
Data were extracted independently by three researchers (LHB, CT and STE). Any disagreement was resolved by consensus. For all eligible studies, we extracted: the number (N) of included CAS or CEA procedures; N patients with symptomatic carotid stenosis, usually defined as ischaemic ocular or cerebral symptoms having occurred in the dependent territory of the carotid artery within the past six months; magnetic field strength (tesla) used in MRI; latency between treatment and post-treatment DWI; N patients with a positive DWI (henceforward referred to as ‘DWI+’), defined as the presence of at least one hyperintense lesion on DWI after treatment which was not present on the pre-treatment DWI and N patients with procedural ischaemic cerebral stroke (henceforward referred to as ‘stroke’), defined as a focal neurological deficit of probable ischaemic vascular cause lasting >24 h occurring up to 30 days after treatment, with exclusion of intracranial haemorrhage, hyperperfusion syndrome and other structural brain disease on neuroimaging. Pure retinal infarction was not included. Transient ischaemic attack (TIA) was not consistently recorded in the studies and was therefore not extracted.
For studies involving CAS, we extracted information on whether a cerebral protection device (CPD) was used and which type of device. For studies involving CEA, we noted whether a standard or eversion technique, a shunt, or a patch was used, and if the procedure was done under local or general anaesthesia. For studies including two or more treatment groups (e.g. studies comparing CAS vs. CEA or studies comparing CAS with vs. without CPD use), data were extracted for each treatment group separately.
Corresponding authors were contacted via e-mail if the published data were not sufficient to determine whether the study was eligible for analysis or not, or to extract the relevant information. Additional data obtained in this way (n = 5 studies) were also considered in this review.
Statistical analysis
To explore the second criterion for surrogacy, we included all groups of patients treated with CAS or CEA from eligible studies for which the proportion or number of patients with DWI+ and stroke was reported (henceforth referred to as ‘treatment groups’) as separate data points. First, a crude correlation coefficient for the association between the observed risks of DWI+ and the observed risks of stroke was calculated across treatment groups. This correlation underestimates the extent of the association between the underlying risks because the imprecision in the observed risks dilutes the association as it does whenever an association is measured between two measurement error-prone variables. To account for this, we utilised a bivariate random effects logistic regression model to estimate the correlation between the underlying log-transformed odds (log odds) of stroke and the underlying log odds of DWI+. A detailed description of this model is given in the supplemental methods.
A number of studies compared two or more different revascularisation procedures with each other. Owing to the relatively small size of these studies, the confidence interval (CI) surrounding the treatment effects with regard to procedural stroke and DWI+ was wide. For these reasons, we refrained from further exploring any correlation between treatment effects on stroke and DWI+ to evaluate the third criterion for surrogacy. Instead, we investigated in a meta-analysis of all studies comparing CAS vs. CEA, whether the treatment effect for DWI+ pointed in the same direction as the treatment effect for procedural stroke (i.e. whether the comparison between CAS and CEA using DWI+ as the outcome favoured the same treatment as the comparison using stroke as the outcome). Data were aggregated using fixed-effect Mantel-Haenszel models and treatment effects calculated as risk ratios with 95% CI, using CAS as the reference treatment. We quantified heterogeneity using the I2 statistic. Review Manager Software (RevMan, version 5.3. Copenhagen: The Nordic Cochrane Centre, The Cochrane Collaboration, 2014) was used.
Reporting standards
Data presentation and reporting in this article is done according to the reporting standards of the PRISMA Statement for Reporting Systematic Reviews and Meta-Analyses.20,21
Results
Included and excluded studies
As of 30 March 2017, the search yielded 2555 results (Figure 2). Full publications were retrieved of 164 studies published up to 31 December 2016. Seventy-nine studies were excluded for the following reasons: MRI to detect cerebral ischaemia did not include DWI sequences, or DWI was not done in all patients (n = 15);22–36 no DWI was performed before treatment (n = 11);37–47 post-procedural DWI was performed more than seven days after treatment (n = 3);48–50 the number of DWI+ patients or procedures could not be extracted (n = 6);51–56 or information on procedural stroke was lacking or patients with procedural stroke were excluded from the analysis (n = 6).49,57–68 One study was excluded because the analysis was restricted to patients with available six months’ follow-up who constituted less than half of the study population (n = 1). 69 One study was excluded since only pre-selected groups of patients showing micro-embolic signals in transcranial Doppler during CEA were included in the final analysis. 70 Another study investigating the use of intravascular ultrasound for carotid plaque characterisation was excluded since the additional device by itself may have contributed to the occurrence of DWI+ or stroke. 71 A further 28 studies were excluded because updated reports from the same cohort including a larger number of patients had subsequently been published and included, or because the studies were secondary analyses of previously included studies that contained no additional data relevant for the present review.72–99
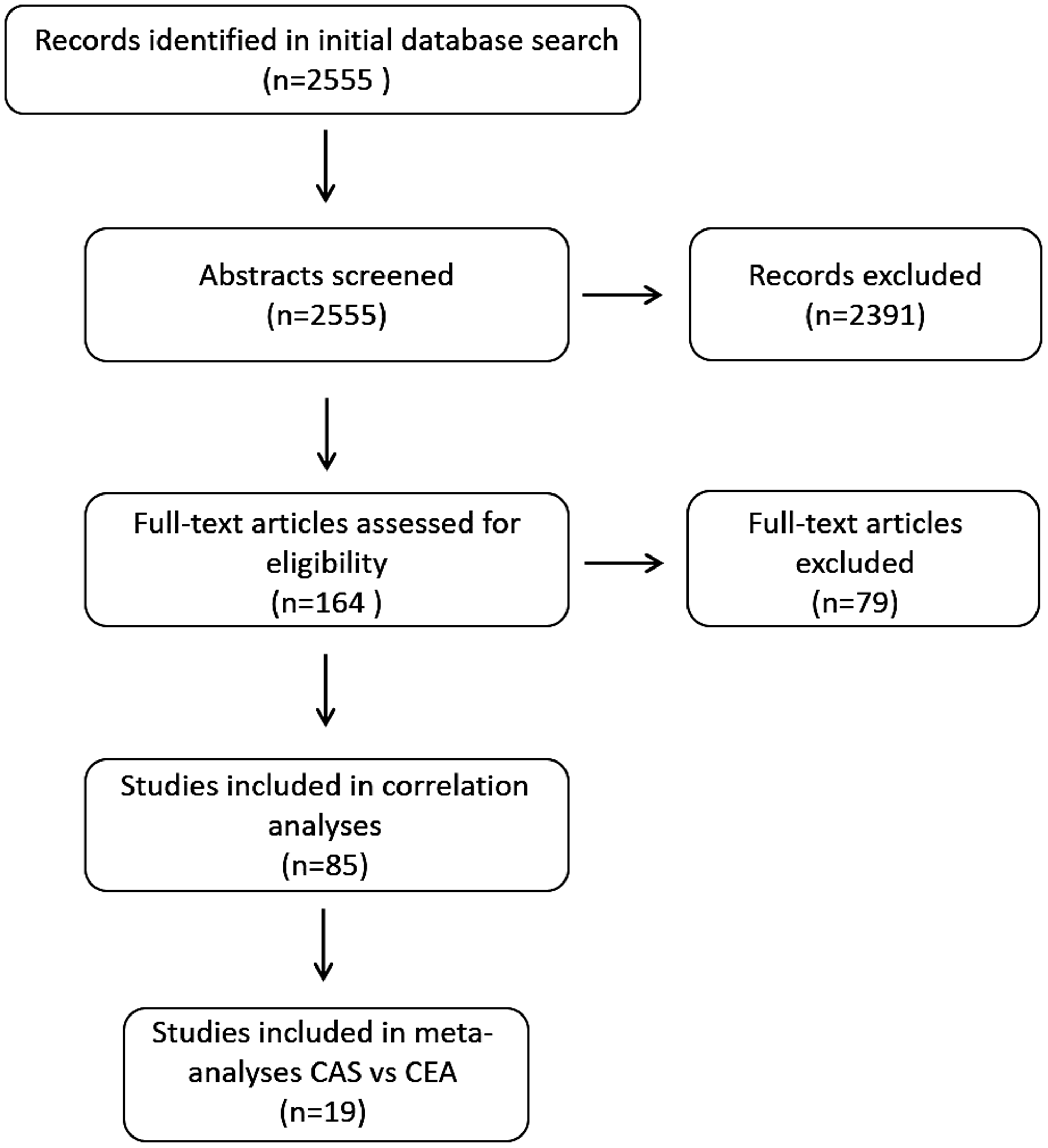
Flow chart of selection of retrieved studies for analysis.
The remaining 85 studies including 6970 carotid revascularisation procedures (CAS: n = 4871; CEA: n = 2099) fulfilled all eligibility criteria and were included in the analysis. Fourteen studies investigated DWI lesions in CEA.100–113 One of these studies retrospectively compared conventional with flow-control-CEA (i.e. clamping of the common carotid artery, the external carotid artery and the superior thyroid artery before dissection of the carotid sheath around the internal carotid artery). 108 Six studies investigated DWI lesions in CAS without CPD,114–119 one of which using a membrane-covered stent. 119 Thirty-eight studies investigated DWI lesions in CAS with CPD:120–157 one study randomly compared membrane-covered vs. bare-metal stents; 129 one study randomly allocated patients to CAS using flow reversal or distal filter protection; 150 one study randomly assigned to either proximal or distal cerebral protection during CAS; 145 one study compared open vs. closed-cell stent type in a randomised design; 147 three studies compared CAS with CPD vs. CAS without CPD158–160 and three compared CAS with filter CPD vs. CAS with balloon CPD.161–163 Three randomised studies, one of which had a 2 × 2 factorial design; 148 investigated clinical and imaging outcomes of CAS according to different regimens of peri-interventional platelet-inhibition148,164 and statin treatment144,148 a further study investigated DWI lesions in CAS using different catheter techniques (use of 7F or 8F catheter or use of a coaxial system with use of a 7F or 8F catheter in conjunction with a 4F or 5F catheter), both with and without using CPD. 165 Nineteen studies compared DWI lesions and clinical outcomes in CEA vs. CAS,166–184 only two of which were randomised.176,184 Details of included studies are provided in supplementary Table 1.
Incidence of positive DWI and procedural ischaemic stroke in CAS and CEA
Data from 85 separate CAS treatment groups reporting on outcomes of 4871 CAS procedures were available from eligible single treatment group and multiple treatment group (comparative) studies combined. Carotid symptom status was available in 3829 CAS procedures, and 2023 of those (53%) were done in symptomatic carotid stenosis. New ischaemic lesions on DWI were found after 1805 procedures (average risk 37%), while procedural ischaemic strokes were reported to have occurred in 131 procedures (2.6%; Table 1).
Average risks of cerebral ischaemia on diffusion-weighted magnetic resonance imaging and procedural ischaemic stroke in carotid artery stenting and carotid endarterectomy.
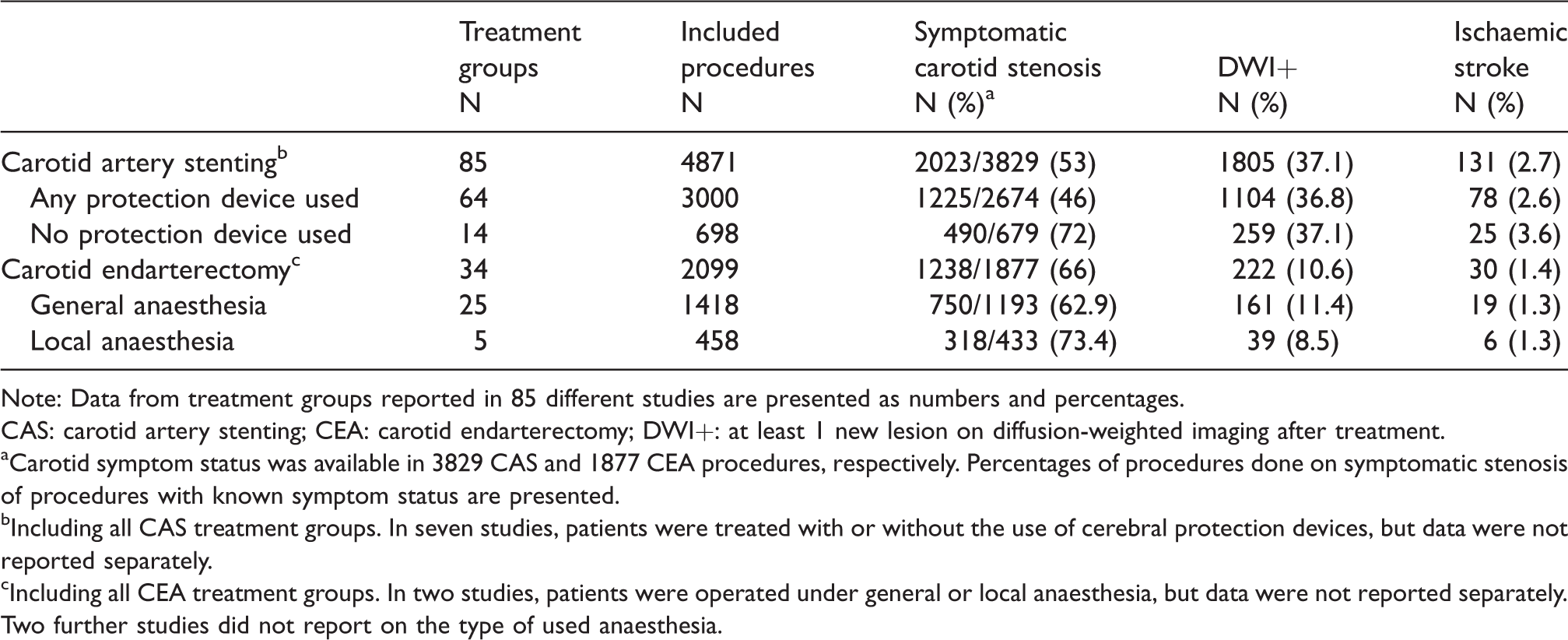
Note: Data from treatment groups reported in 85 different studies are presented as numbers and percentages.
CAS: carotid artery stenting; CEA: carotid endarterectomy; DWI+: at least 1 new lesion on diffusion-weighted imaging after treatment.
aCarotid symptom status was available in 3829 CAS and 1877 CEA procedures, respectively. Percentages of procedures done on symptomatic stenosis of procedures with known symptom status are presented.
bIncluding all CAS treatment groups. In seven studies, patients were treated with or without the use of cerebral protection devices, but data were not reported separately.
cIncluding all CEA treatment groups. In two studies, patients were operated under general or local anaesthesia, but data were not reported separately. Two further studies did not report on the type of used anaesthesia.
Eligible studies provided data for 34 separate CEA treatment groups including 2099 CEA procedures. For 1877 of these CEA procedures, carotid symptom status was available, and 1238 of them (66%) were done in patients with symptomatic carotid stenosis. New DWI lesions were detected after 222 procedures (10.8%) and 30 procedures (1.4%) were complicated by procedural ischaemic strokes (Table 1).
Correlation between risk of cerebral ischaemia on DWI and risk of procedural ischaemic stroke
The crude risks of DWI+ and stroke observed in all 119 treatment groups (CEA and CAS combined) are displayed in Figure 3(a). The crude correlation between these risks was 0.29 (p = 0.0014). Figure 4 illustrates why this crude correlation underestimates the magnitude of the association between the underlying risks. The correlation between the underlying log odds of DWI+ and the log odds of stroke corrected for this underestimation using the bivariate random effects logistic regression model was 0.61 (95% CI 0.27 to 0.87; p = 0.0012). The slope of this relationship was 1.20 (95% CI 0.50 to 2.39), this being an estimate of the change in log odds of DWI+ per 1 unit change in the log odds of stroke. Figure 3(b) converts this estimated linear relationship between the two log odds ratios into the non-linear relationship between the estimated risks. Within the range of procedural stroke risk of between 2% and 7% reported in previous clinical trials of CEA or CAS for symptomatic and asymptomatic carotid stenosis, new ischaemic lesions on post-procedural scans can be expected to occur in about 10 times this proportion. Figure 3(b) shows that for a particular drug or interventional technique in a future randomised controlled trial postulated to reduce procedural stroke risk from 6% to 3%, the predicted reduction in DWI+ risk would be from 56% to 35%. For a standard two-arm clinical trial, with 90% statistical power to detect a difference that is statistically significant at the 5% level using a two-sided test, this equates to a reduction in sample size from 2004 to 228, almost a 90% reduction.
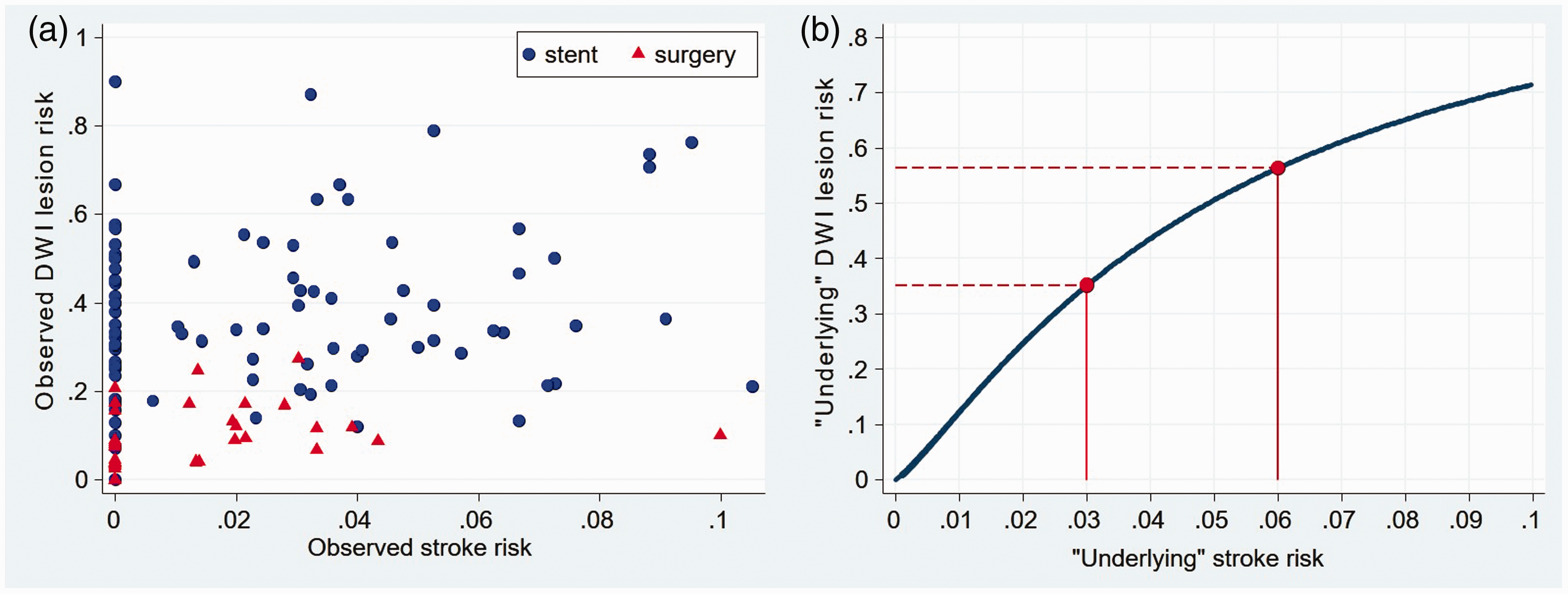
Crude risks of cerebral ischaemia on DWI and procedural ischaemic stroke and fitted association between the underlying risks of these outcomes in 119 groups of patients undergoing carotid revascularisation. (a) Observed crude risks of stroke and ischaemia on diffusion-weighted imaging (DWI) in all 119 treatment groups. Red triangles represent groups of patients treated with carotid endarterectomy and blue dots groups of patients treated with stents. (b) Fitted regression line relating ‘underlying’ (true) risk of DWI lesions to ‘underlying’ (true) risk of stroke in all included studies. Correlation between log odds of DWI lesions and log odds of stroke (coefficient 0.61 (95% CI 0.27, 0.87), p = 0.0012). Red lines show the magnitude of reduction in risk of DWI lesions that might be observed in a pilot trial expected to reduce the risk of procedural stroke from 6% to 3%.
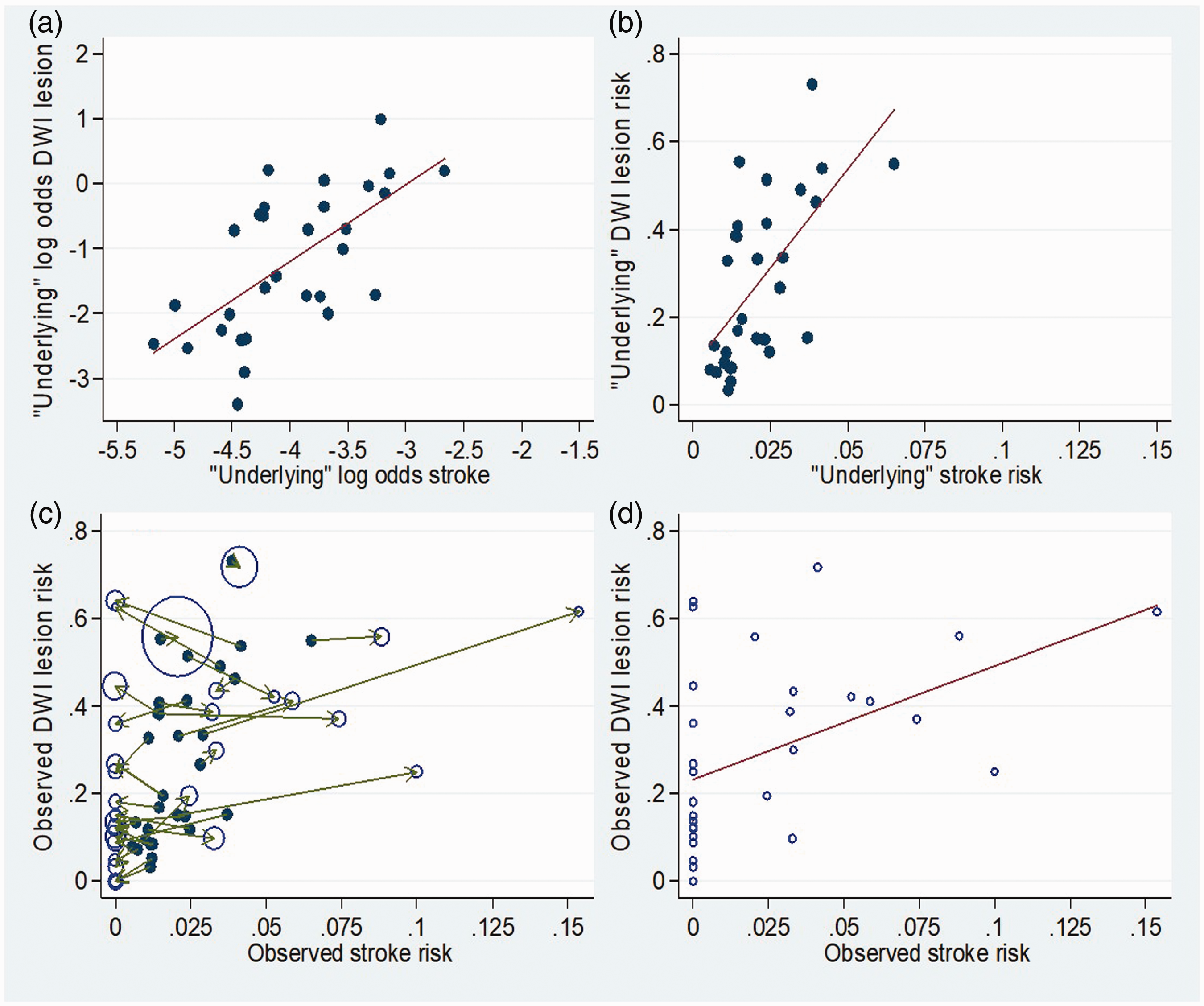
Illustrative example of statistical method using simulated data. (a) Simulated ‘underlying’ (true) log odds of DWI+ (i.e. presence of at least one new DWI brain lesion after treatment) and log odds of procedural ischaemic stroke for 30 studies, with regression line. Simulated data were drawn from a bivariate normal distribution with means, SDs and correlation similar to those estimated using the actual data by the bivariate random effects logistic regression model. This plot therefore represents the ‘corrected’ association between log odds of DWI+ and log odds stroke. (b) Conversion of simulated ‘underlying’ log odds of DWI+ and stroke to ‘underlying’ risks of DWI+ and stroke for all 30 studies, with fitted regression line (risk=exp(log odds)/(exp(log odds)+1). (c) Simulation of observed risks (hollow circles) by addition of sampling error (random error; green arrows) to both the ‘underlying’ (true) risks of DWI+ and stroke (solid circles, as in (b)) through sampling subjects in each of the 30 studies (which have sizes typical of the studies included in our analysis). First observed stroke risk was simulated, and then it was assumed that everyone who has a stroke is DWI+, and finally the number of DWI+ cases amongst those without stroke was simulated. Size of hollow circles for observed risks is relative to the number of patients in that study. Smaller studies tend to be subject to greater sampling error (as indicated by typically longer arrows). (d) Simulated observed risks of DWI+ and stroke for 30 studies, with fitted regression line. This plot represents the crude association between study-specific DWI+ and stroke risks and demonstrates its underestimation compared with the association of ‘underlying’ (true) DWI+ and stroke risks as in Figure b (as both factors are subject to sampling error).
Including only CAS treatment groups, the crude correlation between DWI+ and stroke was 0.19 and the corrected correlation was 0.27 (95% CI –0.19, 0.66, p = 0.24). There were too few studies done on CEA and too little variance in stroke risk between studies to be able to accurately estimate the between study variance in stroke risk (with this actually being estimated as zero). Therefore, the corrected correlation between DWI+ and stroke could not be calculated for CEA.
Comparison of treatment effects on cerebral ischaemia on DWI and procedural ischaemic stroke
Nineteen eligible studies compared DWI findings and clinical outcomes between CAS and CEA. Across these studies, the risk of being DWI+ after CAS was 3.83 times that after CEA (95% 3.17 to 4.63; p < 0.00001; Figure 5), while the risk of ischaemic procedural stroke after CAS was 2.38 times (95%CI 1.44 to 3.94; p = 0.0007; Figure 6) that after CEA.
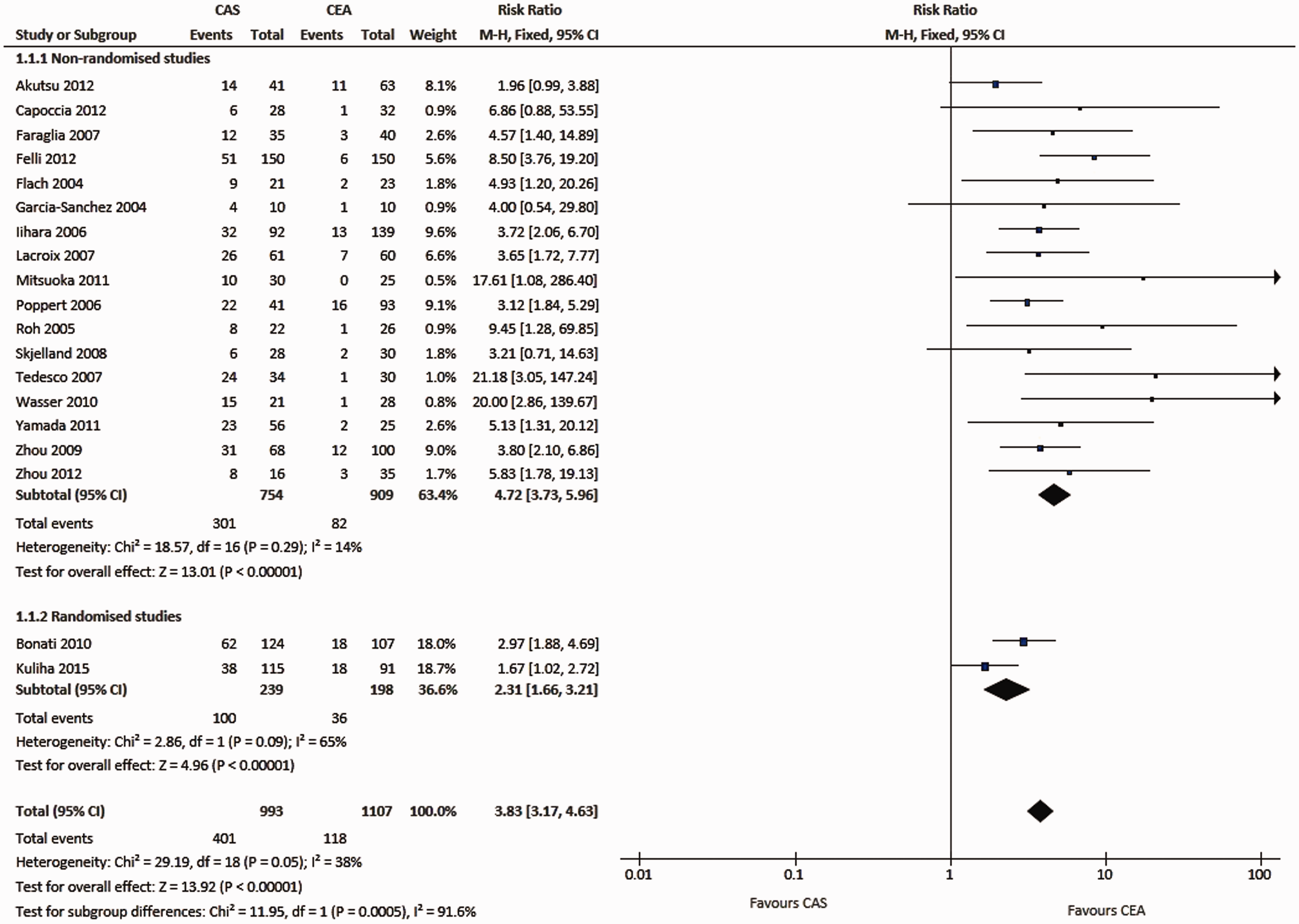
Meta-analysis of studies comparing the risk of new ischaemic brain lesions on diffusion-weighted imaging after carotid artery stenting and carotid endarterectomy. Data are numbers of patients with DWI lesions (‘events’), total numbers of patients and Mantel-Haenszel fixed-effects risk ratios including 95% confidence intervals (CI), with endarterectomy as the reference treatment. Squares on the right represent point estimates of risk ratios at trial level, with 95% CI as horizontal bars. The diamond at the bottom represents the summary risk ratio and 95% CI.
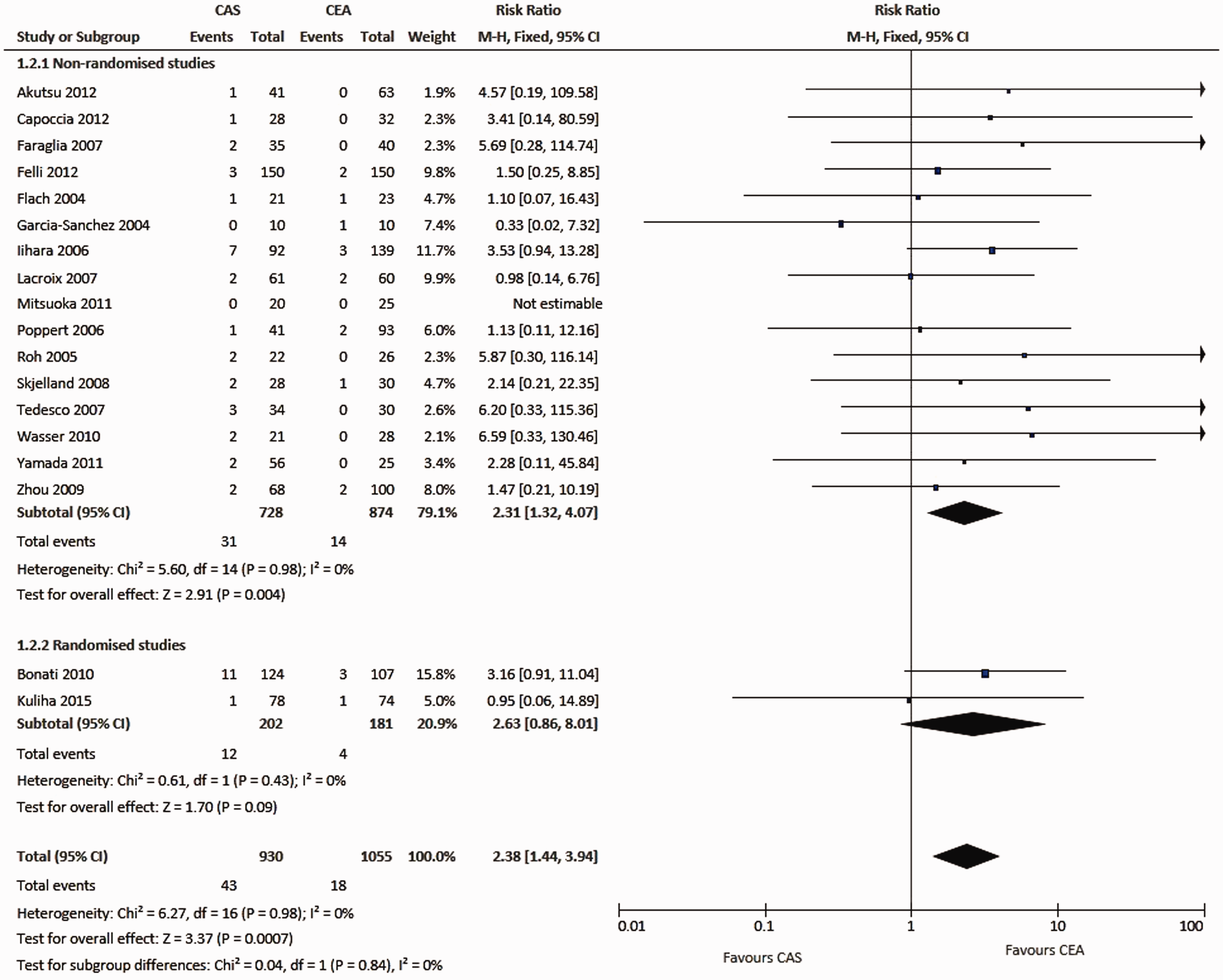
Meta-analysis of studies comparing the risk of procedural ischaemic stroke between carotid artery stenting and carotid endarterectomy. The same studies as in Figure 5 are included. Data are numbers of patients with strokes (‘events’), total numbers of patients and Mantel-Haenszel fixed-effects risk ratios including 95% confidence intervals (CI), with endarterectomy as the reference treatment. Squares on the right represent point estimates of risk ratios at trial level, with 95% CI as horizontal bars. The diamond at the bottom represents the summary risk ratio and 95% CI.
Discussion
In our systematic review of the literature, new ischaemic lesions on DWI were present on average in 37% of patients following CAS compared to 10.8% of patients after CEA; in contrast, the reported risks of procedural ischaemic stroke were only 2.6% and 1.4%, respectively. Across all groups of patients treated with CAS or CEA, the risk of a positive DWI scan after treatment significantly correlated with the risk of procedural stroke. Among those studies comparing CAS vs. CEA, summary treatment effects on the occurrence of DWI lesions pointed in the same direction as summary treatment effects on ischaemic stroke, i.e. the risks of both clinical and radiological cerebral ischaemia were increased in CAS compared to CEA. Does the current evidence therefore support the use of DWI as a surrogate outcome measure for procedural safety in trials of carotid interventions?
The statistical methodology involved in validating surrogate markers is complex and controversial. 185 A set of formal statistical rules for validating surrogate outcome measures has been proposed to reduce observation time in prospective trials of progressive diseases. 186 These rules state among other criteria that the surrogate must predict clinically manifest disease in the future and that the full effect of treatment on the clinical outcome must be explained by the effect of treatment on the surrogate outcome. It is evident that we cannot use these formal criteria to validate DWI lesions as a surrogate outcome measure of procedural stroke in carotid interventions: both the potential surrogate – DWI lesions – and the clinical endpoint – procedural stroke – are short-term outcome measures characterising risk of procedure; the major advantage of DWI as a surrogate outcome is that ischaemic brain lesions are much more common than clinically manifest stroke, allowing reductions in the sample size of pilot trials investigating novel approaches in reducing treatment risks (e.g. surgical technique, stent design, CPDs and peri-procedural medication). Hence, we evaluated the potential surrogacy of DWI against a set of more general rules defined in the ICH Harmonised Tripartite Guideline: Statistical Principals for Clinical Trials. 11
Is cerebral ischaemia on DWI a biologically plausible surrogate outcome measure for procedural ischaemic stroke?
Stroke is the most common serious adverse event occurring in carotid revascularisation procedures. In a meta-analysis of randomised trials comparing CAS vs. CEA for symptomatic carotid stenosis, 94% of all strokes occurring within 30 days of CAS and 81% of all strokes occurring within 30 days of CEA were attributable to ischaemic cerebral infarction and the remaining events to intracerebral haemorrhage. 6
Acute cerebral ischaemia leads to a cascade of events on the biological level, including break-down of electrolyte transport across the cellular membrane with subsequent shift of water from the extracellular to the intracellular space (cytotoxic oedema). The reduction in extracellular water causes a decrease in random translational motion of water molecules. Diffusion-weighted images are generated by measuring this random motion of water molecules by the effect of magnetic gradients on protons. Areas of reduced water diffusion are shown hyperintense in relation to surrounding normal brain tissue. DWI detects brain ischaemia in more than 90% of patients with the final clinical diagnosis of acute ischaemic stroke, and sensitivity and inter-rater reliability are superior to computer tomography or conventional (T2-weighted or FLAIR) MRI.18,187,188 However, DWI may also show ischaemic lesions in the absence of a stroke: hyperintense brain lesions on DWI may be found in about a third patients with TIAs.189,190 Furthermore, brain ischaemia on DWI without associated focal neurological deficit has been shown in patients with carotid stenosis 191 and following therapeutic or investigational procedures on the heart or the carotid artery. 13 DWI therefore identifies a spectrum of acute cerebral ischaemia encompassing asymptomatic lesions, TIA and stroke. Based on its specificity to image processes in the brain directly linked to cellular hypoxia, and its sensitivity in detecting acute cerebral ischaemia, the use of DWI as a surrogate outcome measure for procedural ischaemic stroke seems biologically plausible.
Does ischaemia on DWI correlate with procedural stroke during carotid interventions?
The result of our analysis including all populations treated with CAS or CEA demonstrates a significant correlation between the true risk of stroke and the true risk of cerebral ischaemia (p = 0.0012). A statistically significant association of the potential surrogate and clinical outcome constitutes the basis for a use of the surrogate as an endpoint in clinical trials. Our statistical methodology does not allow us to fully establish surrogacy; however, it does go part of the way towards this since it allows the magnitude of the association between the risk of a stroke and the risk of DWI lesions to be estimated, taking account of the fact that imprecision in the estimates of these two risks will tend to dilute the magnitude of the underlying association. The fact that the observed correlation between the underlying log odds ratio is strong (albeit with a wide 95% CI) is encouraging. The steepness of the association, coupled with the fact that the prevalence of DWI lesions is much greater than that of stroke illustrates that substantial sample size reductions for clinical trials may be possible by switching from a clinically based stroke outcome to MRI-based DWI lesions. Returning to the example given in the Introduction section, instead of 2000 patients (1000 in each treatment group) needed to detect an underlying 3% absolute difference in procedural stroke risk, just over 200 patients would be needed to detect the corresponding difference in DWI lesion risk. The proof of a significant correlation of a potential surrogate outcome measure and the true clinical outcome is crucial but not sufficient by itself to fully validate surrogacy.
Does the effect of treatment on ischaemic lesions on DWI correspond to the effect of treatment on procedural stroke?
Despite the corresponding summary treatment effects on DWI lesions and procedural stroke in studies comparing CAS vs. CEA, there was no clear relationship between the relative risks of DWI positivity and stroke across these studies. The observed number of strokes was very small in most of these comparative studies and estimated relative risks were surrounded by high CIs. Therefore, despite a strong theoretical background supporting the use of DWI as a surrogate outcome measure for procedural stroke in carotid interventions, and evidence for a correlation of DWI lesions with procedural stroke, the existing evidence is insufficient to assess the relationship between the effect of treatment on DWI lesions and on procedural stroke. More data from randomised clinical trials of carotid revascularisation are required to test this criterion. Apart from comparisons of CAS vs. CEA, such randomised trials may also compare various surgical or interventional techniques, such as cerebral protection devices, access routes, or medication. They need to incorporate serial MR imaging including DWI and be of adequate size to detect a sufficient number of clinically manifest strokes. In our experience, a time window of one to seven days before and one to three days after the carotid revascularisation procedure for the pre- and post-procedural scan is suitable to detect new ischaemic brain lesions caused by the procedure while at the same time allowing for enough flexibility in scheduling the scans.
Summarising data from several studies does imply limitations, most importantly the heterogeneity of the included studies. Firstly, the included studies commonly included both patients with symptomatic and asymptomatic carotid stenosis with the proportion of patients having a symptomatic stenosis ranging between 0% and 100% within subgroups (symptomatic stenosis across studies, 53% of all CAS procedures, and 66% of all CEA procedures). Recently, symptomatic carotid stenosis may be more prone to embolisation and subsequent DWI+ during CAS or CEA due to instability of the atherosclerotic plaque. Secondly, the included studies did use different time windows for pre- and post-treatment MRI scanning, which may have influenced the rate of post-procedural ischaemic brain lesions detected on DWI. Thirdly, definitions of what constituted a new ischaemic lesion after treatment differed: some studies purely relied on new hyperintense lesions on post-treatment DWI which had not been present before treatment. In other studies, a corresponding hypointense signal on ADC maps was additionally required. However, very small acute ischaemic lesions appearing hyperintense on DWI may not be identified on ADC maps due to limited spatial resolution. Finally, less than half of the included 85 studies clearly stated that either clinical assessment for procedural complications was done by an independent neurologist or outcome event adjudication was performed; lower reported incidence of procedural stroke in the absence of clinical evaluation by a neurologist is a well-known phenomenon. However, the average risk of procedural stroke in studies with reported event confirmation by a neurologist was only slightly higher than in studies without (2.6% vs. 2.1%).
The importance of DWI lesions occurring during treatment of carotid stenosis may go beyond being a surrogate of procedural stroke. In a prospective, population-based study investigating the association of silent brain infarcts and neurocognitive decline, silent brain infarcts were associated with a higher risk of dementia and a steeper decline in cognitive function. 192 Silent ischaemic brain lesions following coronary artery bypass grafting 193 and intra-cardiac surgery 194 have also been associated with cognitive decline. Only a few small studies performed both cognitive testing and DWI before and after carotid revascularisation.38,86,105,195 These studies identified few patients with post-treatment ischaemia on DWI and were unable to demonstrate or refute a relationship between subclinical ischaemia and cognitive decline. An MRI-based sub-study within the International Carotid Stenting Study investigated multiple aspects of both the occurrence of new DWI lesions and ischaemic events in the procedural period after randomised assignment to CAS or CEA for symptomatic carotid stenosis. 6 After CAS treatment, there was a significantly higher rate of recurrent stroke or TIA during follow-up among patients with new ischaemic brain lesions on DWI after treatment than among those without new DWI lesions. 98 These findings may suggest that new DWI lesions after CAS could further serve as a marker to identify patients at higher risk for future events.
Conclusion
Our findings prove a strong correlation between DWI+ and procedural stroke as clinical outcome measure in carotid interventions. The results of our analyses therefore strengthen the evidence base for the use of DWI+ as a surrogate outcome measure for procedural risk in carotid revascularisation. However, despite including a large number of studies with over 6000 carotid interventions, criteria for validation of a surrogate marker are still only partly satisfied. Further, randomised studies comparing treatment effects on DWI lesions and clinical stroke are needed to fully establish surrogacy.
Supplemental Material
Supplemental material for Silent brain infarcts on diffusion-weighted imaging after carotid revascularisation: A surrogate outcome measure for procedural stroke? A systematic review and meta-analysis
Supplemental Material for Silent brain infarcts on diffusion-weighted imaging after carotid revascularisation: A surrogate outcome measure for procedural stroke? A systematic review and meta-analysis by Christopher Traenka, Stefan T Engelter, Martin M Brown, Joanna Dobson, Chris Frost and Leo H Bonati in European Stroke Journal
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: CT has received funding for travel from Bayer. LHB has received funding for travel and served on scientific advisory boards for Bayer. STE has received funding for travel or speaker honoraria from Bayer and Boehringer Ingelheim. He has served on scientific advisory boards for Bayer, Boehringer Ingelheim, Pfizer/BMS and MindMaze and on the editorial board of Stroke. He has received an educational grant from Pfizer and compensation for educational efforts by Stago. The other authors have no conflicts of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: CT was supported by the University of Basel and the Swiss National Science Foundation. LHB was supported by grants from the Swiss National Science Foundation (PBBSB-116873) and the University of Basel. STE has received an educational grant from Pfizer and compensation for educational efforts by Stago and research support from the Science Funds [Wissenschaftsfonds] of the University Hospital Basel, the University Basel, the Swiss Heart Foundation, and the Swiss National Science Foundation; MMB’s Chair in Stroke Medicine is supported by The Reta Lila Weston Trust for Medical Research.
This work was partly done at University College London Hospital and University College London, who received a proportion of funding from the Department of Health’s National Institute for Health Research Biomedical Research Centres funding scheme.
Ethical approval
Not applicable.
Informed consent
Not applicable.
Guarantor
CT and LHB.
Contributorship
LHB and CT performed the systematic review of the literature. CT designed/conceptualised the study, analysed/interpreted the data, collected data and drafted the article. JD, CF and CT performed statistical analyses. LHB initiated, designed, conceptualised and supervised the study, analysed/interpreted the data, revised the article, and collected data. STE extracted the data. All authors: data collection, critical review of the article, editing article for content. All authors agreed on submission of the present version of the article and reviewed the article and revised it critically for important intellectual content.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
