Abstract
Introduction
Detailed data on the long-term consequences and treatment of stroke are scarce. We aimed to assess the needs and disease burden of community-dwelling stroke patients and their carers and to compare their treatment to evidence-based guidelines by a stroke neurologist.
Methods
We invited long-term stroke patients from two previous acute clinical studies (n = 516) in Berlin, Germany to participate in an observational, cross-sectional study. Participants underwent a comprehensive interview and examination using the Post-Stroke Checklist and validated standard measures of: self-reported needs, quality of life, overall outcome, spasticity, pain, aphasia, cognition, depression, secondary prevention, social needs and caregiver burden.
Results
Fifty-seven participants (median initial National Institutes of Health Stroke Scale score 10 interquartile range 4–12.75) consented to assessment (median 41 months (interquartile range 36–50) after stroke. Modified Rankin Scale was 2 (median; interquartile range 1–3), EuroQoL index value was 0.81 (median; interquartile range 0.70–1.00). The frequencies for disabilities in the major domains were: spasticity 35%; cognition 61%; depression 20%; medication non-compliance 14%. Spasticity (p = 0.008) and social needs (p < 0.001) had the strongest impact on quality of life. The corresponding items in the Post-Stroke Checklist were predictive for low mood (p < 0.001), impaired cognition (p = 0.015), social needs (p = 0.005) and caregiver burden (p = 0.031). In the comprehensive interview, we identified the following needs: medical review (30%), optimization of pharmacotherapy (18%), outpatient therapy (47%) and social work input (33%).
Conclusion
These results suggest significant unmet needs and gaps in health and social care in long-term stroke patients. Further research to develop a comprehensive model for managing stroke aftercare is warranted.
Introduction
Stroke survivors are often affected by long-term disabilities and various complications. 1 Information on these problems is required to optimise management and outcomes, reduce recurrence and to plan and provide stroke services in the community. However, reports on consequences of stroke in the long term (after acute treatment and initial rehabilitation) for stroke survivors living in the community or in nursing homes are limited; and often focus either on patient-reported outcome measures (PROM) or single domains of disability.2–15
Given the increasing prevalence of stroke in an aging population, any interventions to prevent or minimize stroke disability and related complications in long-term care are worthy of further evaluation. Models have been established in older people’s care, 16 which coordinate care, offer patients easily identifiable access points and manage patients using standardised assessments of deficits and individualised treatment plans. A similar model for long-term care after stroke (‘stroke aftercare’) has been suggested.17–22 Several approaches to provide comprehensive assistance to patients after stroke23,24 in order to navigate care once they are discharged from rehabilitation have been described. However, apart from ‘early supported discharge’ which is focussed on reducing length of hospital stay and promoting early return to the community rather than on-going care25–29 no other longer term model is widely recommended.
Many post-stroke complications are preventable or can be ameliorated with treatment. 1 While it has been argued that stroke recovery reaches a plateau after a certain time, 30 there are data to support long-term and late therapy to increase the functional status of patients.31–33 It is therefore reasonable to assume that coordinated long-term management of stroke could benefit some stroke patients.18,22
Our main goals in the ‘Managing Aftercare for Stroke (MAS-I)’ study were to assess the needs, disease burden and current treatments in long-term stroke patients and their caregivers in the urban area of Berlin, Germany. We therefore aimed to assess the feasibility of conducting a comprehensive assessment to characterise stroke outcome, identify long-term stroke needs and to investigate whether stroke patients’ treatment in the chronic phase was in line with clinical guidelines. We also wanted to explore the use of the Post-Stroke Checklist (PSC) as a screening tool for stroke-related health problems in total, and by correlating its individual items to validated tools. Finally, we explored the correlation of individual validated tools with the EuroQoL to establish the impact of individual deficits on quality of life.
Methods
We conducted a cross-sectional observational study of post rehabilitation patient needs and caregiver burden after stroke in a sample of patients in Berlin Germany.
Sampling
To avoid recall bias relating to the initial stroke event, we used purposive sampling of previous participants of two completed hospital-based acute stroke studies (a randomised controlled trial STRAWINSKI NCT01264549 34 ; an observational study PREDICT NCT01079728 35 ) with identical acute event case report forms (CRFs). Both studies recruited patients with acute ischemic stroke (for STRAWINSKI only in the middle cerebral artery (MCA) territory). We invited all previous participants of STRAWINSKI and PREDICT living in the Berlin metropolitan area (n = 516 subjects) to take part and visit our outpatient clinic for a follow-up interview and assessment. No financial incentive was provided, but travel costs were reimbursed.
Outcome measures
After reviewing published literature and consulting experts from the Berlin Stroke Alliance (BSA) and Center for Stroke Research Berlin (CSB), a selection of domains relevant to patient care and needs in the long-term after stroke, was identified. After consultation with a stroke patients’ representative group, these expert-defined domains were supplemented with measures of carer burden. We then assembled validated and clinically useful assessments considered as standards in each of the 11 domains identified and added PROM scales (see Table 1). These included the health-related quality of life scale EuroQoL and the PSC, an easy-to-use PROM of (self-reported) need that screens different domains of stroke-related health problems in the post-rehabilitation phase of care. 21
Domains explored, according assessments and cut-off measures used to define significant deficit.

Assessment in outpatient clinic
We incorporated these scales into an assessment lasting approximately 4 h conducted by a multi-disciplinary team: 30 min stroke neurologist (specifically trained to evaluate spasticity using the modified Ashworth scale in the REPAS (REsistance to PASsive movement system36,37); 30 min nurse; 1 h social worker and 2 h by researchers trained to use standard assessment instruments for aphasia, cognitive deficits and depression.
Comparing treatment plans to guidelines
After the assessment, a stroke neurologist provided participants with an in-depth analysis of their current care and with individual recommendations according to national and international guidelines in treatable domains. 29 The options for recommendations were: none, medical review (need for further diagnostic workup), change in medication, further rehabilitation in a comprehensive rehabilitation centre, outpatient therapy (e.g. physiotherapy, speech therapy) and social worker intervention. We used these recommendations as a surrogate measure of current gaps in the ongoing care of these patients. This treatment plan review was also sent to the patients as a written medical report. While it would have been unethical to document under-provision of healthcare without informing the patients about it, we made sure to complete the study assessment before relaying the feedback to them to avoid any influence on the values measured.
Statistical analyses
Standard descriptive statistics, such as mean, standard deviation, median and interquartile range, were calculated using SPSS 24.0 software (IBM, Armonk, NY) and used to present data. Associations between PROM (PSC and EuroQoL) and deficits (clinical scales) were calculated using Fisher’s exact test, Pearson’s χ2, Mann–Whitney U or independent samples t-test depending on variables included as specified in detail in the result tables. An alpha error ≤ 0.05 was considered statistically significant.
Governance
The study received full ethics committee and data protection approval by the institutional review boards of Charité – Universitaetsmedizin Berlin (reference EA1/183/14), and was registered on clinicaltrials.gov (NCT02320994).
Results
Of 516 subjects from the previous acute stroke studies (above) originally living in Berlin, we received consent from 61 patients, 57 of whom completed their visit to the MAS-I study clinic (Figure 1). We compared the included patients with the pooled cohort of STRAWINSKI and PREDICT to identify any major inclusion bias. Patients included in MAS-I were initially more severely affected by their stroke (admission NIHSS, median (interquartile range (IQR)): 10 (4–12.75) vs. 7 (3–14), p = 0.090) but showed a better early recovery (Modified Rankin Scale (mRS) at three months, median (IQR): 2 (1–4) vs. 3 (1–4), p = 0.016) and were significantly younger (years, mean ± SD: 69.3 ± 9.8 vs. 71.8 ± 12.1, p < 0.001) than patients not included in MAS-I.
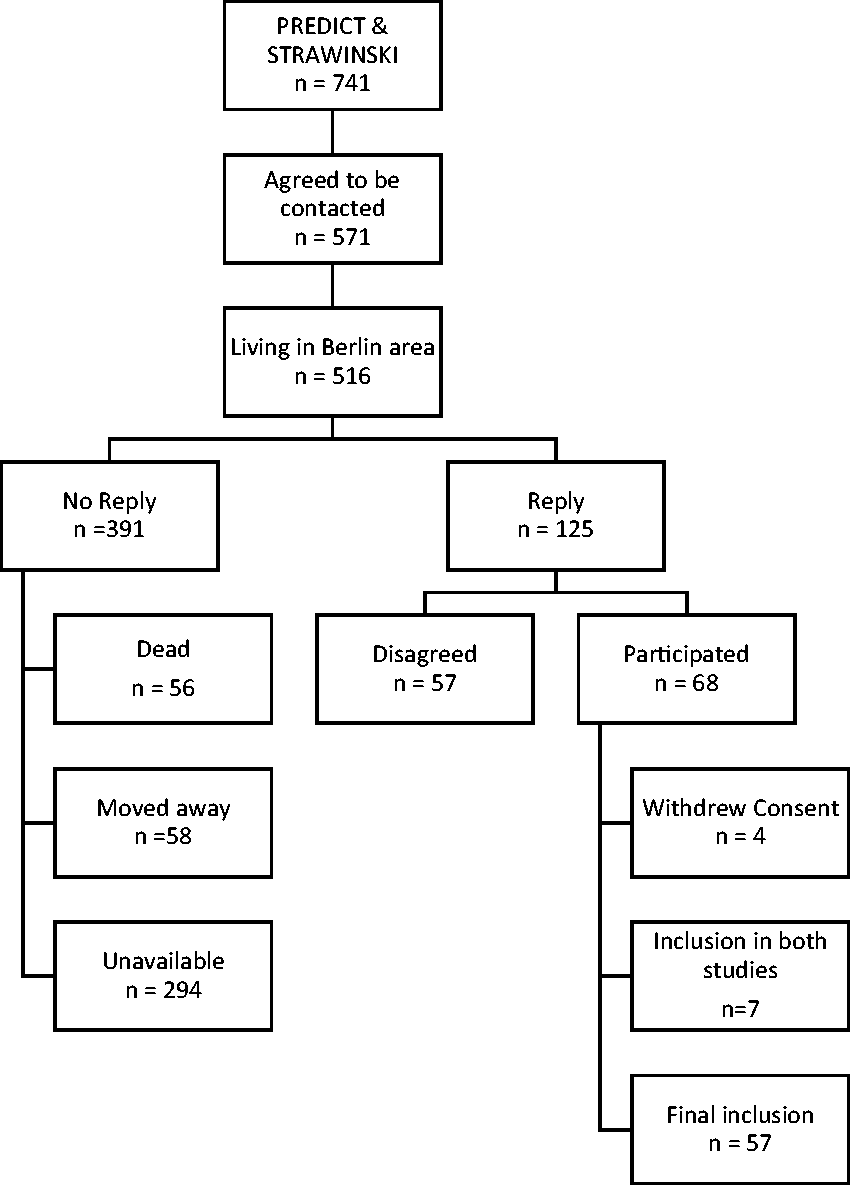
Study flow chart.
Table 2 shows baseline stroke characteristics, comorbidities, complications, secondary prevention and previous treatments. Patients were investigated a median of 41 months (IQR 32–48) after stroke onset. At time of assessment the median mRS was 2 (IQR 1–3), and the EuroQoL index value and visual analogue scale were 0.813 (median, IQR 0.70–1.00) and 70 (median, IQR 50–85), respectively.
Baseline characteristics, comorbidities, complications, secondary prevention and previous treatments in 57 individuals with stroke.
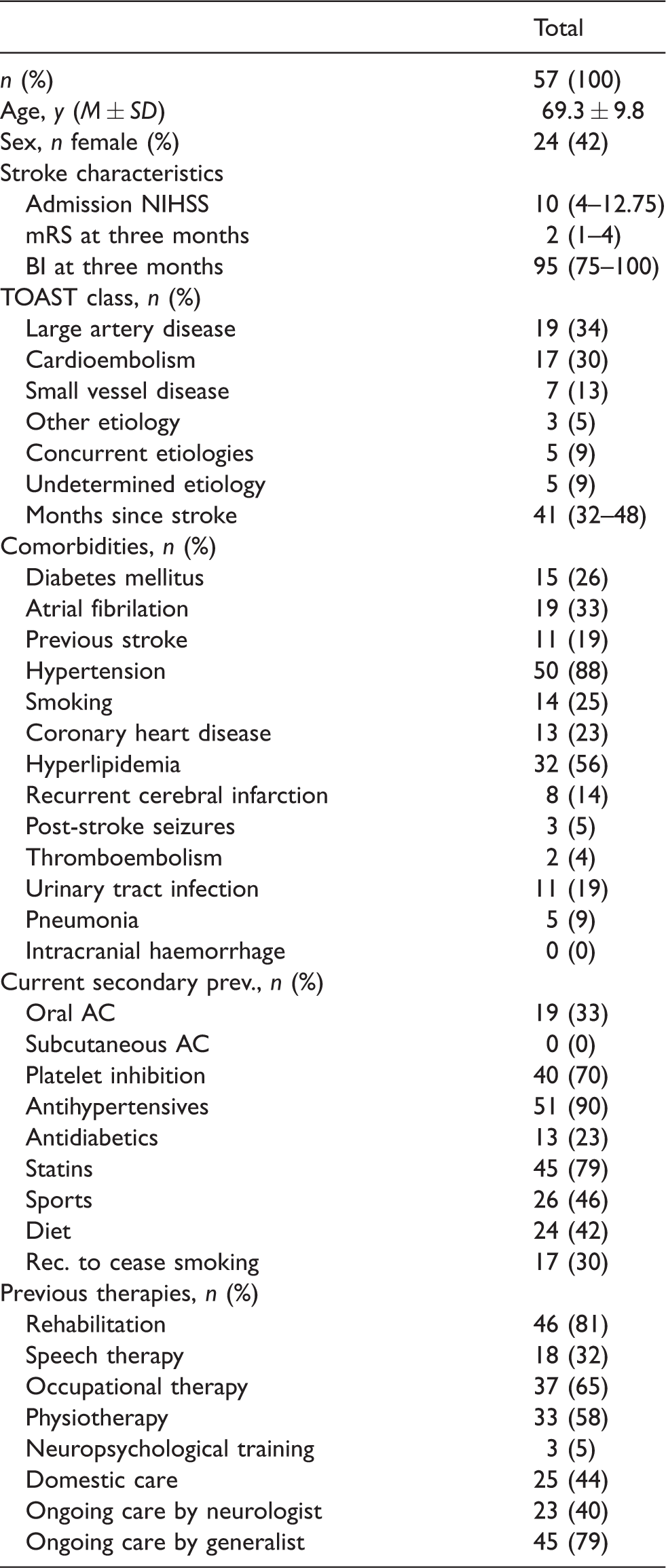
BI: Barthel Index; NIHSS: National Institutes of Health Stroke Scale; mRS: Modified Rankin Scale.
Numbers given are presented as median (interquartile range (IQR)) if not specified otherwise. Prev.: prevention; AC: anticoagulants, Rec.: recommendation; mRS: modified Rankin Scale.
Patient and Carer needs
Table 3 shows patients’ needs according to validated clinical scales including: spasticity (35%), cognitive deficits (61%); depression (20%) and medication non-compliance (14%). Spasticity (p = 0.008) and social needs (p < 0.001) showed the strongest association to quality of life (see Table 4 for further detail). The items from PSC screening for low mood (p < 0.001), impaired cognition (p = 0.015), social needs (p = 0.005) and caregiver burden (p = 0.031) were associated with the validated tools for these items.
Clinical and patient-reported outcome measures at assessment and recommendations by stroke specialist.
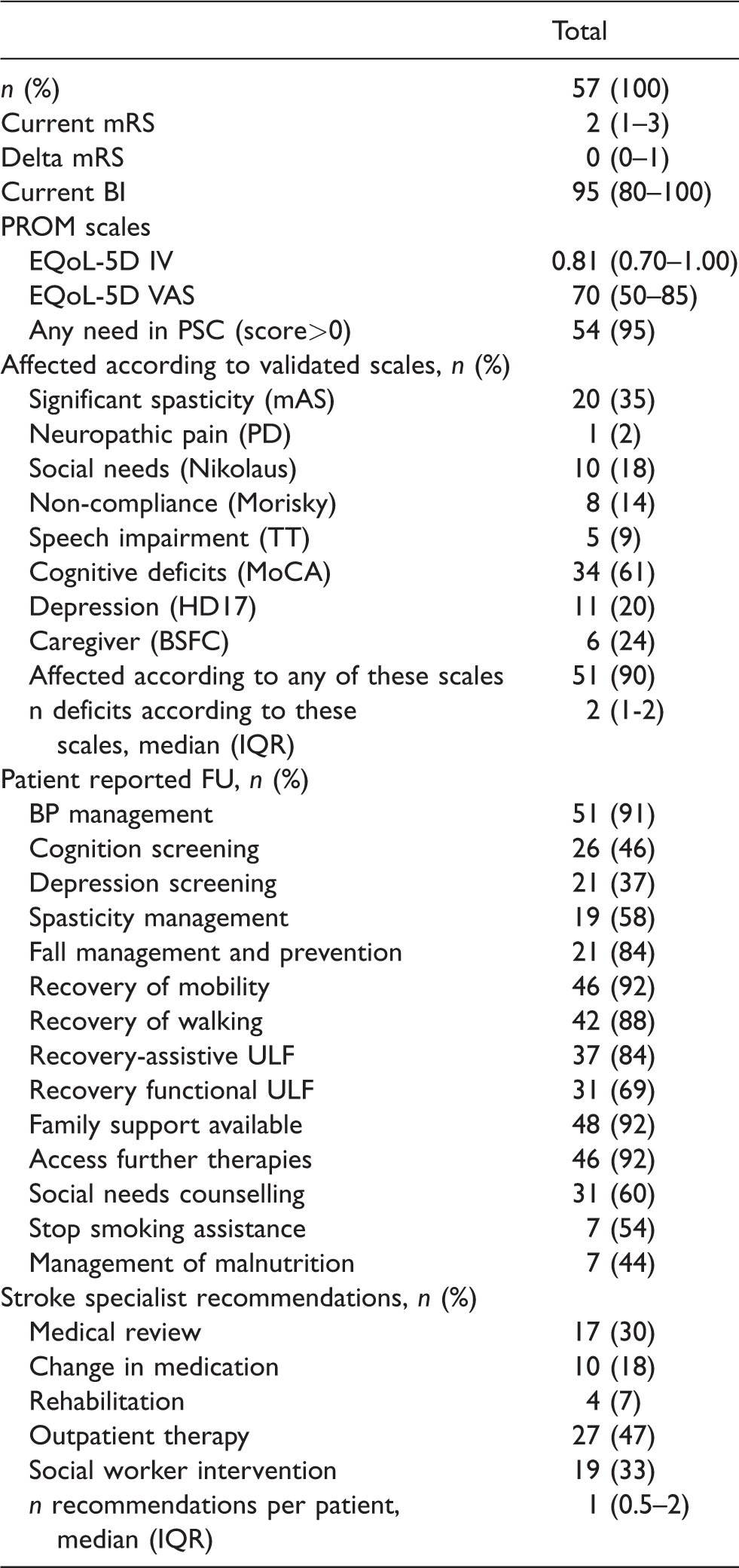
FU: follow-up; ULF: upper limb function; BI: Barthel Index; EQoL-5D EuroQuol; mRS: modified Rankin Scale; BP: blood pressure; PROM: patient-reported outcome measure; IV: index value; VAS: Visual Analogue Scale; PSC: Post-Stroke Checklist; TT: token test; PD: PainDetect; HD17: Hamilton Depression Scale 17 Items; MoCA: Montreal Cognitive Assessment; mAS: modified Ashworth Scale, Nikolaus Social Situation score, Morisky Adherence Scale; BSFC: Burden Scale for Family Caregivers.
Numbers given are presented as median (interquartile range (IQR)) if not specified otherwise.
Association of quality of life as measured by EuroQoL and clinical deficits as well as the sensitivities, specificities; negative and positive predictive values of the PSC to detect stroke-related health problems as measured by validated scales.
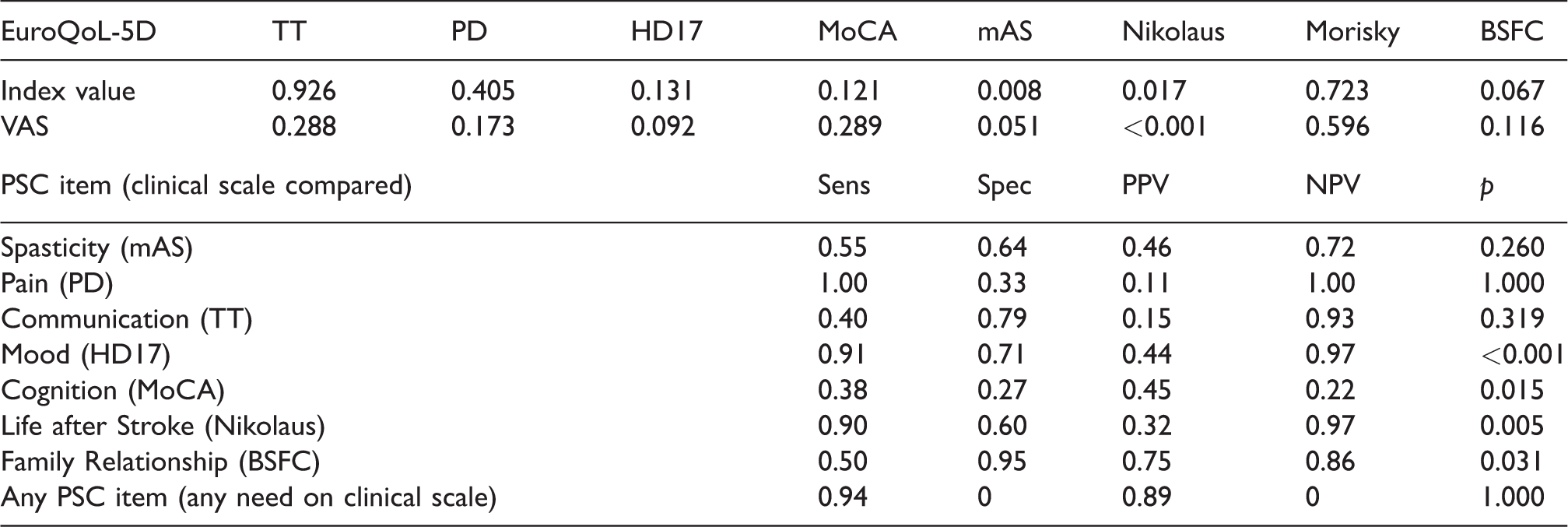
VAS: Visual Analogue Scale; TT: token test; PD: PainDetect; HD17: Hamilton Depression Scale 17 Items; MoCA: Montreal Cognitive Assessment; mAS: modified Ashworth Scale, Nikolaus Social Situation score, Morisky Adherence Scale; BSFC: Burden Scale for Family Caregivers; PSC: Post-Stroke Checklist; Sec.: secondary; ADL: activities of daily living; Sens.: sensitivity; Spec.: specificity; PPV: positive predictive value; NPV: negative predictive value.
Numbers given are p values obtained by Fisher’s exact test unless otherwise specified. For the PROM PSC Items associations were calculated where there are corresponding scales in the clinical scales.
Based on the PSC, 95% of participants demonstrated needs and reported at least one clinically significant stroke-related health problem (Figure 2(a)). Based on the clinical scales, 89.5% had a clinically significant deficit in at least one domain (Figure 2(b)). Approximately two-thirds showed significant cognitive deficits, one-third of patients suffered from significant spasticity and nearly 20% showed a clinically relevant depressive syndrome (Figure 2).
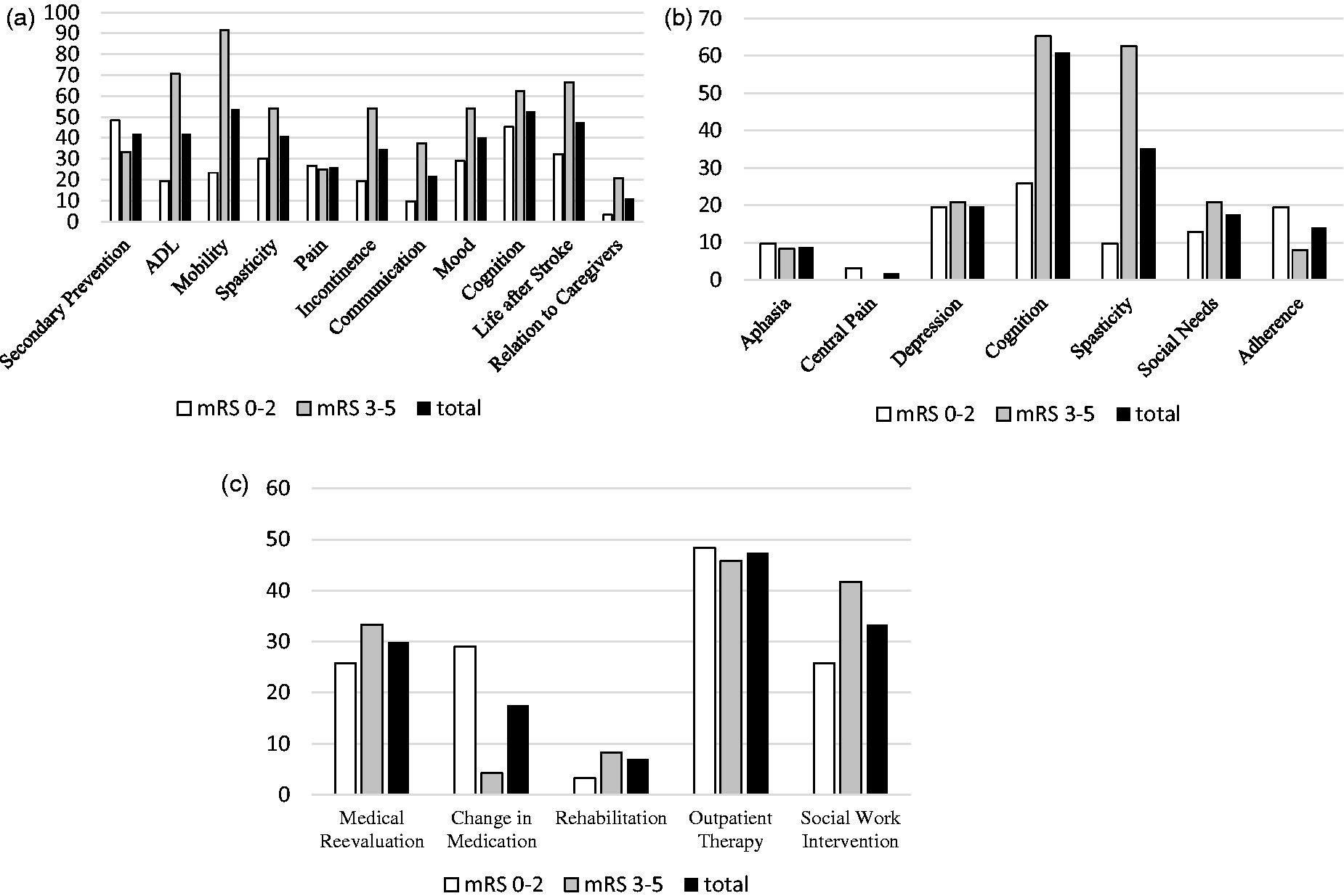
Frequency of (a) patient reported deficits (items from PSC), (b) domains affected according to extensive assessment and (c) of recommendations made in clinic based on guidelines given as % of patients in the cohort.
Burden Scale for Family Caregivers was available in 25 (44%) patients, with 6 (24%) caregivers reporting moderate to high levels of stroke-related caregiver burden.
Care compared to guidelines
Forty per cent of patients were regularly seen by a neurologist and 79% had ongoing general practitioner support. Differences in ongoing care for the patients compared to guideline recommendations were estimated based on the recommendations provided by a stroke specialist following the study assessment. Medical review (30%), social work intervention (33%), additional outpatient therapy (47%), e.g. physiotherapy or speech therapy, and optimisation of pharmacotherapy (18%) were recommended (Table 3 and Figure 2).
Feasibility of assessment
Patients and the multidisciplinary team found the comprehensive assessment to be acceptable and feasible to deliver in an outpatient (ambulatory) setting. There were no drop-outs during the testing. Regarding screening via PSC the scope and cover of the measures varied; not every domain was reflected in the PSC and not every PSC item was reflected in the assessment by clinical scales. Where domains and items overlapped, we found a significant association for mood, cognition, social needs and family/caregiver burden, but not for spasticity, pain and communication. Sensitivity, specificity, positive and negative predictive values for the PSC screening are given in Table 4. Deficits in selected domains, e.g. spasticity, social need, caregiver burden and depression correlate with EuroQoL-5D index values and visual analogue scale, suggesting an impact of these domains on health-related quality of life in chronic stroke (Table 4).
Discussion
Our cross-sectional study of well-characterised patients, drawn from previous prospective studies, demonstrated long-term comorbidities and complications of stroke across a wide number of domains – an area, in which comprehensive data are scarce. Due to marked variability between individuals in characteristics of chronic stroke burden, median and mean values on scores for individual domains may underestimate the health burden of these patients. For example, patients may have normal values on a spasticity scale, but still suffer from significant aphasia. Global scales and PROMs can highlight the current health state in a more representative, yet less specific way.
The overall impact of stroke on health-related quality of life was relevant. Certain domains, especially spasticity and social needs, were strongly associated with a reduced quality of life. Impaired quality of life was reported in a magnitude similar to a previous study, with similar associations of health-related quality of life with physical functioning, depression and cognitive decline. 7
We demonstrated feasibility of a multidisciplinary approach to comprehensive assessment of deficits in a selected sample of patients with chronic stroke. The time allocated to each discipline was sufficient to assess the full range and pattern of complaints, deficits and comorbidities, as well as medical and social needs. Generally, the measures we selected were well tolerated in our sample of outpatients and provide exploratory data on the relationship between outcomes and the performance of measures such as the PSC. However, a screening instrument would greatly help to triage in-depth assessments. We are uncertain if the length or format of assessment contributed to the relatively low response and uptake by stroke patients who decided not to participate.
The PSC was introduced as such a screening measure to identify on-going need in chronic stroke patients.21,38 Due to the high prevalence of clinically relevant deficits in our study, we cannot comment on the overall validity of the PSC as a screening tool. However, individual items of this PROM (mood, cognition, social needs and caregiver burden) are significantly associated with the respective clinical scales. In contrast, association was not significant for the domains of communication and pain, which might be due to the low prevalence of these disabilities in our cohort. Furthermore, the PainDetect scale measures neuropathic pain, whereas patients may report any kind of pain in the PSC, measuring a different kind of burden. Interestingly, the clinical assessment of spasticity was also not significantly associated with PROM, even though reported at a similar frequency. These results (including the sensitivity and specificity values given) should be interpreted with caution, since several subgroups showed low prevalence. Some items of the PSC (activities of daily living, mobility and incontinence) were not represented as individual domains in the clinical assessment and therefore could not be tested for association.
Our findings confirm that although stroke survivors report long-lasting deficits and needs, they also report their outpatient treatment is frequently limited or discontinued.2–14,39 Outpatient therapy was recommended in nearly half of our sample and almost one-fifth of our patients were considered to be on sub-optimal pharmacotherapy based on guideline recommendations.
Our study has the following limitations: The sample-size is limited and there is likely to be recruitment bias from the source cohorts and also from the general stroke population, which was subject to the initial selection criteria of the original studies. Patients in our study were more severely affected than in population-based stroke registries, showing recruitment bias and limiting generalizability to an unselected stroke population. The response rate and uptake were also relatively limited. We tried to accommodate all levels of mobility, but no bed-bound patients were included. Our cross-sectional design does not allow us to consider longitudinal changes. Reporting bias is to be expected in respect of the therapies patients underwent after the acute treatment. Furthermore, care might have been limited by providers who considered prognosis or potential recovery to be limited, or by a lack of patient adherence. We may have missed affected domains that were not included in our assessment battery. On the other hand, no patients reported any additional health issues not covered in our interview schedule. Strengths of this study include the well-characterized original stroke admission and relatively ‘long-term’ follow-up after stroke. The outcome measures reported in this manuscript are the validated standard measures in their respective domain (see Table 1) and simultaneously included PROM, clinical scales and carer measures.
In conclusion, we found unmet needs and a high stroke-related burden in the long-term after stroke in our Berlin sample that requires continued monitoring, treatment and care. Many stroke survivors have multiple conditions, which may be undetected or under-treated according to current evidence and guidance and there may be considerable scope for improvement in management and secondary prevention. These results warrant further study and emphasize the need for a valid screening tool for post-stroke deficits. Such a tool should include a broad range of domains in order to be valid. Despite the study’s limitations, it confirms the feasibility of a comprehensive assessment and has informed the design of a longitudinal interventional study designed to improve long term management of stroke (MAS-II clinicaltrials.gov NCT03097146). Furthermore, the efficacy, effectiveness and cost-effectiveness of a multi-disciplinary treatment approach in long-term aftercare for stroke need to be investigated.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by German Federal Ministry of Education and Research (01EO0801) and German Research Foundation (Exc257).
Ethical approval
This study received approval by the institutional ethics committee of Charité – Universitätsmedizin Berlin (reference EA1/183/14).
Informed consent
All participants gave informed consent either themselves or (in case of inability to do so) via a legal representative.
Guarantor
Prof. Dr. Andreas Meisel,
Contributorship
Benjamin Hotter co-designed the study, analysed the data and wrote the manuscript. Inken Padberg analysed the data and wrote the manuscript. Andrea Liebenau and Petra Knispel co-designed the study and wrote the manuscript. Sabine Heel and Diethard Steube analysed the data and wrote the manuscript. Jörg Wissel, Ian Wellwood and Andreas Meisel co-designed the study, analysed the data and wrote the manuscript.
Acknowledgements
The authors acknowledge the consulting contributions by Prof. Dr. Claudia Wendel and Gisela Kiank, the contribution of members of the CSB's stroke research consumers group (SBHF) group.
