Abstract
Introduction
One of the aims of the European Stroke Organisation (ESO) is to facilitate academic, multinational clinical stroke research. However, despite examples of successful regional and national stroke research networks and collaborative groups, there is no organisational structure at a European level that can facilitate multinational clinical stroke research.
Materials and methods
In a project including a survey and a workshop and involving stroke researchers in the ESO, we sought to identify the challenges faced by existing clinical stroke research networks, to define the purpose and roles of any future European stroke research collaboration, and to propose an organisational structure.
Results
The survey and workshop gave strong support for an alliance model with independent network members, with the purpose of facilitating clinical stroke research through improved coordination and communication, provision of support, education, and advocacy and communication with other stakeholders. The focus of a proposed European clinical stroke research alliance should be multinational randomised-controlled trials in acute care, prevention and rehabilitation, but the alliance could also support other forms of multi-national clinical stroke research.
Conclusion
There is an interest for increased collaboration on multinational clinical stroke research in Europe, in the form of an alliance of independent research networks and collaborative groups. The ESO Trials Network Committee will continue consultation with existing stroke research networks and collaborative groups, and other key stakeholders, to assess the feasibility and support for development of an ESO Trials Alliance.
Introduction

Methods
The project used a mixed methods approach, including a survey among European stroke trialists and a workshop among participants at the ESO Conference in Prague in 2017. Participants were informed of the plan to publish the results. As the project used responses from professionals and information from current practice, no formal ethical approval or written consent was deemed necessary.
Survey
We contacted all national stroke organisations registered in ESO and invited each organisation to send two representatives to a workshop about closer collaboration on clinical stroke research in Europe. We also sent invitations to individual members of ESO. People who registered for the workshop were asked to participate in a web survey. The questions were organised in three themes: (1) types of existing clinical stroke research networks, and challenges faced by these networks; (2) possible purpose and role of a European clinical stroke research collaboration; and (3) possible organisational structure of a European collaboration. For the purpose of this analysis, we only used the responses from survey participants from European countries.
Workshop
The first three themes in the survey were translated into three broad questions for discussion in groups: (1) What are the challenges for clinical stroke research in Europe?; (2) What could be the purpose of a European clinical stroke research collaboration?; and (3) What could be the structure of such a collaboration? In addition, we added a fourth question: (4) What could be the next steps and goals? Participants were divided into four groups of around 30 participants, by alphabetical order of participants’ names, and each group discussed two of the four questions. Conclusions from all eight group discussions were presented and discussed in a plenary session. The eight group discussions and the plenary session were chaired by members of the ESO Trials Network Committee.
Results
In total, 120 people from 37 countries attended the workshop, including 95 people from 27 European countries. Eight European countries were represented by delegates from national stroke organisations. Of the 120 participants, 94 had completed the survey, of whom 80 were from European countries and contributed to the results reported here. Participants in the survey were allowed to give more than one answer to each question, so the number of responses may sum up to more than the number of participants.
Existing networks and their challenges
Types of clinical stroke research networks and collaborative groups in Europe, types of challenges, and funding opportunities, as reported in the survey.
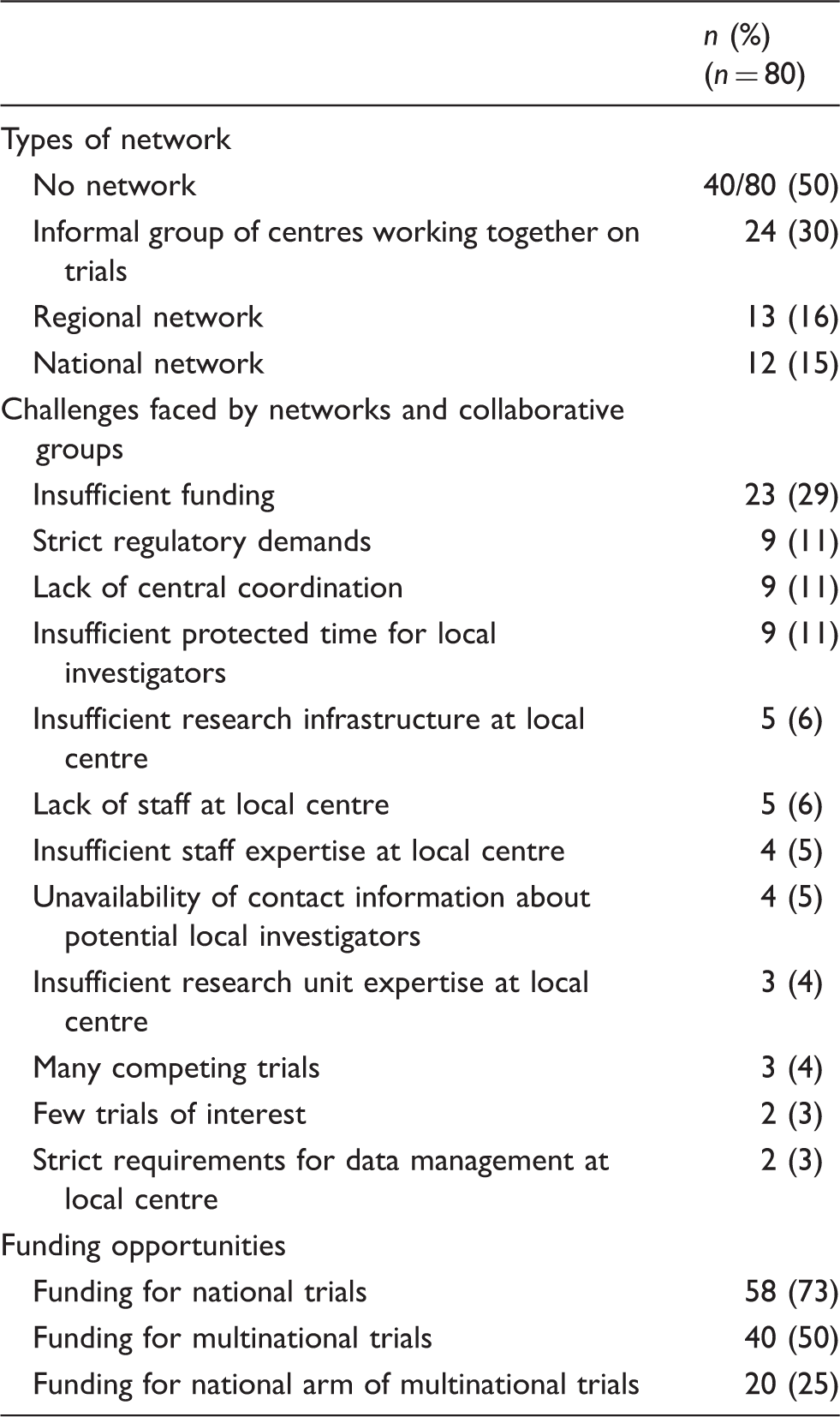
Participants in the survey were allowed to give more than one answer to each question, so the number of responses may sum up to more than the number of participants.
Obtaining funding was identified as the commonest challenge faced by the survey participants. Most participants (58 of 80, 73%) reported that they had been successful in securing funding for national trials (Table 1). However, only 40 of 80 respondents (50%) had received grant funding for multinational trials led from their own country, and only 20 (25%) had been successful in obtaining funding for national participation in multinational trials led from another country.

Purpose and roles of a European clinical stroke research collaboration
Purpose and roles of a European clinical stroke research collaboration, as reported in the survey.
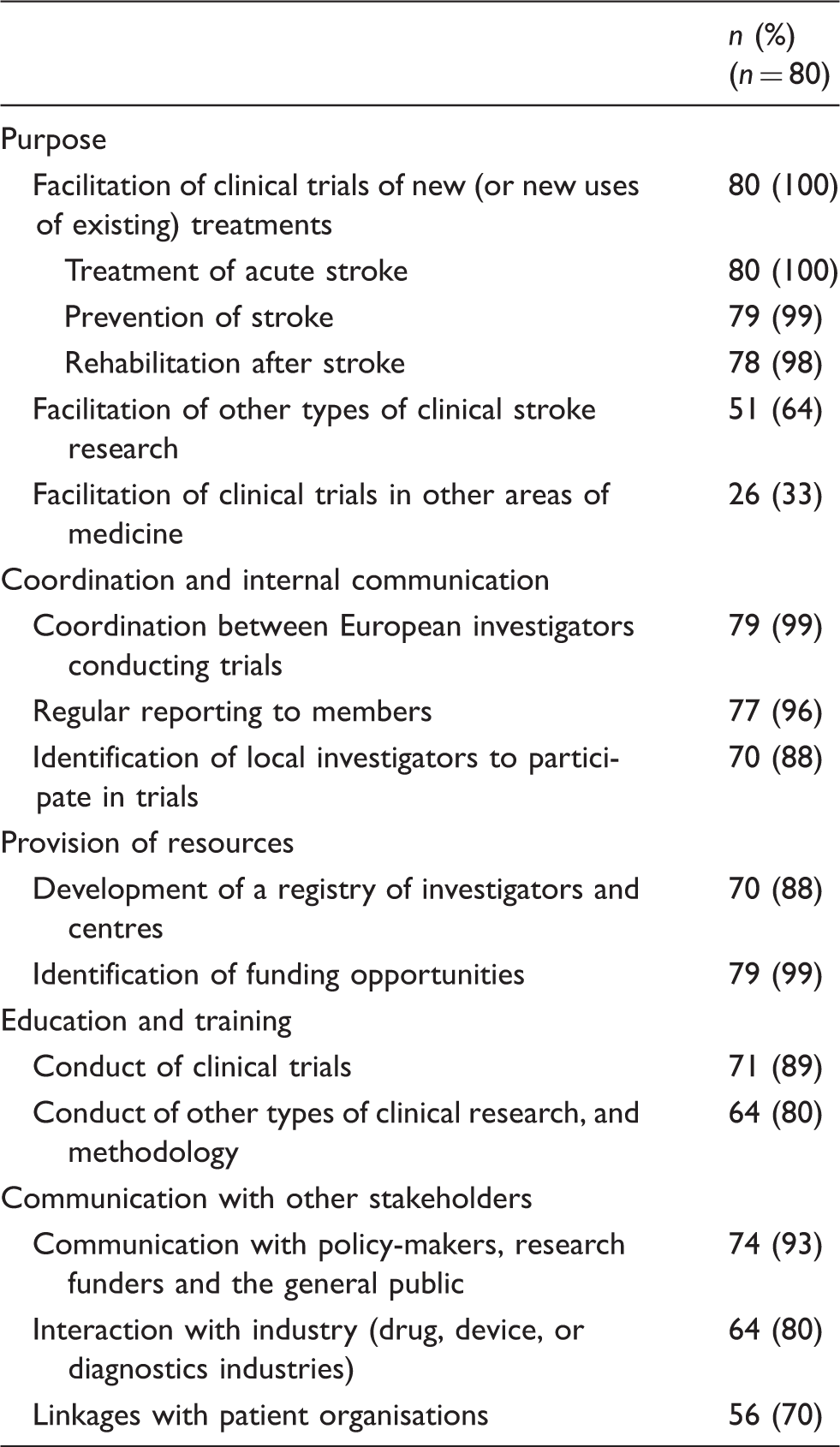
Participants in the survey were allowed to give more than one answer to each question, so the number of responses may sum up to more than the number of participants.
Table 2 also indicates the possible roles of a European clinical stroke research collaboration, as identified by the survey. Coordination of research and communication between members were seen as the main roles of a collaboration, including coordination between European researchers conducting trials (79 of 80 respondents, 99%), regular reporting to members (77 respondents, 96%), and identification of investigators interested in participating in clinical trials (70 of 80 respondents, 88%). Most participants also agreed that the collaboration should provide resources to facilitate the conduct of multinational trials, such as registries of investigators and centres (70 of 80 respondents, 88%), as well as overviews of funding opportunities at national and European levels (79 respondents, 99%). Education and training were also seen as important tasks, including training in the conduct of clinical trials (71 of 80 respondents, 89%) and in research methodology, e.g. epidemiology (64 respondents, 80%). Finally, the majority of participants recommended that the collaboration should play a role in advocacy and communication with other stakeholders, such as policy-makers, research funders and the general public (74 of 80 respondents, 93%), industry (64 respondents, 80%) and patients’ representative groups (56 respondents, 70%).

Structure of a European clinical stroke research collaboration
Structure of a European clinical stroke research collaboration, as reported in the survey.


The next steps: development of a European alliance of clinical stroke research networks
The workshop participants finally discussed the next steps. There was strong support for the ESO Trials Network Committee to develop this project, through consultation with ESO governing bodies and members, existing clinical stroke research networks and collaborative groups, national stroke organisations, research funders and other key stakeholders.
Discussion
In this project involving stroke researchers from 27 European countries, we found that existing clinical stroke research networks face many of the same challenges, and that there is an interest in a closer collaboration, in the form of a European alliance of independent networks and collaborative groups. The participants suggested that the alliance should have a non-hierarchical structure, realistic goals and a gradual and stepwise development, and promote independence of member networks and groups. They also suggested that it should focus on facilitating the conduct of randomised-controlled trials, but could also be inclusive of other types of clinical stroke research.
An alliance model for collaboration has been successful in other areas of clinical research. The International Forum of Acute Care Trialists (InFACT) 12 is a global alliance of regional and national networks for critical care research, funded by small grants from national organisations. It has a small executive committee and a council of members’ representatives, and is not involved in trial management, but operates by fostering research collaboration. There is no “gateway” committee, and trials are not formally approved, but discussed at meetings, and member networks can participate if they are interested. Working groups are formed by researchers with shared interests to address funding and conduct of specific projects. A similar approach is also being adopted by the Global Alliance of Independent Networks focussed on Stroke (GAINS), 13 which is a recent initiative to stimulate international collaboration in clinical stroke research across the world.
This project gathered stroke researchers from the majority of European countries, and used a systematic approach to identify existing challenges and to define the purpose, roles and structure of a possible European stroke research collaboration. However, we acknowledge weaknesses such as the limited number of participants, the limited number of delegates from national stroke organisations, the possibility of bias caused by selective participation and our subjective interpretation of the conclusions at the workshop, and the lack of involvement of other key stakeholders, such as patient organisation representatives and research funders.
In conclusion, we have found that there is an interest for increased collaboration on multinational clinical stroke research in Europe, in the form of an alliance of independent research networks and collaborative groups. Such an alliance could learn from existing alliances in other areas of clinical research and integrate with stroke research networks in other parts of the world, and can potentially lead to more, bigger, and better European clinical stroke studies. The ESO Trials Network Committee will continue consultation with existing stroke research networks, collaborative groups, and other key stakeholders, to assess the feasibility and support for further development of an ESO Trials Alliance.
Footnotes
Acknowledgements
We thank Daniela Niederfeld at the ESO Head Office for help with the survey and the workshop, and Miquel Gallofré for information about the Catalan Stroke Code and Reperfusion Consortium. The workshop was organised by the ESO Trials Network Committee and paid for by the ESO. Members of the ESO Trials Network Committee (2016–2017): Marcel Arnold, Eivind Berge (chairman), Exuperio Diez-Tejedor, Dalius Jatuzis, Peter J. Kelly, Derk W. Krieger, Paul J. Nederkoorn, Peter Sandercock, Christian Stapf, and Christian Weimar. Associate members: Gary A. Ford and Rustam Al-Shahi Salman.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: GAF was Director of the NIHR Stroke Research Network from 2005 to 2013. MA is president of the Swiss Stroke Society which is leading the Swiss Stroke Research Network. PJK is national lead for the HRB Stroke Clinical Trials Network Ireland. The other authors have no competing interests to declare.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The costs of the workshop were covered by ESO.
Informed consent
As the project used responses from professionals and information from current practice, no formal ethical approval or written consent was deemed necessary.
Ethical approval
As the project used responses from professionals and information from current practice, no formal ethical approval or written consent was deemed necessary.
Guarantor
EB.
Contributorship
EB, GAF and PJK planned the survey and the workshop. PJK, SC and CS analysed the survey data. EB wrote the first draft of the report. All authors contributed to the survey and the workshop and commented on the report.
