Abstract
Background
Hyperglycaemia is a frequent complication in acute stroke that has been shown to be independently associated with larger infarct size, haematoma growth, poor clinical outcome and mortality. This Guideline Document presents the European Stroke Organisation (ESO) Guidelines for the management of blood glucose levels in patients with acute ischemic or haemorrhagic stroke.
Methods
The working group identified related questions and developed its recommendations based on evidence from randomised controlled trials following the standard operating procedure of the ESO. This Guideline Document was reviewed and approved by the European Stroke Organisation Guidelines Committee and the European Stroke Organisation Executive Committee.
Results
We found low-quality evidence from clinical trials in ischemic or haemorrhagic stroke exploring the use of intravenous insulin aimed to achieve a tight glycaemic control with different glucose level targets and several other sources of heterogeneity. None of these trials neither the meta-analysis of them have demonstrated any significant benefit of tight glycaemic control with intravenous insulin in acute ischemic or haemorrhagic stroke patients on functional outcome or in survival and they have shown an increased risk for hypoglycaemia.
Conclusions
We suggest against the routine use of tight glycaemic control with intravenous insulin as a means to improve outcomes. The currently available data about the management of glycaemia in patients with acute stroke are limited and the strengths of the recommendations are therefore weak. Nevertheless, this does not prevent that hyperglycaemia in acute stroke patients could be treated as any other hospitalised patient.
Introduction
Hyperglycaemia is a frequent complication in the acute phase of stroke, affecting up to 50% of patients, both in patients with diabetes mellitus (DM) but also in those without a prior diagnosis of DM.1–3 There is a strong evidence for the association of high glucose levels on deleterious effects during the acute phase of stroke, as it is an independent predictor of larger infarct size, poor clinical outcome and higher risk of mortality. 4 In acute ischemic stroke (IS) patients, it could counterbalance the benefit of recanalisation therapies such as intravenous (IV) thrombolysis5–12 or mechanical thrombectomy.13,14 Although less data are currently available, it has been unequivocally shown that hyperglycaemia is independently associated with poor outcome also in patients with acute haemorrhagic stroke or cerebral venous thrombosis.15–18
Noteworthy, poor outcome linked to hyperglycaemia is not exclusive to stroke patients as it has been reported in hospitalised, mostly critically ill, patients, with or without DM. 19 Thus, international guidelines for inpatient glycaemic control have been developed. 19 An early randomised controlled trial in critically ill patients admitted to intensive care units suggested that intensive treatment of hyperglycaemia with target glucose levels between 4.4 and 6.0 mmol/L (80 and 100 mg/dL) was safe and improved hospital outcomes by reducing in-hospital complications. 20 These results led to the development of clinical trials in several diseases 21 trying to evaluate the possible benefit of tight glycaemic control with IV insulin on outcome and stroke was not an exception.
The aim of this guideline is to update the ESO guidelines 22 and develop evidence-based recommendations for the management of glucose levels in acute stroke patients and, in particular, whether a tight glycaemic control with IV insulin should be preferred in acute stroke patients over a standard glycaemic control.
Methods
The ESO Guidelines Committee invited the lead author (ED-T) to develop and chair a working group (WG) of experts in glucose management in acute stroke. The WG consisted of ED-T, BF, GN, GT, JP with the collaboration of BT from the Cochrane Stroke Group for the systematic literature search. The composition of the WG was approved by the ESO Guidelines Committee and the Executive Committee.
This guideline has been developed following the ESO standard operating procedure for the development of clinical guidelines, 23 which recommends to follow the Grading of Recommendations Assessment, Development and Evaluation (GRADE) methodology. 24
The PICO questions (the PICO acronym stands for Population, Intervention, Comparator, Outcome) were established by consensus of all the WG members. The selected outcomes were rated by importance using a nine-degree scale (7–9 critical, 4–6 important and 1–3 of limited importance).
The WG formulated 10 PICO questions, five related to IS and five to haemorrhagic stroke. Survival and functional outcomes were rated as of critical importance (average nine points) whilst hypoglycaemia (symptomatic or not), infarct or hematoma growth and the development of hypokalaemia were rated as important, but not critical for decision-making (Appendix 1). For each PICO question, subgroup analysis according to prior diagnosis of DM or to the presence of hyperglycaemia on baseline was pre-specified.
The search strategy was formulated according to the PICO questions. Each of them is presented in the results section. In July 2015, BT searched the literature for randomised trials, controlled trials, meta-analyses and systematic reviews of clinical trials using the following databases: Cochrane Stroke Group Trials Register, Cochrane library databases, MEDLINE, EMBASE and CINAHL. For the purpose of this guideline, both ischemic and haemorrhagic strokes were included in the literature search although the final selection of included studies was done separately for ischemic and haemorrhagic strokes according to the formulation of the PICO questions. The details of literature search process are available in online supplementary Appendix 2.
Duplicates were identified and removed and manual search of all references was performed to identify eligible studies. For each PICO question, two authors independently screened the titles and abstracts of the publications and assessed the full text of potentially eligible studies.
Eligible studies were assessed by two members of the WG for each PICO question to extract and analyse the data. In the case that specific data were not reported in an eligible study, its corresponding author was contacted. In case of no response, the co-authors of the study were also contacted. If no answer was received after several attempts, data were considered as missing and the study was not included in the analysis.
For the analysis of the extracted data, we used the Review Manager 5 software (Version 5.3.5; Copenhagen: The Nordic Cochrane Centre, The Cochrane Collaboration, 2014). Data analysis was performed on a random-effects basis, and results were summarised as risk ratios (RRs) and 95% confidence intervals (CIs). Heterogeneity across studies was evaluated with the I2 test. Publication bias was assessed with the help of funnel plots. The results of data analysis were imported into the GRADEpro Guideline Development Tool (McMaster University, 2015; developed by Evidence Prime, Inc.).
Quality of evidence was then graded for each PICO question. The direction, the strength and the formulation of the recommendation were determined according to the ESO SOP. Whenever the WG considered that further clarification on a PICO question would be appropriate, an ‘Additional Information’ box was added.
Results
Literature search retrieved a total of 5897 results. After duplicate removal, 4355 manuscripts were screened based on title/abstract review and 15 manuscripts were selected for further reading. Five manuscripts were excluded at this step.25–29 The flowchart with reasons for exclusion is provided in Figure 1. Thus, a total of 10 articles were finally selected.
Flow diagram of the systematic review search. *One manuscript excluded as this was only published as conference proceedings.
25
One manuscript excluded due to nonconfirmation of stroke prior randomisation
26
and another one due to active treatment with IV insulin in the control group.
27
Two manuscripts28,29 excluded due to the impossibility to obtain separate data for ischemic and haemorrhagic stroke and data collection not currently available after contacting the authors.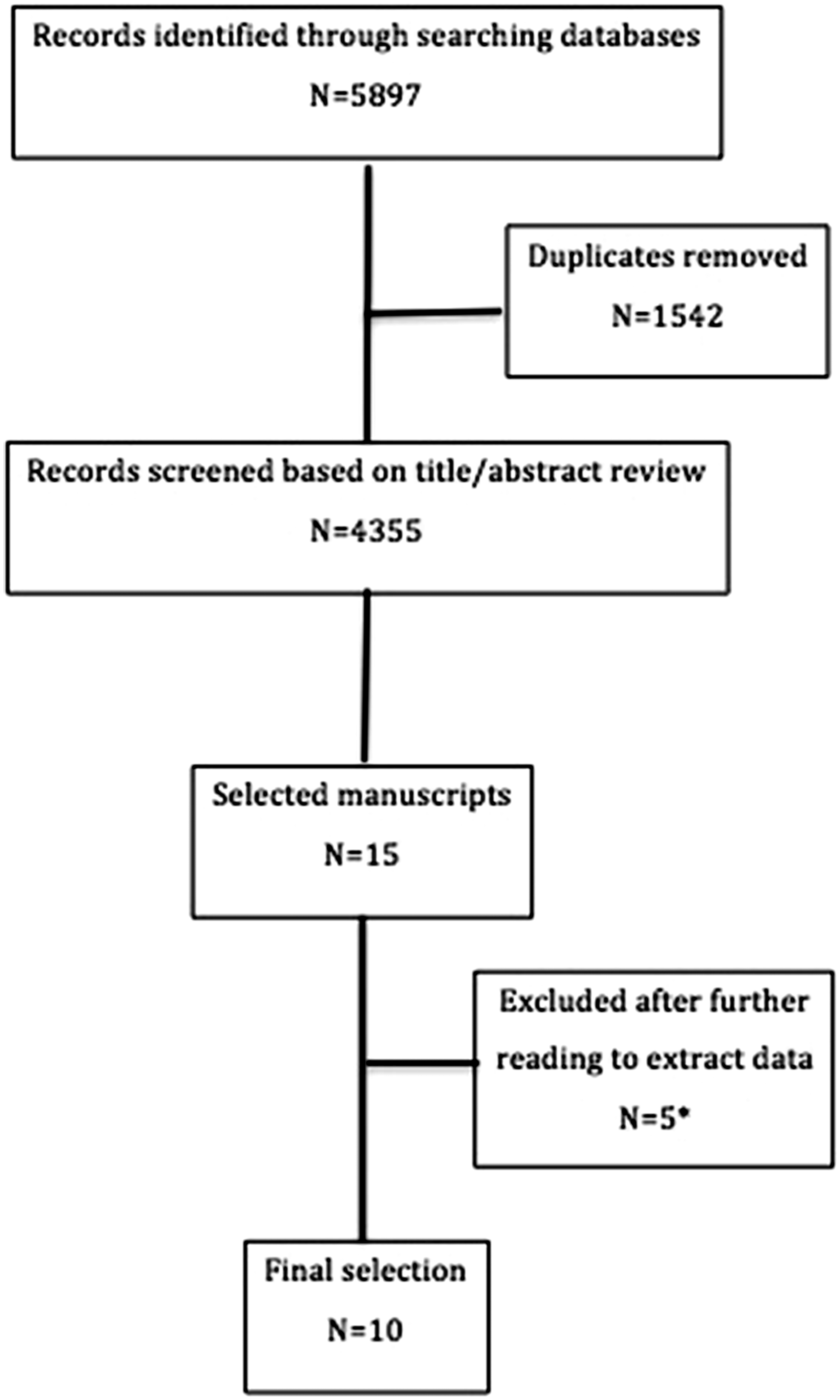
Main characteristics of the included studies.
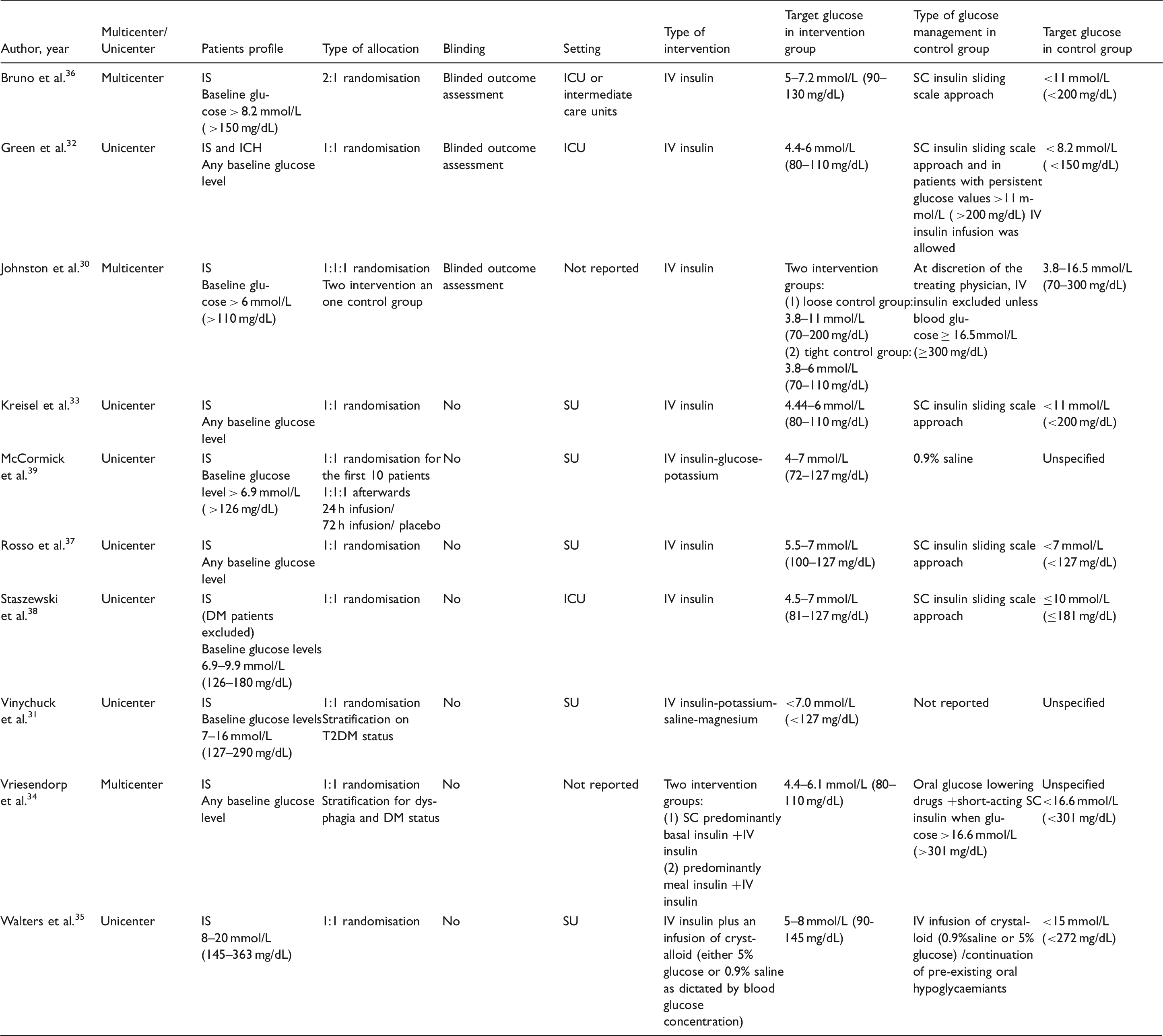
EMS: means Emergency Medical System; ICU: intensive care unit; IS: ischemic stroke; SU: stroke unit; T2DM: type-2 diabetes mellitus.
Importantly, there was substantial heterogeneity regarding the control arm of each study. Indeed, controls received subcutaneous (SC) insulin with a sliding approach in five studies,32,33,36–38 could receive oral lowering glucose agents in two studies34,35 and did not receive any glucose-lowering treatment in two studies.31,39 An additional source of heterogeneity was the various definitions of hyperglycaemia or baseline glucose level required for inclusion, namely 6 mmol/L (110 mg/dL), 30 6.9 mmol/L (126 mg/dL),38,39 7 mmol/L (127 mg/dL), 31 8 mmol/L (145 mg/dL) 35 or 8.2 mmol/L (150 mg/dL). 36 In four studies, patients with any baseline glucose level could be enrolleded.32–34,37
Ischemic stroke
PICO 1: In acute IS patients with hyperglycaemia, does treatment with IV insulin for tight glucose control compared to no treatment/SC insulin improve functional outcome?
A total of eight clinical trials on glucose management in acute IS, comprising 560 IS patients reported functional outcomes30–33,36–39 There was heterogeneity among studies regarding the type of intervention, the threshold blood glucose for inclusion, the target glucose level in the intervention group, the type of glucose management in the control group, the time to start of treatment, the duration of treatment and the time for outcome analysis, which ranged between 30 days and 4 months (Table 1). Modified Rankin Scale (mRS) score at the end of follow-up was reported in seven of the included studies,30,32,33,36–39 whilst Vinychuck et al.
31
reported Barthel Index (BI). Figure 2(a) shows the forest plot for comparison between the clinical trials regarding good functional outcome, defined as mRS < 3 or BI ≤ 50. The meta-analysis showed no statistically significant difference in rates of good functional outcome between patients treated with insulin per clinical trial protocol and controls (RR 1.09; 95%CI 0.87–1.37) with no sign of statistical heterogeneity. The quality of evidence was downgraded to low due to the heterogeneity between studies and the serious risk of bias as all studies had a performance bias (non-blinded treatment) and some of them also had an outcome bias (non-blinded outcome evaluation).
Forest plot analysis for ischemic stroke. (a) Good functional outcome at the end of follow-up, (b) survival at the end of follow-up, (c) hypoglycaemic events (<3–4 mmol/L) and (d) symptomatic hypoglycaemic events.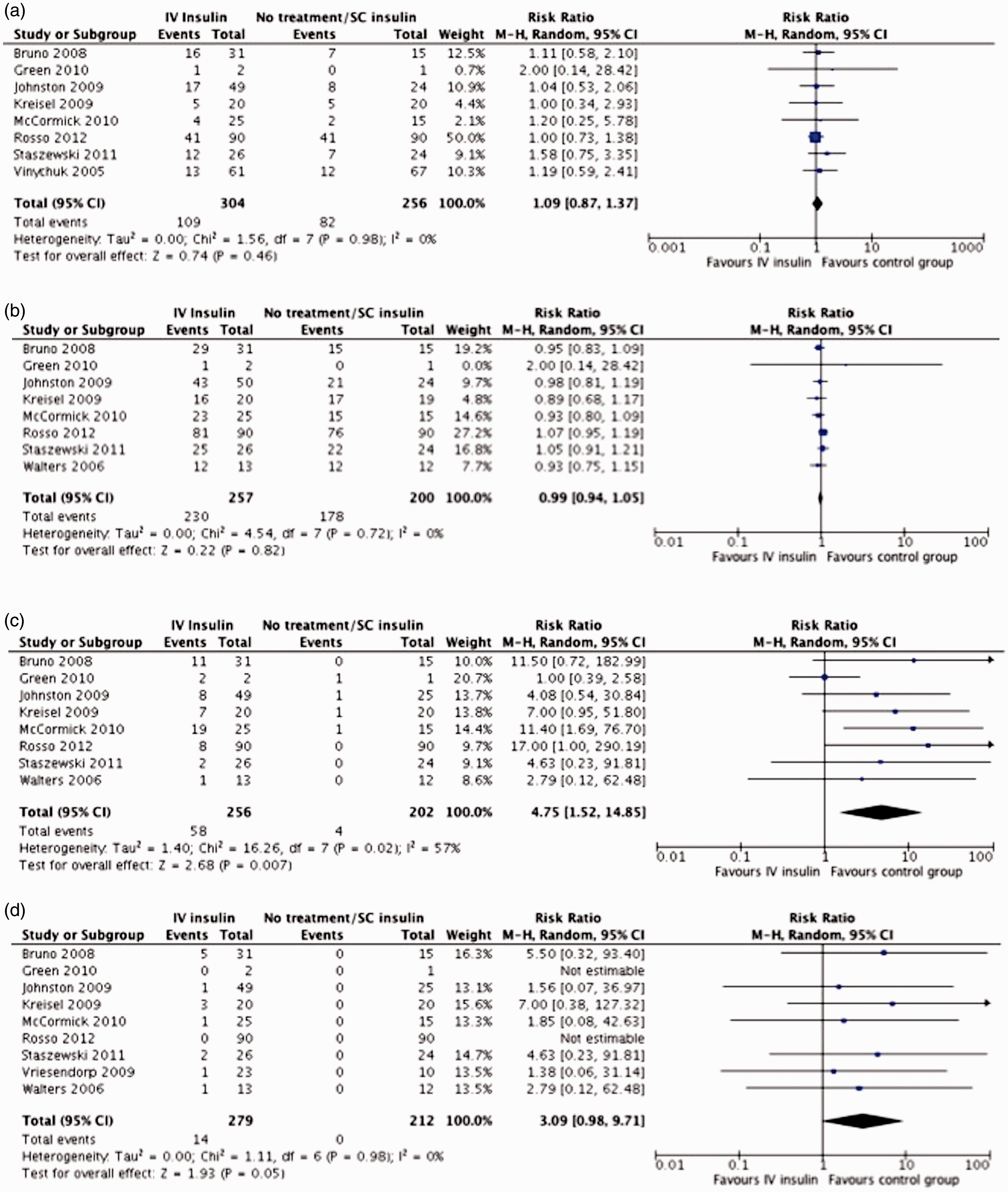
Subgroup analysis
Only one trial reported functional outcome analysis stratified on prior diagnosis of DM.31 A total of 76 patients with DM were included in the analysis that retrieved no significant difference between the intervention and control group (RR 1.25; 95%CI 0.54–2.89).
A total of 52 patients without prior diagnosis of DM were included in the analysis, without any difference in the risk of poor functional outcome at the end of follow-up period between groups (RR 1.35; 95%CI 0.41–4.47).
Five trials selected patients with high glucose levels on baseline31,35,36,38,39 and four of them31,36,38,39 reported outcome analysis with no significant differences between groups (RR 1.25;95%CI 0.85–1.84) (Figure 3(a)).
Forest plot analysis for ischemic stroke patients with hyperglycaemia at baseline. (a) Good functional outcome at the end of follow-up, (b) survival at the end of follow-up, (c) hypoglycaemic events (<3–4 mmol/L) and (d) symptomatic hypoglycaemic events.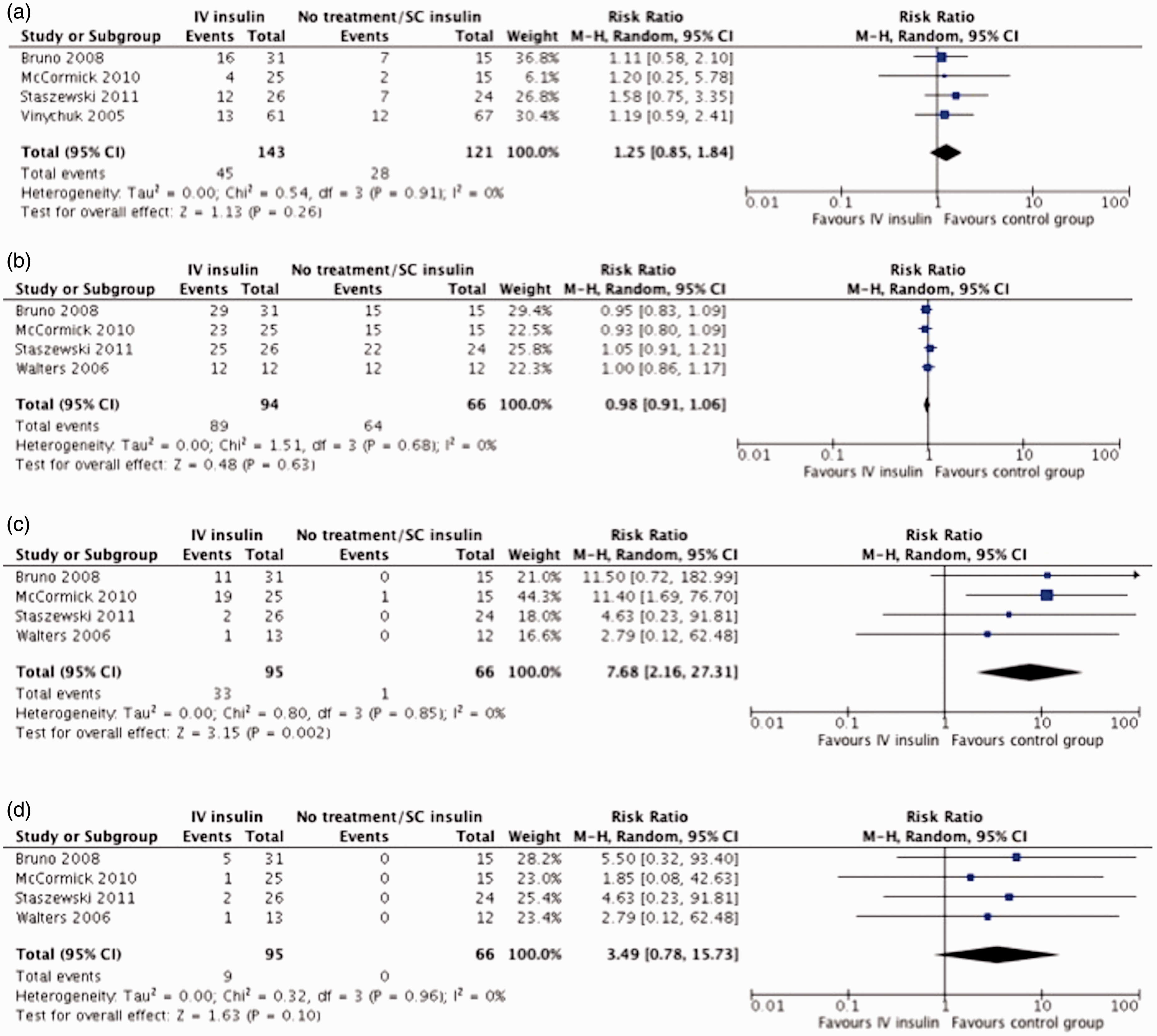
Additional information
Data from prospective observational studies40–42 and from a systematic review of cohort studies 43 have clearly shown an increased risk of poor functional outcome in patients with IS who develop post-stroke hyperglycaemia, independently of other prognostic factors, especially in those patients with persisting hyperglycaemia. 44
PICO 2: In acute IS patients with hyperglycaemia, does treatment with IV insulin for tight glucose control compared to no treatment/SC insulin improve survival?
Eight studies30–33,35–39 reported data on the number of deaths at the end of follow-up, which ranged between 30 days35,38,39 and 4 months. 33 There was heterogeneity among studies regarding the type of intervention, the target glucose in the intervention group and the type of glucose management in the control group (Table 1). The meta-analysis showed no significant difference in survival rates between patients treated with IV insulin for intensive glycaemic reduction and controls managed by the standard protocol or without specific glucose-lowering treatment (RR 0.99; 95%CI 0.94–1.05), with no sign of statistical heterogeneity (Figure 2(b)). The quality of evidence was downgraded to low due to serious risk of bias as all of the studies had a performance bias (non-blinded treatment).
Subgroup analysis
None of the trials reported separate data on mortality for patients with or without prior diagnosis of DM.
Five trials selected patients with high glucose levels on baseline31,35,36,38,39 and four of them35,36,38,39 reported data on survival at the end of follow-up with no significant differences between groups (RR 0.98; 95%CI 0.91–1.06) (Figure 3(b)).
Additional information
Data from prospective observational studies41,45,46 and a systematic review of cohort studies 43 have clearly shown an increased risk of death in patients with IS who develop post-stroke hyperglycaemia, independently of other prognostic factors. However, to date no clinical trial has demonstrated any effect of insulin on the risk of death. This may be due to the small sample size of previous trials, making them clearly unpowered. In fact, the UK-Glucose Insulin in Stroke Trial (GIST-UK) trialists 28 estimated that a sample size of 2355 patients would be needed to detect a significant difference in mortality, while our review only included a total of 457 patients. One limitation of our review is the exclusion of the largest clinical trial (GIST)28,29 that included 933 patients but was deemed to be excluded by the WG due to the impossibility to obtain separate data for ischemic and haemorrhagic stroke, even though the authors were contacted. That study was prematurely stopped and no difference in mortality was found between the intervention and control groups. Nevertheless, there are several studies suggesting that normalisation of glucose levels in patients with acute stroke could confer a survival benefit.45,47,48
PICO 3: In acute IS patients with hyperglycaemia, does treatment with IV insulin for tight glucose control compared to no treatment/SC insulin increase the risk of any hypoglycaemia?
A total of nine studies reported data on hypoglycaemia events.30,32–39 Definitions of hypoglycaemia varied between <3 mmol/L (54 mg/dL) and <4 mmol/L (72 mg/dL) whilst two studies31,35 did not specify glucose levels threshold for hypoglycaemia definition. One study was excluded from the first meta-analysis on any hypoglycaemia event as it reported number of events related to the number of glucose evaluations rather than to the number of patients. 34 Patients in the intervention groups were at higher risk of any hypoglycaemic event (RR 4.75 95%CI 1.52–14.85). There was substantial statistical heterogeneity (I2 = 57%) (Figure 2(c)).
Patients in the intervention groups also had a higher risk of symptomatic hypoglycaemic events (RR 3.09; 95%CI 0.98–9.71, Figure 2(d)).
Subgroup analysis
Two studies reported separate data on hypoglycaemic events in patients with or without prior diagnosis of DM.33,36 However, one study had to be excluded for the meta-analysis of hypoglycaemic and symptomatic hypoglycaemic events, because number of events but not number of patients with hypoglycaemia were reported. 33 Data from Bruno et al.36 provided an RR for hypoglycaemic events in patients with prior diagnosis of DM of 8.63 (0.55–132) and of 5.5 (0.32–93.4) for symptomatic hypoglycaemia events. No data to estimate the RR of hypoglycaemic events (either symptomatic or not) in patients without diagnosis of DM were available.
Five trials selected patients with high glucose levels on baseline31,35,36,38,39 and four of them35,36,38,39 reported data on hypoglycaemic events showing a higher risk of any hypoglycaemic event (RR 7.68;95%CI 2.16–27.31) (Figure 3(c)) and a non-significant trend to higher risk of symptomatic hypoglycaemic events (RR 3.49; 95%CI 0.78–15.7) (Figure 3(d)).
PICO 4: In acute IS patients with hyperglycaemia, does treatment with IV insulin for tight glucose control compared to no treatment/SC insulin reduce infarct growth?
Only two clinical trials measured infarct growth37,39 with controversial findings. McCormick et al. 39 in a clinical trial involving 40 patients failed to demonstrate any significant difference in absolute or relative lesion growth between baseline and either day 3 or day 7. However, Rosso et al. 37 in a study involving 160 patients who underwent a control MRI showed significantly larger infarct growth in the intervention group (median 27.9 cm3 IQR 3.4–64.2 vs. 10.8 cm3 IQR 2.6–41; 60% of difference; p = 0.04). In a multivariable analysis conducted to investigate those factors associated with infarct growth, intensive insulin therapy, baseline NIHSS and admission DWI were included in the final model, but not hypoglycaemic events. No meta-analysis could be performed due to the lack of the needed data to do it.
Additional information
It has been reported from prospective cohort studies that persistent hyperglycaemia is associated with infarct growth.40,49
PICO 5: In acute IS patients with hyperglycaemia, does treatment with IV insulin for tight glucose control compared to no treatment/SC insulin increases the risk of hypokalaemia?
None of the clinical trials that studied the effect of insulin treatment in acute ischaemic stroke evaluated the risk of hypokalaemia. However, two studies reported data on serum potassium values during the treatment with no significant differences in serum potassium value or in change in serum potassium levels during the protocol treatment between the intervention and control groups.35,36
Additional information
Few studies have reported the frequency of hypokalaemia in IS patients treated with IV insulin. An observational study analysing safety and feasibility of a IV insulin infusion protocol reported hypokalaemia (defined as serum potassium <3.5 mMol) in up to 18.5% of acute IS patients who received IV insulin with a target of preprandrial glycaemia of 4–6 mmol/L (72–109 mg/dL). 50
Recommendation
In patients with acute IS, we suggest against the routine use of IV insulin to achieve a tight glycaemic control as a means to improve functional outcome, survival or infarct growth.
Additional comments for clinical practice
Summary of Findings (SoF) table for ischemic stroke.
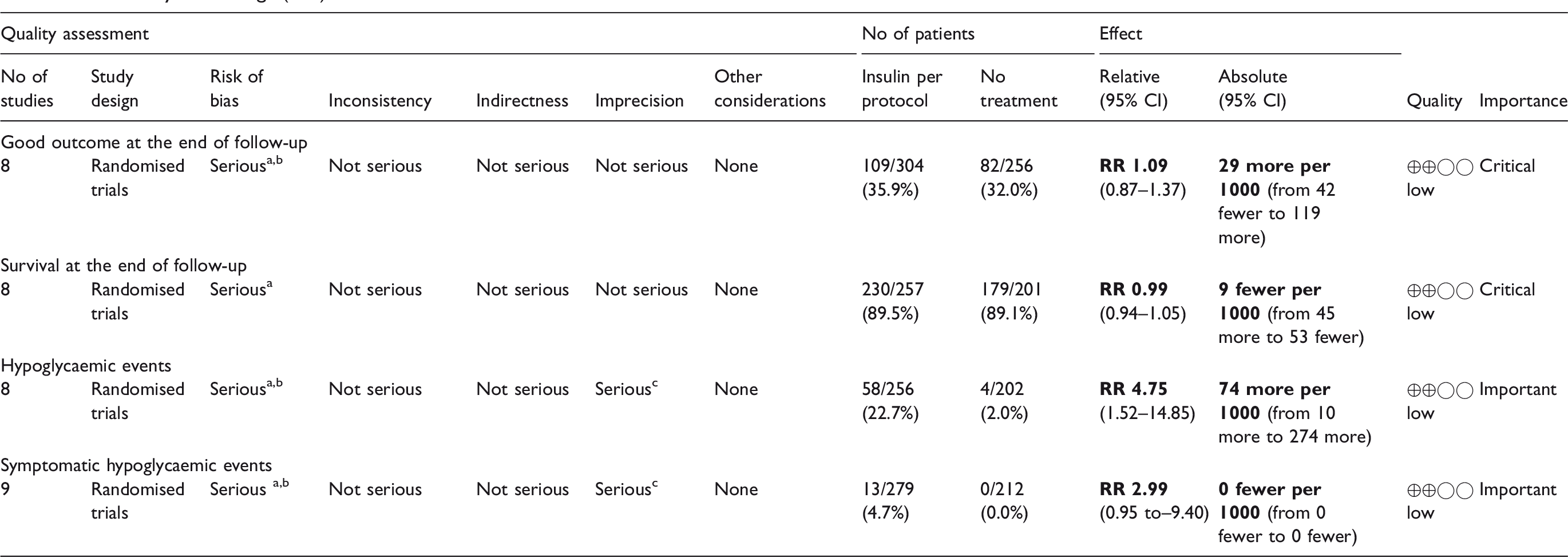
CI: confidence interval; RR: risk ratio; OR: odds ratio.
Performance bias.
Non-blinded outcome analysis.
Low number of events.
Nevertheless, taking into account the clear risk of functional dependence and mortality associated with hyperglycaemia in patients with IS, the lack of evidence of any benefit of tight glucose reduction with IV insulin due to the poor quality of the clinical trials developed to date, does not prevent that IS patients with hyperglycaemia could be treated as any other in-hospitalised patient with hyperglycaemia aiming at standard glycaemic control. 19 Expert-based recommendations suggest as the preferable approach for hyperglycaemia management in critically ill patients like acute stroke the use of IV insulin therapy titrated to achieve a target glucose level between 7.8 and 10.0 mmol/L, avoiding more intensive targets that can result in a higher risk of hypoglycaemia.19,51 There is also a general recommendation against the use of SC sliding-scale insulin or the so-called correction insulin based on the use of rapid-acting insulin in critically ill patients as the sole regimen as it is not evidence based and ineffective in the majority of the patients. 52 In fact, a systematic review that focused on the evaluation of efficacy of this approach yielded a total of 52 clinical trials, none of which described a benefit of the SC sliding-scale strategy. 53 After the acute phase of stroke when the patient is considered to be clinically stable, basal insulin or a basal plus bolus correction insulin regimen is the preferred treatment for patients with poor oral intake whilst basal, nutritional and correction components is the preferred strategy for non critically ill patients with good nutritional intake. 19 On the other side, noninsulin agents are also considered as inappropriate in most hospitalised patients. 19
Haemorrhagic stroke
PICO 6: In acute haemorrhagic stroke patients with hyperglycaemia, does treatment with IV insulin for tight glucose control compared to no treatment/SC insulin improve functional outcome?
Only one study provided data to perform the analysis on functional outcome in patients with acute haemorrhagic stroke. 32 This study randomised critically ill neurological patients to intensive IV insulin therapy aimed to maintain glucose levels between 4.4–6.0 mmol/L (80–110 mg/dL), or to conventional treatment to keep levels <8.3 mmol/L (151 mg/dL). A total of 25 included patients had haemorrhagic stroke (18 with intracranial haemorrhage and 7 with subarachnoid haemorrhage). Functional outcome was measured using the mRS at 90 days, either by non-blinded telephone assessment or by face-to-face evaluation in those patients who were still hospitalised at the time of follow-up. No significant difference in the risk of good outcome was found (RR 0.72; 95%CI 0.14–3.61).
Subgroup analysis
A total of 10 patients had prior diagnosis of DM in the study by Green et al. 32 However, no data on functional outcome at three months are available in the control group. The analysis of functional outcome in the subgroup of 17 patients without a prior diagnosis of DM showed an RR of 0.56(95% CI 0.06–5.09) for good outcome.
Additional information
Several prospective observational studies 54 and post hoc analysis of clinical trials on blood pressure management, like the ATACH, 55 the INTERACT 16 and the SAMURAI-ICH 56 trials, have reported an association between hyperglycaemia and a poor functional outcome in patients with haemorrhagic stroke and one of them has shown a lower risk of poor outcome in patients with declining glucose serum concentrations over time. 55
PICO 7: In acute haemorrhagic stroke patients with hyperglycaemia, does treatment with IV insulin for tight glucose control compared to no treatment/SC insulin improve survival?
Green et al. 32 provided data on survival in acute haemorrhagic stroke patients; however, no differences were found with regard to the allocated group of blood glucose management (RR 0.81; 95%IC 0.40–1.65).
Subgroup analysis
Only eight patients had a prior diagnosis of DM and no differences were found in the comparison of survival between the interventional treatment and the control group (RR 0.50; 95%CI 0.07–3.55). Similarly, no differences were found in the subgroup of 17 patients without a prior diagnosis of DM (RR 0.94; 95%CI 0.46–1.90).
Additional information
Data from prospective observational studies and meta-analysis have clearly shown the relationship between hyperglycaemia and an increased risk of death in patients with primary intracerebral haemorrhage17,54,57–59 as well as in subarachnoid haemorrhage. 60 However, very few clinical trials have analysed the effect of insulin on the survival of patients with haemorrhagic stroke. Some of these studies had to be excluded from our systematic review: one including patients with aneurysmal subarachnoid haemorrhage due to active treatment with IV insulin both in the intervention and in the control group 27 and the GIST trial due to the impossibility to obtain separate data for ischemic and haemorrhagic stroke and data collection not currently available after contacting the authors.28,29 None of these studies were powered enough to detect any impact of insulin on survival, similarly to the study by Green et al. 32 included in this review, with only 25 patients.
PICO 8: In acute haemorrhagic stroke patients with hyperglycaemia, does treatment with IV insulin for tight glucose control compared to no treatment/SC insulin increase risk of any hypoglycaemia?
Hypoglycaemic events were found in only 4 of 25 acute haemorrhagic stroke patients included in the study by Green et al. 32 none of which were symptomatic (RR 3.25; 95%CI 0.39-27.15).
Subgroup analysis
Three patients with prior diagnosis of DM and one without DM diagnosis developed hypoglycaemic events (none symptomatic), representing an RR for hypoglycaemic events of 2.0 (95%CI 0.28–14.20) for patients with prior diagnosis of DM and of 3.33 (95%CI 0.15–71.90) for patients without a history of DM.
PICO 9: In acute haemorrhagic stroke patients with hyperglycaemia, does treatment with IV insulin for tight glucose control compared to no treatment/SC insulin reduce hematoma growth?
Hematoma growth was not evaluated by the only clinical trial included in the systematic review that evaluated the effect of insulin treatment in haemorrhagic stroke. 32
Additional information
Post hoc analyses of clinical trials on blood pressure management have reported data on the effect of glucose levels on infarct growth,16,55,61 with discrepant results. The ATACH investigators showed that a decline in serum glucose concentration correlated with a reduction in the proportion of subjects with hematoma expansion, suggesting that this could be a modifiable factor to decrease hematoma growth. 55 In contrast, the recently published analysis from the INTERACT2 study did not find any difference in hematoma growth in patients with or without hyperglycaemia. 16
PICO 10: In acute haemorrhagic stroke patients with hyperglycaemia, does treatment with IV insulin for tight glucose control compared to no treatment/SC insulin increase the risk of hypokalaemia?
The only study in which the effect of insulin treatment was evaluated in patients suffering from haemorrhagic strokes did not report data on hypokalaemia development. 32
Recommendation
In patients with acute haemorrhagic stroke, we suggest against the routine use of IV insulin to achieve a tight glycaemic control as a means to improve functional outcome or survival.
Additional comments for clinical practice
Summary of Findings (SoF) Table for haemorrhagic stroke.
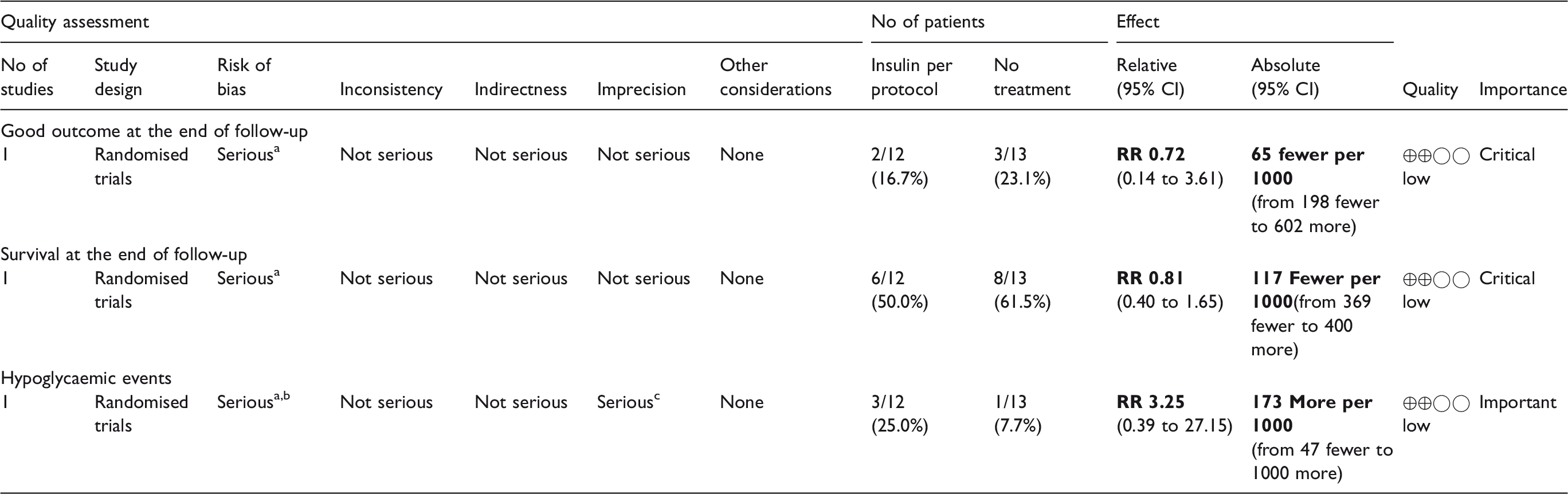
CI: confidence interval; RR: Risk ratio; OR: odds ratio.
Performance bias.
Non-blinded outcome analysis.
Low number of events.
Discussion
Data from clinical trials are hampered by their insufficient statistical power to detect an effect of insulin treatment on outcomes, as well as heterogeneity in trial design, some studies including only patients with hyperglycaemia (with additional heterogeneity in its definition) whilst other also including patients with normal glucose levels. However, as a conclusion of the systematic review and meta-analysis that we performed, no evidence of any benefit of IV insulin titrated to tight glycaemic control was found on outcomes of patients with ischemic or haemorrhagic strokes, and a significantly higher risk of hypoglycaemic events was observed. This fact led the WG to recommend against the use of IV insulin treatment aiming at tight glycaemic control.
One of the critical open questions in glucose management in acute stroke patients is the optimal target glucose to counterbalance its deleterious effect. Some of the clinical trials have chosen the intensive glucose reduction approach,29,30,32–34 but this strategy has been shown to be deleterious, being associated to higher mortality in other populations like in the critically ill patients.21,62 In fact, a recently published network meta-analysis evaluating the optimal target for acute glycaemic control in critically ill patients has shown that target glucose levels of <5.5 mmol/L (100 mg/dL) and 6–7.9 mmol/L (110–144 mg/dL) were associated with a higher risk of hypoglycaemia compared to target levels of 7.9–9.9 mmol/L (144–180 mg/dL). 63 Thus, future clinical trials should be aimed to achieve a ‘physiologic’ normalisation with well-controlled IV insulin infusion rather than intensive reduction of glucose levels and avoiding roller-coaster effects produced by intermittent SC insulin administration that are not currently recommended to manage hyperglycaemia in any inpatient. 19 Future studies should also select only patients with glucose levels higher than the threshold associated with poorer outcomes, that has been suggested to be around 8.6 mmol/L (155 mg/dL). 41
An important concern with the use of IV insulin is the risk of hypoglycaemic events. The development of moderate or severe hypoglycaemia and multiple hypoglycaemic events in critically ill patients have been found to be associated with increased mortality.64,65 In acute stroke patients, glucose levels lower than 3.7 mmol/L (67 mg/dL) within the first 24 h were associated with higher risk or poor functional outcome 42 and it has been suggested that the possible benefit of intensive insulin therapy may be negated by the development of hypoglycaemic episodes. 66 It is known that after a brain injury, there is an increase in metabolic demand and, as glucose is the primary energy substrate for the brain, it is especially vulnerable to glucose deficits. Thus, a tight blood glucose reduction to levels that are considered within the normal ranges in otherwise healthy people could induce brain glycopenia in acute stroke patients and could even trigger a metabolic crisis in the ischemic brain. 67 Thus, for clinical practice, a moderate reduction of glucose levels with close monitoring to avoid hypoglycaemic events might be reasonable as it is currently recommended for hospitalised patients. 19
Another important challenge is the frequent lack of care of hyperglycaemia in acute stroke patients that has been reported in several studies44,68,69 which could be favoured by the lack of strong recommendations on glucose management due to the limitations of the clinical trials as discussed before. However, it would be a serious error to conclude that the lack of evidence of benefit from tight control IV insulin in acute stroke would mean that acute stroke patients should not be treated with IV insulin aiming at non-intensive standard glycaemic targets similarly to other hospitalised patients who develop hyperglycaemia, and taking into consideration the glucose levels thresholds for poor outcomes provided by observational studies.40–42 With this approach, the risk of hypoglycaemia could be minimised. In this sense, it is worrisome that the detailed analysis of the standard management group in clinical trials provided in Table 1 clearly reflects the lack of clear protocols for glucose management as well as the frequent lack of care of hyperglycaemia in acute stroke patients in clinical practice. Further efforts to improve conventional glucose management in acute stroke patients are needed.
One objective of the guidelines for management would be to provide practical recommendations to guide physicians in each patient and although we would like to do that type of recommendations, the lack of evidence of any significant effect on stroke outcome with insulin treatment as a result of the systematic review and meta-analysis performed, as well as the used methodology to formulate the recommendations (based in the GRADE system) impede us to provide any practical evidence-based recommendation. However, knowledge on the brain metabolism of glucose, 67 the pharmacological properties of insulin as well as data from observational studies 48 and the expert-based consensus on in-hospital management of hyperglycaemia in general, 19 could provide us some practical tips regarding the type of insulin, the threshold level to start corrective treatment and the optimal target of glucose levels.
One of the open questions is the optimal target for glucose values, or saying it in another words, when to start corrective treatment. Several observational studies have tried to answer this question and the optimal value could be between 7 and 10 mmol/L (140–180 mg/dL) for the majority of the patients,41,42,70 and a bit lower, around 6 mmol/L (109 mg/dL) for non-diabetic patients. 71 The ongoing Stroke Hyperglycemia Insulin Network Effort (SHINE) trial defines hyperglycemia as glucose levels >6.1 mmol/L (110 mg/dL) in patients with known diabetes and >8.3 mmol/L (150 mg/dL) in those without known DM. 72
As mentioned before, expert-based recommendations suggest as the preferable approach for hyperglycaemia management in critically ill patients the use of IV insulin therapy titrated to achieve a target glucose level between 7.8 and 10.0 mmol/L (141–180 mg/dL), avoiding more intensive targets that can result in a higher risk of hypoglycaemia.19,51 There is also a general recommendation against the use of SC sliding-scale insulin or the so-called correction insulin based on the use of rapid-acting insulin in critically ill patients as the sole regimen as it is not evidence based and ineffective in the majority of the patients.52,53 On the other side, noninsulin agents are also considered as inappropriate in most hospitalised patients. 19
Our systematic review and meta-analysis had several limitations and being the most important is the unavailability of data from the GIST trial which is larger clinical trial on blood glucose management in acute stroke.28,29 This was due to the unfortunate death of the GIST trial statistician who was in care of the study data base and no other investigator to whom we could contact had access to it. Nevertheless, as mentioned before, it is highly likely that the inclusion of the GIST data (if we could have them) would not alter our results and conclusions as the main results of that trial are coincident with those of our systematic review as well as of those from prior meta-analysis66,73 on blood glucose management in acute stroke which included the GIST data despite that up to 16% of the patients had an hemorrhagic stroke.
In conclusion, according to the available evidence, in patients with acute ischemic or haemorrhagic stroke, routine IV insulin titrated to achieve a tight glycaemic control do not have a significant influence on death or in functional outcome but increases the risk of any hypoglycaemia event as well as symptomatic hypoglycaemia. The currently available data about the management of glycaemia in patients with acute stroke are limited and the strengths of the recommendations are therefore weak. Nevertheless, this does not prevent that hyperglycaemia in acute stroke patients could be treated as any other hospitalised patient.
Footnotes
Acknowledgments
We would like to thank to Deborah Green for her kindness in providing us with the different data analysis of ischemic and haemorrhagic stroke.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Not applicable
Informed consent
Not applicable
Guarantor
ED-T
Contributorship
ED-T lead the working group. ED-T, BF, GN, JP and GT were involved in protocol development, literature review, data analysis, writing and approval of the final versión of the manuscript. BT researched literature. The members of the ESO Guidelines Committee approved the final version of the manuscript (Appendix 3).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
