Abstract
Highlights
This formative research presents an innovative infrastructure designed to scale patient decision aids through capacity-building tools and standardized templates.
Stakeholder feedback suggests that the platform could enable more timely and resource-efficient development of decision aids, although its effectiveness has yet to be evaluated.
The platform’s collaborative features could foster partnerships between developers, clinicians, and patients by promoting a shared understanding of the process and making roles and decisions about content more transparent.
To increase confidence in the quality of decision aids, future work should introduce a validation process for decision aids and integrate evidence-synthesis guidance into the platform.

Keywords
Patient decision aids (PtDAs) are evidence-based tools that prepare patients and their family or friend caregivers to actively participate in shared decision making with their health care provider. 1 PtDAs provide information about options and associated benefits/harms and help guide patient decisions by complementing and facilitating counseling by the health care provider.2,3 They have been shown to improve knowledge and lead to more accurate risk perceptions. 1 With the impetus from research funding, as well as national or regional programs and policies, PtDAs are now increasingly in demand among clinicians and health care organizations worldwide in a wide array of clinical contexts. 4 This growing demand is not limited to higher-income countries; there is also pressure to improve access in countries where person-centered care and shared decision making are emerging, such as Mexico and Malaysia. 4 Providing clinicians and policy makers in these contexts the means to develop, adapt, and update PtDAs could significantly support the implementation of shared decision making, 5 which is increasingly recognized as part of value-based health care.6–8
However, the widespread adoption of PtDAs is limited by their availability. From a practical perspective, the current production—primarily led by researchers—is insufficient to meet demand. A key barrier is the specialized expertise required to develop PtDAs that meet the International Patient Decision Aid Standards (IPDAS),2,9 ensuring their quality and effectiveness. In addition, PtDA development is resource intensive and complex, requiring evidence review 10 and the involvement of multiple stakeholders such as researchers, patients, clinicians, and health care managers.11–13 Achieving early stakeholder involvement, despite its benefits in creating ownership and buy-in,14–20 adds significant complexity and demands to the PtDA development process, requiring multiple cycles of evaluation and feedback between users and developers in a user-centered approach,20,21 which also requires substantial resources. Financial costs further constrain PtDA development. Users increasingly expect interactive, Web-based PtDAs,13,22 which are more expensive to develop, especially when considering recommended user testing. 23 Moreover, ongoing resources are required to keep PtDAs up to date. 24
Beyond these significant practical challenges, the widespread adoption and successful integration of PtDAs into routine clinical practice also fundamentally depend on understanding the complex social and organizational processes through which new innovations become embedded within existing contexts. This often-overlooked dynamic, termed
Efforts to scale-up PtDA production are often not well documented. Some scientific articles and PtDA directories highlight active PtDA developers,27–30 mostly academic researchers who often lack the resources to update their PtDAs postresearch. Government agencies, professional organizations, and public bodies also develop PtDAs, but in limited numbers, and they are subject to policy changes. There are also professional PtDA developers who aim to scale and sustain PtDA development through revenue generation, either nonprofit or for-profit. For example, EBSCO offers subscription-based PtDAs with DynaMed evidence summaries, and MAGICapp offers features for developing PtDAs based on clinical practice guidelines. Healthwise offers numerous free interactive PtDAs, although its business model is unclear. These developers typically operate centralized PtDA development services funded by subscription fees from health care organizations, clinicians, or patients. However, the business models are not always transparent on their Web sites.
To address these multifaceted barriers—including the need for specialized expertise, high resource intensity, the limitations of centralized and often unsustainable development models, and unclear business models—we set out to develop an innovative infrastructure: a digital platform called PADA (A Collaborative Platform for Authors of Healthcare Decision Aids; Giguere). This platform promotes the decentralization of PtDA development by building capacity and offering a validated generic template, thereby optimizing resources and overcoming key obstacles to widespread PtDA availability.
This study aimed to develop and optimize a mock-up of the platform with key stakeholders and to identify factors that could influence its adoption by knowledge users in order to ultimately support its future implementation. We drew on the NPT to describe these factors and guide the iterative optimization of this innovation in collaboration with end users and other stakeholders.
Methods
Study Design
We used a people-centered and collaborative approach to design an alpha version of the platform. We then optimized it in an improved beta version by conducting individual interviews with key stakeholders to explore their perceptions of the platform in a qualitative, descriptive study design.
This study is part of a larger project that also examined participants’ perceptions of the PtDA template generated through the platform (manuscript currently in preparation). We prepared this report in compliance with COREQ guidelines. 31 All participants signed an information and consent form.
Innovation Design
We structured the design of the platform using the Design Council’s Framework for Innovation, 32 a recent revision of the Double Diamond model from 2004, which promotes a commitment to innovation from a people-centered and collaborative perspective and comprises 4 phases: Discover and Define (in a challenge-oriented diamond) and Develop and Deliver (in an outcome-oriented diamond) (Figure 1).

Adaptation of the Framework for Innovation 32 to guide the iterative design of the platform by the Design and Development team through people-centered engagement of key stakeholders.
A core design and development team led the iterative design process, which was supported by input from an advisory panel. The core design and development team consisted of the project leader, who is a content expert (A.G.); a computer analyst (P.F.); a user interface/user experience designer (E.B.); and the first author, a public health student (J.B.). The advisory panel consisted of potential end users (C.B., P.B., K.S., F.L., B.D.T., D.S., K.H.), patient representatives (J.L., K.P.), clinicians (P.B., F.L., K.S., D.S., S.E.S., B.V.), a technical expert (P.D.), and implementation experts (E.D., A.G., F.L., D.S., S.E.S.). Several team members are part of the IPDAS collaboration (A.G., C.B., F.L., D.S.).
Discover and Define
In keeping with the Design Council Framework’s “Discover” phase, we began by articulating the need for a scalable PtDA development platform. The project leader’s (A.G.’s) first-hand experience (more than 30 PtDAs created) and insights from her research network highlighted a recurring demand from government agencies, practice-based research networks, and academic teams for timely, high-quality PtDA development. These requests often exceeded the scope of ad hoc academic collaboration.
We then conducted a comprehensive analysis of existing PtDA development initiatives, as described in the introduction. This analysis included academic, government, and commercial offerings (e.g., DynaMed, MAGICapp, Healthwise, The Ottawa Hospital Research Institute) and confirmed that there is a significant unmet need in this area.
Then, as per the “Define” phase of the framework—informed by the identified unmet needs, theoretical frameworks,11,33 and the evidence-based IPDAS criteria 9 —the design and development team specified the core user needs and challenges that the platform sought to address. This stage also involved defining the platform’s core requirements, including key functionalities and users’ stories.
This preliminary description of the core requirements was subsequently reviewed by members of the advisory panel, who were invited to provide written feedback in a shared online document, enabling them to build on one another’s perspectives.
Develop and Deliver
Based on the refined description of the platform obtained through the Discover and Define phases, the design and development team (E.B.) developed a high-fidelity mock-up of the platform using Figma (version 11, Figma Inc., San Francisco). This mock-up was a detailed visual representation that provided users with a clear conceptualization of the future platform’s interface and user experience, without requiring a fully functional prototype. The mock-up was meant to serve as a critical formative assessment tool, allowing for early and cost-effective feedback on the proposed design before significant development resources were committed.
Members of the advisory panel subsequently reviewed this alpha mock-up, providing feedback either individually or in a group setting. Their collective input led to the development of an improved mock-up version.
We then used a systematic and iterative approach to improve the mock-up based on key stakeholders’ assessments of the platform, as described in the next sections.
Recruitment
Through the team’s professional networks, we recruited a convenient maximum-variation sample of key stakeholders in PtDA development, striving to diversify their continent of residence, native language, and gender. We defined a key stakeholder as an individual who had participated or planned to participate in the development of a PtDA, for example, as a project manager, scientific reviewer, patient or clinical expert, or health care manager, or in a more specialized role (e.g., biostatistician, translator, graphic designer, or health literacy expert). To participate, interested stakeholders had to be at least 18 y old; speak fluent English, French, or Spanish; be able to read English; and have access to a computer with a camera and a stable Internet connection. People who had a hierarchical relationship with any member of the research team were ineligible.
Invitations were made through French-, Spanish-, and English-speaking shared decision-making networks and the Facebook page of the Shared-Decision-Making Society. We sought to recruit a sample of 24 participants, or until saturation, which we defined as the point at which no new main theme emerges. 34
Data Collection
The data collection and analysis, as well as the optimization of the platform, were based on the NPT as recommended by the authors and users of the theory.25,35,36 The application of NPT to the project can be found in Supplementary Material 1.
Upon recruitment, participants completed an online survey to describe their sociodemographic characteristics and experience in PtDA development.
Participants were asked for feedback on the mock-up in individual, think-aloud interviews 37 via videoconference, which lasted approximately 1 h and were video-recorded and transcribed verbatim. We showed participants the PtDA template created with the platform at the beginning of the interview so that they could understand its purpose. Participants were then asked to complete specific tasks related to capturing and revising the text of a PtDA using the mock-up while sharing their screen. The moderator encouraged participants to say aloud what they were thinking during the process, answered participants’ questions, and noted when they needed help. This strategy allowed for user feedback on usability (e.g., navigation, terminology).
After completing the different tasks, participants were asked a series of open-ended questions to assess their perception of factors that might affect the uptake of the platform. These questions followed a semi-structured interview guide based on NPT, 26 which was pilot tested in our research team (Supplementary Material 2). A logbook was kept throughout the study to document observations that were not recorded.
The first author (J.B.) conducted the interviews and had no relationship with the participants prior to the study. She was new to the field of shared decision making and had no assumptions about what the participants’ responses would be. She conducted this work as part of her graduate training in research and public health.
Between interviews, the issues raised were discussed among team members to find solutions to improve the mock-up. The number of interviews conducted before integrating changes into the mock-up depended on the level of importance of the issues raised, reflecting the team’s previous experiences, resources, and literature on user-centered design.38,39 This user-centered design process continued until we reached saturation, as defined above.
Data Analysis
Three researchers conducted mixed deductive–inductive thematic qualitative analysis of the recording transcripts 40 using NVivo 41 : a master’s student in public health (J.B.), a bachelor’s student in psychology (L.Z.M.), and a researcher (PhD) in public health (A.G.). We used a deductive approach initially by predefining nodes for each of the 4 NPT constructs (Coherence, Cognitive Participation, Collective Action, Reflexive Monitoring). 25 Under each construct, we also created nodes for its subcomponents (e.g., Enrollment, Legitimation, Activation under Cognitive Participation), forming a parent–child node structure in NVivo. As we analyzed transcripts, we inductively created additional child nodes—representing emergent subthemes—nested under the relevant component node. By organizing nodes into parent (construct) and child (component or subtheme) relationships, we captured both theory-driven and emergent insights.
Usability issues surfaced through moderator-guided think-aloud tasks (e.g., navigation confusion, unclear labels, interruptions) and were inductively coded under Collective Action, namely, under the Interactional Workability component within this construct, aligning real-time user-centered feedback with the work to operationalize PtDA creation using the platform.
The 3 researchers first independently analyzed part of a first interview and established consensus on some subthemes. Then, they analyzed a second part of an interview based on this consensus, met again, and so on until they had reached an acceptable understanding of the research objectives and themes. After this initial analysis, 1 person (J.B.) analyzed each of the remaining interviews and a second person reviewed the subthemes and corresponding transcripts (L.Z.M.). Any subthemes that emerged were reviewed and agreed on by consensus between the 3 researchers.
Results
Discover and Define Findings
In the initial Discover and Define stages, we identified 3 core challenges faced by developers of PtDAs: 1) ensuring that PtDAs meet the rigorous quality standards outlined by IPDAS, in order to be both useful to patients and work as intended 9 ; 2) actively engaging a diverse group of stakeholders—including patients, clinicians, and decision makers—through a structured, iterative process to incorporate feedback and enhance the relevance and adoption of the PtDA 21 ; and 3) offering interactive PtDAs online, which often requires significant technical and financial resources that may not be readily available in all settings.
Our landscape analysis of existing PtDA development initiatives highlighted a lack of transparent, flexible, and broadly accessible tools that empower diverse stakeholders to develop high-quality, sustainable PtDAs. Specifically, while some platforms offered structured evidence-based content or evidence summaries, they often lacked robust features for multistakeholder collaboration, flexible content development beyond clinical guidelines, and transparent business models for sustained operations.
Supplementary Material 3 describes the detailed requirement specifications, personas, and user stories that were developed during these formative phases and subsequently used to create the mock-up.
The Platform
The platform is an interactive and user-friendly online environment designed to support the systematic development of PtDAs. It integrates previously validated printable and Web-based templates that fulfill the IPDAS criteria.42,43 A description of the platform is available on the PADA Web site. 44
The platform is structured around 3 main modules (see Table 1 for a detailed list of all functionalities): the Edition module guides authors through the content development process for their PtDA, the Revision module facilitates collaborative work among an unlimited number of stakeholders on the same PtDA, and the Consultation module allows end users (clinicians or patients) to view the developed PtDAs. Notably, the Revision module was conceived to meet one of the IPDAS criteria of the development domain (i.e., potential users were involved in designing, developing, and/or refining a prototype). 45 In addition, one feature of the Edition module was specifically focused on empowering PtDA authors to fulfil IPDAS criteria in their development processes, namely, “Checking the PtDA content against IPDAS criteria and health literacy design principles” (Table 1).
Description of the Different Modules, Intended Users, and Main Functionalities of the Platform a
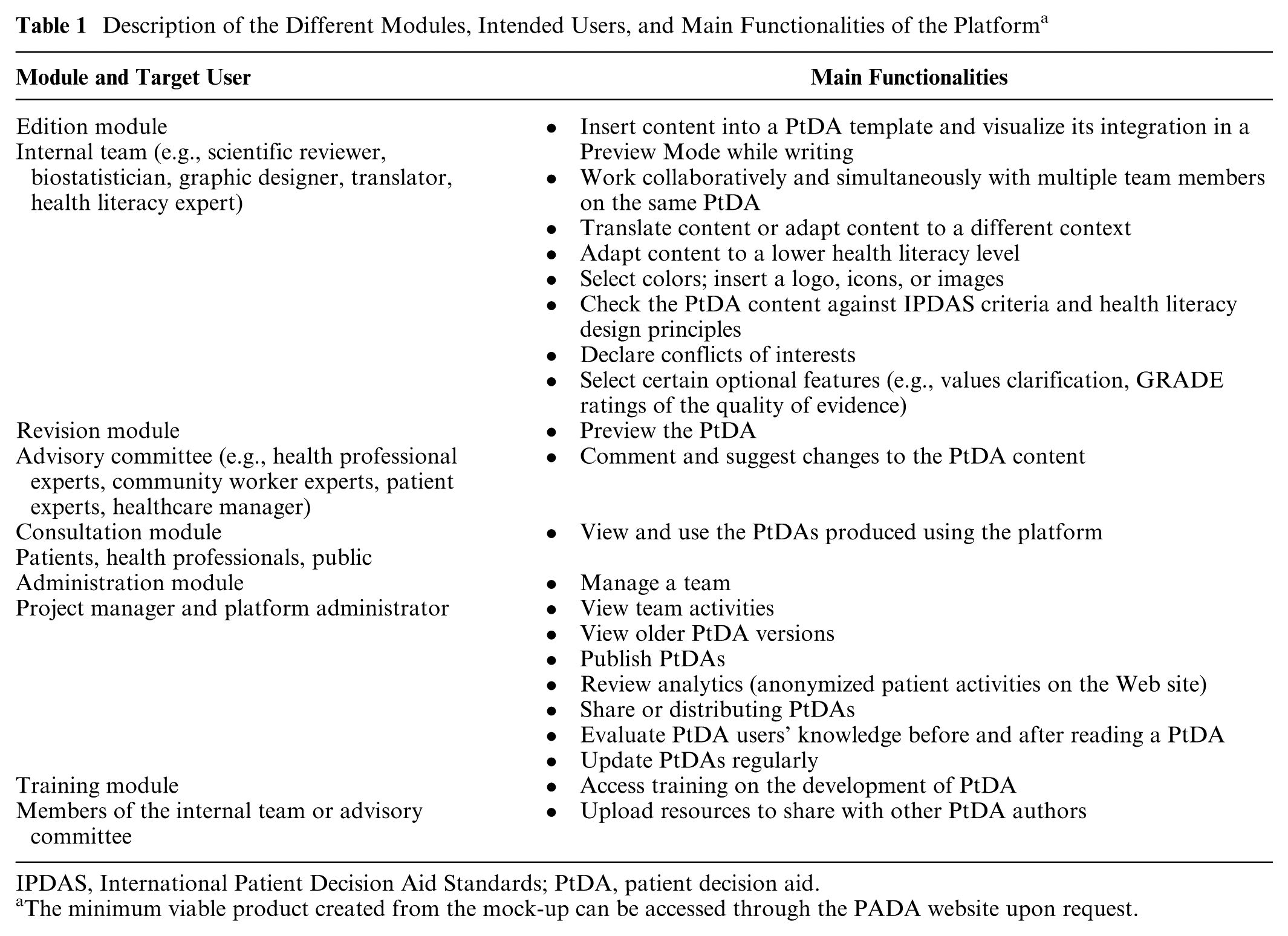
IPDAS, International Patient Decision Aid Standards; PtDA, patient decision aid.
The minimum viable product created from the mock-up can be accessed through the PADA website upon request.
The proposed business model to sustain the platform involves authors paying for licenses to create freely accessible PtDAs.
Similar to MAGICapp, the platform will provide resources for structuring evidence-based tools but with a distinct emphasis on user-centered design by offering dedicated revision features for input from advisory committee members and end users. Unlike MAGICapp, the content of the PtDAs produced will not be tied to clinical guidelines, allowing for formats that deviate from traditional recommendation-based messaging.
Develop and Deliver Findings
During the user reviews of the alpha mock-up version, the advisory panel and subsequent interview participants provided valuable feedback.
We significantly modified the mock-up on 3 occasions: after the advisory panel review, after 8 participant interviews, and then at the end, after all 20 interviews were completed. Several minor modifications were also made to the mock-up over the course of the process. These modifications are listed in Supplementary Material 4.
Participant Characteristics
We interviewed 20 participants including researchers, clinician–researchers, patient experts, and health care managers (Table 2). Thirteen interviews were conducted in English and 7 in French. Fifteen participants lived in high-income countries and 5 in upper-middle-income countries, as defined by the World Bank. 46 While most participants had developed 2 to 5 PtDAs, their experience ranged from 0 to 80 PtDAs developed. The 2 participants who had never developed a PtDA were planning to develop one.
Characteristics of Study Participants

Factors Influencing the Adoption of the Platform
We reported our findings according to each construct, dividing them among positive and negative perceptions. The most striking results are presented in the text below, and more information is available in Tables 3 to 6.
Participants’ Positive (+) and Negative (−) Perceptions of the Platform Relative to the Coherence Construct

PtDA, patient decision aid.
Participants’ Positive (+) and Negative (−) Perceptions of the Platform Relative to the Cognitive Participation Construct

PtDA, patient decision aid.
Participants’ Positive (+) and Negative (−) Perceptions of the Platform Relative to the Collective Action Construct

PtDA, patient decision aid.
Participants’ Positive (+) and Negative (−) perceptions of the Platform Relative to the Reflexive Monitoring Construct

PtDA, decision aid; SDM, shared decision making.
Coherence
Under the Coherence theme, we have included participants’ comments on how they understand the platform and what it would mean for them to use it to develop PtDAs (Table 3).
Participants clearly understood the unique features of the platform, for example, for collaboration between authors and for revising the PtDA in a “preview” mode at any point during its development.
They found the PtDA templates useful, and some even considered them to be the biggest advantage of the platform, as they are evidence based and save time. One of the patient participants thought that it could be limiting to offer only a few PtDA templates and suggested that several should be offered to better meet needs, for example, by organizing the information differently.
Participants also appreciated the fact that the platform worked similarly to other text-editing applications they were already using, such as Google Docs™ or Microsoft Word™, creating a sense of familiarity.
One participant mentioned that he felt the platform offered something different from the way he currently worked:
What we did with our decision aids is we developed a content management system, as you did. But our content management is only developed to show the content as it is [without formatting], but it’s not to create certain sections or certain tools [complete PtDAs]. So, I think, this one is much more interactive and much more . . . flexible than ours is. (EN-06, researcher with >10 PtDAs)
Participants felt that the review features of the platform would promote a shared understanding of the aims and expected benefits of the platform, particularly for patient experts.
Participants also considered the significance of the platform for their individual work as authors. Many found the platform helpful, such as this participant:
I think that it will make our work much easier, especially if you had features to present icon arrays; it will make my life easier because it is really difficult sometimes to talk to the designer, what to do, what you want, what do you need. So, I think if you can do it by yourself, it will be really nice. (EN-05, researcher with 2–5 PtDAs)
Participants appreciated the practicality of the platform and felt that it could be integrated into their routine practice. However, some mentioned that some of the features require familiarization.
They also felt that the platform would help in the long run to create high-quality Web-based PtDAs, even with limited resources:
So, compared to those I have developed, I started from scratch, I designed it, they look very nice, but at the end of developing, I’m so tired because it took so much time. It looks good, but it is not sustainable. . . . So, this kind of multiple templates, though it might not look so . . . as fancy or as tailored, it achieves its purpose of developing a PDA very quickly and effectively. (EN-04, clinician with 5 PtDAs)
One participant suggested adding a step in which the content of the PtDA is written in a separate document before being transferred to the platform.
Cognitive Participation
The Cognitive Participation theme reflects participants’ views on how the platform supports PtDA developers in building and maintaining a community of practice around its use.
The initial reaction of many participants to the platform was that they would be willing to push for its use. Several of them expressed interest in using the platform as soon as it is available.
It would be nice if, once you have completed this project, we could use the platform. (EN-03, clinician with 2 PtDAs)
Participants felt that they could rely on the available Web-based and printable PtDA templates (Table 4).
While participants expected that offering PtDAs on the Web sites of renowned institutions would help to better reach patients, they were also concerned that the PtDAs would not meet their government’s requirements to be published on a national platform. Participants also mentioned that the platform should enable the promotion of their PtDAs through reputable institutions, suggesting that the publication and distribution processes should consider the needs of individual institutions (e.g., governments, universities, and hospitals).
One participant was concerned that requiring mandatory training for authors might hinder the platform’s adoption. Referring to an existing online training program for PtDA developers,
47
they questioned whether such an approach would be appropriate:
In Ottawa, there is a kind of credit course, a tutorial. At the end you’re asked a lot of questions, and you get a certificate, a kind of diploma. Should we do the same? But would that slow down the use [of the platform]? I don’t know, but maybe it’s a recommendation. I don’t know. (FR-14, health care manager with 1 PtDA)
Participants also felt that the structure offered by the platform would support collective work and user participation:
So, I click on “Aspirin for cardiovascular disease,” and there’s a paragraph that tells me what stage the project is at. If I have a concern, I can ask, “Please add the finance director from such-and-such Integrated Care and Social Services Center as a collaborator, because he had to make decisions and present this to the board of directors, which wasn’t easy.” Then, I have space to write all the points that concern me to move the project forward. The platform allows for all these dimensions. (FR-13, patient without PtDA)
Some participants provided insights into the value of the platform for all types of users:
I think it will be useful for different types of authors, for the first-time author or even the experienced author who has developed many decision aids. . . For a first-time author, it is more to guide the author to follow important elements in decision aids and to follow the steps. (EN-11, clinician–researcher with 4 PtDAs)
Some participants also noted that less experienced authors and health managers would benefit most from it:
For example, if I were a doctor, a pediatrician, and I would really like to do a decision aid for my patients, but I don’t have the resources, I don’t have time to create a whole Web site, I would be very happy to use it. (EN-06, researcher with >10 PtDAs)
In contrast, some participants felt that authors with more experience in developing PtDAs might be reluctant to adopt the platform because they are already using another PtDA template:
All of our decisions aids are in the same font and in the same colors and the pictures are similar and so on and so on. Of course, if you asked me: Would you like to change all your decision aids to our template? I’m afraid I wouldn’t. (EN-06, researcher with >10 PtDAs)
In general, participants appreciated the fact that the platform would support input from all types of stakeholders. They felt that the platform would facilitate collaboration with patient experts, organizations, and health managers:
I’m happy to see that this [the platform’s way of working on PtDA content] allows for more details, it is more customizable for both [collaborators], or all three, or however many partners there are. A caregiver partner can then add the things that are important for them. (EN-02, patient with 3 PtDAs)
Participants also saw the platform as a way to facilitate PtDA updates and keep them relevant, which they described as an ongoing concern:
Moderator: “What is different in this platform from conventional methods to develop decision aids?” Participant: “I think there’s a certain plasticity, so it’s very plastic. It feels like you can play around with different things before it gets launched. It also means that the . . . it kind of removes the fear of . . . in the past, when we apply for funding for decision aid, there’s always that: “who’s going to do the updating? We’re going to print these booklets, how long are these booklets going to be useful for?” This one, it seems that there’s more longevity for the decision aid, bringing the promise that the decision aid could go on for a much longer time than just showing it once. That’s my feeling. I think it was quite positive, actually. (EN-12, researcher with 6 PtDAs)
Some participants were ready to promote the platform to their collaborators, teams, or research centers. In contrast, others mentioned specific features to be added beforehand, such as the possibility of adding logos to PtDAs or having access to several PtDA templates.
Collective Action and Usability
The Collective Action theme summarizes participant views on operationalizing the processes to create PtDAs using the platform, such as tasks, resources, and interactions between the authors. We have integrated all comments on the usability of the platform here.
Overall, participants appreciated the fact that the platform’s revision features allow them to work with an advisory committee, which was highlighted by this participant:
Because it really enables, especially nonexperts, to say: “Okay, I’m reading this and I have this reaction,” and so it is real world, representing what a user would feel about it. (EN-20, researcher with > 10 PtDAs)
In addition, some participants felt that the platform’s features could support all types of stakeholders by enabling them to work autonomously and efficiently and to share their complementary expertise (Table 5).
Several participants mentioned that it would be valuable if the platform offered the possibility to develop PtDAs in several languages, and these comments touched on the NPT themes (Tables 3–5). Under the Collective Action theme, some explained that having multiple language options would improve access to PtDA by patients in different contexts. Some participants also mentioned the importance of being able to work in several languages, and one of them felt that there should be an automatic translation option.
Several participants saw it as a strength that the platform could support the training of authors and ensure the quality of PtDAs. For instance, one individual pointed out that some authors may not be comfortable with synthesizing and presenting evidence:
Do you have anything, like some guidelines, about how to get the evidence or results from studies into the decision aid? Because that’s the most important, and also the most difficult thing, with all decision aids. (EN-22, clinician–researcher with 4 PtDAs)
While most participants felt that the platform’s revision features would enhance collaboration, additional features such as version tracking and sharing of reference materials were also suggested to enhance collaboration. Some also suggested that adding functionality to replicate or adapt existing PtDAs while tracking authorship would benefit collaborative work, and some recommended that the platform provide professional coaching and counseling.
Participants mentioned that the lack of resources (e.g., skills, funds, and time) would be an incentive to use the platform, but it could also limit the quality of the PtDAs produced.
Several participants felt that the costs to access the platform and create the PtDA might pose a barrier:
With a template like this, we can just put in our content. I think that will be very good. I’m just wondering whether you would allow a user to use it for free. (EN-11, clinician–researcher with 4 PtDAs)
Lastly, several participants appreciated the flexibility of the platform—in terms of languages, institutions, formats, and populations—to adapt the PtDAs to their different contexts; however, some pointed out certain challenges in implementing PtDAs in electronic medical records.
Usability
Several minor usability issues were identified, such as unclear navigation, low visibility of key features (e.g., Infotips, preview), and misunderstandings regarding the required content. These issues were relatively easy to resolve. A detailed summary of the factors influencing the usability of the platform can be found in Supplementary Material 5.
Overall, the platform was perceived as user friendly. By the last round of interviews, adjustments had been made based on participant feedback, and the features and navigation were considered intuitive. For example, participants appreciated the built-in “Infotips” that provided clear instructions for completing each section. They also unanimously praised the planned feature displaying PtDA’s readability level.
In previous rounds, some participants expressed a desire for the platform to support inexperienced authors in complying with IPDAS standards when creating content. In response, we added a checklist that allows authors to self-assess their PtDA against the IPDAS standards, similar to the approach used in the Ottawa Decision Aid Library Inventory. 48 Participants responded favorably to this feature in subsequent rounds.
Most of the negative feedback related to missing elements. In particular, participants wanted greater flexibility in the inclusion of images, videos, graphics, testimonials, and icon arrays throughout all sections of the PtDA.
Reflexive Monitoring
The fourth construct, Reflexive Monitoring, summarizes participants’ insights into monitoring and evaluating the effects of the platform on PtDA development processes and patient outcomes (Table 6).
One participant felt that the platform could improve PtDA development and help disseminate trustworthy and evidence-based PtDAs:
I think it has . . . the potential to have a significant impact on the creation of decision aids. (FR-14, health care manager with 1 PtDA)
Some participants mentioned that offering certification of the PtDA could help build trust in the platform and the resulting decision aids. Some also felt that trust in the platform could be improved if the PtDAs were reviewed by qualified reviewers before publication.
The ability of the platform to provide detailed login/access information (e.g., patient usage of PtDAs) was considered valuable, as long as user data management remains transparent.
Participants also saw the platform as beneficial for teams lacking full access to the necessary expertise:
It saves time; it saves resources. For example, we didn’t have a graphic designer, so we had to hire someone from outside the institution, and that took a lot of time. (FR-03, clinician with 3 PtDAs)
Lastly, the participants said they could imagine adopting the platform in the long term. One patient expert felt that the platform could support successful collaboration between researchers and patients by guiding the patients on the types of input researchers want:
So, sometimes questions with the best of intentions to determine values and beliefs resonate differently with us [patients]. So, these are the things that I look for: the way you’re putting things forward, how are we going to hear it? How are we going to interpret them? So, this gives me great confidence. (EN-02, patient with 3 PtDAs)
Saturation
We estimated that we reached data saturation as there were no new themes created in the last 4 interviews. 34
Discussion
In seeking to understand the factors that influence the uptake of a collaborative platform for the development of PtDAs, we have identified 5 key determinants of success: that scaling PtDA development requires adaptive platforms that offer a range of flexible templates, that meaningful co-design depends on interactive feedback modalities, that global implementation requires a focus on guided cultural adaptation to ensure equity, that a platform must function as a sociotechnical ecosystem that integrates training and quality assurance, and that such platforms can serve to democratize PtDA development by bridging resource gaps. Each of these determinants is discussed below.
Participants appreciated the platform’s evidence-based PtDA templates because they ensure adherence to IPDAS and improve development efficiency. Converging with this finding, a recent systematic review suggested that the use of templates for PtDA creation could reduce the need for iterations and extensive revisions, thus facilitating PtDA scaling. 21 In addition, PtDA templates have been successfully used in several large-scale implementation studies.17,49,50 However, our study reveals a critical insight for scalability in practice. We found that a “one-size-fits-all” approach is insufficient; participants emphasized that, to be useful, the platform must function as an adaptive system by offering a range of flexible and diverse PtDA templates suited for different clinical contexts. This adaptability is essential to allow the customization needed to support successful scaling of shared decision making across varied settings.
Our research highlights a significant flaw in the traditional PtDA co-design process, which often relies on stakeholders commenting on text-heavy Word documents. Participants believed the platform’s features to preview and comment on a fully designed PtDA would foster a shared understanding of the PtDA development process among users and allow for more valuable feedback, especially from inexperienced authors. People understand information better when it incorporates colors,51,52 a clear layout,51–54 images or graphics,51,55 and paragraphs with white space.52,53 Moreover, a previous large-scale PtDA implementation study found that key stakeholders’ understanding of their respective roles is crucial for participatory design. 56 Participants also valued the fact that the platform enabled iterative and simultaneous revisions by multiple stakeholders, including patients, clinicians, health managers, and designers, as this enhanced collaboration would support more meaningful interactions and result in PtDAs that are better aligned with user needs. A previous study on user-centered design for the development of mobile health services has shown that collaboration between designers and stakeholders indeed improves product usability. 23
Participants identified several factors influencing the platform’s successful implementation, emphasizing the need for flexibility in branding, hosting, and PtDA costs. They also discussed the need for embedding PtDAs in electronic medical records or integrating them into government programs, which have been identified as key to PtDA implementation in previous studies.57–59 Several participants also appreciated the platform’s translation potential to meet the needs of patients in different contexts. Although translation could enable a wider range of PtDAs at a lower cost, 27 it must take into account not only language differences but also cultural norms and health-literacy variations. A recent report from Malaysia, for example, highlighted the challenges of developing and implementing PtDAs due to the country’s multiethnic population (Malay, Chinese, and Indian communities), different religious beliefs about health, and the influential role of spouses and family members in medical decision making. 60 Relying solely on machine translation and automated literacy assessments could therefore overlook these nuances and even exacerbate social inequalities. 61 To mitigate these risks, future platform development must include not only translation capabilities but also educational support and guidance to promote evidence-based cultural adaptation processes as described in the PACE framework. 62 Automated literacy assessment also does not ensure true accessibility, even if more comprehensive automated tools are developed. 63
Our study shows that a development tool alone is insufficient to ensure user confidence and adoption; instead, the platform must function as a sociotechnical ecosystem that integrates training, coaching, and quality assurance pathways. Participants expressed the need for training on evidence synthesis and risk communication, echoing a systematic review that concluded that scaling strategies are successful when supported by both infrastructure and human resources. 64 They emphasized that PtDAs must reflect the best available research 65 and be communicated clearly 10 —expectations that training modules or coaching could help meet by guiding the evidence selection, synthesis, and presentation, following best-practice guidelines. 66 Training could include links to existing programs focused on PtDA development.48,67 From an NPT perspective, such training or coaching could strengthen user confidence in the quality of PtDAs and their impact on patient outcomes. The platform could also support evidence transparency, by requiring developers to include a literature synthesis report with each PtDA, which can be downloadable as a PDF so that users can easily access the supporting evidence. Participants also mentioned that confidence would increase if qualified reviewers assessed PtDAs prior to publication, which is consistent with concerns that accelerating PtDA development could reduce formal academic assessment. 68 Current PtDA certification initiatives in the United States could serve as inspiration.69,70 As the platform is aimed at researchers, among others, it could also facilitate compliance with the SUNDAE reporting guidelines, which aim to improve the quality of publications evaluating PtDAs. 71 To facilitate compliance, the platform could offer a “SUNDAE Wizard” that prompts developers to complete each item on the checklist. Overall, these features position the platform not only as a production tool but also as a structured system that fosters capacity building and the development of trustworthy, high-quality PtDAs.
Finally, our work identifies the platform’s potential to democratize PtDA development and promote global health equity. Participants felt that while all PtDA authors could benefit from the platform’s community of practice, it was most critical for those with less experience or fewer resources. Lack of experience may limit an author’s ability to meet quality criteria, 2 theoretically assess user needs, 33 and take a systematic, iterative approach to soliciting user feedback.13,21 The financial and human resources required13,39 may also burden the occasional or inexperienced author. The platform could efficiently guide these authors while reducing development costs. In so doing, the collaborative digital platform could strategically address the global imbalance in shared decision-making implementation, 4 thereby positioning itself as a key policy tool for building capacity and increasing the availability of relevant PtDAs in settings where they are needed most.
Limitations
We assembled a relatively diverse sample in terms of gender and stakeholder type, but with limited diversity in language and country types, which limits the transferability of our findings. Our difficulties in recruiting authors from middle- and low-income countries reflect current challenges in implementing shared decision making globally. 4 For example, shared decision making in Latin American countries often does not include PtDAs,4,72 and this impeded our recruitment efforts there. We did not recruit participants who were unaware of shared decision making; however, some of the participants had not yet developed PtDAs. Therefore, our results may not apply to people who are not aware of shared decision making at all.
Another limitation is that we did not formally evaluate each user suggestion against the IPDAS criteria. Instead, we used the IPDAS standards as a nonnegotiable filter throughout the development of the platform and included only usability or engagement improvements that maintained the core, evidence-based principles.
Conclusions
In this formative study, we identified user requirements and initial perceptions of a PtDA development platform designed to streamline capacity building and collaboration. Stakeholders indicated that translation support, template libraries, and embedded guidance could address common resource and coordination challenges. Next steps include creating a beta version of the platform, potentially as a minimum viable product; usability testing with early adopters in their own environments, to identify any remaining issues in practice; then evaluating the effect on development efficiency and—ultimately—patient engagement and outcomes. By grounding future work in rigorous evaluation, we hope to determine whether this platform can meaningfully advance the scalability and implementation of high-quality PtDAs in routine care.
Supplemental Material
sj-docx-1-mpp-10.1177_23814683251409185 – Supplemental material for Factors Influencing the Adoption of a Patient Decision Aid Development Platform: A Qualitative Study
Supplemental material, sj-docx-1-mpp-10.1177_23814683251409185 for Factors Influencing the Adoption of a Patient Decision Aid Development Platform: A Qualitative Study by Julie Bélanger, Carissa Bonner, Paulina Bravo, émilie Dionne, Katherine Hastings, France Légaré, Karina Prévost, Kevin Selby, Dawn Stacey, Sharon E. Straus, Brett D. Thombs and Anik Giguere in MDM Policy & Practice
Supplemental Material
sj-pdf-2-mpp-10.1177_23814683251409185 – Supplemental material for Factors Influencing the Adoption of a Patient Decision Aid Development Platform: A Qualitative Study
Supplemental material, sj-pdf-2-mpp-10.1177_23814683251409185 for Factors Influencing the Adoption of a Patient Decision Aid Development Platform: A Qualitative Study by Julie Bélanger, Carissa Bonner, Paulina Bravo, émilie Dionne, Katherine Hastings, France Légaré, Karina Prévost, Kevin Selby, Dawn Stacey, Sharon E. Straus, Brett D. Thombs and Anik Giguere in MDM Policy & Practice
Supplemental Material
sj-docx-3-mpp-10.1177_23814683251409185 – Supplemental material for Factors Influencing the Adoption of a Patient Decision Aid Development Platform: A Qualitative Study
Supplemental material, sj-docx-3-mpp-10.1177_23814683251409185 for Factors Influencing the Adoption of a Patient Decision Aid Development Platform: A Qualitative Study by Julie Bélanger, Carissa Bonner, Paulina Bravo, émilie Dionne, Katherine Hastings, France Légaré, Karina Prévost, Kevin Selby, Dawn Stacey, Sharon E. Straus, Brett D. Thombs and Anik Giguere in MDM Policy & Practice
Supplemental Material
sj-docx-4-mpp-10.1177_23814683251409185 – Supplemental material for Factors Influencing the Adoption of a Patient Decision Aid Development Platform: A Qualitative Study
Supplemental material, sj-docx-4-mpp-10.1177_23814683251409185 for Factors Influencing the Adoption of a Patient Decision Aid Development Platform: A Qualitative Study by Julie Bélanger, Carissa Bonner, Paulina Bravo, émilie Dionne, Katherine Hastings, France Légaré, Karina Prévost, Kevin Selby, Dawn Stacey, Sharon E. Straus, Brett D. Thombs and Anik Giguere in MDM Policy & Practice
Supplemental Material
sj-docx-5-mpp-10.1177_23814683251409185 – Supplemental material for Factors Influencing the Adoption of a Patient Decision Aid Development Platform: A Qualitative Study
Supplemental material, sj-docx-5-mpp-10.1177_23814683251409185 for Factors Influencing the Adoption of a Patient Decision Aid Development Platform: A Qualitative Study by Julie Bélanger, Carissa Bonner, Paulina Bravo, émilie Dionne, Katherine Hastings, France Légaré, Karina Prévost, Kevin Selby, Dawn Stacey, Sharon E. Straus, Brett D. Thombs and Anik Giguere in MDM Policy & Practice
Footnotes
Acknowledgements
We would like to thank Philipe Fekete (P.F.) and Émilie Beaulieu (E.B.) from the Web agency Toumoro for their collaboration in the development of the PADA mock-up and Philippe Després (P.D.) and Jean Légaré (J.L.) for their support in planning this study. We also thank Anne-Gabrielle Ouellet for transcribing the interviews, Lizette Zamora Martinez (L.Z.M.) for transcriptions and qualitative analysis and Beatriz Valera (B.V.) for translating the research documents into Spanish and assisting with data collection.
This work was presented at the International Shared Decision Making Conference in Kolding, Denmark, in June 2022 and at the meeting of the Association Canadienne Française pour l’avancement des Savoirs (ACFAS) in Québec City, Canada, in May 2022.
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article. The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The development of PADA was funded by the Unité de Soutien SSA Québec. JB also received scholarships from the Québec Excellence Centre on Aging and the VITAM Research Center on Sustainable Health.
Ethical Considerations
The study protocol was approved by the sectoral ethics committee for research in health sciences at Université Laval (approval No. 2021-161/06-07-2021).
Consent to Participate
All participants signed an information and consent form. Consent to participate was written.
Consent for Publication
Not applicable.
Data Availability
The research data will be made available upon request to the corresponding author, Anik Giguere.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
