Abstract
Highlights
Our study demonstrates that discrete-choice experiments (DCEs) are valuable for capturing health-related preferences among patients with prostate cancer.
Contextual factors, such as efficacy and the country-specific health care system in which choices are presented, influence the ability to pool DCE data across countries.
DCEs have the potential to enhance patient-centered care, shaping the future of evidence-based health care decision making.
This is a visual representation of the abstract.
Keywords
Worldwide, prostate cancer (PC) is the second most common cancer among men, with an estimated 1.4 million new cases reported in 2020. 1 In 2022, the age-standardized incidence rates of PC estimated in France and Spain were 82.3 and 66.5 per 100,000, respectively, while these rates were lower in Japan, South Korea, and China, at 50.1, 29.3, and 9.7 per 100,000, respectively.2,3 PC treatment and management guidelines to optimize care are available in various countries. In most cases, patients with high-risk localized PC (HRLPC) are treated with external beam radiation therapy and long-term androgen-deprivation therapy (ADT). 4 Patients with metastatic hormone-sensitive PC (mHSPC) receive ADT in combination with novel hormonal therapies (i.e., abiraterone acetate with prednisone, apalutamide, enzalutamide, or darolutamide) or in combination with docetaxel or prostate radiotherapy.5–7
Patients differ in terms of their expected response to treatment as well as their experiences with treatment-related adverse events and route or frequency of drug administration. Evidence from previous assessments among patients with PC has shown that differences in preferences can be expected across countries, 8 in part due to differing access to care or in populations with different baseline comorbidities. 9 Thus, the relative importance of treatment aspects can be heterogeneous. Understanding patients’ treatment preferences for the outcomes and requirements of these treatment options becomes crucial when long-term treatment commitments from patients are necessary. Failing to understand patient preferences could affect patients’ quality of life (QoL) over time, both functionally and psychologically. 10 Patient–physician communication and shared decision making may lead to minimal posttreatment decisional regret in patients. 10
Several previous studies across many countries in Europe and Asia have reported on the treatment preferences of patients with PC.11–13 These studies showed that patients have strong preferences for treatment effectiveness or that they were mainly concerned about bone pain or fatigue, depending on how advanced their disease was.11–13 Similarly, in the United States, Canada, and the United Kingdom, representative of English-speaking countries, patients indicated the highest preference for treatment efficacy, followed by a strong preference to avoid treatment-related risk of serious infections. 9 Treatment-related convenience factors were considered, such as route of drug administration and frequency of monitoring visits, and were as important as some treatment-related side effects, such as skin rash, nausea, and fatigue. 8 A Swiss study reported strong patient preference for experiencing a survival benefit overall, with some patients strongly preferring the absence of adverse effects. 9 The heterogeneity in patient preferences may have a relevant impact on treatment choices in those with HSPC. 9 Given previous findings that patients in different countries exhibit common concerns around attributes of PC medications (i.e., efficacy 8 and survival 9 ), we used a discrete-choice experiment (DCE) to investigate the degree to which this was the case among countries with significant cultural and health system-level differences.
In DCEs, patients are asked to convey the relative importance of treatment attributes in terms of how the attributes affect their choice among experimentally constructed treatments. Specifically, DCEs quantify the relative value that patients assign to various treatment attributes based on how choices vary with changes in those attributes. 14 Patients’ treatment preferences often vary with their cultural background and health care system and with the socioeconomic support they receive.15–18 Thus, the treatment choices consistent with patient preferences may differ considerably across countries despite disease biology remaining largely the same. Instead of measuring the degree to which the preferences are the same across countries on average, understanding the degree to which a single model of preferences is sufficient to characterize the types of preference patterns across countries may inform the feasibility of generalizing treatment preferences across geographies.
Usual evaluations of differences in preferences across countries focus on differences in mean preferences. However, it has recently been proposed that such an approach might overstate differences. Instead, a single model of preferences can be sufficient to characterize the preferences of respondents in different groups and can offer a more meaningful evaluation of preference heterogeneity. 19
The objective of this study was to elicit and quantify the preferences and tradeoffs patients make when selecting chemohormonal treatment options in 5 culturally diverse countries: France, Spain, China, South Korea, and Japan. We aimed to provide a detailed description of the differing treatment attributes to aid shared decision making in support of treatment decisions among patients with HRLPC and mHSPC. Data from this study may allow health care providers to understand patient diversity with respect to treatment preferences in different populations across geographies. Identifying common preference patterns across countries can also facilitate conversations between patients and physicians on treatment selection in the hope of improving treatment adherence and, eventually, treatment outcomes.
Materials and Methods
Survey Development
Treatment aspects valuable for patients were identified via a targeted literature review, focus group discussions with patients, and clinical expert consultations. Efficacy (5-y overall survival), tolerability (fatigue, skin rash, neurotoxicity, and common chemotherapy-related toxicities), and treatment convenience (route, frequency, and setting of drug administration; concomitant use of steroids; and monitoring requirements) were key treatment attributes. These attributes and select clinically plausible treatment outcomes (attribute levels; Supplementary Table S1) were used to construct experimentally controlled treatment profiles for choice questions in the survey (Supplementary Figure S1).
The survey was initially developed in English for administration in the United States, United Kingdom, and Canada. Translation of the English instrument was performed and validated through a series of interviews in each of the non–English-speaking countries involved in the study. Upon validating the translation, we implemented the survey using the same experimental designs used to populate the choice questions in the English surveys. These were 2 statistically efficient experimental designs created to estimate preference weights with a limited number of questions. Some examples include the following: (1) for those who are not currently on hormone therapy or ADT: “Suppose your cancer gets worse and your doctor suggests that you start taking ADT. Your doctor also suggests that you consider taking ADT with or without an additional medicine”; (2) for those who are currently on hormone therapy or ADT and taking >1 medicine as part of hormone therapy: “Suppose that your current treatment for prostate cancer stops working. Your doctor suggests that you start taking ADT, and that you consider adding an additional medicine to ADT”; and (3) for those who are currently on hormone therapy or ADT and not taking >1 medicine as part of hormone therapy: “Suppose that your treatments for prostate cancer stop working. Your doctor suggests that you start a new hormone therapy (ADT), and that you consider adding an additional medicine to ADT.”
Treatment-related side effects associated with chemotherapy and nonsteroidal antiandrogens in combination with ADT were included as attributes. These included common adverse effects related to mineralocorticoid excess (i.e., hypertension, hypokalemia, and edema), hormonal effects (i.e., fatigue and hot flushes), and liver toxicity reported with abiraterone acetate plus prednisone; rash, hypothyroidism, and ischemic heart disease reported with apalutamide plus ADT; fatigue, seizures, and hypertension reported with enzalutamide plus ADT; fatigue, pain in the extremities, and rash reported with darolutamide plus ADT; and nausea, peripheral neuropathy, and serious infections associated with chemotherapy. Each experimentally constructed choice question had 3 treatment alternatives. “ADT alone” represents a baseline choice for patients, and the other 2 choices show the treatment profiles of additional medications in combination with ADT. Respondents were asked to choose the most preferred option of the three. Since it was difficult for respondents to process all 8 attributes in a single choice question, 2 DCE modules were developed: one to capture patient tradeoffs between efficacy and tolerability and the other to understand patient tradeoffs between efficacy and convenience. This method improved choice consistency and decreased the dropout rate in a DCE. 20
After the translation and before the interviews, the survey instrument was back-translated into country-specific languages and culturally adapted. The country-specific survey instruments were then refined and updated based on patient feedback from one-on-one pretest interviews. An example comprehension question, upon being shown an icon array depicting 3 of 10 (30%) shaded figures receiving ADT (Supplementary Table S2): “How many patients are expected to be alive 5 years after taking ADT?” The DCE survey was developed in accordance with good-practice guidance 21 and was reviewed and approved by the Duke University Health System Institutional Review Board (Pro 00106523). The DCE survey was administered in France, Spain, China, South Korea, and Japan between September 5 and December 11, 2022. Electronic consent (e-consent) was collected from the participants. Respondents were not allowed to complete the survey until they clicked to consent to participate in the research.
Experimental Design
The experimental designs for both modules were developed in accordance with ISPOR guidance on good practice for experimental designs of DCEs.21,22 In an effort to allow for meaningful responses by the patients in the study, 2 distinct modules were created to isolate questions regarding preferences as they related to health outcomes and convenience factors. The 2 modules shared the 5-y survival attribute and attribute levels. By constraining the preference weights for this attribute to be the same across modules, we were able to build a bridge that facilitates estimating both sets of preferences together.
Combinations of attribute levels for each hypothetical treatment profile to populate the DCE choice questions were determined using the SAS experimental-design algorithms. 23 Both modules were optimized to enhance the statistical efficiency (D-optimality) of the choice questions. For module 1, an experimental design was generated with 36 questions grouped into 6 blocks of 6 questions each. For module 2, another experimental design was developed with 36 questions grouped into 9 blocks of 4 questions each. Each respondent answered up to 14 questions: 6 questions from the module 1 design, 4 questions from the first assigned module 2 block, 2 questions from the second assigned module 2 block, and, if willing, 2 more questions from the second assigned module 2 block.
Study Population
Patients were recruited through an online health panel managed by Global Perspectives and in partnership with local physicians who treat these patients. Those patients willing to participate in the DCE survey were identified by a treating physician and were contacted by the study team to evaluate their eligibility. For attribute selection and pretest interviews, adults (≥18 y of age) diagnosed with mHSPC, with or without previous experience with ADT, who could read and understand the survey language, and who provided informed consent were included. For the online survey, adults (≥18 y of age) diagnosed with HRLPC or mHSPC, with or without previous experience with ADT, who could read and understand the survey language, and who provided informed consent were included.
The study sample size was determined following the general rules for sample size determination proposed by Orme 24 and Yang et al. 25 The expected necessary sample size ranged between 35 and 174.24,25 Orme’s rule suggested the final sample sizes for each country were adequate: China, n = 100; France, n = 79; Japan, n = 100; South Korea, n = 100; Spain, n = 91.
Statistical Analysis
One of the study objectives was to identify the similarities and dissimilarities in patient preferences among culturally and socioeconomically diverse countries. For this purpose, a broader evaluation of preference heterogeneity was conducted by pooling responses from all countries in a latent-class random-parameters logit (LCRPL) model with 8 classes to explore the overlapping and unique treatment choices of patients from France, Spain, China, South Korea, and Japan (modeling details are described in the supplementary methods).26,27 To quantify the values respondents placed on treatment attributes, relative importance weights were computed by class. The relative importance measures were normalized to facilitate comparisons across the classes. The study also aimed to determine whether the clinical characteristics of patients from the 5 countries explained the heterogeneity observed in the patterns of their treatment choices. For this, odds ratios (ORs) were calculated to evaluate the impact of covariates on the class membership assignment of patients. Data analysis was conducted using Latent GOLD 5.1 (Statistical Innovations, Arlington, MA, USA).
Results
A total of 468 patients across the 5 countries—79 patients from France, 91 patients from Spain, and 100 patients each from China, South Korea, and Japan—completed the survey. The median time to survey completion varied between 10.2 and 36.2 min. Tables 1 and 2 show the demographic and clinical characteristics of the study population, highlighting heterogeneity in aspects related to PC and treatments across the 5 countries. Survey respondents from Japan were the oldest among the 5 countries, with a mean (standard deviation [s]) age of 69.9 (7.3) y, whereas those from South Korea were the youngest, with a mean (s) age of 60.1 (7.6) y. In Japan, 60% of respondents had a bachelor’s degree, while only 13% in China did (Table 1). In terms of employment, there was considerable variation among the countries, with 44% of respondents in China being employed (highest) but only 20.9% of respondents in Spain being employed (lowest) (Table 1). The average time since PC diagnosis varied between 1.2 and 5.9 y among all respondents. The average time since PC diagnosis was less than 2 y among only Chinese respondents (Table 2). In 59% to 89% of the respondents, the cancer had spread to areas around the prostate, and in 11% to 42% of the respondents, it had spread to other organs, such as the bones, lungs, and brain (Table 2). There was considerable variation (8.0%–42.9%) across countries in the number of respondents who had stopped PC treatment in the past. Among those who stopped treatment, 12.5% to 43.6% of the respondents did so because the treatment did not stop cancer progression, whereas 12.5% to 40.9% of the respondents stopped due to a side effect (Table 2). The proportion of patients who experienced fatigue varied substantially, from 35% in South Korea to 91% in China; among those who experienced fatigue, there was considerable variation in terms of the proportions of patients with limiting fatigue, nonlimiting fatigue, or fatigue that necessitated help with everyday chores. In total, 72% of respondents in China received prior steroid treatment, whereas only 24% of respondents in Japan were exposed to prior steroid treatment (Table 2).
Demographics of Patient Respondents in the 5 Countries

Q1/Q3, quartile 1/3.
P < 0.001; **P < 0.01
Clinical Characteristics of Patient Respondents with PC in the 5 Countries
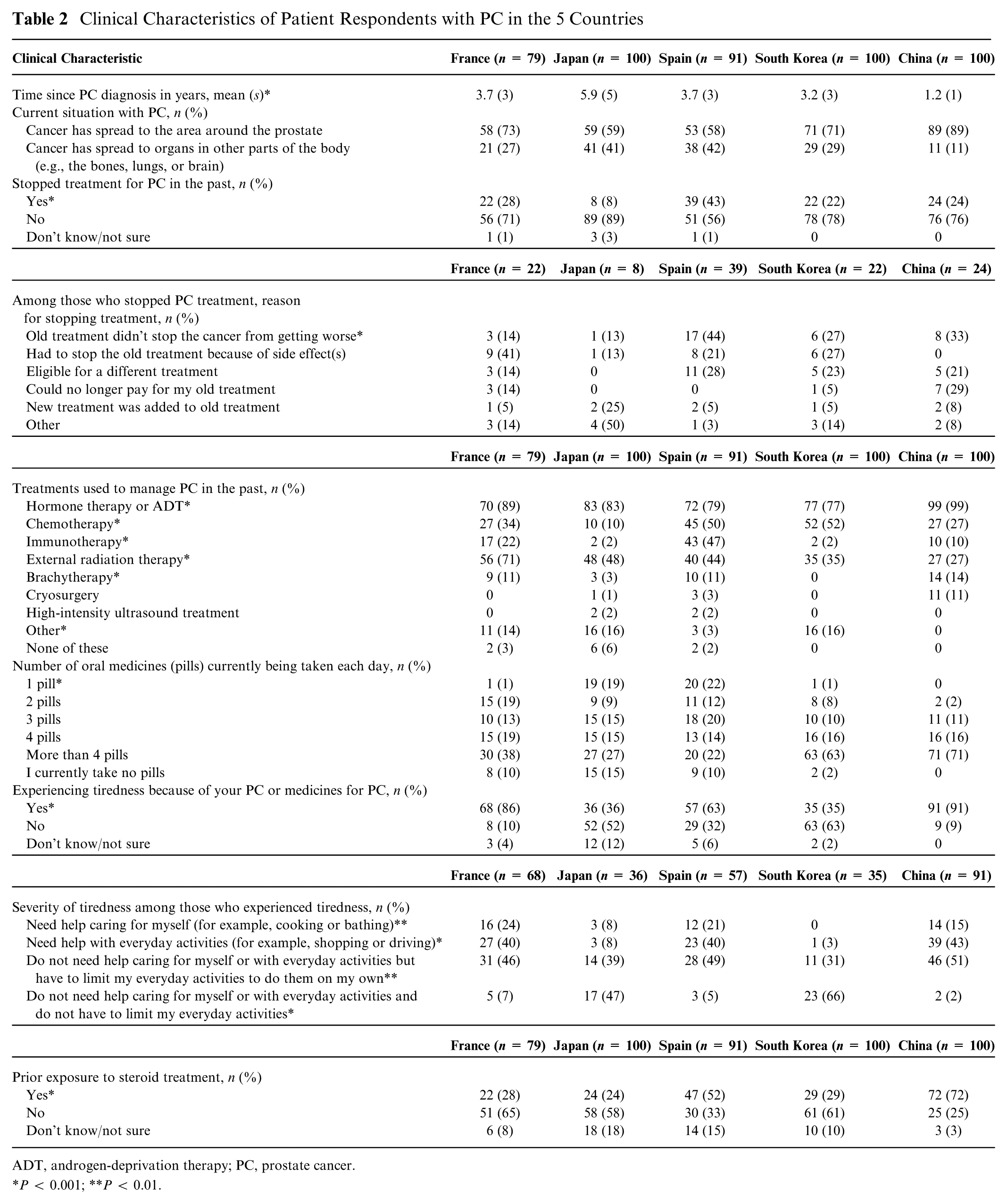
ADT, androgen-deprivation therapy; PC, prostate cancer.
P < 0.001; **P < 0.01.
Eight-LCRPL Model
Multicountry analysis using the 8-LCRPL model estimated probabilistic membership to 8 classes based on the similarities and dissimilarities in the patterns of treatment preferences among patients from all 5 countries (Table 3). The 8-class modeling approach was based on a novel approach to assess the poolability of preference estimates based on the similarity of preference types within a population, rather than the consistency of means across cohorts. 19 Unlike traditional tests that compare sample means across cohorts, the latent-class approach we used evaluates whether at least some respondents share a common distribution of preferences across cohorts. This approach leverages latent-class analysis to sort respondents from different cohorts into cohort-specific classes (5 classes) and global sample classes that aggregate responses from all cohorts (3 classes). Among the global classes, we considered a task-non-attendant class (also sometimes called garbage class) in which respondents offered no systematic information on attribute preferences. Two additional global classes were included based on model fit. Only these 2 global classes included random parameters and represented distinct preference profiles that were present in all countries. Parameters were correlated only to the extent they were estimated for specific classes. We ran individual country models with random parameters for each attribute level, which suggests we were powered to accommodate at least 32 parameters per country. This is less than the number of parameters in the 8-class model.
Overall Membership Probabilities for the 8-Class Model (N = 468)
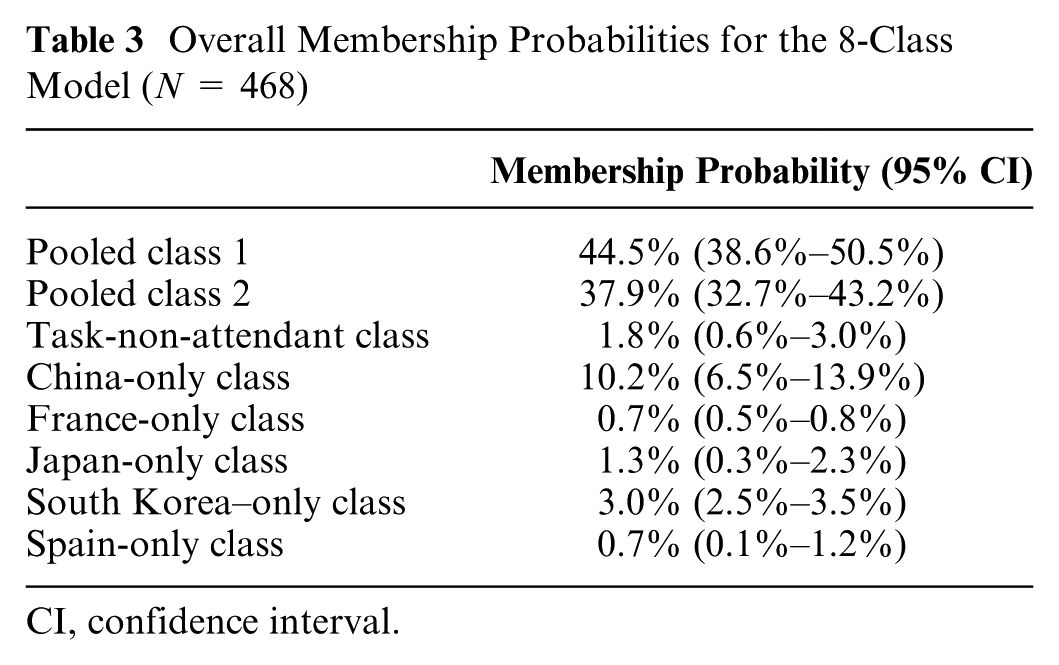
CI, confidence interval.
Results from the LCRPL model showed that the preferences of most respondents across the 5 countries had a modal probability of being in pooled class 1 (44.5%) and pooled class 2 (37.9%) (Table 3). The preference patterns of patients in these 2 pooled classes were similar in that patients had the highest preference for survival over side effects and treatment convenience factors (Supplementary Figure S2). In pooled class 1, the top factors of concern for patients were skin rash, fatigue, central nervous system (CNS) problems, and use of steroids, while for patients in pooled class 2, chemotherapy-associated problems were the main concerns (Supplementary Figure S2). Membership to country-specific classes (China only, France only, Japan only, South Korea only, and Spain only) identified patients’ treatment choices that were specific and unique for that country and that could not be pooled with any other classes (Table 3). Membership to the task-non-attendant class (1.8%) identified a choice behavior in which attention was not paid to all treatment attributes in the choice questions. The distribution of respondents to the 8 classes by country showed that patients had an 82.4% chance of being in pooled classes 1 and 2 (Table 3), suggesting that survey respondents across the 5 countries had overlapping treatment preferences (Tables 3 and 4). China had the highest assignment of modal probabilities to their country-specific class (47.7%), implying that the treatment preferences of Chinese patients were systematically more distinct than those observed in other countries (Table 4). The distribution of country membership to the pooled classes was comparable. South Korean respondents contributed the most to pooled class 1 (32%), whereas Japan, South Korea, and China all contributed (21%) the most to pooled class 2 (Table 5). By individual country, most of the respondents in the task-non-attendant class were from France (61.3%), followed by Japan (37.3%) (Table 5).
Distribution of Classes in the 5 Countries

Distribution of Countries by Class

Priorities of the Patients in the 2 Pooled Classes
Preference patterns in both pooled classes were similar in terms of the importance patients gave to chance of survival with treatment. Survival was the most important treatment attribute for patients with pooled class 1 preferences (47%; 95% confidence interval [CI]: 2%–93%) and pooled class 2 preferences (47%; 95% CI: 30%–64%) (Figure 1a and b, Supplementary Table S3). Patient preferences for the survival level associated with alternative treatments (70%, 50%, and 40%) was higher than for “ADT alone” (30%), and their error bars were nonoverlapping (Supplementary Figure S2). The data further showed clear differences in the priorities patients assigned to factors other than survival. The 2 pooled classes are distinct from each other in terms of importance placed on other key treatment attributes (Figure 1a and b, Supplementary Table S3) by the patients. Although the main factors of concern varied among the 5 countries, the 2 classes had overlapping treatment concerns. In pooled class 1, the main factors of concern were skin rash (11%), followed by CNS problems (10%), and steroid use and tiredness (9%) (Figure 1a, Supplementary Table S3). In pooled class 2, the main factors of concern included chemotherapy problems, such as nausea, weakness, tingling, pain, and serious infections (30%), followed by skin rash (8%) and CNS problems (7%) (Figure 1b, Supplementary Table S3).

(a) Relative importance weights for patient preferences in pooled class 1. (b) Relative importance weights for patient preferences in pooled class 2.
The analysis used patient covariates in the 8-LCRPL model to characterize which patients are more likely to be included in each of the pooled classes and the task-non-attendant class. Respondent characteristics, such as nonlimiting fatigue (OR = 17.74; P = 0.007), diagnosis within the past 2 y (OR = 7.62; P = 0.013) and high school education or less (OR = 2.99; P = 0.042), were significantly associated with the task-non-attendant class (Supplementary Table S4).
Covariates, such as nonlimiting fatigue (OR = 10.16; P = 0.001), diagnosis within the past 2 y (OR = 4.46; P = 0.002), prior use of ADT (OR = 4.43; P = 0.002), and having a graduate degree (OR = 3.25; P = 0.005), were significantly associated with membership to pooled class 1 (Supplementary Table S5). This means that patients with a graduate degree who had received their diagnosis recently had experienced nonlimiting fatigue, had received prior ADT treatment, and had stopped treatment in the past were more likely to be concerned about skin rash, CNS problems, tiredness, and steroid use. Having a graduate degree (OR = 2.54; P = 0.032) and nonlimiting fatigue (OR = 7.17; P = 0.006) were the 2 respondent characteristics significantly associated with membership to pooled class 2 (Supplementary Table S6). Thus, patients with a graduate degree with or without fatigue were more likely to be concerned about serious infections, weakness, tingling, pain, and nausea associated with chemotherapy.
Discussion
To our knowledge, this is the first multinational DCE study that elicited treatment preferences from patients with HRLPC and mHSPC from 5 countries in Europe and Asia. An earlier report of this study discussed the treatment preferences of patients with HRLPC and mHSPC across 3 countries with shared language and cultural backgrounds (United States, Canada, and United Kingdom) 8 and found that approximately 60% of patients who participated in the survey across the 3 countries could be pooled together based on similarities in treatment-choice patterns. Data from that report showed patients believed that using ADT alone is insufficient when more effective treatments are available. Further, although efficacy significantly affected treatment choices, treatment-related convenience factors were as important as some safety concerns. 8 Previous studies reported on patients at different clinical stages of PC, such as hormone-sensitive PC, 12 castration-resistant PC,11,28 and localized PC, 29 while the current study focused on the treatment choices of patients with HRLPC and mHSPC.
The burden of new PC cases (age-standardized rates per 100,000) in the 5 countries in this study is highly variable. These countries also differ in terms of disease burden, cultural history, societal structure, economy, and health care system. Across the 5 countries, given the attribute levels presented in the DCE questions, treatment efficacy or survival was the most important treatment attribute for patients. However, the relative importance of different attributes influencing patient preferences in a DCE is dependent on the choice of attribute levels. For instance, while route or frequency of administration had a significant effect on decision making in the DCE, it also had a low relative importance when compared with treatment efficacy. Patients believed that, given the alternatives that can improve survival, the use of ADT alone is not sufficient. This is in line with findings from that earlier report. 8 Despite differences in culture, demographics, and the clinical characteristics of patient respondents across countries, the data showed 2 unique patterns of treatment preferences (pooled class 1 and pooled class 2). These 2 groups captured the views of patients with similar preferences among the 5 countries. Patients within pooled classes 1 and 2 prioritized survival (efficacy) over other treatment attributes.
Although China, Japan, and South Korea may be relatively more aligned culturally and historically, our findings indicate distinct preference patterns among patients in these countries. The preferences of Japanese patients closely aligned with the pooled classes, similar to European respondents, whereas Chinese respondents were the most likely to exhibit unique, country-specific preferences. When Chinese patients’ preferences did align with the pooled classes, they were more commonly represented by pooled class 1. The pattern seen among South Korean patients more closely resembled China than Japan.
For patients with pooled class 1 preferences, it was important to avoid skin rash, CNS problems, tiredness, and steroid use. For patients with pooled class 2 preferences, it was important to avoid chemotherapy-related problems, such as serious infections, weakness, tingling sensations, pain, and nausea. Regardless of diversity in terms of culture and socioeconomic conditions, patients with preferences for pooled class 1 and pooled class 2 displayed treatment choices representative of the majority of the respondent population from the 5 countries. The similarities observed in patients with preferences for these 2 pooled classes may represent a snapshot of the type of treatment choices one could expect from patients with PC across geographies and cultures. The heterogeneity in patient preferences captured in this study was consistent with that observed in another study in Swiss patients with mHSPC, in which 2 distinct patient groups were identified. 9 Interestingly, in that study, the group showing strong preference for the absence of adverse effects was substantially larger than the group that strongly preferred survival. 9 In an Australian DCE study, people with cancer were reluctant to make tradeoffs between survival and QoL; however, the same study also suggested that the side effects that participants were most willing to avoid and trade off against survival were nausea, pain, and reduced mobility. 30 These studies, as well as our earlier report 8 and the findings from the present study, emphasize the existence of heterogeneity as well as similarities in patient preferences among countries. It is worth noting that the differences across countries appear to emerge both from variations in the types of preferences identified and their distributions across countries.
We used the demographic and clinical characteristics of patients as covariates to explain class assignments, but we did not succeed in segregating the 2 pooled classes based on clinically relevant covariates. This suggests that clinical factors are not necessarily associated with patient preferences for treatments. Patient covariates that were significantly associated with country-specific class assignments (Supplementary Table S7) show that several diverse clinical factors drive country-specific class assignments, supporting the idea that the treatment preferences of patients across the countries in our study may vary depending on cultural differences and patients’ socioeconomic status rather than institutional factors.
Groups of patients may have similar preferences across countries but with differing mean preferences, in part due to differing health care systems and socioeconomic support. 8 Our results highlight the importance of capturing the preferences of patients with PC, as clinical factors alone may not be indicative of preferred treatment choices. Further, respondents with graduate degrees were more likely to provide systematic responses that aligned with global preference distributions, which was negatively correlated with belonging to the task-non-attendant class. This is particularly critical given the long-term treatment commitments that are needed from patients and how patients’ QoL, both functionally and psychologically, can be affected by a mismatch between patient preferences and treatment options. 10
Like patient preferences, treatment-related shared decision making also varies across cultures and social groups. Despite the limited involvement of patients in decision making, especially in Asian societies, joint decision making was reported to be associated with higher well-being and perceived quality of care. 31 Hence, physicians who are armed with knowledge about patients’ treatment preferences and mindful of their sociocultural circumstances may be more effective in communicating with their patients and better at matching treatment options with patient preferences. Thus, tailoring shared decision support to specific patient groups/phenotypes may be a more manageable and effective approach compared with a single/one-size-fits-all or overly personalized decision aids. Exploring methods, such as pre–decision support surveys, to identify to which group a patient likely belongs may help to streamline the provision of appropriate materials or discussions about ways to address patients’ concerns ahead of the realization of concerning outcomes. This approach may enhance the utility of decision-making tools by ensuring their adaptability and effectiveness across diverse countries and patient populations.
The present study has some limitations. As the survey included hypothetical treatments, patient preferences elicited may be different from those elicited with actual treatments available in the real world. However, the survey was structured and formatted to represent real-world decision making as much as possible and to elicit patient preferences in a reliable manner. In addition, to address potential data quality concerns, we specifically controlled for task-non-attendance in one of the classes in the pooled analysis. Patients in this class provided no systematic information regarding attribute preferences. While the model assigned respondents to this class probabilistically, it also down-weighted their influence on other model classes based on their probability of being nonattendant. Notably, time to completion was not found to be correlated with membership in the task-non-attendant class. Lastly, the enrolled patient sample from each country may not be fully representative of all patients with HRLPC or mHSPC.
Conclusion
Respondents in this study believed that, given the treatment alternatives that can improve 5-y survival, the use of ADT alone is not sufficient. Across all 5 countries in this study, efficacy was the most significant driver of patients’ treatment choices. Beyond treatment efficacy, there were 2 distinct patterns of treatment preferences across all countries. These 2 patterns differed in terms of their concerns about treatment side effects, convenience, and chemotherapy-associated problems. Skin rash, CNS problems, steroid use, and tiredness were the main concerns for patients with pooled class 1 preferences, whereas, for patients with pooled class 2 preferences, chemotherapy-related problems, such as serious infections, weakness, tingling, pain, and nausea, were key concerns. The similarities in preference for treatment attributes in patients among the 5 countries may have implications for shared decision making and treatment adherence. The observed heterogeneity and lack of clinical predictors of preference patterns highlight the need for physicians to tailor treatment strategies on the basis of patients’ personal and socioeconomic circumstances before making a therapeutic recommendation. This study highlights the need to encourage communication between patients and clinicians about treatment choices and decisions, since clinical factors may not be predictive of patient preferences.
Supplemental Material
sj-docx-1-mpp-10.1177_23814683251386887 – Supplemental material for Treatment Preferences among Patients with Hormone-Sensitive Prostate Cancer in France, Spain, China, South Korea, and Japan: A Discrete-Choice Experiment
Supplemental material, sj-docx-1-mpp-10.1177_23814683251386887 for Treatment Preferences among Patients with Hormone-Sensitive Prostate Cancer in France, Spain, China, South Korea, and Japan: A Discrete-Choice Experiment by Juan Marcos Gonzalez, Arijit Ganguli, Alicia K. Morgans, Bertrand F. Tombal, Sebastien J. Hotte, Hiroyoshi Suzuki, Daniel Ng, Charles D. Scales, Matthew J. Wallace, Jui-Chen Yang and Daniel J. George in MDM Policy & Practice
Footnotes
Acknowledgements
The authors acknowledge the contribution made by Kiran Grover during survey development. The authors would also like to thank Lakshman Puli (PhD), Olga Klibanov (PharmD, CMPP), and Rucha Kurtkoti (MSc) from IQVIA for medical writing and editorial support.
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Arijit Ganguli is an employee of Astellas Pharma Inc. and owns stocks in AbbVie. Daniel Ng is an employee of Astellas. Daniel J. George received research grants and contracts from Astellas, AstraZeneca, BMS, CORVUS, Exelixis, Janssen, Novartis, Pfizer, and Surface Oncology. He also received consulting fees from ABRX, Astellas, IdeOncology, Janssen, Propella TX, Sanofi, AstraZeneca, Bayer, Exelixis, Merck, MJH Assoc, Pfizer, Seattle Gen, WebMD, and Sumitovant Biopharma. Daniel J. George also received honoraria for lectures and speaker bureaus from Bayer, Exelixis, Sanofi, UroGPO, IdeOncology, MJH Assoc, Pfizer, and UroToday. He provided expert testimonies for Exelixis and WilmerHale and received meetings/travel support from Exelixis, Bayer, Sanofi, and UroToday. Daniel George is part of the advisory boards for Astellas, AstraZeneca, Bayer, Elsai, Merck, MJH Assoc, Nektar, Pfizer, Propella TX, and Sumitovant Biopharma. Sebastien J. Hotte received research grants or contracts from Astellas, Bayer, Janssen, and Novartis. He is a consultant for Astellas, Bayer, Janssen, and Novartis. Matthew J. Wallace is a Duke University employee, and Duke University received funding from Astellas. Bertrand F. Tombal received advisory fees and clinical trial support from Bayer, and he is an advisor for Astellas, Bayer, Ferring, Janssen, Myovant, and Pfizer. He is also a consultant and recipient of honoraria for lectures and speakers’ bureaus, participates in advisory boards, and has a leadership role at Idem 4. He is also in a leadership role at EORTC and is a board member for ISSECAM. Hiroyoshi Suzuki received honoraria from Astellas. Jui-Chen Yang is a consultant for Duke University. Juan Marcos Gonzalez is an employee of Duke University, which received funding for this study and has a research contract with Astellas. Charles D. Scales Jr is an employee of Duke University, which received funding for this study and has a research contract with Astellas. Alicia Morgans is a consultant for Astellas, AstraZeneca, AAA, Bayer, Exelixis, Janssen, Myovant, Novartis, Pfizer, Telix, Tolmar, and Sanofi. She received honoraria from Novartis, Sanofi, Telix, AstraZeneca, AAA, and Pfizer and received meetings/travel support from Sanofi, Pfizer, and Telix; she is also on the advisory board of Gilead. The competing interests declared here do not alter our adherence to MDM Policy & Practice’s policies on sharing data and materials. The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors received no specific funding for this work. This study was funded by Astellas Pharma Inc. and Pfizer Inc., the co-developers of enzalutamide. Astellas Pharma Inc. provided funding to Duke University for this study as well as support in the form of employee salaries for Arjit Ganguli and Daniel Ng. The funding agreement ensured the authors’ independence in designing the study, collecting and interpreting the data, writing, reviewing, and publishing the key findings. Astellas Pharma Inc. participated in study design, data collection, data analysis, data interpretation, and writing of the report. Pfizer Inc. did not play any role in study design, data collection, data analysis, data interpretation, or writing of the report. The specific roles of the authors are detailed above. Medical writing and editorial services from IQVIA were funded by Astellas Pharma Inc.
Ethical Considerations
The study was conducted in accordance with the protocol, applicable International Council for Harmonization (ICH) of technical requirements for pharmaceuticals for human use guidelines for Good Clinical Practice, and ISPOR guidance on the design of discrete choice experiments. The study followed applicable regulations, guidelines governing clinical study conduct, and ethical principles as per the Declaration of Helsinki. The protocol for interviews was reviewed and approved by the institutional review board of a large academic institution. Individual patient medical information and sensitive personal information obtained as a result of this study were kept confidential, and data related to patient identification numbers and/or initials, when used, were anonymized.
Consent to Participate
Written informed consent was obtained from the participants for participation in the study.
Consent for Publication
Written informed consent was obtained from the participants for publication of the study.
Data Availability
All data generated or analyzed during this study, which support the findings of this study, are included within this article and its supplementary information files. Researchers may access analysis not present in the article from the corresponding author upon reasonable request. Details for how researchers may request access to anonymized participant-level data, trial-level data, and protocols from Astellas-sponsored clinical trials can be found at ![]() .
.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
