Abstract
Highlights
This study provides the first content-validated version of the IPDAS Minimal Criteria in Portuguese, addressing a significant gap in shared decision-making tools.
The instrument supports Portuguese-speaking researchers and clinicians in systematically assessing decision aids for quality and usability.
These findings highlight the importance of cultural adaptation in ensuring the applicability and effectiveness of decision support tools across different populations.
This study advances the field of decision-making research by fostering equitable access to high-quality decision aids in clinical practice.
Keywords
In the past, physicians have held a dominant role in health care decisions, particularly in contexts involving medical interventions such as treatments, screening, or procedures. Historically, their recommendations were guided by clinical expertise and the assumption that they knew what was best for their patients. This approach is known as the “paternalistic model,” which is grounded in Parsons’ conceptualization of the sick role. 1 Under this model, physicians make decisions based on what they believe is in the patient’s best interest without necessarily considering the patient’s preferences.
However, over recent decades, there has been a paradigm shift toward more collaborative models of care. Shared decision making is a dynamic and collaborative process in which clinicians and patients work together to make health care decisions, integrating the best available clinical evidence with the patient’s own values, goals, and preferences. 2 This approach recognizes patients as active agents in their care, promoting not only their autonomy but also their engagement in decisions that respect their health.3,4 This consultation model is especially important when more than 1 medically reasonable option exists and when no single option is clearly superior (i.e., in situations of clinical equipoise). In such cases, choosing among alternatives requires not only clinical reasoning but also an understanding of what matters most to the patient.
To support shared decision making, interventions known as patient decision aids (PDAs) were designed. These tools provide evidence-based information about the risks and benefits of different options and help patients understand their options, anticipate potential outcomes, and clarify what matters most to them.5,6 Evidence from a systematic review of 209 randomized controlled trials demonstrates that patients exposed to decision aids feel more informed, better understand the benefits and harms of their options, and are more likely to engage in decision making.7,8 While these interventions are not essential for shared decision making, they can help make the process easier and ensure that the patient remains at the center of the decision-making process.
However, despite their benefits, most of these instruments are developed in English, limiting access for patients in other linguistic and cultural settings, including Portuguese-speaking populations.
A recent Portuguese study revealed that many family doctors confuse PDAs with clinical decision support tools. 9 While both aim to improve care quality, PDAs are focused on informing and empowering patients in preference-sensitive decisions, whereas clinical decision support tools assist health care professionals in applying clinical guidelines or algorithms to optimize medical decisions. Despite some confusion between these concepts, the integration of PDAs in primary health care was received positively, with barriers to implementation including insufficient funding, time constraints, and the absence of validated Portuguese-language translations. 9
Concerns about the quality and consistency of PDAs first arose in 2003 when evidence showed how they could influence patient choices. 10 The International Patient Decision Aid Standards (IPDAS) Collaboration, an international and multidisciplinary group, developed a set of criteria to assess the quality of PDAs, which has since been refined and used globally.6,10–13 Among these, the IPDAS Minimal Criteria (v4.0, 2013) identify the essential elements a PDA must include to support high-quality decision making. Although versions of these criteria exist in other languages, such as Spanish, Chinese, and Japanese, no validated version exists in Portuguese.
This study aimed to translate, culturally adapt, and assess the content validity of the IPDAS Minimal Criteria instrument (v4.0, 2013) for the Portuguese population, addressing a significant gap and providing a validated tool to support the evaluation and development of decision aids in Portuguese-speaking health care settings.
Methods
Study Design
The translation, cultural adaptation, and content validation of the IPDAS Minimal Criteria v4.0 (2013) for the Portuguese population were conducted based on the methodology of Sousa and Rojjanasrirat. 14 Cultural adaptation is more than a linguistic process; it ensures that translated instruments maintain conceptual, semantic, and content equivalence within the cultural context of the target population.14–17
Translation, Cultural Adaptation, and Content Validation of the Instrument
Before beginning the translation process, the authors sought and received permission for the translation and adaptation of the instrument from the IPDAS Steering Committee, which granted this authorization via e-mail.
Once authorization had been granted, some changes were proposed to the original version of the instrument, including the addition of a new qualifying criterion stating the target audience of the decision aid, as suggested by Martin et al. 18 Furthermore, for the qualifying criterion, “The PDA describes the features of options to help patients imagine what it is like to experience their physical, emotional, and social effects,” we were informed that the committee allows the use of an explicit values clarification exercise as an alternative to this item. This adjustment acknowledges that decision aids meeting this description would be longer compared with those using a values clarification exercise. Therefore, the research team decided to include this alternative in the criteria. In addition, the committee recommended using a dichotomous scale to evaluate certification and quality criteria. According to the IPDAS Steering Committee co-lead, most people find the 4-point response scale difficult to follow without training and instead prefer a yes/no response. Following these amendments, we obtained permission to use the modified version of the original IPDAS instrument in the linguistic validation process. The modified version of the criteria translated for this study is provided in Appendix 1.
The research team decided to use the methodology of Sousa and Rojjanasrirat 14 since it had been suggested when permission to translate the instrument was granted. Table 1 summarizes the recommended 7-step cross-cultural adaptation and validation process. Steps 1 to 5 encompass the translation and cultural adaptation of the instrument, whereas steps 6 and 7 focus on validating the translated instrument. In this initial study, we followed steps 1 to 5 to create a translated, culturally adapted, and content validated instrument in Portuguese. Steps 6 and 7 will be addressed in future psychometric validation studies.
Summary of the Translation, Cultural Adaptation, and Content Validation Process of the Portuguese IPDAS Minimal Criteria Instrument

I-CVI, Item-Level Content Validity Index; IPDAS, International Patient Decision Aid Standards; S-CVI/Ave, Scale-Level Content Validity Index.
Step 1: Forward translation
In the first stage, 2 forward translations of the instruments from the original language (English) to the target language (the language to which we were translating the instrument, Portuguese, in this case) were produced by 2 independent bilingual professional translators whose mother tongue was Portuguese. The translators had different backgrounds: one was a specialized medical translator, knowledgeable about health care terminology and the content area of the instrument construct, and the other was neither knowledgeable about medical terminology nor familiar with the construct of the instrument. Both translators sought to reflect the usual spoken language and cultural nuances of the general population. From these translations, 2 versions were obtained: Translation 1 (TL1) and Translation 2 (TL2).
Step 2: Synthesis I
A third independent bilingual and bicultural professional medical translator compared TL1 with TL2 as well as both translations with the original version of the instrument. Using a committee approach, the 2 translators of step 1, the translator from this step (step 2), and 2 members of the research team met via Zoom to discuss any ambiguities and discrepancies and synthesize the results of the 2 translations. They then produced a preliminary initial translated version (PI-TL) of the instrument.
Step 3: Blind back-translation
Working from the PI-TL of the questionnaire and blinded to the original version, 2 independent professional translators (whose mother tongue was English, the language of the source instrument) translated the questionnaire back into their original language, producing 2 back-translated versions of the instrument (B-TL1 and B-TL2). The 2 translators had different backgrounds: one specialized in medical translation (and back-translation) while the other was not familiar with medical translation.
The 2 translators who performed the back-translations in step 3 were different from those who performed the initial forward translations in step 1. This ensured independent evaluation and minimized the risk of bias.
Step 4: Synthesis II
At this stage, a multidisciplinary committee compared the 2 back-translations of the instrument with each other and with the original instrument during a meeting held on Zoom. Following the recommendations of Sousa and Rojjanasrirat, 14 the multidisciplinary committee consisted of 1 methodologist (a member of the research team), 1 health care professional familiar with the domains of the instrument’s construct (also a member of the research team), and all translators involved in steps 1 to 3. The committee’s role was to evaluate the conceptual, semantic, and content equivalence of the back-translated instruments and develop a prefinal version of the instrument (P-FTL) for pilot testing and psychometric testing.
Step 5: Pilot test of the prefinal version (cognitive debriefing)
This phase ensured that the adapted version retained its equivalence in the applied situation. To determine the conceptual equivalence of the translated instrument, an expert panel comprising 10 members knowledgeable about the content areas of the instrument construct and whose mother language was Portuguese evaluated the quantitative and qualitative content validity of the P-FTL.
A letter of invitation to participate in the expert panel was sent via e-mail to each potential member. This panel included 2 family doctors with more than 5 y of practice, 2 family doctors with less than 5 y of practice, 3 researchers who had published at least 1 paper related to PDAs, and 3 researchers with experience in translating, adapting, and validating instruments.
Two weeks before the meeting, the authors sent the English and prefinal Portuguese versions of the IPDASi, a document with scales to evaluate the clarity and content relevance of the instrument, and a Google Form questionnaire to record their responses to the members who accepted the invitation (Appendix 2).
The questionnaire used in the pilot test to evaluate content validity comprised 2 sections: 1) an introductory section explaining the objectives of this step, followed by a clarity quantitative assessment question for each criterion and an open-ended question for qualitative comments and 2) a section with the scale to assess the content relevance of the instrument, again followed by an open comment box.
To evaluate the clarity of the P-FTL items, each expert rated the instrument criteria as clear or unclear. Then, the percentage agreement and Fleiss’ kappa values were calculated. The minimum interrater agreement for the sample was defined as 80%. Criteria found to be “unclear” by at least 20% of the panel were flagged for revision. These items were discussed and adjusted accordingly during the expert panel meeting. 19 To further evaluate the consistency of the expert ratings, Fleiss’ kappa was calculated. This statistic measures interrater agreement beyond what would be expected by chance and is particularly suitable when more than 2 raters are involved. The interpretation of kappa values typically follows the Landis and Koch classification: values less than 0.20 indicate slight agreement, 0.21 to 0.40 fair, 0.41 to 0.60 moderate, 0.61 to 0.80 substantial, and greater than 0.80 almost perfect agreement. In the content validity studies, kappa values greater than 0.60 are generally considered acceptable, although values greater than 0.75 are preferred for strong agreement. 20
The expert panel was also asked to evaluate each item of the instrument for content relevance using a 4-point Likert-type scale: 1 = not relevant, 2 = somewhat relevant—item needs some revision, 3 = quite relevant but needs minor revision, and 4 = very relevant. The expert panel was instructed to compare the Portuguese version of each item with the original English version, focusing on both semantic equivalence and cultural appropriateness. Ratings of 1 or 2 were considered content invalid, while ratings of 3 or 4 were considered content valid. Items classified as 1 or 2 by any panel member were considered indications that an item required revision. All such items were discussed and revised during the expert panel meeting.21,22
To quantify content validity, the Content Validity Mean Score, the Item-Level Content Validity Index (I-CVI), and Scale-Level Content Validity Index (S-CVI/Ave) were calculated. The Content Validity Mean Score was calculated as the mean score for each item across all reviewers. This mean score is not a psychometric indicator in itself but provides an additional descriptive measure of how each item was perceived by the panel. The Content Validity Indices reflect the proportion of experts who consider each item relevant. The I-CVI was calculated for each criterion as the number of experts giving a rating of 3 or 4 for each item (i.e., content valid) divided by the total number of experts of the expert panel. Values range from 0 to 1, where I-CVI >0.79 indicates the item is relevant and between 0.70 and 0.79 means the item needs revisions; and if the value is <0.70, the item should be eliminated. 22 S-CVI/Ave was then computed as the average of all I-CVI values. A value ≥0.90 was considered indicative of excellent overall content validity. An I-CVI ≥0.78 and S-CVI/Ave ≥0.90 were the minimum acceptable indices.21–23
To increase confidence in the content validity of the instrument, Fleiss’ kappa was also determined for relevance ratings.
Finally, a qualitative assessment was conducted during the expert panel meeting. Members were asked to compare each translated item to the original in terms of meaning, clarity, and cultural relevance. They were encouraged to reflect on grammar, vocabulary, and overall phrasing and to suggest improvements where needed. Discrepancies in interpretation or suggested changes were resolved through discussion and consensus building among the panel members, moderated by a member of the research team. This process ensured that both the linguistic and conceptual integrity of the instrument were maintained in the Portuguese context.
Sample Size and Sampling
The research team used an expert panel composed of 10 members for the pilot test of the prefinal version of the translated IPDASi (step 5). The literature recommends using an expert panel composed of 6 to 10 members to evaluate the clarity of the items and content equivalence of the translated instrument.21,23
Data Collection Methods and Sources
The translation and cultural adaptation process occurred from September to December 2023. Data collection from the pilot test occurred from January to February 2024 and was then entered into a computer database.
The Synthesis meetings involved the translators and 2 members of the research team and were used to resolve discrepancies and ensure cultural adaptation of the instrument. The expert panel, composed of 10 members not involved in the earlier steps, was responsible for evaluating the clarity and content relevance of the translated instrument. Quantitative data were recorded using a structured online Google Form, while qualitative feedback was captured through open-ended comments and interactive discussions during the final expert panel meeting.
Synthesis I, Synthesis II, and pilot test meetings were held via the Zoom platform. Participants gave verbal consent to participate at the beginning of each Zoom meeting.
Data Analysis
Quantitative variables were described by the mean, standard deviation, and minimum and maximum values (
To evaluate the clarity of each item, a dichotomous scale (clear or unclear) was used. The overall and specific percentage agreements and Fleiss’ kappa were then calculated with the corresponding 95% confidence interval (CI). To evaluate the content validity of the instrument, we used both the I-CVI and the S-CVI/Ave, along with Fleiss’ kappa statistic, with the respective 95% CI.
Qualitative data analysis occurred during the expert panel meeting. All 10 panel members were asked to examine each criterion in the Portuguese version alongside its original English version. They provided feedback on semantic equivalence, cultural appropriateness, grammar, and phrasing. When discrepancies or uncertainties arose, they were discussed openly among the panel until consensus was reached. This structured discussion ensured alignment with best practices for cognitive debriefing and qualitative validation in instrument adaptation.
Patient and Public Involvement
Patients and the public were not involved, given that the IPDAS is primarily used by researchers and health care professionals.
Role of the Funding Source for the Study
The funding source had no role in the study design; data collection, analysis, or interpretation; manuscript preparation; or the decision to submit the manuscript for publication. The authors declare that this research was conducted independently of any influence from funding organizations.
Results
Translation and Cultural Adaptation of the Instrument (Steps 1 to 4)
During the Synthesis I Zoom meeting, the PI-TL was analyzed, and several ambiguities were addressed. For instance, feedback received during the meeting highlighted that some terms were direct translations from English and required adjustments. An example of this is the term “qualifications,” which was translated literally as qualificações; however, the correct term used by Portuguese speakers is habilitações. This change improved the wording of some items, enhancing their clarity and linguistic adequacy. Appendix 3 contains all the changes made at the Synthesis I meeting.
At the Synthesis II meeting, when the multidisciplinary committee compared the 2 back-translations of the instrument with each other and with the original instrument, no discrepancies were observed. As a result, no further changes were made to the PI-TL, and the P-FTL of the instrument was then ready for pilot testing.
Validation of the Instrument (Step 5: Pilot Test of the Prefinal Version)
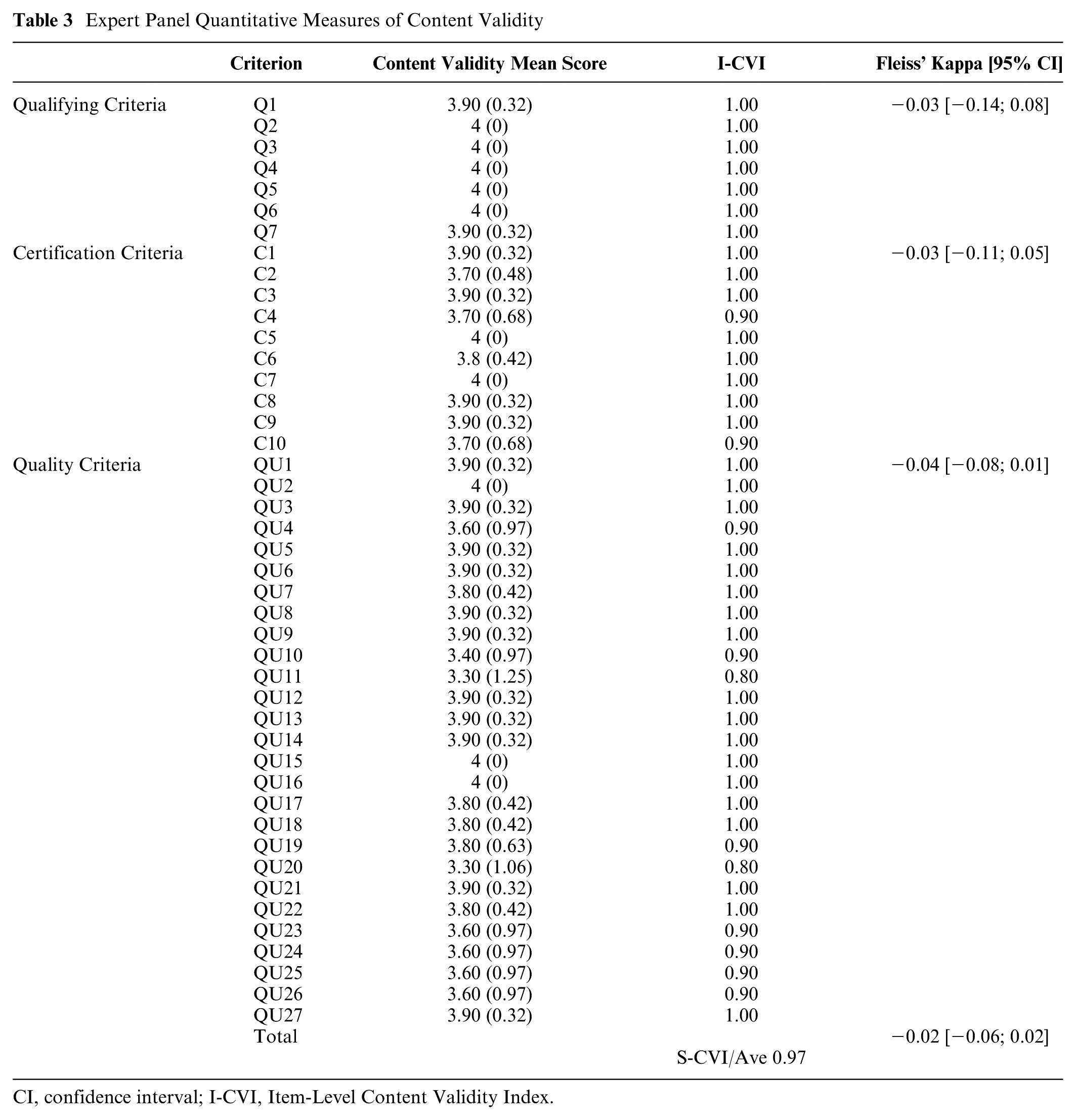
At the expert panel meeting, each of the 44 criteria were individually analyzed, discussed, and commented on. The panel sought to adapt each criterion to the linguistic specificities of Portuguese from Portugal and successfully reached a consensus solution for all items of the instrument under analysis. The quantitative measures of validity are summarized in Tables 2 and 3.
Expert Panel Quantitative Measures of Clarity

CI, confidence interval.
Expert Panel Quantitative Measures of Content Validity
CI, confidence interval; I-CVI, Item-Level Content Validity Index.
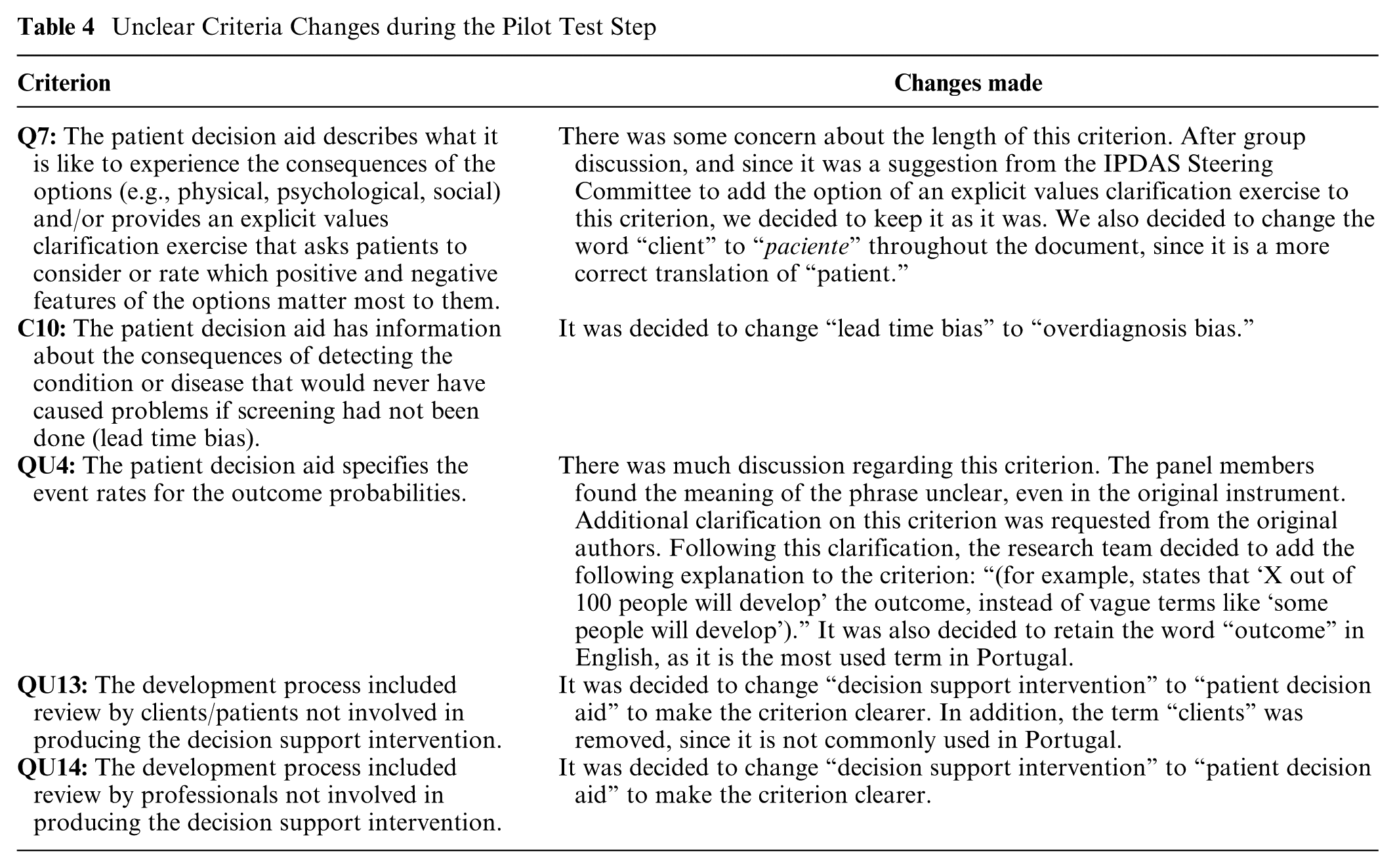
Criteria Q7, C10, QU4, QU13, and QU14 were found to be unclear by at least 20% of the panel and were revised and reevaluated during the expert panel meeting. Changes to these criteria are listed in Table 4. The overall agreement was 0.93 [0.89; 0.97], and the calculated Fleiss’ kappa was 0.03 [−0.01; 0.08].
Unclear Criteria Changes during the Pilot Test Step
Eleven criteria (C4, C10, QU4, QU10, QU11, QU19, QU20, QU23, QU24, QU25, and QU26) were rated as content invalid by at least 1 panel member (i.e., received a score of 1 or 2 on the 4-point relevance scale) and were revised during the expert panel meeting. In this context, “content invalid” indicates that the item was perceived as lacking sufficient relevance or requiring substantial modification to improve semantic equivalence and cultural appropriateness in the Portuguese version.
The Content Validity Mean Score ranged from 3.30 ± 1.06 to 4.00 ± 0.00. The I-CVI score was >0.79 for all criteria. The S-CVI calculated for the entire instrument was 0.97, and the calculated Fleiss’ kappa was −0.02 [−0.06; 0.02].
The final Portuguese version of the IPDASi is shown in Table 5.
Portuguese Version of the IPDAS Minimal Criteria v4.0 (2013) Instrument

IPDAS, International Patient Decision Aid Standards.
Original: IPDAS Minimal Criteria v4.0 (2013) instrument. Traduzido com autorização dos autores originais.
Autores originais: Natalie Joseph-Williams, Robert Newcombe, Mary Politi, Marie-Anne Durand, Stephanie Sivell, Dawn Stacey, Annette O’Connor, Robert J. Volk, Adrian Edwards, Carol Bennett, Michael Pignone, Richard Thomson, Glyn Elwyn.
Referência: Toward minimum standards for certifying patient decision aids: a modified Delphi consensus process. Med Decis Making. 2014;34(6):699–710. DOI: 10.1177/0272989X13501721
Traduzido por: Micaela Gregório, Andreia Teixeira, Mariana Teixeira, Inês Marques, Ana Sofia Correia, Phillippa Bennett, Helen Carter, Dawn Stacey, Carlos Martins.
Data de aprovação como versão portuguesa: 05/03/2024
Discussion
This study contributes new evidence regarding the translation, cultural adaptation, and content validation of the IPDAS Minimal Criteria v4.0 (2013) for use in the Portuguese context. The translation process followed a structured methodology, including forward and backward translation, multidisciplinary committee review, and pilot testing with an expert panel. Although our findings support the semantic and cultural relevance of the Portuguese version, we acknowledge that this study focused on linguistic and contextual adaptation rather than broader frameworks of cultural adaptation.
The cultural adaptation process played a crucial role in enhancing item clarity and contextual appropriateness. For instance, the term “qualificações” (a literal translation of “qualifications”) was replaced with “habilitações,” which is more commonly used in Portugal to refer to educational background. Similarly, technical or ambiguous phrases were reworded to reflect plain language more accessible to end users, ensuring that the adapted instrument better reflects local idioms and cultural norms. This adaptation underscores the importance of engaging native speakers and experts in the translation process to preserve both conceptual and practical relevance.
Although the expert panel reached high overall agreement on item clarity and relevance, Fleiss’ kappa values were low. This apparent contradiction is not unusual in validation studies using dichotomous or ordinal rating scales. Fleiss’ kappa measures interrater agreement beyond chance and is known to be sensitive to the prevalence of ratings within categories. In our case, the predominance of ratings such as “clear” or “very relevant”—indicating high agreement—reduced kappa values due to limited variability. According to common interpretative thresholds, 20 our values would be considered poor (<0.20); however, when combined with high percentage agreement and content validity indices, they offer a more nuanced and reassuring picture of item quality.24,25
The expert panel identified several unclear criteria that were revised and reevaluated, underscoring the importance of expert input. The revised items did not change the meaning of the original items.
Eleven criteria were initially rated as content invalid by at least 1 expert, meaning they were scored as either “not relevant” or “somewhat relevant—needs revision.” These items were carefully discussed and revised during the expert panel meeting. Following revisions, all criteria achieved acceptable content validity scores, as reflected in high I-CVI and S-CVI/Ave indices. This supports the appropriateness of iterative refinement based on expert feedback. Nonetheless, the limited variability in response categories on the relevance scale likely contributed again to the low Fleiss’ kappa for content validity.
Although this study focused on the linguistic and cultural adaptation of the instrument for European Portuguese, the translated tool may serve as a foundation for future adaptations in other Portuguese-speaking regions. Further validation would be necessary to ensure appropriateness for Brazilian Portuguese speakers, given important lexical and contextual differences.
Conclusion
This study provides initial evidence supporting the semantic and contextual relevance of a Portuguese version of the IPDAS Minimal Criteria instrument. Through a structured process of translation, adaptation, and expert review, the instrument demonstrated strong content validity and high agreement regarding clarity and relevance of its items. While the results are encouraging, the scope of this study was limited to linguistic and contextual adaptation; broader aspects of cultural adaptation were not evaluated.
The findings of this study demonstrate the critical role of expert panels in the translation and cultural adaptation of instruments such as the IPDASi. Despite limitations in interrater agreement due to methodological constraints of the rating scales used, the combined results from content validity indices and agreement measures support the use of this instrument for evaluating PDAs in Portugal.
This study addresses the significant gap in the availability of high-quality decision aids in European Portuguese to facilitate better patient involvement in health care decisions. Future studies should focus on broader validation efforts to establish the psychometric properties of the Portuguese IPDASi and ensure its reliability and validity in practice.
Not applicable.
Supplemental Material
sj-docx-1-mpp-10.1177_23814683251386451 – Supplemental material for Translation, Cultural Adaptation, and Validation of the International Patient Decision Aid Standards Minimal Criteria Instrument for the Portuguese Population
Supplemental material, sj-docx-1-mpp-10.1177_23814683251386451 for Translation, Cultural Adaptation, and Validation of the International Patient Decision Aid Standards Minimal Criteria Instrument for the Portuguese Population by Micaela Gregório, Andreia Teixeira, Mariana Teixeira, Inês Marques, Ana Sofia Correia, Phillippa May Bennett, Helen Carter, Dawn Stacey and Carlos Martins in MDM Policy & Practice
Footnotes
Acknowledgements
The authors would like to acknowledge the valuable contributions of the expert panel members who participated in the pretest phase for their professionalism and hard work. We would like to thank Editage (![]() ) for English language editing. The authors affirm that all individuals who contributed significantly to the scientific content or technical aspects of the study have been acknowledged.
) for English language editing. The authors affirm that all individuals who contributed significantly to the scientific content or technical aspects of the study have been acknowledged.
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This article was supported by National Funds through FCT - Fundação para a Ciência e a Tecnologia,I.P., within CINTESIS, R&D Unit (reference UIDP/4255/2020). The funding agreement ensured the authors’ independence in designing the study, interpreting the data, writing, and publishing the report.
Ethical Considerations
The study protocol was reviewed and approved by the Ethics Committee of the São João Hospital Centre/Faculty of Medicine of the University of Porto (approval No. 107/CEFMUP/2023) in June 2023. All procedures were conducted in compliance with the Declaration of Helsinki. Informed consent was obtained from all participants prior to their inclusion in the study.
Consent to Participate
Participants gave verbal consent to participate in the beginning of each Zoom meeting.
Consent for Publication
Not applicable.
Data Availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
