Abstract
Highlights
A new oral cancer simulation model with risk factors including degrees of smoking and alcohol exposure, oral lesion features, and sex incorporates more accurate and precise representation of patient risk categories.
We evaluated screening strategies for oral potentially malignant disorders with or without risk-stratified biopsy referral in both the general population and subpopulations defined by degrees of smoking and alcohol exposure.
Men with a high degree of both smoking and alcohol exposure exhibited a significant reduction in cancer-specific deaths and cancer incidence from screening programs for oral potentially malignant disorders.
Screening with risk-stratified biopsy, using a surgical treatment threshold of moderate dysplasia or worse, yielded the greatest efficiency in term of biopsies needed to detect 1 treatable case.
Keywords
The incidence of and mortality from oral cavity and pharyngeal cancers has not improved over recent decades, and the prognosis remains poor despite advances in cancer treatment.1,2 Oral cavity cancers constitute two-thirds of all oral cavity and pharyngeal cancers, and more than 90% of these are oral squamous cell carcinoma (OSCC).1,3–5 The US Cancer Statistics Public Use Database reported a steady rise in the burden of OSCC cases, with 10,616 in 2001 to 13,856 in 2017 among all ages. 6 The age-adjusted incidence of OSCC also steadily increased from 2005 to 2019, with an annual percentage change of 1.07%, based on the data from the National Program of Cancer Registries and Surveillance, Epidemiology, and End Results (SEER) Program. 7 For OSCC, survival is highly dependent on the cancer stage at diagnosis; 5-y survival for cancer stage I and II combined is 74% compared with 38% in cancer stage III and IV combined.8–11 Thus, the detection of early-stage OSCC or dysplasia, which are at risk of malignant transformation, represent a major opportunity for better patient mortality and morbidity. Visual–tactile examination entails clinicians taking a look in the oral cavity using their hands to manually reposition and aims to discover oral potentially malignant disorders (OPMDs), specifically leukoplakia and erythroplakia, which may represent both premalignant or malignant lesions but also include some benign lesions with overlapping appearance. Even when performed by trained experts, 12 inaccurate clinical assessment can lead to false positives and excess referrals for biopsy, the gold standard for diagnosis, or false negatives and nonreferral. These limitations represent major barriers to the design and implementation of potential screening programs.
The American Dental Association has recommended visual–tactile oral examination for all adults as part of initial, routine, and emergency dental visits. 13 Despite the recommendation, visual–tactile examination is inconsistently performed,14–16 and only 29% of US adults have reported ever receiving an oral cancer examination. 16 Fear of excess unnecessary referrals and patient burden contribute to such reluctance on the part of dental providers, given the rarity of oral cancer in the general population.14,15 Screening programs in some countries such as India 17 and Taiwan 18 are based on the visual–tactile examination of high-risk individuals (i.e., those with risk factors such as tobacco, areca nut, or heavy alcohol use), 19 but in the United States, questions remain about the specific population that would likely experience favorable benefits compared with harms and the potential screening schedule associated with optimal clinical utility. Since trials for less common cancers are limited by cost and duration, disease simulation modeling is applied to understand whether test and population characteristics exist that support screening trials. Such models estimate the comparative effectiveness of test-and-treat decisions in terms of reduction of late-stage disease burden, life expectancy (LE), and also overall diagnostic efficiency.20,21
Risk stratification based on risk exposures and OPMD features could be a key determinant of precision oral cavity screening. Men have a 28% increased odds of oral lesions 22 and an 87% higher risk of developing OSCC compared with women,22,23 and heavy alcohol consumption and high smoking intensity are consistently associated with increased odds of developing OSCC.24–26 The combination of alcohol consumption and tobacco smoking also produces a synergistic effect. 24 In addition, patients with OPMDs with clinical features such as nonhomogeneous texture, mixed red/white color, size exceeding 200 mm2, and high-risk anatomic site carry an increased risk of malignant transformation (MT).27–29
Our purpose was to develop a new disease simulation model for the development and progression of OPMDs and oral cavity cancers in US men and women at 40 y of age. The model incorporates specific risks for evaluating more precise screening and management pathways. We evaluated the effectiveness of potential screening, beginning at age 50 y, by visual–tactile examination conducted during a routine dental visit, in both the overall population (all risk groups) and subpopulations ranging from very low to high risk according to degrees of smoking and alcohol exposure, using the medical sector perspective. 30
Methods
Simulation of Cancers of the Oral cavity and Risk Exposures (SCORE) is a discrete time, state transition microsimulation model that expanded upon a predecessor model 31 with an inclusion of behavioral risk factors and OPMD features. The model simulated a closed hypothetical cohort of 40-y-old US men and women using a lifetime horizon. Modeling from age 40 y allows for the accumulation and progression of OPMDs prior to screening initiation at age 50 y, capturing the influence of behavioral risk exposures on lesion development and progression that may affect screening outcomes. The individual-level characteristics of the cohort include smoking and alcohol exposure, presence/absence of OPMD, and various OPMD features. This article provides a description of SCORE, including model structure, model inputs, and associated assumptions, and is provided in accordance with the model reporting guidelines (ISPOR-SMDM).30,32–35
Natural History Model Overview
SCORE models the natural history of cancer development and progression in patients presenting both with and without OPMDs using a monthly cycle. The model also includes benign disorders with visual characteristics that overlap those with dysplastic OPMDs and cancers. The model simulated patients with the same age and sex and assigned individual risk behaviors and OPMD features that inform differential risks of disease development and progression. For patients presenting with OPMDs, SCORE assigned OPMD features including phenotypes, appearance, anatomic sites, and initial disease severity by randomly sampling from the categorical distributions informed by oral cancer screening studies (Table 1). The spectrum of disease severity (disease states) included benign disorders such as reactive/frictional keratoses; dysplastic OPMDs with grades ranging from mild, moderate, and severe dysplasia or carcinoma in situ; and OSCC with stages ranging from stage I to IV. Disease transitions follow a Markov model with nonhomogeneous individual transition matrix based on individual-level characteristics and OPMD features, resulting in probabilistic model outcomes. SCORE was implemented in Python 3.9.
Base-Case Parameters for the US Population
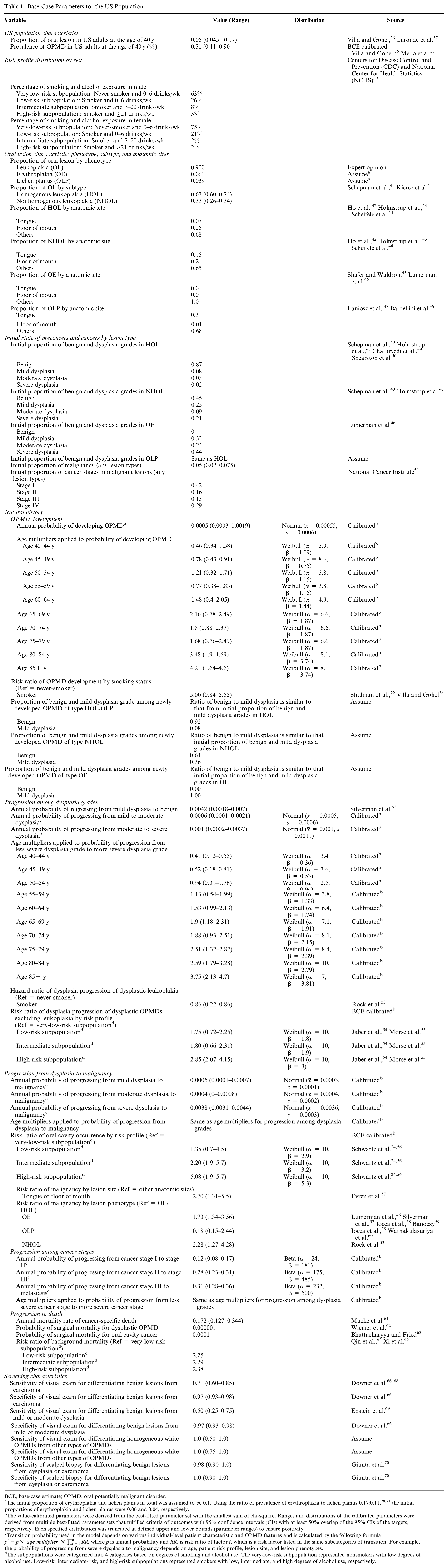
BCE, base-case estimate; OPMD, oral potentially malignant disorder.
The initial proportion of erythroplakia and lichen planus in total was assumed to be 0.1. Using the ratio of prevalence of erythroplakia to lichen planus 0.17:0.11,38,71 the initial proportions of erythroplakia and lichen planus were 0.06 and 0.04, respectively.
The value-calibrated parameters were derived from the best-fitted parameter set with the smallest sum of chi-square. Ranges and distributions of the calibrated parameters were derived from multiple best-fitted parameter sets that fulfilled criteria of outcomes with 95% confidence intervals (CIs) with at least 50% overlap of the 95% CIs of the targets, respectively. Each specified distribution was truncated at defined upper and lower bounds (parameter ranges) to ensure positivity.
Transition probability used in the model depends on various individual-level patient characteristic and OPMD features and is calculated by the following formula:
The subpopulations were categorized into 4 categories based on degrees of smoking and alcohol use. The very-low-risk subpopulation represented nonsmokers with low degrees of alcohol use. Low-risk, intermediate-risk, and high-risk subpopulations represented smokers with low, intermediate, and high degrees of alcohol use, respectively.
Dysplastic OPMDs progressed by increasing in grades of dysplasia or to OSCC stages based on the conditional transition probability of the current disease state (Figure 1). Mild dysplasia was susceptible to either regressing to a normal state or to progression in dysplasia grade. 52 During each monthly cycle, all patients were subject to background mortality, where we applied the US life table and risk ratios of background mortality to account for higher mortality rates in those with higher degrees of exposure to smoking and alcohol.64,65 Only patients with metastasis were additionally subject to oral cancer death. We assumed the transition probabilities in adults aged ≥40 y increased with age following 1) increasing observational oral cavity cancer incidence by age from SEER 51 and 2) the association between older ages and MT rates based on Cox proportional hazard analysis using data on patients with OPMDs from a large retrospective study. 60
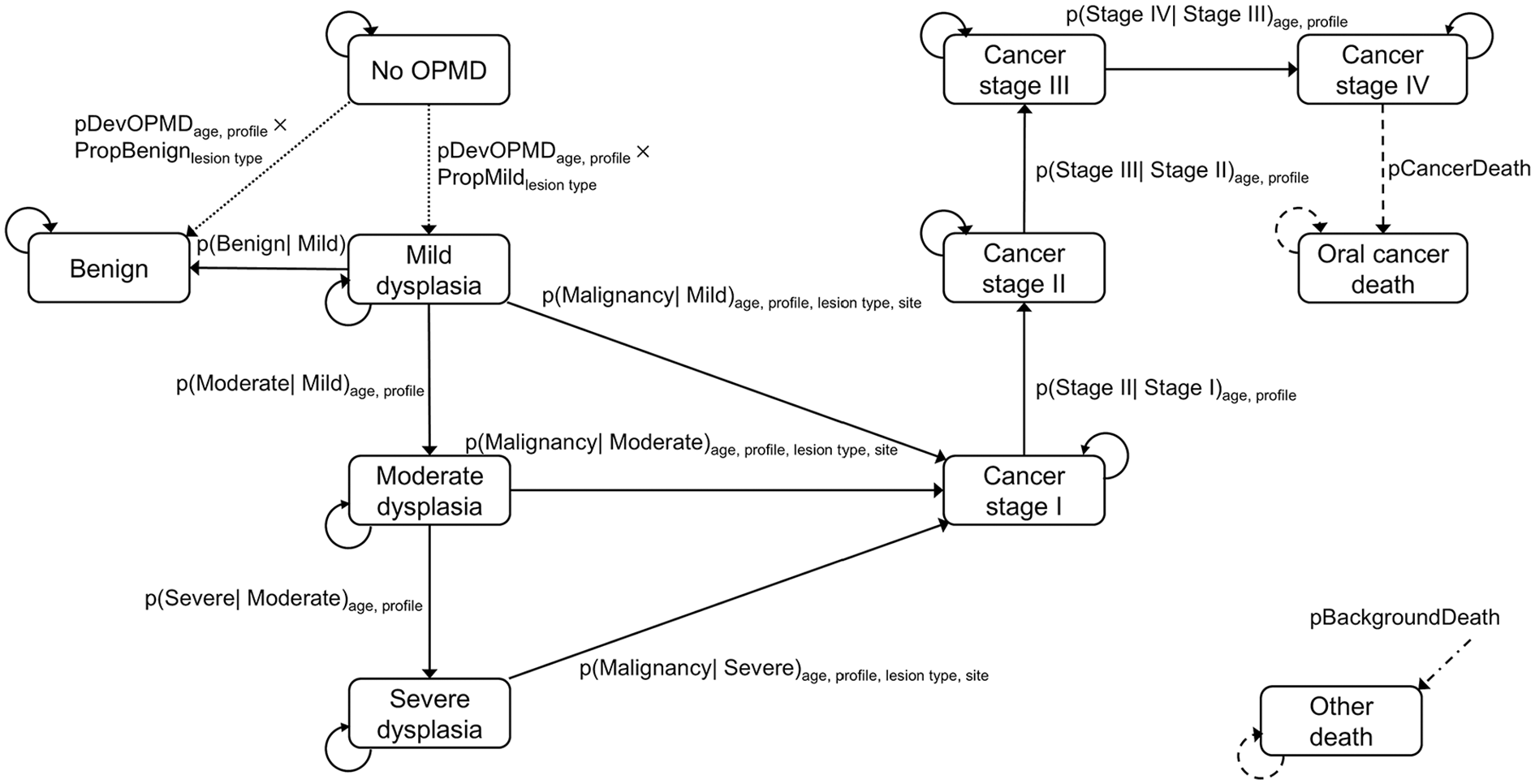
State transition diagram for the natural history model of benign disorders, dysplastic oral potentially malignant disorders (OPMDs), and oral squamous cell carcinoma (OSCC).
Patient Risk Factors and OPMD Features
As smoking and alcohol exposure are well-established risk factors associated with the development of OPMD 36 and MT,24,56 we constructed 4 patient risk profiles capturing the two factors. The joint prevalence of smoking and alcohol use across degrees of exposure by sex were informed by our assessment of the National Health and Nutrition Examination Survey (NHANES) longitudinal series 2009–2018 39 ; for example, 63% of men were nonsmokers, with the lowest degree of alcohol exposure (very low-risk subpopulation), while the remaining 27% were smokers from low to high degrees of alcohol exposure (subpopulations of low, intermediate, and high risk) (Table 1). Smokers in the three risk profiles were defined as long-time smokers (lifetime smoking duration ≥21 y) with both low smoking intensity (1–20 cigarettes per day) and high smoking intensity (≥21 cigarettes per day). Rates of OPMD formation and progression in dysplasia or cancer differed depending on risk exposures. Compared with nonsmokers, smokers were more likely to develop dysplastic OPMDs and those with higher degrees of alcohol exposure were associated with a higher risk of OSCC occurrence.24,54–56 However, leukoplakia, the most prevalent type of OPMD, exhibited higher rates of progression from dysplasia to OSCC when developing in the absence of smoking and alcohol use, as previously reported. 53
We modeled the development of 3 common phenotypes of OPMDs that are defined by the main features of color and texture: leukoplakia (further delineated as homogeneous and nonhomogeneous leukoplakia), lichen planus, and erythroplakia.45,72–74 Evidence has shown that nonwhite color or nonhomogeneous texture are among high-risk features for MT. 75 For example, erythroplakia (i.e., red-colored OPMDs) exhibits the highest rate of MT among the 3 common phenotypes. 58 A meta-analysis reported the overall rates of MT of 33.1%, 9.5%, and 1.4% in erythroplakia, leukoplakia, and lichen planus, respectively. Within the 2 subtypes of leukoplakia, the nonhomogeneous subtype is associated with a 2.3 times higher rate of MT. 53 In addition to color and texture, certain anatomic sites also portend greater risk; OPMDs on the tongue or floor of mouth exhibit 2.7 times higher risk of MT than those located at other sites do. 57 Accordingly, we incorporated OPMD phenotypes associated with different rates of MT into the model, with a representative spectrum of histology and anatomic location (Table 1).
Model Calibration
To ensure the model reflects trends in oral cavity cancer, we calibrated SCORE using SEER data from 2004 to 2010. We used more recent SEER data (2011–2017) for subsequent validation, described in the next section. To represent current population risk behaviors, we incorporated smoking and alcohol use data from NHANES 2009–2018.
For the overall population comprising 4 subpopulations of very-low-risk to high-risk individuals (Table 1), we calibrated SCORE to several targets: 1) age-specific oral cavity cancer incidence, 2) OSCC stage distribution at the age of diagnosis of 50 y, and 3) cancer-specific mortality risk derived from SEER 17 data based on oral cancer cavity cases diagnosed in 2004 to 2010. 51 Thirty-six clinical parameters relevant to natural history were simultaneously calibrated, including the prevalence of OPMD for adults at age 40 y, the probability of OPMD development, the probability of progression in dysplasia grades, and probabilities of progression of dysplasia to malignancy and to successive cancer stages (Table 1). Differential rates of OSCC development by patient risk profile were generated using a risk ratio specific to each profile compared with never-smokers. To search for the best-fitted parameters, the sum of chi-square was used as the goodness-of-fit measure.76,77 To generate the model outcomes, we simulated the overall population of 2 million with 500 repetition iterations accounting for first-order uncertainty (Supplementary Table S1). SCORE was calibrated for US men and women separately using a heuristic search consisting of 3 steps: 1) eliminating infeasible parameter spaces, 2) reducing parameter spaces using Bayesian optimization with an objective function of minimizing the sum of the chi-square between the model outcome and targets,78,79 and 3) iteratively performing Latin-hypercube sampling on the reduced parameter spaces. Correlation coefficients were analyzed to assess the independence assumption of parameters. We define the best-fitted parameter sets as those that fulfilled 2 criteria: 1) the mean of model outcomes must fall within the 95% confidence interval (CI) of the calibration target, and 2) the 95% CI of the model outcomes must overlap by at least 50% with the 95% CIs of the targets. We then fit distributions across all the best-fitted parameters. The parameter set with the smallest sum of chi-square is defined as the base-case parameter set. We derive the ranges of calibrated parameters from the best-fitted parameter sets.
Model Validation
We validated SCORE against age-specific oral cavity cancer incidence, stage distribution, and cancer-specific mortality derived from SEER data from 2011 to 2017. 51 The predefined validation criterion is overlapping 95% CIs between the model outcomes and targets for incidence and mortality and standard error (SE) ≤0.5% for stage distribution, respectively. We additionally validated our model for the overall population of men by comparing SCORE’s LE in 50-y-old men to the SEER Oral Cancer Survival Calculator (OCSC).80,81 This calculator provides 3 submodels to estimate LE without cancer depending on patients’ age and health conditions and uses a competing risk model to estimate the probabilities of death from oral cancer and death from other causes using National Health Interview Survey. 82 To validate, we assessed whether model LE fell within the ranges generated using the SEER OCSC.
Application: Evaluation of Representative Screening Strategies
To provide an example of the application with SCORE, we evaluated 4 hypothetical screening strategies against no screening. Thus, 5 strategies were compared: 1) No Screening, 2) Biopsy Referral for all OPMDs with Moderate Treatment Threshold: Screening with biopsy referral for all OPMDs with surgical treatment threshold of moderate dysplasia or worse, 3) Biopsy Referral for all OPMDs with Severe Treatment Threshold: Screening with biopsy referral for all OPMDs with surgical treatment threshold of severe dysplasia or worse, 4) Risk-stratified Referral for Biopsy with Moderate Treatment Threshold: Screening with decision to refer for biopsy based on a positive smoking history or for OPMDs that are nonhomogeneous, nonwhite, or at a high-risk site 75 with a surgical treatment threshold of moderate dysplasia or worse, and 5) Risk-stratified Referral for Biopsy with Severe Treatment Threshold: same biopsy referral criteria as 4) but with surgical treatment threshold of severe dysplasia or worse (Figure 2 and Supplementary Table S2). With no screening, only patients with cancer presented to care, underwent biopsy, and then received treatment according to OSCC stage at diagnosis. For the screening strategies, screening was assumed to initiate at the age of 50 y, occurred every 3 y in the base-case analysis, and ended at the age of 75 y. We assumed patients have 100% adherence to dentist visits for screening and received the visual–tactile examinations during their visits. When an initial risk stratification was included in the visual–tactile examination, only OPMDs with the concerning features were referred for biopsy. Positive biopsies were treated with surgical excision while lesser disease states underwent surveillance. Two definitions of a positive biopsy, namely, moderate and severe treatment thresholds, were considered in the 4 management strategies. When moderate dysplasia was the threshold for positivity, surgical excision was performed for moderate or severe dysplasia or localized carcinoma. Meanwhile, surveillance was undertaken for nondysplastic OPMDs and mild dysplasia. When the threshold was severe dysplasia, severe dysplasia and carcinoma were then treated with surgery. The surveillance duration was 5 y and occurred every 6 mo during the first year and every year thereafter.

Screening schematics for the 5 strategies.
In the base case, we assumed that the sensitivity and specificity of visually differentiating homogeneous white OPMDs from other types of OPMDs (red or nonhomogeneous white) was 100% in the risk-stratified referral for biopsy strategies. In the sensitivity analysis, the sensitivity and specificity of visually differentiating homogeneous white OPMDs from other OPMDs varied from 100% to 71% and 97%, respectively, such that the performance was similar to distinguishing between benign and malignant lesions on the standard visual tactile examination. 66 Patients were subject to surgical mortality rates according to the literature.62,63 For each strategy, we simulated each subpopulation of 2 million patients and 500 repetition iterations with the starting age of 40 y using a lifetime horizon and all-cause mortality from the 2011 US life table. 83 The outcomes of the overall population were derived from the weighted average of subpopulation outcomes using a distribution of 4 subpopulations—very-low, low, intermediate, and high risks, defined by degrees of smoking and alcohol exposure (Table 1)—as corresponding weights. Using the medical sector perspective, 30 we evaluated the primary outcomes of LE in patients who developed OPMDs from age 40 to 60 y and the intermediate outcomes of cancer incidence, cancer-specific deaths, and the number of biopsies accumulated over lifetime. The evaluation was conducted for the overall cohort of men and women separately in the base-case analysis. To identify efficient strategies, we compared the tradeoff of benefit gain and harm of screening strategies, both against no screening. Benefit gains were calculated as a reduction in the cumulative cancer incidence and cancer-specific deaths. Harm was calculated as an increase in the number of biopsies. The strategies were sorted by the order of benefits, and any strategies that were strongly dominated by others were ruled out (less benefit and more harm). We calculated the incremental benefit–harm ratio for each strategy by comparing it with the next most effective strategy. The strategy was efficient if it provided higher benefit with less harm as compared with alternative strategies.84,85 The identified efficient strategies were presented as a frontier plot, where the dominated strategies were under the frontier.
For the scenario analyses, we evaluated 2 scenarios: 1) a longer screening interval of 10 y and 2) infrequent dental visits in which an adherence to a dental visit was reduced from 100% to 50%.86,87 Sensitivity analysis was performed by varying major parameter values (1-way deterministic analysis) to assess the impact on model results. Major parameters included the prevalence of OPMD at the age of 40 y, clinical parameters relevant to natural history, and screening characteristic parameters (Table 1).
Results
Model Calibration
SCORE was well-calibrated to all 3 targets from SEER. Among the 100 best-fitted parameter sets, we found weak correlations among the 36 calibrated parameters, supporting the validity of independence assumption. Specifically, of 630 calibrated parameter pairs representing all unique combinations of 2 parameters selected from 36 calibrated parameters (i.e., 36 choose 2, without repetition;

Model calibration results for the overall population comparing Simulation of Cancers of the Oral cavity and Risk Exposures (SCORE) model outcomes against Surveillance, Epidemiology, and End Results (SEER) 2004–2010.
Model Validation
In the overall population, SCORE provided well-matched outcomes to observations from SEER 2011–2017 with the sum of chi-squares ranging from 2.97 to 13.03 (SE: 0.35) (Supplementary Figure S1). The 95% CIs of cancer incidence rates from SCORE overlapped with those from SEER 2011–2017 in all age groups with a slightly overestimated incidence in age 55 to 60 y and age 75 to 85+ y (Supplementary Figure S1A). Compared with SEER, SCORE estimated a closely matched distribution of stages at diagnosis with SE ≤ 0.42% across all stages (Supplementary Figure S1B). In addition, SEER 2011–2017 reported the 5-y cancer-specific survival rate at stage IV cancer of 38.4% (95% CI: 33.3%–43.6%), while SCORE simulated the 5-y rate of 41.0% (95% CI: 33.1%–48.8%). In addition, for 50-y-old men, SCORE estimated an LE without cancer of 28.5 y (95% CI: 26.8–30.3) and 19.8 y (95% CI: 17.8–22.2) in nonsmokers and smokers, respectively. The estimated LE fell within SEER OCSC reported ranges of 23 to 34 y for nonsmokers and 17 to 30 y for current smokers.
Application: Evaluation of Representative Screening Strategies
All screening strategies yielded similar LE in patients who developed OPMDs from age 40 to 60 y of 36.37±0.01 life-years (+0.03 y compared with no screening) in the general population of 50-y-old men and 30.97 ± 0.01 life-years (+0.02 y compared with no screening) in the high-risk subpopulation (Table 2). Screening, whether with or without risk-stratified biopsy, and paired with a treatment threshold of moderate dysplasia, provided the highest reduction in cumulative oral cancer incidence of 30%. All screening strategies yielded a similar reduction in cancer-specific death of at least 20% compared with no screening. Compared with screening with biopsy referral for all OPMDs, screening with risk-stratified biopsy referral yielded 30% fewer biopsies (1,300–1,314 v. 1,877–1,977 biopsies per 100,000 patients), leading to 8 to 12 biopsies needed to diagnose 1 treatable condition in persons receiving first biopsies and with minimal difference in LE in patients aged 40 to 60 y who developed OPMDs, cancer incidence, or cancer-specific death.
Comparison of Primary and Intermediate Outcomes of Screening and No Screening Strategies for the Base Case of Screening Starting at Age 50 y in US Men with a 3-y Screening Interval

LE, life expectancy; OPMDs, oral potentially malignant disorders. All outcomes are reported as mean (95% interpercentile range).
The numerator is the number of patients with OPMDs receiving first biopsies, and the denominator is the number of treatable cases from first biopsies.
Treatable cases include only early-stage cancers consisting of cancer stage I and II.
Treatable cases include precancers consisting of moderate and severe dysplasia and early-stage cancers.
Treatable cases include precancers of severe dysplasia only and early-stage cancers.
For the first scenario analysis, we evaluated an increase in screening intervals to 10 y from the initial assumption of 3 y. This further reduced the biopsy burden by approximately 244 to 409 biopsies per 100,000 individuals without a substantial change in LE in patients aged 40 to 60 y who developed OPMDs or overall cancer incidence but with a reduction in early-stage cancer detection by 10% to 13% compared with 3-y screening intervals (depending on the chosen treatment threshold) (Supplementary Table S3). In the second scenario analysis, we evaluated screening strategies with 3-y screening intervals with a decrease in adherence to a dental visit to 50% from the initial assumption of 100%. Compared with the base-case analysis, there was no substantial change in LE in patients aged 40 to 60 y who developed OPMDs or overall cancer incidence (Supplementary Table S4). However, there was a reduction in early-stage cancer detection by 7% to 9% in the general population and by 7% to 10% in the high-risk population, respectively (depending on the chosen treatment threshold). For all scenarios, there were minimal differences in the overall LE across all 5 strategies (Supplementary Table S6).
In women, similar to men, screening with risk-stratified biopsy strategies yielded a significantly lower biopsy burden (960–961 v. 1,402–1,468 biopsies per 100,000 patients) with similar LE (−0.01 y), cumulative oral cancer incidence, and cancer-specific death when compared with screening with biopsy referral for all OPMDs (Supplementary Table S5). In addition, screening with risk-stratified biopsy strategy and a treatment threshold of moderate dysplasia led to reductions in the cumulative number of detected treatable cases of 3% to 6% in women and 3% to 4% in men, compared with biopsy referral for all OPMDs, depending on treatment threshold.
Furthermore, we investigated the outcomes by patient risk profiles. Considering the incidence reduction and biopsy burden in very-low-risk men, both screening with and without risk-stratified biopsy and paired with a treatment threshold of moderate dysplasia were on the frontier (Figure 4). Specifically, screening with a biopsy referral for all OPMDs yielded an additional reduction in the incidence of 0.27 per 100,000 patients with an increment in biopsies of 308 per 100,000 patients compared with risk-stratified biopsy (Figure 4A). Similar results were observed in low-risk men. In intermediate-risk and high-risk men, screening with risk-stratified biopsy with a treatment threshold of moderate dysplasia yielded similar benefits and harms to screening with a biopsy referral for all OPMDs at the same treatment threshold. Screening with biopsy referral for all OPMDs with a moderate treatment threshold was on the frontier, providing an additional incidence reduction of only 0.15 per 100,000 patients, compared with screening with risk-stratified biopsy at the same treatment threshold (Figure 4A). However, when comparing cancer-specific death averted to biopsy burden, screening with risk-stratified biopsy with a treatment threshold of moderate dysplasia was on the frontier, with only an additional 0.12 cancer-specific deaths averted per 100,000 patients (Figure 4B). Screening with and without a risk-stratified biopsy with a treatment threshold of moderate dysplasia yielded the highest biopsy efficiency of 7 to 13 biopsies needed to detect 1 treatable case across all 4 subpopulations (Supplementary Figure S2).

Comparison between the following benefit gained and burden of screening strategies compared with no screening.
Sensitivity Analysis
In one-way sensitivity analysis, each major parameter was varied across a plausible range. The OSCC incidence and disease-specific death were affected mostly by changes in the probability of developing an OPMD, the probability of progressing from mild dysplasia to malignancy, the probability of progressing from moderate to severe dysplasia (Supplementary Figures S3 and S4). The comparative favorability of strategies remained stable.
Discussion
We developed, calibrated, and validated a new microsimulation model (SCORE) of the natural history of OPMD development and progression of OPMD and OSCC to enable virtual trials in US men and women aged 40 y and their subpopulations. SCORE differs from other existing models with the incorporation of major behavioral risk factors and OPMD features that are associated with disease development and progression and allowed for various phenotypes of OPMD development in a cohort of patients under simplified assumptions. As such, SCORE can be used to bridge the gap in population screening evaluation for the United States and evaluating various OPMD screening and management strategies that use behavioral risk factors and/or OPMD features. Accordingly, we evaluated the potential benefits and harms of alternate screening strategies.
Our results showed that screening with risk-stratified biopsy led to a minimal improvement in primary and intermediate outcomes in very-low-risk persons. However, both screening and no screening strategies led to a similar overall LE in the general population and subpopulations, primarily due to the low prevalence of OPMDs among US men and women and the high proportion of benign disorders among OPMDs. Screening strategies, whether with or without risk-stratified biopsy, yielded comparably significant improvement in cancer-specific death and cancer incidence in high-risk persons. Compared with screening strategies with biopsy referral for all OPMDs, screening strategies with risk-stratified biopsy could greatly reduce biopsy burden in patients with low exposure to smoking and alcohol use while maintaining similar levels of reduction in cancer incidence and cancer-specific death and increase in biopsy efficiency. This finding underscores the importance of the use of risk stratification of OPMDs in management decisions. Furthermore, risk stratification models that improve upon simple decision rules based on low versus non-low-risk OPMD features may further improve the benefits and harms considerations for screening. In addition, SCORE demonstrated that population screening for OPMDs best improved cancer detection when risk-stratified biopsy tempered the procedure burden and enabled diagnostic yield comparable with established cancer screening programs such as cervical cancer screening. 88 We believe that SCORE may be useful for guiding health policy and identify high-priority clinical trials in precision oral cancer screening.
A prior decision analytics study by Dedhia et al. 89 examined a yearly community-based screening program in 40-y-old US high-risk men who regularly use tobacco and/or alcohol over a time horizon of 40 y and found that screening is more effective and less costly compared with no screening. Our model emphasizes additional risk factors that are associated with malignant transformation: degrees of smoking and alcohol exposure ranging from very low to high, dysplasia grade spectrum in “precancer,” and OPMD features of color, texture, and sites. Specifically, SCORE modeled various dysplasia grades exhibiting within precancer, while Dedhia et al. modeled only one “precancer” state. However, both models considered the potential regression of dysplastic OPMD to a healthy state. SCORE also assumed older ages were associated with higher probabilities of disease progression, whereas Dedhia et al. assumed constant probabilities of progression. SCORE uniquely simulates US patients with and without OPMD development and incorporates both patient risk factors and OPMD features that were influential to OPMD development and MT.
Our study had several limitations. Model assumptions of disease development and progression were simplified using a multiplicative effect for behavioral risk factors and OPMD features observed to elevate the risk of malignancy. The existing literature reported a greater than multiplicative effect when only behavioral risk factors were considered in association with risk of malignancy, 90 although confounding and selection bias may also be reflected. Some of the model parameters including the initial proportions of OPMD anatomic sites and grades by each OPMD phenotype were informed by US survey data on oral lesions or described OPMD characteristics in patients with OPMDs, and misclassification errors are possible.40,42–44,46–49 As OPMDs were not common in younger patients, there is a limited literature on screening studies for younger age groups. Although mortality risks should be adjusted for major comorbidities and smoking and alcohol exposures, we include only an increase in all-cause death due to smoking and alcohol due to the lack of comorbidity-adjusted life tables that accounted for smoking and alcohol exposure.
In conclusion, SCORE is a new microsimulation tool for the virtual trial for specific risk groups who may undergo screening and management for OPMDs. We applied SCORE to the US population and key risk-stratified subpopulations, and our results point to specific future research needs evaluating screening strategies augmented by rule-based biopsy referral using risk factors and specifically the possibility that more advanced testing technologies could further increase the efficiency in biopsy. By enabling analyses that assess the downstream impact of risk factors, OPMD features, screening regimen, and other test performance, SCORE will be available to address additional policy and practice questions for improving oral cancer control.
Supplemental Material
sj-docx-1-mpp-10.1177_23814683251353226 – Supplemental material for Simulation Modeling of Oral Cancer Development with Risk Stratification: How Potential Screening Programs Can Be Evaluated
Supplemental material, sj-docx-1-mpp-10.1177_23814683251353226 for Simulation Modeling of Oral Cancer Development with Risk Stratification: How Potential Screening Programs Can Be Evaluated by Mutita Siriruchatanon, Emily R. Brooks, Alexander R. Kerr, Denise M. Laronde, Miriam P. Rosin and Stella K. Kang in MDM Policy & Practice
Footnotes
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr. Kang reports royalties from Wolters Kluwer for unrelated work. The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Financial support for this study was provided entirely by a grant from the National Institutes of Health/National Institute of Dental and Craniofacial Research (R01DE030169, principal investigator: Kang). The funding agreement ensured the authors’ independence in designing the study, interpreting the data, writing, and publishing the report. The data generated in this study are not publicly available due to protections of patient privacy and the scope of informed consent, but summaries of the data may be available upon reasonable request from the corresponding author
Ethical Considerations
This study is based entirely on simulated data generated using the authors’ simulation model. Model inputs were derived from previously published studies, all of which are appropriately referenced. The study does not involve any human participants, identifiable personal data, or real-world experimental interventions. Therefore, ethical approval from an institutional review board was not required. As the data were fully simulated, there are no ethical concerns related to human or animal research.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Data Availability
The data generated in this study are not publicly available due to protections of patient privacy and the scope of informed consent, but summaries of the data may be available upon reasonable request from the corresponding author.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
