Abstract
Highlights
ACIP recommends shared clinical decision making for MenB vaccination.
Data were collected from young adults and parents of adolescents by online survey.
We measured values and consultation preferences on MenB disease and vaccination.
Young adults/parents strongly preferred doctor-initiated MenB vaccine discussion.
Respondents were willing to pay for a MenB vaccine.
Keywords
Invasive meningococcal disease (IMD) is a serious infection caused by the bacteria Neisseria meningitidis, with the average case fatality rate ranging from 10% to 20% 1 ; up to 20% of survivors can experience permanent or long-term sequelae, including severe neurologic, visual, or hearing impairments.1,2 Adolescents and young adults are a particularly vulnerable population, with high meningococcal carriage rates that peak at 19 y of age.3,4 The age-typical social-mixing behaviors of adolescents and young adults (eg, kissing, living in close quarters) contribute to the spread of the disease among this population. 5 According to the most recently available (2019) US Centers for Disease Control and Prevention (CDC) Enhanced Meningococcal Surveillance data, most IMD cases in the United States are caused by serogroup B (MenB), including 49% of IMD cases among individuals 16 to 23 y of age and 26% of all IMD cases across age groups. 6
Two vaccines are required for protection against the 5 predominant disease-causing meningococcal serogroups.7,8 The US Advisory Committee on Immunization Practices (ACIP) recommends routine MenACWY vaccination at 11 to 12 y of age followed by a booster dose at 16 y of age. 9 MenB vaccination is recommended at 16 to 23 y of age (16–18 y preferred) on the basis of shared clinical decision making, and the CDC has provided guidance explaining this recommendation.9,10 In 2021, MenACWY vaccination coverage was 89.0% for ≥1 dose and 60.0% for ≥2 doses, whereas MenB vaccination coverage was 31.4% for ≥1 dose. 11
The ACIP uses the Evidence to Recommendations (EtR) framework, adopted in 2018, to gather and evaluate evidence to develop vaccination recommendations.12,13 The EtR framework provides a structured and transparent way to inform clinical recommendations, coverage decisions, and health system or public health recommendations/decisions based on clinical evidence of vaccine effectiveness, patient values and preferences, comparative health benefits, and acceptability among stakeholders.13–16 In 2011, the CDC engaged with representatives from stakeholder groups and members of the public regarding meningococcal vaccines in infants and toddlers, but not adolescents, to examine values about the use of licensed vaccines to protect children from rare but severe illness. 17 When MenB vaccines were first recommended in 2015, no formal studies were conducted as part of the EtR process to investigate the values and preferences of the adolescent vaccine population. 18 Now that the ACIP is placing more emphasis on patient values and preferences, including their willingness to pay (WTP) for vaccines, it is important to use stated-preference surveys to support the formal decision-making process. 15
Our objective was to quantify the perceptions of young adults and their parents about serogroup B disease and vaccines and the value they placed on vaccination using standard stated-preference study methods recommended by the ACIP. 19 Specifically, given the ACIP recommendation for shared clinical decision making between the patient and their health care provider (HCP), this study examined the value families place on information shared by a provider regarding the option of MenB vaccination and quantified the amount patients would be willing to pay for a discussion with their HCP about MenB vaccination and the amount they would be willing to pay for the vaccine itself.
Methods
Design and Respondents
The study design followed good research practices for health-related choice-experiment studies. 20 A detailed description of the information provided to the respondents has been published elsewhere. 21 Briefly, this included information about both the disease and the vaccine to help protect against the disease (ie, the effect of the disease, how many people get the disease each year, how the disease spreads, how long the vaccine lasts and, finally, the cost of the vaccine). A draft survey was pretested and modified in 25 “think-aloud” interviews. Participants were paid $75 for a 1-h interview.
The final survey was administered online by Ipsos Knowledge Panel, an international survey-research company. Respondents were recruited via e-mail or through a personalized online portal from the Ipsos Knowledge Panel. Respondents in the Ipsos Knowledge Panel received modest compensation in accordance with standard practice in the industry. This online national opt-in consumer panel is a probability-based, representative, random sample of US households. Young adults (18–25 y of age) or parents/guardians of young adults aged 12 to 25 y were eligible to participate and were required to be able to read and understand English. All respondents provided explicit consent before participating in the study. The Web-enabled stated-preference survey22,23 was conducted across the United States from August 22, 2019, to October 2, 2019.
Instrument
The Web-enabled survey instrument used contingent-valuation (CV) questions to quantify the monetary value that young adults placed on 1) additional time to consult with their doctor about MenB vaccination and 2) being vaccinated with the MenB vaccine in general. For parents of young adults, the CV questions quantified the monetary value of 1) additional time to consult with their doctor about MenB vaccination for their teenage or young adult children and 2) MenB vaccination for their teenage and young adult children. A double-bounded CV format was used. Respondents were randomly assigned 1 of 5 starting bids/prices ($50, $100, $200, $500, or $1,000). If they responded “Yes,” they were asked if they would pay an amount twice as large as the starting cost for additional time with their doctor to discuss MenB vaccination or for a MenB vaccine. If a respondent answered “No” to the initial bid, they were asked if they would pay an amount half as large. If they again responded “No,” they were asked if they would pay more than zero dollars.
The survey also collected data on respondents’ social and demographic characteristics and their opinions regarding vaccines in general (i.e., vaccine capabilities for preventing and protecting against a disease). Respondents were asked to rate whether they agreed or disagreed with the following statements:
“My doctor has a moral obligation to tell me about a disease like MenB and the vaccine that can prevent it.”
“I trust my doctor to tell me what I need to know to make good decisions about my [my child’s] health.”
“I do not need to consult with a doctor if I have questions about my [my child’s] health.”
Respondents also were asked to describe their reaction to a hypothetical situation in which their doctor did not discuss the possibility of MenB vaccination with them and they found out about the vaccine later. Respondents were asked to choose which of 8 possible reactions ranging from “unconcerned” to “angry or disgusted” best described how they would feel if this were to happen. The 3 statements above and the hypothetical-situation response categories were based on open-ended responses obtained in parent and young-adult interviews during the qualitative research phase of the study. These questions are best interpreted as exploratory research.
Respondents also were asked a series of additional questions to evaluate their knowledge about vaccination and incidence rates of various infectious diseases (e.g., seasonal flu, whooping cough, meningitis, shingles). The study also included an additional module with a discrete-choice experiment focused on respondents’ preferences for vaccines to protect against a generic disease with low incidence but severe outcomes, with results reported in a separate manuscript (questionnaire available upon request). 21
Data Analysis
Descriptive analyses were conducted to examine the probability of reactions to the doctor choosing not to discuss MenB vaccines with young adults and parents. We estimated several parametric and nonparametric WTP split-sample models for young adults, parents of a child younger than 18 y, and parents of a child 18 y and older using both the single-bounded and double-bounded CV data. A well-known problem with estimating CV models is that the WTP of respondents who accept the highest bid offered is in an open-ended interval. Hence, mean values can be sensitive to the shape of the right-hand tail of the assumed error distribution. We estimated parametric models using logistic, normal, log-logistic, log-normal, and Weibull distributions. Covariates were included to help explain variations in the probabilities. The means of the WTP for additional time with the doctor for discussing MenB vaccine and WTP for MenB vaccination also were examined. A multivariate analysis was further performed to explore factors associated with WTP. Confidence intervals were obtained by the Krinsky-Robb method with 10,000 draws on the variance-covariance matrix. 24
Results
Study Population and Demographics
A total of 2,388 respondents were recruited from the Ipsos general-population panel and provided valid study data. Approximately 3% (n = 69) of the respondents provided invalid preference data (ie, data in which respondents provided conflicting responses) and were excluded from the analysis. Table 1 shows that 1,185 were parents of children 12 to 25 y of age and 1,203 were young adults 18 to 25 y of age. Most respondents were White (parent, 89.4%; young adult, 67.3%) and female (parent, 50.1%; young adult, 76.1%).
Demographics

n, number of respondents in the specified group; NA, not applicable.
Respondents’ Reaction If Doctor Did Not Discuss MenB Vaccines
Parents and young adults were asked to respond to the question, “Suppose your doctor did not discuss the vaccine with you, so you never knew it was an option. You found out later, when a friend asked whether you got the vaccine. How would you feel about this situation?” We found no differences between the parent groups, so we have combined them in this analysis. Figure 1 summarizes the similar reactions of parents and young adults. Although 30% of parents and 33% of young adults indicated they would feel “concerned,” 41% and 28% of parents and 36% and 31% of young adults would feel more concerned (“disappointed,” “anxious”/“afraid,” “upset,” or “angry”/“disgusted”) and less concerned (“unconcerned,” “calm,” or “understanding”), respectively. Thus, approximately 70% of respondents indicated their reaction to a doctor choosing not to discuss MenB vaccines with them would be negative (concerned or more concerned).

Reaction to doctor choosing not to discuss MenB vaccine.
The results from the multivariate analysis are found in Figure 2 and Supplementary Tables S1 and S2. Compared with parents who said they would be “unconcerned,” parents who indicated they would be “upset” or “concerned” were more likely to 1) be women older than 55 y, 2) have answered knowledge questions about disease incidence accurately, 3) have agreed that doctors should provide information about MenB vaccines, 4) be willing to pay for more time with their doctor to discuss a MenB vaccine, and 5) be willing to pay for a MenB vaccine. Young adults who indicated a reaction of “concerned” were more likely to agree that doctors should provide information about MenB vaccines and be willing to pay for a MenB vaccine. Both parents and young adults who said they would be “concerned” were less likely to have had a personal discussion about MenB vaccines with their doctors. Parent and young adult respondents were also less likely to agree or strongly agree that they “don’t need a doctor to answer their questions” if they indicated a response of “concerned” or “upset” compared with those who indicated that they would be “unconcerned.” In addition, parents who indicated a response of “concerned” or “upset” and young adult respondents who indicated a response of “concerned” were more likely to accept any of the offered bids for a MenB vaccine compared with those who responded as “unconcerned.”
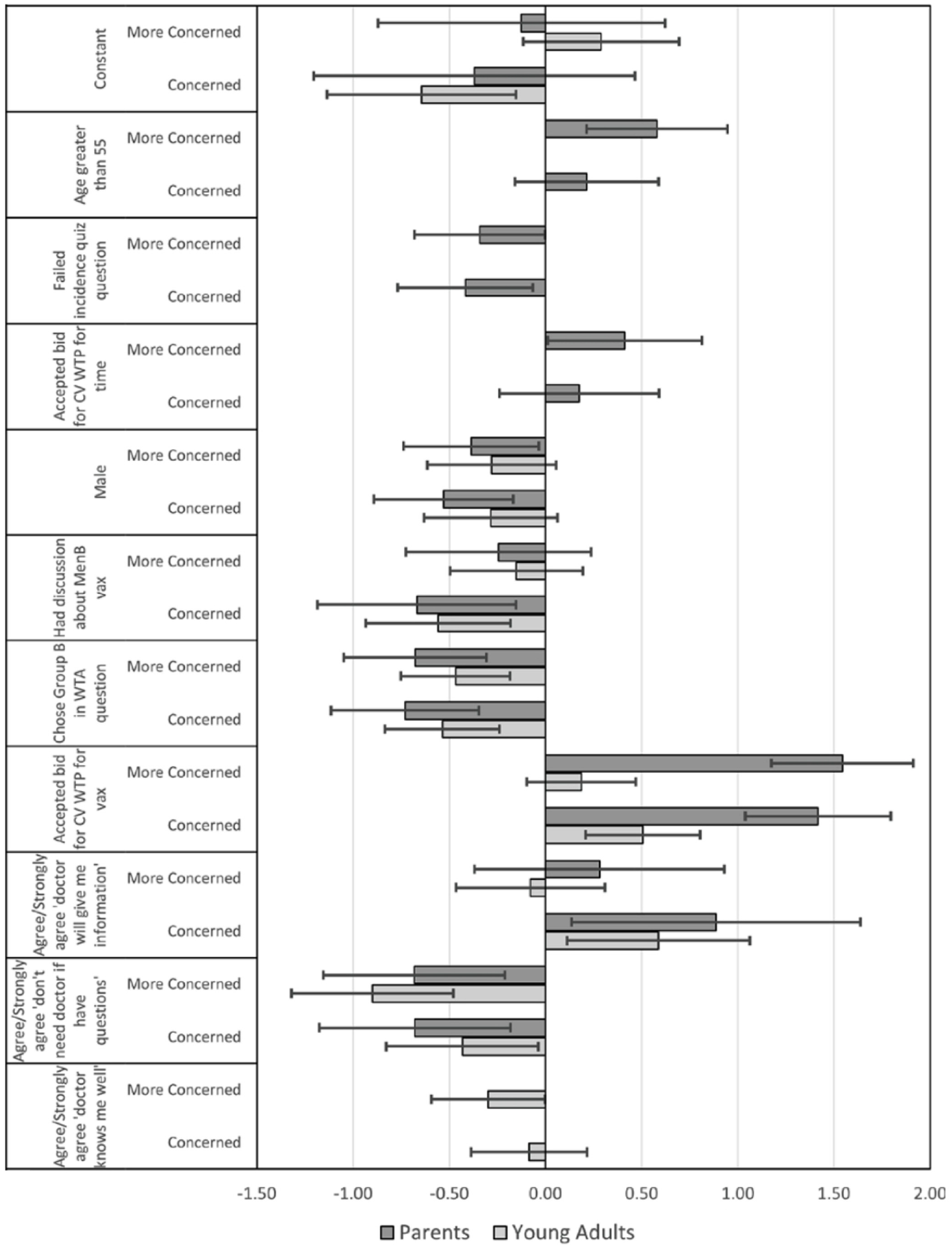
Multinomial-logit log-odds estimates of “upset” or “concerned” versus “unconcerned.”
WTP for MenB Vaccination and for Additional Time with Their Doctor
We found that mean estimates varied considerably among assumed error distributions. Also, the single-bounded estimates were smaller than the double-bounded estimates. However, medians were similar between single- and double-bounded models, and a conservative measure of means based on truncating the distributions at the highest bid also was similar. For vaccination, distributions for our best lower-bound estimates based on model fit were logistic for the young-adult sample and normal for both parent samples. For more time with a doctor, distributions were log-normal for young adults and parents with a child younger than 18 y and logistic for parents with a child 18 y and older.
Table 2a and b compare WTP for vaccination and time with doctor for the 2 parent groups with young adults. The truncated means are our best lower-bound value estimates of $129, $154, and $157 for young adults, parents with a child younger than 18 y, and parents with a child aged 18 y or older, respectively. Note that the truncated means are very similar to the nonparametric estimates. Also, we clearly reject the hypothesis that parents of younger and older children have different vaccine and time values.
a. Willingness to Pay (United States Dollars) for MenB Vaccination and 95% Confidence Intervals Compared with Statistical and Nonparametric Models

b. Willingness to Pay (United States Dollars) for More Time with the Doctor and 95% Confidence Intervals Compared with Statistical and Nonparametric Models

In Table 2b, large variances result in very long tails to the right for young adults and parents of children younger than 18 y, resulting in implausible mean values and extremely wide confidence intervals. However, similar to the vaccine results, the truncated means have tighter confidence intervals. Also, the point estimates for the truncated means again are similar to the lower-bound nonparametric values. Our best lower-bound estimates for more time with the doctor are $63, $81, and $88 for young adults, parents with a child younger than 18 y, and parents with a child 18 y or older, respectively.
Figure 3 shows how WTP was influenced by various factors. There were no significant differences among the 3 subsamples except for income less than $30,000 and having had a discussion with a doctor. The low-income effect was negative for parents with older children, and the doctor-discussion effect was positive for young adults. Provaccination attitude, income >$100,000, being willing to pay for more time with the doctor, and having a doctor who knows them positively influenced parents’ WTP for a MenB vaccine for their child. Antivaccination attitude (vaccine hesitancy) and income <$30,000 negatively influenced parental WTP for a MenB vaccine. For young adult respondents, significant covariates that positively influenced WTP for a MenB vaccine included provaccination attitude, an opinion that the doctor has a moral obligation to discuss the vaccine, and having a discussion with the doctor. An antivaccination attitude and being married negatively influenced young adults’ WTP for a MenB vaccine. For estimation details, see Supplementary Table S3. Because of the relatively poor estimates for time with the doctor, we are not reporting the covariate analysis. However, full model estimates can be found in Supplementary Table S4.

Factors associated with WTP for MenB vaccination.
Discussion
To our knowledge, this is the first formal assessment of population preferences for MenB vaccination in the United States using ACIP-prescribed methodology. We examined how respondents would feel if their HCP failed to discuss with them serogroup B disease and MenB vaccination, as required by the ACIP recommendation for shared clinical decision making. We also estimated respondents’ WTP for 1) additional time to discuss these issues with their physician and 2) MenB vaccination. The principal findings from this study were 1) most respondents would have a negative reaction if their doctor neglected to discuss MenB vaccination with them, indicating that they placed primary responsibility upon their doctor to initiate such a discussion; 2) most respondents felt entitled to such a discussion with their doctor without having to pay extra for the additional time that this required; and 3) young adults’ WTP for MenB vaccination was nearly $300 and parents’ WTP was more than $400 to vaccinate their teenage or young adult child.
In the United States, the first example of a nonroutine “clinical decision making” ACIP recommendation for an entire age group (i.e., adolescents) was for MenB vaccines in 2015.18,25 Since 2005, routine vaccination with conjugate MenACWY vaccines continues to be recommended for all adolescents.18,25 For nonroutine recommendations, ACIP’s terminology has evolved from “permissive” to “category B” and to “individual clinical decision-making.”25,26 As of June 2019, the term has been referred to as “shared clinical decision-making.”25,26 Subsequently, shared clinical decision-making recommendations have been applied to additional vaccines and age groups, such as human papilloma virus vaccines for individuals >26 y of age and pneumococcal conjugate vaccines for individuals ≥65 y of age.27,28
Shared clinical decision making is now used in multiple clinical situations and is probably a familiar concept to most providers. However, barriers to effective MenB vaccination discussions between physicians and patients can include limitations on physicians’ time, some patients’ general vaccine hesitancy, and clinical assumptions about the relevance of vaccines for diseases with low incidence, even those with severe consequences. Health care discussions involve shared decision making between the clinician and the patient and are considered essential for ensuring that clinical care is in agreement with the patients’ values and preferences. 29 Vaccine discussions and decisions in the United States are driven by the ACIP’s recommendations, which rely on evidence on the balance of benefits and harms, the type or quality of the evidence, the values and preferences of the people affected, and health economic analyses. 12 Recognizing the uniqueness of the vaccination discussion, the ACIP’s EtR framework requires gathering available evidence on target population values and preferences related to vaccination and comparative health benefits and risks. 15
Our results suggest that, specifically within the context of MenB vaccination, young adults and parents expect their doctors to proactively inform them about MenB disease and the option to vaccinate. Respondents were not, however, willing to pay for additional time for such a discussion, which may indicate that they presume this should be covered during their existing routine visits. This expectation appears consistent with the shared clinical decision-making recommendation for the MenB vaccine. Thus, given the limited time available for such visits, HCPs might consider reevaluating priorities for how to use this time. More generally, the growing use of clinical decision-making recommendations and accumulating evidence regarding the implementation experience with MenB vaccination necessitate empowering HCPs with clear guidelines and developing a cohesive set of outcome measures (eg, patient satisfaction, rates of disease, disparities among demographic groups) that can be used to evaluate the practice of shared clinical decision making and the public health impact of these recommendations. 30 Furthermore, when we grouped the parents according to the age of their child (<18 y old v. ≥18 y old), we found no evidence of a difference in vaccine values between the subgroups. It is possible that in both cases, parents act in a similar “consulting” role within the shared clinical decision-making process, although future studies are necessary to clarify this result. This finding can, however, inform vaccine-policy decisions beyond those surrounding MenB vaccination as shared clinical decision making becomes a more common recommendation.
Studies have shown that a recommendation from an HCP is the most influential variable associated with vaccination.31–33 Parents rely on their providers for vaccination recommendations, and 5 y of implementation experience with the ACIP nonroutine recommendation for MenB vaccination has shown a lack of understanding among providers and parents, that discussion can be limited, and disparities exist.31,34–36 Meningococcal disease is preventable with vaccination; however, the experience with MenB vaccination indicates that physicians struggle with the interpretation, communication, and implementation of ACIP’s clinical decision-making recommendation, leading to disparate vaccine-prescribing practices.35–37 Consequently and despite parental willingness to vaccinate, evidence shows sociodemographic inequities in MenB vaccine access and vaccine receipt.34,38,39 In a nationally representative survey, parental awareness of MenB vaccines was low (43%) and significantly lower among males, those of Hispanic or non-White race, and parents with less educational attainment. 39 Regional studies examining immunization records have also shown that MenB vaccination was positively associated with White race, having private insurance, and MenACWY vaccine receipt. 38
All stated-preference studies are limited by the hypothetical nature of the scenarios in the survey and the possibility that respondents could make different choices if faced with the complexities of real-life decisions. To minimize hypothetical bias, the survey instrument used in this study was designed to simulate choices respondents could be asked to make and to ensure that the decision context aligned with their lived experiences. Although we took care to explain that costs shown in the choice questions were personal out-of-pocket costs, it is possible that respondents incorrectly assumed the WTP question was referring to an increased insurance cost rather than a direct out-of-pocket expense. While the Knowledge Panel is representative of the general US population, a potential limitation is that individuals who opted in to participate in this survey may not have been representative of the population. However, given the large sample size of more than 2,000 respondents, the survey sample was highly diverse, which facilitated extensive covariate analysis to understand how preferences are affected by vaccine attitudes and knowledge as well as sociodemographic characteristics.
This study was conducted before the onset of the COVID-19 pandemic, so the preference assessments do not account for the effects of the pandemic on perceptions and attitudes related to infectious disease and vaccination. Had this survey been fielded today, the results may be different. In the COVID-19 era, vaccine hesitancy is likely exacerbated, which could affect how respondents answer survey questions related to vaccine preferences. 40 This is particularly true for the emotional responses, which were already considered exploratory prior to the pandemic. The COVID-19 pandemic has sensitized regulators to the importance of the public’s preferences, and every ACIP recommendation in the last 2 y has included information on vaccine preferences. 41 Future studies should take into account the impact of COVID-19 on vaccine preferences to best inform ACIP recommendations.
Overall, our results demonstrate that young adults and their parents place a high value on the MenB vaccine and expect their HCP to initiate a discussion with them regarding vaccination. This highlights the importance of provider engagement in informing their patients of vaccination options and reemphasizes the need for substantial provider support and education to enable them to implement the ACIP recommendation for shared clinical decision making with respect to MenB vaccination. Results also demonstrate the usefulness of including formal stakeholder preference data using ACIP-prescribed methodology in the EtR process. These findings help to close the evidence gap between patient preferences related to MenB vaccination and shared clinical decision making. Our hope is that the results of this survey will support future development of ACIP recommendations by improving the application of input from all stakeholders not only for the next-generation pentavalent meningococcal vaccines but also for other vaccines for which shared clinical decision-making policies are implemented (or being considered).
Supplemental Material
sj-docx-1-mpp-10.1177_23814683241264280 – Supplemental material for Young Adult and Parent Willingness to Pay for Meningococcal Serogroup B Vaccination
Supplemental material, sj-docx-1-mpp-10.1177_23814683241264280 for Young Adult and Parent Willingness to Pay for Meningococcal Serogroup B Vaccination by Liping Huang, Amit Srivastava, Angelyn Fairchild, Dale Whittington and Reed Johnson in MDM Policy & Practice
Footnotes
Acknowledgements
The authors would like to thank Vincenza Snow, MD, and Jessica Presa, MD, of Pfizer Inc for providing helpful feedback on the manuscript. Editorial/medical writing support was provided by Lindsey Kirkland, PhD, of ICON (Blue Bell, PA, USA) and was funded by Pfizer Inc. Dr. Huang affirms that she has listed everyone who contributed significantly to the work. This study was sponsored by Pfizer Inc. Pfizer was involved in the concept and design of the article; collection, analysis, and interpretation of the data; writing the report; and the decision to submit the article for publication.
The work was presented at IDWeek 2020, SAHM 2020. The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: LH is a Pfizer employee and may hold stock options. AS is currently employed by Orbital Therapeutics; he was a Pfizer employee at the time of manuscript development and may hold stock options. AF reports Pfizer research support for consulting services. DW has no conflicts to report. RJ reports Pfizer research support to Duke University. The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was sponsored by Pfizer Inc. The funding agreement ensured the authors’ independence in designing the study, interpreting the data, writing, and publishing the report.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
