Abstract
Highlights
Shared involvement around chemotherapy decisions remains limited for patients diagnosed with colon cancer.
Sociodemographic (age, race, gender), interpersonal (medical mistrust), and intrapersonal (decisional self-efficacy, perception of choice) factors that influence preferred involvement in chemotherapy decision making may differ from those influencing actual involvement in chemotherapy decision making.
Shared involvement in chemotherapy decisions may look different than currently conceptualized, notably when uncertainty around the benefits exists.
The multimodal nature of colon cancer (CC) treatment pathways, coupled with uncertainty and lack of consensus around the benefits of adjuvant chemotherapy for stage II CC, introduces challenges for treatment decision making. 1 Engaging CC patients in the process of shared decision making (SDM) around their treatment is a key component of patient-centered cancer care, especially when decisions involve more than 1 treatment option, limited or conflicting evidence, or with high degrees of uncertainty.2–4 SDM has been associated with increased knowledge, satisfaction, decreased sense of uncertainty, and is seen as a relevant strategy for empowering historically disenfranchised populations by improving quality of patient–provider communication, engagement, and satisfaction with care received. Research suggests cancer patients want to be involved in the cancer treatment decision-making process.5,6 However, the degree to which cancer patients’ preferred involvement matches their actual involvement varies, 7 and evidence regarding the extent to which CC patients are involved in treatment decisions is limited.8–10
Multiple factors play a role in preferences for and experiences of SDM.11–13 It is well documented that the racial and ethnic minority populations’ experiences with SDM differ. These differences have been attributed to complex sociodemographic, cultural, and systemic factors, such as barriers in cross-cultural communication, health literacy, racism, and strained relationships between patients and providers, that result in mistrust and miscommunication.14,15 Significant differences by gender have also been observed with respect to communication styles 16 and involvement in decision making, but these differences are less clear. 17 For example, prior studies have shown that women tend to prefer more SDM compared with men, while other studies found no significant differences for decision making by gender.5,18 While the literature supports the need to further examine the role of demographic variables including race and gender, these variables are complex sociocultural constructs and should be examined alongside other variables and processes that account for differences in communication and SDM.
In addition to demographic indicators, disease-related, interpersonal, and intrapersonal factors may also play an important role in SDM. 13 The survival benefit of adjuvant chemotherapy for stage II CC patients is inconclusive in contrast to stage III CC patients, making chemotherapy treatment decisions for stage II patients more complex and challenging.19,20 In addition, patient perceptions at the interpersonal and intrapersonal level, such as confidence in one’s ability to make effective treatment decisions (decisional self-efficacy),17,21 perception of choice, 22 and trust in providers and the health care system,23,24 may also influence treatment decision making and involvement.
To date, few studies have explored how these various factors contribute to preferences for and actual experiences with chemotherapy treatment decision making among diverse CC patients. Hence, the purpose of this study was to explore preferred and actual involvement in chemotherapy treatment decisions among stage II and III CC patients by sociodemographic, interpersonal, and intrapersonal communication factors. We also explored variation in interpersonal and intrapersonal factors by race and gender.
Methods
This exploratory, cross-sectional analysis was part of a larger longitudinal study to understand factors contributing to chemotherapy decision making among CC patients. This study recruited a convenience sample of stage II and III CC patients from 2 cancer centers located in Northern Manhattan on a rolling basis. Eligible participants had to be within 6 months of a new CC diagnosis, over the age of 18 y, and proficient in English or Spanish. Eligible participants were approached to obtain consent and completed/scheduled a baseline survey either by phone or in person. The study protocol was approved by Columbia University Irving Medical Center and Mount Sinai Institutional Review Board.
Between 2013 and 2016, we administered a baseline survey to collect self-reported sociodemographic (e.g., age, race, ethnicity, education), clinical (e.g., cancer stage, undergo chemotherapy), interpersonal, and intrapersonal variables/constructs relevant to chemotherapy treatment decision making using valid and reliable measures when available.7,25–28 Due to small sample sizes, we categorized race as “White” and “Other,” with “Other” including cancer patients who self-identified as Black, Asian, American Indian or Alaska Native, Native Hawaiian or Pacific Islander, other/free response. Detailed descriptions of selected variables, measures, and analytic approach can be found in Supplementary Table 1.
Decision Involvement
We assessed patients’ overall preferred and actual involvement in cancer treatment decision making using the 5-item Control Preferences Scale. 26 For this analysis, actual and preferred involvement were categorized as patient controlled ( “You prefer to make or you made the decision about treatment with little or no input from your doctors” or “You prefer to make or you made the decision after considering your doctor’s opinion”), shared (“You prefer or you and your doctors made the decision together”), or physician controlled (“You prefer that your doctor or your doctor made the decision after considering your opinion” or “You prefer that your doctor or your doctor made the decision with little or no input from you”).
Interpersonal Communication Factors
The patient’s perception of trust including dependability, confidence in knowledge and skills, and reliability of information was assessed using the 10-item Trust in Physicians Scale 26 response options ranged from 1 to 5 (strongly disagree to strongly agree), with higher scores representing greater trust. Medical mistrust was assessed using the 3-item subscale discrimination and group disparities in health care and the 3-item subscale lack of support from health care providers from the 12-item Group-based Medical Mistrust Scale. 27 Response options ranged from 1 to 5 (strongly disagree to strongly agree), with higher scores representing greater mistrust.
Intrapersonal Communication Factors
We assessed the patient’s confidence in their ability to make decisions about their treatment using the 11-item Decisional Self-efficacy scale, with higher scores indicating higher levels of self-efficacy. 28 Patient’s perception of choice whether or not to receive chemotherapy was assessed using the single item, “When discussing options for treatment, did you feel there was much choice for you?” with the response options of “yes” and “no.” We also asked if the patients spoke with their doctor about chemotherapy as part of their CC treatment.
Of 88 eligible patients, 59 completed the survey (response rate = 67%). Three surveys were removed from this analysis due to missing data on key variables. Our final analytic sample size was 56. Summary statistics were calculated to describe the distribution of variables. Differences in actual and preferred decisional involvement and differences in interpersonal and intrapersonal communication factors by race and gender were assessed using Fisher’s exact chi-square, t tests, and analysis of variance (ANOVA). All analyses were conducted using SAS, 29 with P < 0.05 considered statistically significant.
Results
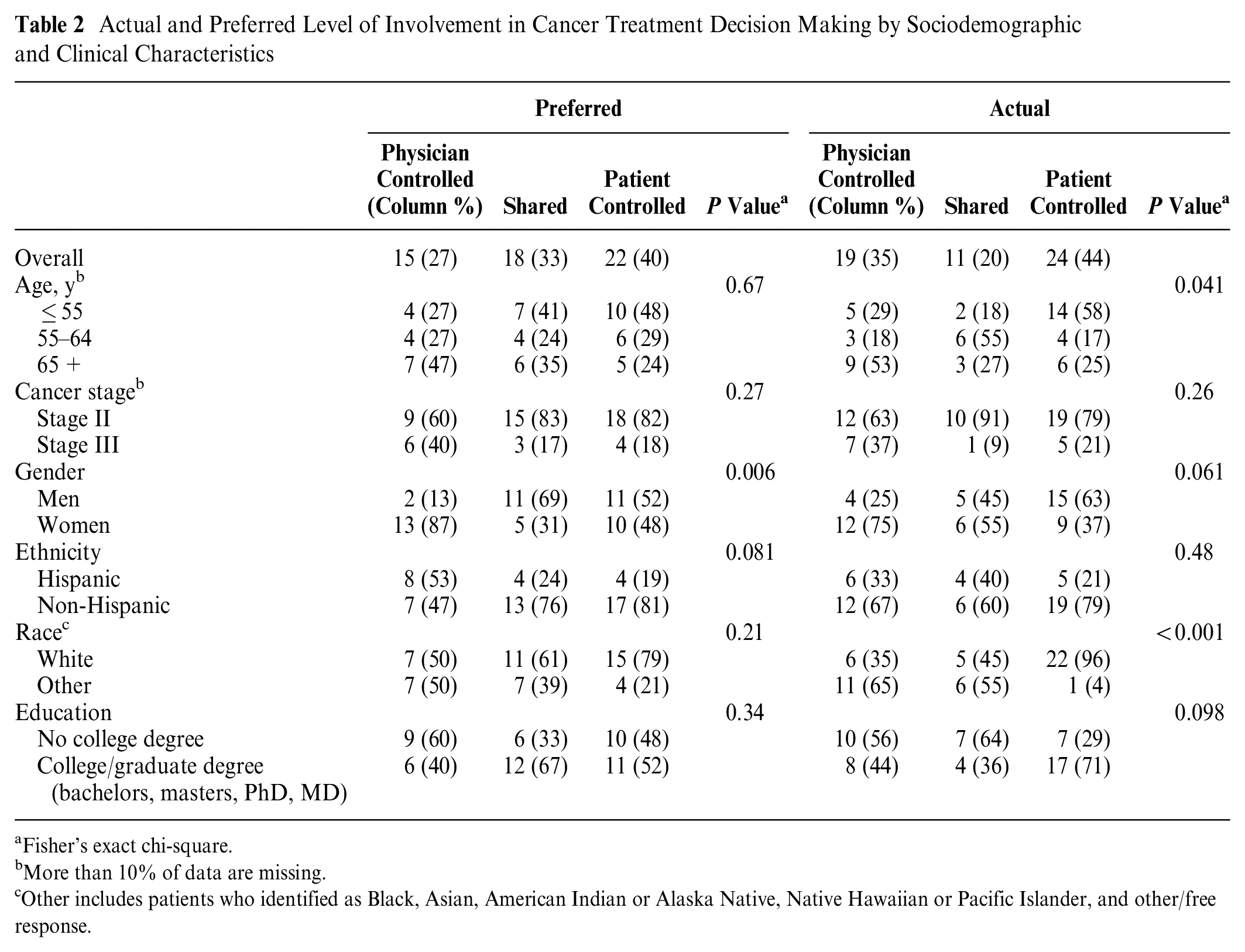
Per Table 1, 75% of patients were diagnosed with stage II CC, 53% identified as women, 70% identified as non-Hispanic, and 63% identified as White. Overall, 50% of patients preferred decisional involvement aligned with their actual involvement. We assessed differences in preferred and actual involvement in chemotherapy decision making by sociodemographic characteristics using Fisher’s exact chi-square (Table 2). Overall, only 20% of CC patients reported their actual involvement as shared. We also observed significant differences in preferred levels of involvement by gender, with women reporting significantly higher preferences for physician-controlled decision making (87% of women v. 13% of men; P = 0.006), while men reported higher preferences for shared (69% of men v. 31% of women; P = 0.006) decision making. Significant differences in actual involvement in decision making was observed by age and race (Table 2).
Self-reported Baseline Survey Characteristics of Colon Cancer Patients (N = 56)

Other includes patients who identified as Black, Asian, American Indian or Alaska Native, Native Hawaiian or Pacific Islander, and other/free response.
Actual and Preferred Level of Involvement in Cancer Treatment Decision Making by Sociodemographic and Clinical Characteristics
Fisher’s exact chi-square.
More than 10% of data are missing.
Other includes patients who identified as Black, Asian, American Indian or Alaska Native, Native Hawaiian or Pacific Islander, and other/free response.
Specifically, patients whose race was categorized as Other and patients over the age of 65 y experienced more physician-controlled involvement (65% Other v. 35% White, P < 0.001; 53% 65+ y v. 18% 55–64 y and 29% ≤ 55 y of age, P = 0.041), while patients identifying as White and patients ≤ 55 y of age experienced more patient-controlled decision making (96% White v. 4% Other, P < 0.001; 58% ≤ 55 y v. 17% 55–64 y and 25.0% 65+ y, P = 0.041). We did not observe significant differences in actual or preferred decision making by cancer stage.
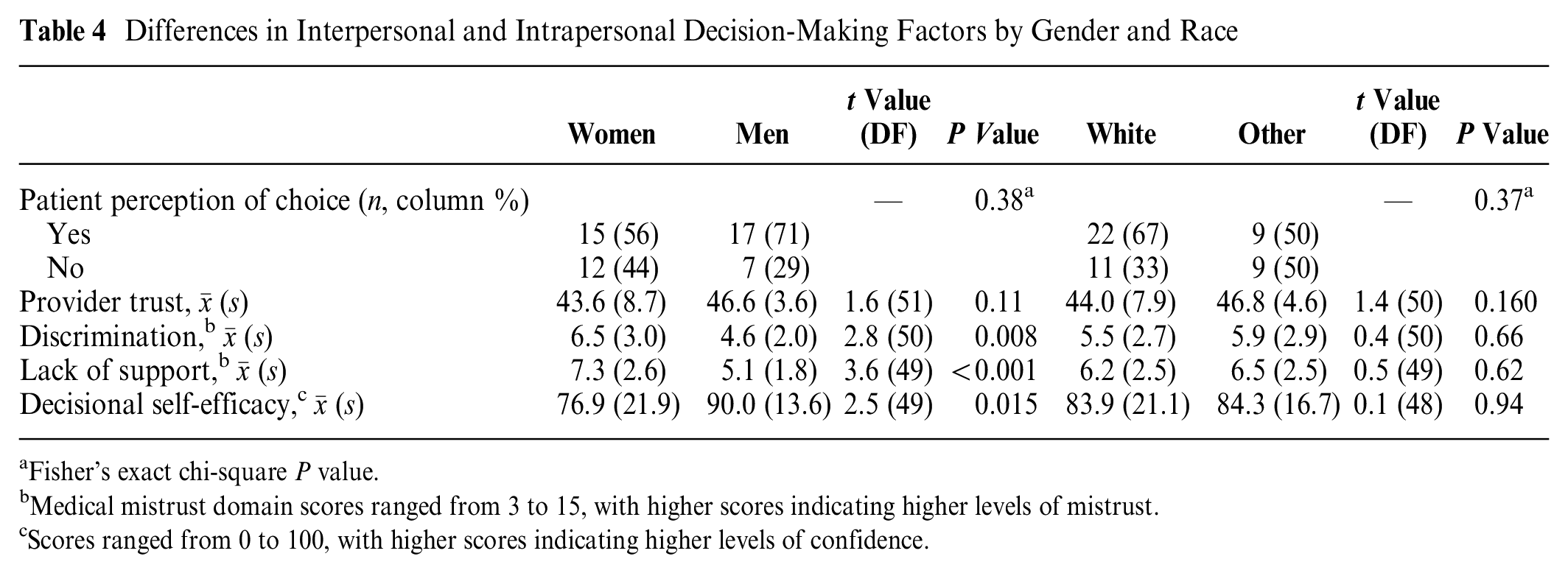
Next, we assessed differences in preferred and actual involvement in chemotherapy decision making by interpersonal and intrapersonal decision-making factors (Table 3) using Fisher’s exact chi-square for categorical variables (e.g., perceived choice) and ANOVA for continuous variables (e.g., provider trust, discrimination, lack of support, and decisional self-efficacy). We observed significant differences in preferred involvement by decisional self-efficacy (F = 4.4 [2], P = 0.018), with higher mean decisional self-efficacy reported among patients preferring shared involvement (mean 93.4 shared v. 77.6 physician controlled and 77.7 patient controlled). There were also significant differences in actual involvement by perception of choice, with patients perceiving “no” choice reporting more physician-controlled involvement (66% “no” choice v. 33% “yes” choice, P = 0.017. Next, we assessed differences in interpersonal and intrapersonal communication factors by race and gender using a t test for continuous variables and Fisher’s exact chi-square for categorical variables (Table 4). We did not observe significant differences in interpersonal and intrapersonal communication factors by race. However, we did observe significant differences in medical mistrust subdomains of discrimination (t = 2.8 [50], P = 0.008) and lack of support (t = 3.6 [49]; P < 0.001) by gender, with women reporting higher mean levels of discrimination (mean 6.5 v. 4.6) and lack of support (mean 7.3 v. 5.1) compared with men. Finally, we observed significant differences in decisional self-efficacy (t = 2.5 [49], P = 0.015), with women reporting lower mean levels compared with men (mean 76.9 v. 90.0).
Difference in Actual and Preferred Involvement in Cancer Treatment Decision Making by Interpersonal and Intrapersonal Decision-Making Factors

Fisher’s exact chi-square
Medical mistrust domain scores ranged from 3 to 15, with higher scores indicating higher levels of mistrust.
Scores ranged from 0 to 100, with higher scores indicating higher levels of confidence
Differences in Interpersonal and Intrapersonal Decision-Making Factors by Gender and Race
Fisher’s exact chi-square P value.
Medical mistrust domain scores ranged from 3 to 15, with higher scores indicating higher levels of mistrust.
Scores ranged from 0 to 100, with higher scores indicating higher levels of confidence.
Discussion
Overall, 20% of CC patients reported shared involvement in their actual chemotherapy treatment decisions, and half had alignment between their preferred and actual involvement. Together, these findings suggest insufficient shared and concordant chemotherapy decision making among CC patients facing uncertain and complex treatment options. Our results also suggest that gender and decisional self-efficacy may play a role in preferred involvement, while age, race, and perception of choice may play a role in actual involvement in cancer treatment decision making. No significant differences in preferred or actual involvement in cancer treatment decision making were observed by cancer stage, which may be due to power limitations. Finally, we did not observe significant differences in interpersonal and intrapersonal communication factors by race, but several differences were noted by gender.
We found that 50% of CC patients preferred involvement aligned with their actual involvement in chemotherapy treatment decision making, and similar to prior research, CC patients tend to perceive their actual involvement in treatment decisions as patient controlled.30,31 Discordance between patients’ preferred and actual roles in treatment decision making may lead to reduced patient satisfaction and overall decreased quality of life. There is inconclusive evidence and uncertainty around the effective management of stage II CC,32,33 which makes up a large majority of our study sample. Interventions to increase the physician’s knowledge of treatment options and to facilitate patient–provider communication, such as decision aids, may help facilitate SDM for CC patients.34,35 Yet, this may be too simplistic of an approach for stage II CC patients given the complexity and uncertainty surrounding the benefits of chemotherapy for this population.36,37 More research is needed to identify strategies that allow physicians to effectively communicate treatment options under clinical uncertainty and to elicit the decision-making preferences of patients.
We found that women preferred more physician-controlled decisions than men did,5,38 and they reported lower levels of decisional self-efficacy and higher levels of medical mistrust.25,39–41 It is possible that women in our sample felt ill-equipped to make decisions about their treatment due to inadequate communication, lack of trust in information received, or the belief that doctors do not have their best interests in mind. Our findings add to the mixed literature to date highlighting the complexity of using gender to understand differences in actual and preferred involvement in treatment decision making. While our study is one of the first to show differences in decisional involvement and factors potentially contributing to differences in involvement, such as medical mistrust and decisional self-efficacy by gender, more research is needed to understand the complex factors driving differences observed by gender.
We also found that CC patients whose race was categorized as “Other” (Black, Asian, American Indian or Alaska Native, or other/free response) experienced more physician-controlled decisions, despite more than 60% indicating preferences for shared or patient-controlled decision making.14,42 Most CC patients whose race was categorized as “Other” self-identified as Black or “Other,” with the majority writing in “Hispanic” as their race. This is particularly relevant given the evidence of racial and ethnic disparities in cancer care and may be due to complex and multifactorial factors such as literacy barriers, poor patient–provider communication, language barriers, and poorer health outcomes.14,15,42 Some studies suggest that physicians do worse at explaining treatment options with racial and ethnic minority populations and may implicitly assume that patients with poor health outcomes may feel more dependent or willing to relinquish control, making it more challenging for these populations to engage in decisions.42,43 Interestingly, we did not observe significant differences in decisional involvement by ethnicity or differences in perceptions of choice, provider trust, medical mistrust, or decisional self-efficacy by race. This may be due to how we categorized race for this analysis given small cell sizes but may also suggest that providers are not eliciting preferences for involvement in CC treatment decision making for racial minority populations. Like gender, race is a complex sociocultural construct, and more empirical work with larger and diverse samples is needed to disentangle the role race on CC chemotherapy treatment decision making.
We note several limitations. The cross-sectional design allowed for capturing individual reports at a single point in time and thus limits causal interpretation. Our sample size was small, limiting generalizability, and this may explain the lack of differences observed by race and notably cancer stage. However, our sample was more diverse and provided important insights for generating hypotheses and into the complexities of treatment decision making for CC patients. In addition, the validated tool used to measure decisional involvement measured decision making at a single point in time and might not have fully captured the complexity around SDM for CC patients, particularly those with stage II cancer. Specifically, nearly 40% of stage II CC patients selected that they made the final decision but strongly considered their doctor’s opinion. Although categorized as patient-controlled involvement in this analysis, it is possible that our sample perceived this as a form of shared involvement. Importantly, about 26% of stage II CC patients selected that the physician made the final decision, but no patients selected this statement as their preference, emphasizing the importance of physician-targeted interventions to elicit preferences for treatment decision making.
This study adds to the limited research on how sociodemographic, interpersonal, and intrapersonal factors are related to chemotherapy decision making among a relatively diverse sample of stage II and III CC patients. SDM remains a key element of high-quality cancer care but may look different for CC patients when uncertainty exists. Efforts, such as the development of decision support tools and training for providers to elicit preferences for involvement while providing information about the harms, benefits, and uncertainties around treatment modalities, should be explored to improve SDM under clinical uncertainty. Given that SDM is an ongoing process, additional research is needed to capture changes in actual involvement in decision making over multiple timepoints. Moreover, more research is needed to explore how CC patients perceive shared involvement and the role of various factors, notably gender, race, medical mistrust, and decisional self-efficacy, across the communication continuum to inform more personalized approaches that facilitate SDM.
Supplemental Material
sj-docx-1-mpp-10.1177_23814683231163189 – Supplemental material for Involvement in Chemotherapy Decision Making among Patients with Stage II and III Colon Cancer
Supplemental material, sj-docx-1-mpp-10.1177_23814683231163189 for Involvement in Chemotherapy Decision Making among Patients with Stage II and III Colon Cancer by Jessica D. Austin, Elizabeth Shelton, Danielle M. Crookes, Alfred I. Neugut and Rachel C. Shelton in MDM Policy & Practice
Footnotes
Acknowledgements
We thank the recruiters at Mount Sinai and the Herbert Irving Comprehensive Cancer Center.
Correction (October 2023):
This article has been updated to list Parisa Tehranifar as the fourth author.
This work was completed at Columbia University Mailman School of Public Health. The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr. Neugut has consulted for Otsuka, GlaxoSmithKline, Eisai, United Biosource Corp, and Hospira and sits on the medical advisory board of EHE Intl. He receives grant support from Otsuka. All other authors have no other interests to disclose. The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Financial support for this work was provided by the American Cancer Society (grant No. 124793-MRSG-13-152-01-CPPB) and the National Center for Advancing Translational Sciences, National Institutes of Health (grant Nos. UL1 TR000040, UL1 RR024156). The funding agreement ensured the authors’ independence in designing the study, interpreting the data, writing, and publishing the report.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
