Abstract
Introduction
The worldwide incidence of melanoma continues to increase, with annual incidence rising as rapidly as 4% to 6% per year in most White populations, with the greatest burden occurring in Australasian, North America, and European populations.1,2 In clinical practice, the American Joint Committee on Cancer (AJCC) staging criteria based on tumor, node, and metastatic (TNM) status is used to risk stratify and treat patients. 3 The 10-year melanoma-specific survival rate of those with the earliest stage of disease, stage IA, is 98%, and stage IB is ≥92% (AJCC 8th edition). 3 Specific data on melanoma recurrence and new primary tumors is less well-defined. 4 Consensus on standardized follow-up care is lacking, leading to considerable variation in practice across countries.5,6 For example, the Cancer Council Australia Melanoma Guidelines Working Party recommend that stage I patients have annual visit for 10 years with a medical professional, whereas German, Dutch, and British guidelines stratify surveillance by AJCC substage (i.e., IA or IB).
The National Institute for Health and Care Excellence (NICE) “Melanoma: Assessment and Management” guideline 2015 advise that stage I melanoma patients are followed-up to detect signs of recurrence by history and physical examination. 7 This assumes that the clinician undertakes a visual inspection using a dermoscopy to examine any suspicious moles. Specifically, for stage IA the recommendations suggest follow-up 2 to 4 times for 1 year and then discharge, while for stage IB follow-up is 3-monthly for 3 years and then 6-monthly to 5 years. More recently, in 2019, NICE considered all evidence in a consultation process and decided that updates of melanoma guidelines including follow-up are necessary. 8 An important consideration of melanoma surveillance is that self-examination by patients and their partners can detect melanoma recurrence outside of active surveillance regimes but also may lead to overconsumption of care. 9
A recent Australian economic evaluation of surveillance strategies does so without disaggregating between substages. 10 Our study has been undertaken comparing different surveillance strategies for patients with AJCC stage IA and IB melanoma separately as the risk of further disease are different. The study aimed to identify various candidate surveillance strategies and compare them with the current recommendations by NICE in a cost-utility analysis framework in terms of incremental costs and effectiveness.
Methods
The economic model is based on published data, expert opinion, and individual patient data (n = 161) obtained from the University Hospital of North Durham, England (NREC 19/NE/004) that was collected from 2004 with follow-up to 2018. A National Health Service (NHS) perspective was taken.
Model Structure
Microsimulation models were built in TreeAge Pro 2019 R1.0 (Williamstown, MA, USA) to evaluate candidate surveillance strategies for stage IA and IB melanoma patients separately. The two models comprised four health states: disease free, recurrence (diagnosed stage IA-IV incorporating tunnel states per AJCC substage if undetected), death from melanoma, and death from other causes. The model uses discrete time with a cycle of 1-month. After initial surgical excision, individuals entered the model in the disease-free health state. Within each monthly cycle, a recurrence could occur, be detected and diagnosed in the scheduled follow-up visit by a clinician. There was also a probability that recurrence would be self-detected by the patient or their partner. If this occurred there would be an unscheduled emergency visit. If a suspected recurrence was identified by a clinician or patient, patients then followed a care pathway, modelled as a decision-tree, based on current practice and the NICE guideline. 7 The detected suspicious lesion had a probability of being one of three outcomes: either being a recurrence, “false alarm,” or a false diagnosis. 9 It was also possible that a recurrence might occur but that it remain undetected in that and subsequent time periods. If it remained undetected then disease progression could occur. This is depicted as a tunnel state in the model (see the supplementary appendix for more details on the model development process).
The model has a lifetime time horizon with both costs and effects discounted using a 3.5% annual discount rate. 11 Costs were estimated in pounds Sterling from an NHS and personal social service (PSS) perspective for the financial year 2017–2018.
Plausible Strategies
Organized surveillance for melanoma patients was deemed essential. The selection of the most plausible candidate strategies within the NHS health care system was based on an iterative consultation process with the clinical expert team. Nonrealistic strategies such as those involving prognostic risk models (not suitably validated to be part of a surveillance strategy), use of diagnostic imaging techniques (not supported by evidence 12 ), and surveillance led by general practitioners (based on lack of capacity and training in dermoscopy) were excluded. The three variables used to define variations between surveillance strategies were clinical specialty, surveillance intervals, and duration of follow-up. The clinical specialty options comprised patients seen by a dermatologist, surgeon, or specialist dermatological nurse (known as cancer nurse specialist [CNS] within the NHS). The frequency of follow-up considered were every 3, 4, 6, or 12 months. The duration of follow-up considered for stage IA was 1, 2, 5, and 10 years, while for stage IB, 1, 3, 5, 10, and 20 years. Given the computational burden of running the model with large probabilistic sensitivity analysis (PSA) simulations for the original number of strategies, the model was first run deterministically to identify 20 strategies that were considered to have the most promise to be cost-effective for patients initially treated for stage IA and IB melanoma. The 20 strategies included in the analysis are presented as the base-case analysis in Table 1 and include a mix of strategies that differ between substage (e.g., 2-year intervals for IA and 3-year intervals for IB).
Strategies for Surveillance of Stage IA and IB Included in the Base-Case Analysis

NICE, National Institute for Health and Care Excellence.
Model Inputs
The model was based on individual-level data of patients surgically treated for early-stage melanoma (mean age 56 years, range 17–98 years) from University Hospital of North Durham (2007–2019). For subsequent recurrences, the care pathway included sentinel lymph node biopsy for staging purposes and if positive subsequent lymph node dissection was performed. The costs of treating advanced stage III and IV disease with newer systematic targeted therapies and immunotherapies was based upon the list price. 13 For many parameters, values from the literature were obtained and supplemented with expert opinion. Model inputs are summarized in Table S1 with the following brief explanatory commentary.
Patient Behavior
In a Dutch randomized control trial study (N = 180) comparing surveillance regimens, eight of 17 recurrences (48%) in the first year time were diagnosed via self-detection by the patient or their partner. 14 However, our clinical experts considered a higher value of 60% to be more reflective of UK clinical practice. Another Dutch study reported that almost 80% of patients (538/699) with a Breslow thickness less than 1 mm reported more frequent follow-up visits than guideline recommendations. 15 Local clinical experts considered that 85% of patients would make a “false alarm” emergency clinic visits in a given year. Annual probabilities of self-detection and “false alarm” emergency visits were converted into a monthly probability.
Epidemiology
National melanoma epidemiology statistics, such as summary stage of melanoma incidence, were obtained from the National Cancer Registration and Analysis Service run by Public Health England. 16 However, more granular incidence information (i.e., by substage IA, IB, IIA, IIB, etc.) was needed and this came from a large German registry-based study. 17
Diagnostic Accuracy of Practitioners
A Cochrane review exploring the diagnostic accuracy of clinicians was identified and a meta-analysis of the different staff categories was conducted. 18 For dermatologists, 11 studies from mainland Europe were pooled together; for surgical oncologists, two Italian studies were pooled together; and for specialist dermatological nurse, only one US study was identified based on eight physician assistants (see supplemental appendix for more details). Mean sensitivity and specificity values of these groups were estimated.
Diagnostic Accuracy of Staging Disease
The sensitivity and specificity of local biopsy were obtained from an observational study which aimed to investigate how accurate and reproducible the results of pathologists’ diagnosis of melanocytic skin lesions are. 19 The sensitivity and specificity of sentinel lymph node biopsy was obtained from the accompanying systematic review of the clinical evidence for the NICE guideline. 7 Patients staged as AJCC III or IV melanoma are based on computed tomography imaging with the sensitivity and specificity based upon the results of a meta-analysis. 20
Disease Progression/Transition Probabilities
Data for the natural history of undetected or untreated melanoma were obtained from an expert elicitation study. 21 Clinical experts from the United Kingdom (n = 8) were asked for their beliefs about the probability of progression from each of the starting stages stated (i.e., in situ to stage IV) to any other stage and death. Six-month transition probabilities were obtained from the expert elicitation study that asked, “Imagine a cohort of 100 patients with stage X undiagnosed and hence untreated disease. After 6 months, the patients may be in any of the following stages.” Experts assigned probabilities using the quantile method, where median and upper and lower 95% credible limits were elicited. For our model, monthly transition probabilities were subsequently assigned.
Recurrence Probabilities
Probabilities for disease and new primary cancers for AJCC stages IA to IIC over 10 years were obtained from an Australian study. 22 Data extracted from the study were used to estimate shape and scale parameters of the Weibull distribution for AJCC stage IA recurrence curve and used to calculate the transition probabilities for recurrence. Recurrence rates for the remaining melanoma stages were computed as a function of the probability of recurrence of stage IA using the distribution of the hazard rate of each stage up to stage IIC. 22 Recurrence rates for AJCC stages III and IV were assumed to be identical as those of stage IIC.
Mortality Probabilities
Based on the individual-level data (Durham cohort), age-specific, all-cause mortality rates were derived from general population statistics 23 with adjustment for melanoma-specific risk of death. 24 The monthly probability values of mortality from melanoma are presented in Table S3. These values are based on calculations from Wilson and colleagues. 24 The odds ratio of survival was a function of disease stage at diagnosis. This model assumed that stage IA disease has no impact on overall survival, then the monthly probability of death is calculated as the age/gender baseline rate for the general population, 23 adjusted for the odds ratio.
Resource Use and Unit Costs
Data on the costs incurred for the different surveillance regimens and their consequences were obtained from NHS Reference Costs. 25 The costs of drug treatment, targeted therapy for AJCC stage III and immunotherapy for AJCC stage IV disease were obtained from the British National Formulary as per current clinical treatment regimen.13,26
Health Utilities
A systematic review and meta-analysis of melanoma utility weights based on 33 health-related quality of life studies was identified which included studies from Australia, Europe, and North America. 27 The majority of studies used SF-36 (Short Form-36) and QLQ-C30 (Quality of Life Questionnaire), from which health-related quality of life scores reported were converted to EQ-5D utilities using published mapping algorithms. The ED-5Q is internationally accepted as the preeminent tool for estimating health state utilities and the use of EQ-5D scores is a requirement for many health technology assessments agencies worldwide. They sought to define posttreatment utilities by stage with time of data collection component up to 3 months, 3 to 12 months, and greater than 12 months. Utility values were converted to monthly utilities and were used in the economic model. The justification in using these values was that our model sought to capture the change in utility over time in order to estimate quality-adjusted life years (QALYs). 28
Model Analysis
Probabilistic sensitivity analysis demonstrates the parameter uncertainty in a decision problem and is part of best practice modelling guidelines. 29 Guidance on the number of simulations required in a model is seldom defined with the de facto standard of 1000 to 10,000 simulations likely to be sufficient, 30 and given computational restriction, we ran base-case analyses based on 1000 patients with 1000 PSA simulations including half-cycle correction. The model allows us to estimate the expected cost, QALYs for each strategy. Cost-effectiveness acceptability curves are plotted over various willingness-to-pay (WTP) thresholds for a QALY. One-way sensitivity analysis was conducted on probability of self-detection, probability of recurrence, utility scores, and specialist dermatological nurse/CNS diagnostic accuracy using selected strategies based on the base-case analysis.
Value of Information Analyses
The main uncertainties in the model are explored through the expected value of partial perfect information (EVPPI) analysis. EVPPI relates to the maximum cost of further research in removing the uncertainty in a set of values for a single or groups of parameters to get more precise estimates. Individual EVPPI was first calculated and then multiplied by the size of the population affected over the affected time horizon. For the estimation of the size of the population, the annual prevalence was obtained from National Cancer Registration and Analysis Service and proportion of substage from the Leiter et al. study.16,17 Two-level simulations were conducted to estimate EVPPI. The first level happened within the microsimulation by randomly selecting individuals of different age and sex from the individual-level data. Then, each selected individual was simulated 10,000 times (PSA), and values for the parameters were selected from prespecified distributions. Results from the PSA were then used within the Sheffield Accelerated Value of Information tool to estimate the EVPPI. 31
Four groups of parameters were considered in the EVPPI analysis: health utility values, diagnostic accuracy of health care professional to detect a melanoma, probability of transitioning between stages, and recurrence of melanoma.
Results
From the deterministic analysis, strategies involving dermatologists are likely to be preferred. Only a few strategies involving surgeons could potentially be cost-effective. No strategies involving specialist dermatological nurse/CNS were likely to be considered cost-effective and therefore not considered for the PSA base-case analysis. This is due to the lower diagnostic specificity assessment value, which results in patients been referred for further assessment by medical specialists (i.e., dermatologists or surgeons) instead of being discharged, which results in additional costs being incurred. See supplementary appendix for more details on results section.
Base-Case Analysis (PSA)
The base-case results for the 20 active surveillance strategies for stage IA and IB are presented in ranked order based on lowest mean costs in Table 2. Owing to similar effectiveness (QALYs), there is little to choose between strategies in terms of net monetary benefit (NMB). The probabilities of being cost-effective for different threshold values for society’s willingness-to-pay for a QALY are also presented. Given that 20 strategies are compared, it would be expected that each strategy would have a 5% chance of being considered cost-effective if equally likely. Using the lower bound of the conventional NICE threshold of £20,000 to £30,000/QALY gained, the strategy with the highest probability of being cost-effective is less than 23% and therefore even though 20 strategies were compared this is not a sufficiently compelling argument to change current recommendations. The cost-effectiveness planes comparing the strategies are shown in Figure 1.
NICE, National Institute for Health and Care Excellence; NMB, net monetary benefit; QALY, quality-adjusted life year.
Abs. dominated = absolutely dominated—this strategy is always more costly and less beneficial than comparator strategies. Ext dominated = extendedly dominated has a higher incremental cost-effectiveness ratio (relative to a third option) and fewer benefits than the alternative. Nondominated strategies highlighted in boldface.
QALY are approximate values reported to 4 decimal places. ICER and NMB reported as per software rankings report which utilizes more decimal places.
For IA, Strategy 22 (common baseline as it is least costly option) is the comparison or reference strategy and ICER are compared to preceding undominated strategy.
For IB, Strategy 77 (common baseline as it is least costly option) is the comparison or reference strategy are compared to preceding undominated strategy.

(A and B) Cost-effectiveness plane showing the mean cost and effectiveness (QALYs) for selected strategies for stages IA and IB.
For stage IA, strategy 22 (follow-up once at 12 months by a dermatologist) is on average the least costly surveillance strategy (£8467 per person) with 14.72 QALYs over the life-course. This strategy has the highest NMB (£285,894) at a WTP threshold of £20,000 per QALY gain with a probability of being cost-effective of 22%. The recommended NICE strategy (strategy 1, surveillance every 3 months for 1 year) has the fourth highest probability of being cost-effective at 12%. As society’s willingness-to-pay for a QALY gain increases, more resource-intensive strategies become progressively more cost-effective (Figure 2A). For example, strategy 14 (surveillance every 4 months for the first year then every 12 months for the next 9 years) has higher probability (>10%) at £50,000/QALY gained threshold.
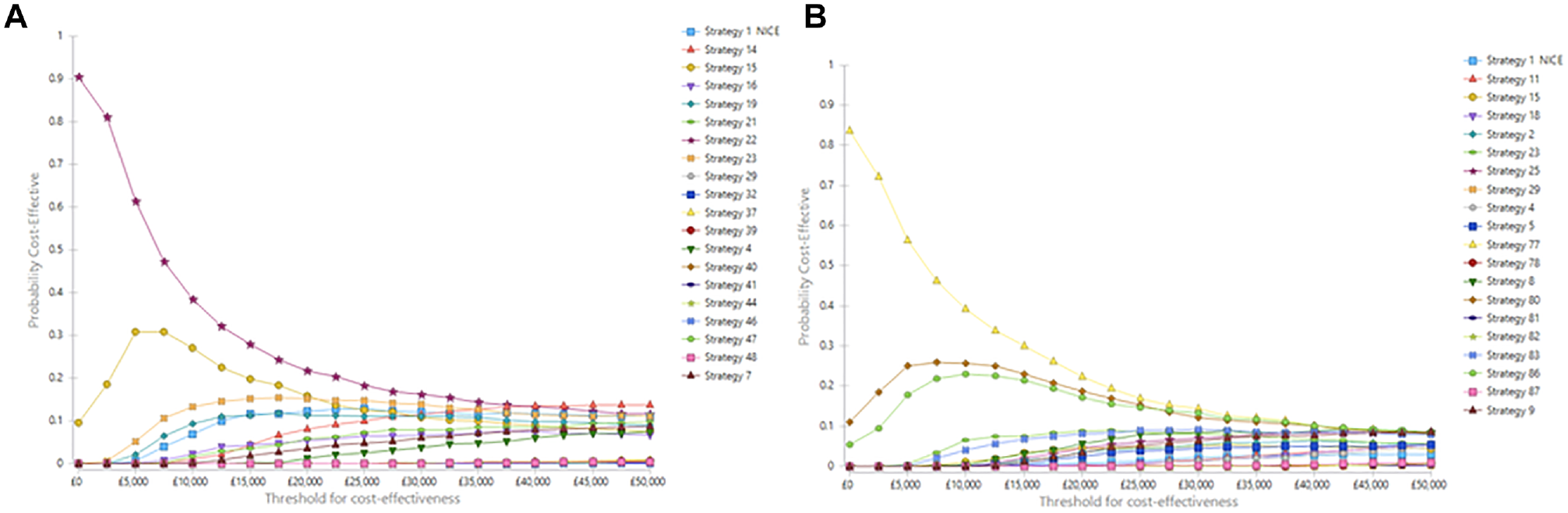
(A and B) Cost-effectiveness acceptability curve for stages IA and IB surveillance strategies
For stage IB, strategy 77, follow-up once for 1 year by a dermatologist, is on average the least costly strategy (£9517) with 14.57 QALYs (Table 2) over the life-course. This strategy has the highest probability of being cost-effective (22%) at the £20,000 per QALY gained threshold. Only two other strategies had a probability of being cost-effective of over 10%—strategy 80 (surveillance by a dermatologist every 12 months for 2 years) and strategy 86 (surveillance by a dermatologist every 6 months for 1 year) at this threshold. These strategies are less intensive than the NICE recommended strategy. None of the strategies led by a surgeon had a probability of being cost-effective above 1% over any of society’s WTP values for a QALY for stage I patients. As society’s willingness-to-pay for a QALY gain increases, more resource-intensive strategies become progressively more cost-effective (Figure 2B).
Expected Value of Partial Perfect Information (EVPPI) for Pertinent Parameters

This lifetime economic model allowed for a count estimate of recurrences and melanoma-related deaths for each strategy. Critically, for a cohort of 10,000 patients, the model estimates that the number of melanoma-related deaths to be 271 for stage IA under the NICE strategy and 369 for strategy 22 (the cost-effective strategy). For stage IB under the NICE strategy, an estimated 493 melanoma-related deaths compared to 672 under strategy 77 (the cost-effective strategy).
Sensitivity Analyses
When selected strategies were subject to one-way sensitivity analysis, only slight difference were noted. Increasing self-detection probabilities only made less intensive strategies more favorable than the NICE strategy. If recurrence probability increased, the benefits of surveillance will be in terms of faster detection of recurrence. If the absolute utility value associated with having a recurrence or new primary is above 0, then all strategies show a positive NMB. Without data to the contrary, most second primaries are like recurrences and are also diagnosed when they are at stage I disease (∼70%; see Table S4 for staging distribution of incidence case in England). For a less intensive surveillance strategy led by a nurse specialist, the specificity will need to be practically equivalent to dermatologist to have the same NMB.
Value of Information Analyses
In 2017, there were 8555 patients diagnosed with stage I melanoma. 16 The split in IA and IB was 63.37% as per German registry dataset. 17 The EVPPI of the four groups of parameters over a 20-year time horizon is presented in Table 3. Research that aims to remove the uncertainty associated with natural history would be most welcomed in both substages.
Discussion
Cancer surveillance schemes often lack a sound scientific base and impose a significant economic burden to health care systems and societies. 32 This study presents an economic evaluation exploring surveillance strategies for stage I melanoma patients in England. Our analyses found that less intensive clinical follow-up strategies may be more cost-effective than the strategies recommended in the NICE guidelines for stage IA and IB melanoma survivors. The main determinant of cost-effectiveness is the mean lifetime cost of the surveillance strategy as little difference is estimated in QALYs. This is not surprising for two reasons. First, the risk of further disease for people treated with stage I disease is quite low; and second, there is a very high level of self-diagnosis, which means that any form of active surveillance is limited in terms of the benefits it provides. Limited data prevent us from promoting an optimal strategy that can be strongly recommended for implementation in local guidelines.
Our findings complement two randomized control trials (RCTs) reported in the literature. First, the ongoing melanoma follow-up (MELFO) RCT in the Netherlands compares outcomes of a reduced follow-up schedule for patients with stage IB-IIC melanoma to the Dutch guidelines. 33 After 3 years, no statistically significant difference in disease-related outcomes (i.e., recurrence-free survival, locoregional, and/or distant disease or second primaries) were found. 33 The second RCT focused on the utility of self-examination by patients and partners in Chicago, USA, and found that those who performed more frequent self-examinations were more likely to identify new melanomas and consequently reduce the burden on physicians. 9
Our economic evaluation comes with both strengths and limitations. We created a de novo economic model specifically to answer the question: What is the optimal surveillance for stage I survivors? The model’s structure was informed by the clinical and patient members of the research team using an iterative consultation process. Model parameters and their values were obtained in a rigorous systematic manner. The model considers patient behavior in the form of self-detection and heightened anxiety that results in “false alarm” emergency visits. Perhaps the greatest limitation is the inherent problems in combining data from multiple sources, which result in a lack of precision in the estimates from the analyses. There is a paucity of research on quality-of-life of melanoma survivors. 34 Health state “utilities” values were based on patients with first primary melanoma diagnosis rather than recurrence. This implies that values are similar for patients with a history of melanoma to those newly diagnosed which may be erroneous. The lack of data of clinical accuracy of clinicians working in the NHS is a noteworthy limitation. Only restricted strategies were analyzed and by omitting relevant comparators an underestimation of ICERs may have occurred. 35 As there is a plethora of ways a surveillance strategy could be organized, a structured process was followed. However, it is possible that this process may have omitted a relevant comparator, but the key findings are unlikely to change. This we believe is an area of further methodological research. Finally, for computational burden reasons, this microsimulation was based on 1000 patients with 1000 PSA simulations (run time ∼7 days), which might not be sufficient to overcome any model instability.
The value of information analysis indicates that further research is extremely valuable (especially considered over a 20-year timeframe) and warranted in removing uncertainty around the diagnostic accuracy of health care professionals, the probability of transitioning between stages, health state utility values, and the recurrence of melanoma to make a confident decision to change NICE guidance. Additionally, future research to update the model based on the 2018 AJCC 8th edition staging criteria would be welcomed. 3 The key practical differences is that the definition of stage I disease has changed where classification of T1 melanomas was modified whereby mitotic rate is no longer a staging criterion and 0.8 mm, not 1.0 mm, has become the thickness boundary between T1a and T1b. In a small study from Australia (n = 208), 47% of T1b patients under AJCC 7th edition were downgraded to T1a under AJCC 8th edition. 36 This will make the value of active surveillance even more questionable for those with a stage IA diagnosis.
For dermatological nurse/CNS to be considered the main health care provider in surveillance, further work would be needed to determine and, if necessary, support the development of their diagnostic performance with extensive dermoscopy training. Specialist nurses providing surveillance would be less costly than dermatologists and it may be relatively easier to increase the cadre of nurses able to provide surveillance compared with increasing the numbers of dermatologists. Until diagnostic accuracy evidence is provided, the costs of the additional confirmatory follow up by dermatologists more than outweighs the lower cost of nurse provided melanoma surveillance.
In conclusion, our analyses suggest the benefits of NICE surveillance strategies for melanoma patients diagnosed with stage I disease are minimal compared to less intensive strategies. However, the underpinning model values to support any changes in the NHS lacks empirical evidence from the United Kingdom.
Supplemental Material
sj-docx-1-mpp-10.1177_23814683211069988 – Supplemental material for Optimal Surveillance Strategies for Early-Stage Cutaneous Melanoma Post Primary Tumor Excision: An Economic Evaluation
Supplemental material, sj-docx-1-mpp-10.1177_23814683211069988 for Optimal Surveillance Strategies for Early-Stage Cutaneous Melanoma Post Primary Tumor Excision: An Economic Evaluation by Vasileios Kontogiannis, Diarmuid Coughlan, Mehdi Javanbakht, Patience Kunonga, Fiona Beyer, Catherine Richmond, Andy Bryant, Dalvir Bajwa, Robert A. Ellis and Luke Vale in MDM Policy & Practice
Footnotes
Acknowledgements
The authors would like to thank colleagues Margaret Astin and Dawn Craig.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Financial support for this study was provided by the National Institute of Health Research (NIHR) Health Technology Assessment Programme, Grant Number: 16/166/05. The funding agreement ensured the authors’ independence in designing the study, interpreting the data, writing, and publishing the report.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.