Abstract
Introduction
When contemplating introduction of a new health care intervention for the first time, an accurate estimation of its full costs based on real-world data will usually not be available. Whereas it is increasingly recognized that rational decision making on the allocation of health care resources requires comprehensive assessments of the outcomes and benefits as well as the costs of interventions, it may be necessary to base decisions on provisional, approximate data.1,2 One approach to resolve this dilemma is to examine the costs of interventions that are similar to the one under consideration and which have already been implemented in the jurisdiction of interest or elsewhere.1,2
A case in point is the newly developed RTS,S vaccine candidate against malaria, which is considered for introduction in several African sub-Saharan countries, where the disease burden of malaria is still heavy. Despite sustained progress in the fight against malaria with an estimated decrease in malaria deaths worldwide of 60% since 2000, estimations from the World Health Organization (WHO) indicate that around 438,000 individuals died of malaria in 2015. 3 More than 90% of these deaths occurred in sub-Saharan Africa and most of them were children under the age of 5 years.
The RTS,S vaccine candidate received a positive evaluation by the European regulatory authorities and WHO is planning to conduct a pilot implementation of the vaccine in three sub-Saharan countries with moderate to high malaria transmission intensity. 3 To inform the consideration about including the RTS,S vaccine candidate as part of the Expanded Program on Immunization of these countries, estimates of the anticipated incremental costs of doing so are highly relevant and important. 4
The purpose of this study was to have a clear overview of the methods and estimates for vaccine implementation costs from the scientific literature. This review supports the design and allows comparison with results obtained from a field study conducted in five African sub-Saharan countries (Burkina Faso, Ghana, Kenya, Mozambique, and Tanzania) to derive an estimate of the anticipated costs of introducing the RTS,S vaccine candidate in sub-Saharan countries. This field study is described in the accompanying paper. 5 This review of the literature focuses on studies reporting the costs of recent vaccine programs in these countries. Ideally, such studies should be using micro-costing principles with itemized costs and separate reporting of quantities of resources and their unit prices. However, relatively few studies based on these principles have been published until now, although the importance of using this approach is increasingly recognized and guidelines for their performance, reporting, and appraisal are under preparation.6,7 We grant that the scarcity of studies in this area may be somewhat a function of our limited search (we used only one database). We therefore applied less strict criteria for inclusion of studies, but at minimum, studies should report itemized costs or a cost per dose delivered to be selected. Further inclusion criteria are detailed in the next section.
Methods
Systematic Search Strategy
PubMed was searched for relevant articles published between 2000 and the end of 2018 using the following search string:
(((((((vaccine OR vaccination) OR immunization) AND (economic OR cost)) AND (“2000/1/1”[Date - Publication]: “2018/12/31”[Date - Publication]))))) AND (Africa OR country x). The countries specified were Benin, Burkina Faso, Burundi, Cameroon, Central African Republic, Chad, The Democratic Republic of Congo, Côte d’Ivoire/Ivory Coast, Djibouti, Eritrea, Ethiopia, Gambia, Ghana, Guinea, Guinea Bissau, Kenya, Lesotho, Liberia, Madagascar, Malawi, Mali, Mauritania, Mozambique, Niger, Rwanda, Senegal, Sierra Leone, Somalia, Sudan, Tanzania, Togo, Uganda, Zambia, and Zimbabwe.
Screening
First, the title of the identified articles was screened, then the abstract of the retained possibly relevant articles, and finally the full text of papers retained after the two screening steps. The reference lists of the retained articles for full text screening were also examined and possibly relevant ones included in the screening steps. Papers were excluded if they specifically focused on non-GAVI (Global Alliance for Vaccines and Immunization) or non-African countries, if they were about nonhuman vaccines, and if they were written in languages other than English and French.
The criteria for selection of articles were: 1) costing study performed in a GAVI-supported sub-Saharan African country (or more than one) in order to have a more homogeneous set of countries in terms of income level and health system status; 2) study performed between 2000 and 2018; 3) costing performed for a human vaccine; 4) costs itemized and quantified; itemized costs reported in monetary terms or as percentages of an overall cost figure reported in monetary terms; and 5) possibility to calculate a comprehensive cost per dose administered, excluding the costs of vaccine.
Data Extraction
The data extracted from each selected study include the authors’ report of the costs of vaccination per dose. If the cost per dose was presented in the article, this figure was used as such. If the reported cost figure included the cost of the vaccine, we subtracted the vaccine cost from this or the proportion of non-vaccine costs in the total costs was applied. If a study reported the cost per fully vaccinated child (FVC), this figure was divided by the number of doses required for a FVC and, if necessary, the cost of the vaccine was deducted as well. For studies reporting detailed, itemized costs, the cost per dose was calculated as the sum of the itemized costs excluding the costs of vaccine and wastage. We did not have an a priori definition for wastage, and took wastage as the value defined by the study authors. In one particular case, the study reported a cost figure calculated as the weighted average of administration in urban and rural facilities based on the number of facilities of the respective type. 8 For this study, we recalculated the cost per dose using the weighted average of the number of doses administered in urban versus rural facilities.
Categorization
For cost items, the following categories were used and their proportion of the total cost per dose calculated to the extent possible: human resources, transportation, administration, equipment, sensitization, training, and surveillance. Explicit definitions for each category were not created; instead, we “agnostically” relied on the definitions used by authors themselves, and used common sense for classifying those expenditures that did not use the same jargon as our categories (e.g., “awareness” was classified as “sensitization,”“salaries” were classified as “human resources,” etc.). These categories were used to follow an approach similar to the one developed for the field study as described in the accompanying paper.
Perspective and Challenges
We only took into account the cost from the program/providers’ perspective. Though this excludes a great deal of the true economic cost of vaccine programs, this limited perspective is most relevant to the aim of this study, and most applicable to estimating potential program costs for the rollout of an intervention.
If incremental or marginal costs were mentioned in a study, that figure was preferably used rather than an average economic cost per dose in order to account for the actual budgetary requirement for the introduction of a new vaccine in the program. Though incremental costs are more variable and are highly contingent on local capacity, this approach was in line with our study’s aim than looking at full economic program costs. However, distinctions between cost types such as fixed and variable, start-up, and recurrent were not retained, because they were used inconsistently across studies, sometimes overlapping, sometimes insufficiently categorized, and sometimes in incompatible ways.
Some studies do not specify the vaccine purchase cost separately but combine it with injection material and other supplies. In such cases, we decided to eliminate the entire cost item including both vaccine and injection material costs, given that the costs of injection supplies generally are small compared with the vaccine purchase cost. Some studies do not explicitly mention the vaccine price,9–13 but they may mention the source of data; in such cases, we retrieved it from the data source, mostly the UNICEF website. 14
In other situations, it was not possible to remove the cost of wastage because it was not separately reported in the study and was therefore a nonobserved component of the final cost. If a study reported wastage separately, this item was not included in our calculation of the cost per dose.
Wastage is a major cost that is included in most studies but not always based on field data and often included in the vaccine cost and not reported separately. Wastage costs may be very high according to some studies12,15 and there are several difficulties involved in handling them. Wastage costs depend mainly on the vaccine price and the level of the health care system at which the wastage occurs, with variation between routine vaccinations in health facilities and outreach activities or vaccination campaigns. Wastage also varies with the number of doses per vial, the service volume in terms of number of vaccinations administered, and the vaccine characteristics. As a consequence, the costs related to wastage vary widely and may be difficult to capture fully. For these reasons we did not include wastage as a separate cost category in the cost estimation.
Monetary Homogenization
All the studies report costs in US dollars (US$) for a base year, usually the year the cost data were collected. All the cost figures were converted to 2015 US$ using data from the World Bank. 16 This conversion was performed in 3 steps: 1) conversion from US$ to the local currency unit (LCU) for the base year reported in the study; 2) taking inflation into account by applying the consumer price index increase in the LCU from the base year until and including 2015; and 3) converting the inflation corrected figure back to 2015 US$. The search query was devised by CS. The initial search was carried out by JB; iterative screening was carried out by both JB and CS.
Results
Screening Results
The process of articles selection is summarized in Figure 1 with details on the reasons for exclusion. The initial search returned 2119 articles. Based on the title alone, 941 were retained and 1178 were eliminated for one of the following reasons (from the most to least common reason): 1) no or insufficient economic content, 2) not vaccination of humans, 3) not about a vaccine, 4) not about an African GAVI country, 5) article withdrawn, or 6) not in English or French.

Selection of articles.
The abstract of the 941 retained articles were read, and based on this, 778 articles were excluded for one of the following reasons (from the most to least common reason): 1) no cost researched, retrieved, reported or not in relation with vaccine administration; 2) cost not related to vaccine delivery or was for delivery in a specific emergency context; 3) no abstract found or not in English/French or article withdrawn; 4) not vaccination in humans; 5) not in African GAVI country; 6) vaccination of special subgroups not including children; 7) opinion paper, review, qualitative study, meeting report; or 8) not about a vaccine.
The remaining 163 articles were screened based on the full text and 108 were excluded based on one of the following reasons (from the most to least common reason): 1) referring to another article (added or already included in the articles reviewed), 2) no delivery cost or no doses reported, 3) limited to specific cost items (often injection or logistics), 4) based on assumptions or models only, 5) special vaccination context (e.g., refugees, outbreak, emergency), 6) referring to another article that could not be found/included, 7) not in African country, 8) the vaccine price could not be segregated from the cost, or 9) data not available or accessible in journal supplement.
In addition to the 55 articles selected by this procedure, three more were included after examination of eight articles found in the reference lists of selected articles. The other five were excluded because they focused on special emergency situations (2), only logistics costs (1), or did not allow elimination of the vaccine purchase cost (1).
The 58 articles contained 80 data points, that is, combinations of country, vaccine type, and vaccination approach (routine or campaign). The 22 countries included had a preponderance of East African countries, and the most frequently represented were Tanzania (9/80), Uganda (9), Zambia (8), Ethiopia (7), Ghana (6), Kenya (4), and Burkina Faso (4). One study aggregated data for 27 African countries, 17 and another one aggregated data on HPV immunization for 10 African countries. 18
Altogether 11 different vaccines including a category defined as “multiple vaccines” when vaccines could not be distinguished were examined, and those costed in the 58 data points on routine vaccination were multiple vaccines (17), measles (8), malaria (8), rotavirus (7), human papillomavirus (HPV; 8), hepatitis B virus (HBV; 5), pneumococcal conjugate vaccine (PCV; 4), and meningitis (1). The vaccines included in the 22 data points for campaign-type vaccination were predominantly measles (8), cholera (7), and meningitis (3), with the remaining including typhoid, yellow fever, and multiple vaccines.
Most studies used the so-called ingredients approach as costing method, combining field data collection with documented unit prices for each cost item. One clear example of this is the EPIC (Extended Program of Immunization Costing and Financing) project, which collected data for routine vaccination cost using the same consistent methods in around 50 vaccine delivery facilities in each of four African GAVI countries (and two non-African).8,19,20 Of the 80 data points, 55 (69%) are based on primary cost data collected in field studies, with some variation in the extent of field data collection and in some cases limited to interviews with health workers.
For a minority of studies,17,20–25 the costing is based on existing budget plans (such as comprehensive multi-year plans (cMYP) or financial plans for immunization of the Ministry of Health). An average cost of administration is then calculated based on the total cost and the number of doses planned. One study in Nigeria 26 is based on the cost of administration estimated in a study in Tanzania, 27 with an adaptation limited to the difference in purchasing power.
Several studies of vaccination campaigns are based on all the costs incurred over a relatively short time period in the campaign and recorded in financial accounts or reports on the campaign.28–33
A number of studies are actually mainly cost-effectiveness analyses just reporting the cost of vaccination used as an input, sometimes based on primary collection of field data.9,21,24,25,34–41 The initial PubMed search returned several more cost-effectiveness studies but these were excluded because they based their cost estimates on cost data from papers included in the review. The cost-effectiveness analyses retained in the review have either collected primary cost data or are based on other studies.
Analysis
The studies are summarized in Table 1 with a brief indication of the methods used by the authors for the cost estimation and the estimated average cost per dose in 2015 US$. All the costs extracted are estimated from the perspective of the public health authorities.
Summary Table of Costing Studies, Cost per Dose in 2015 US Dollars
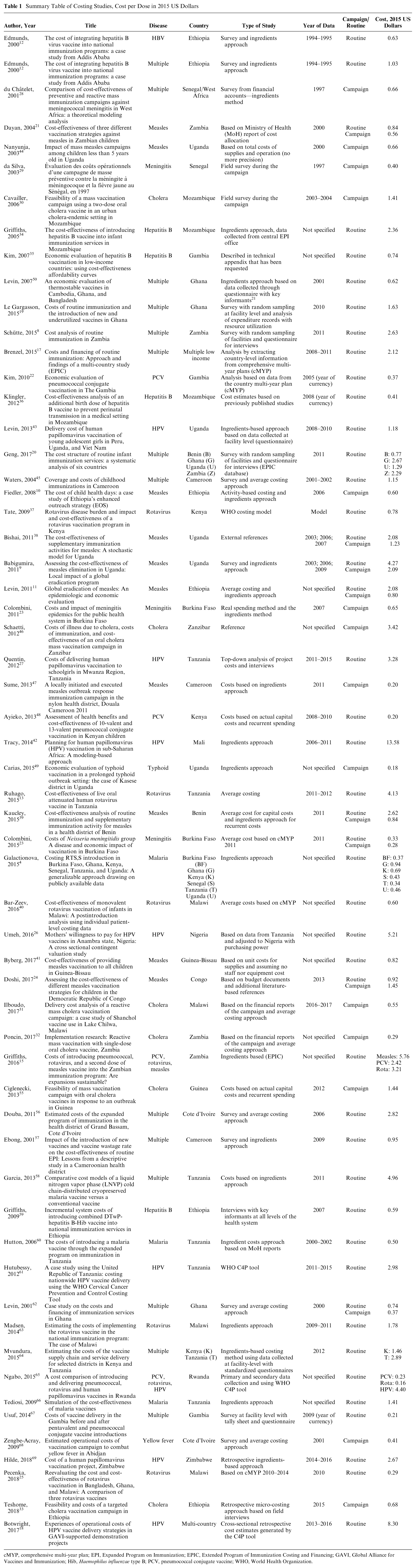
cMYP, comprehensive multi-year plan; EPI, Expanded Program on Immunization; EPIC, Extended Program of Immunization Costing and Financing; GAVI, Global Alliance for Vaccines and Immunization; Hib,
The aggregate cost results are summarized in Table 2, with overall average and median cost per dose of US$1.68 and US$0.88, respectively, and a range from US$0.16 to US$13.58. For routine vaccinations, the average and median cost per dose are US$1.99 and US$1.17, respectively, and for campaign-type vaccinations US$0.88 and US$0.66, respectively. The histogram in Figure 2 shows the distribution of cost results in intervals of US$0.25. For both types of vaccination approach, the major part of the average cost results is in the interval US$0.25 to US$1.50, with 33/58 of the routine vaccination studies and 20/22 of the campaign-type studies.
Summary of Cost per Dose Delivered


Number of data points reporting average cost in cost intervals of US$ 0.25.
The cost per dose varies with the type of vaccine as shown in Table 3. HPV is an outlier with average and median costs of US$5.20 and US$3.84, respectively, for routine vaccinations, far above the corresponding cost figures for other routine vaccination programs. These aggregates are very much determined by a single study in Mali with a cost of US$13.58, 42 but even disregarding that study the costs are high, with a range of average costs for the remaining five HPV studies from US$1.18 43 to US$5.21. 26 A multi-country study found an average cost of US$8.30 per dose administered. 18 The reason for the elevated costs may be that HPV vaccines are typically delivered through a bundling with school-based or outreach programs, which require more training and personnel. If HPV vaccinations are excluded from the aggregation, the average cost for routine programs would be US$1.47 instead of US$1.99.
Cost per Dose Delivered for Different Types of Vaccine, 2015 US$

HBV, hepatitis B vaccine; HPV, human papilloma virus; PCV, pneumococcal conjugate vaccine.
There was a tendency for the newer studies to be more comprehensive including further cost categories such as the costs of social mobilization/sensitization and surveillance programs. The results regarding the proportion of the total cost per dose accounted for by each cost category are based on the studies for which these data were available. Sixteen of the 58 articles do not report any data on cost categories,9,22,23,25,28,35,37,38,40,41,44–49 and one study only provides details on transportation, representing 17% of the total cost per dose. 50 Table 4 presents the number of data points for each cost category and its average proportion of the cost per dose based on the available data (Note: the average proportions are not supposed to sum to 100 across the cost categories due to the gaps in the reporting of categories). Human resources and transportation are the most frequently reported categories and surveillance the least. The average proportions vary somewhat between the vaccination approaches, in particular, for human resources, administration, and equipment. Overall, human resources account for almost half (44%) of the average cost per dose followed by administration, transportation, and building/equipment (each about 20%).
Mean Proportion of Cost per Dose for Each Cost Category from the Studies Reporting Each Item
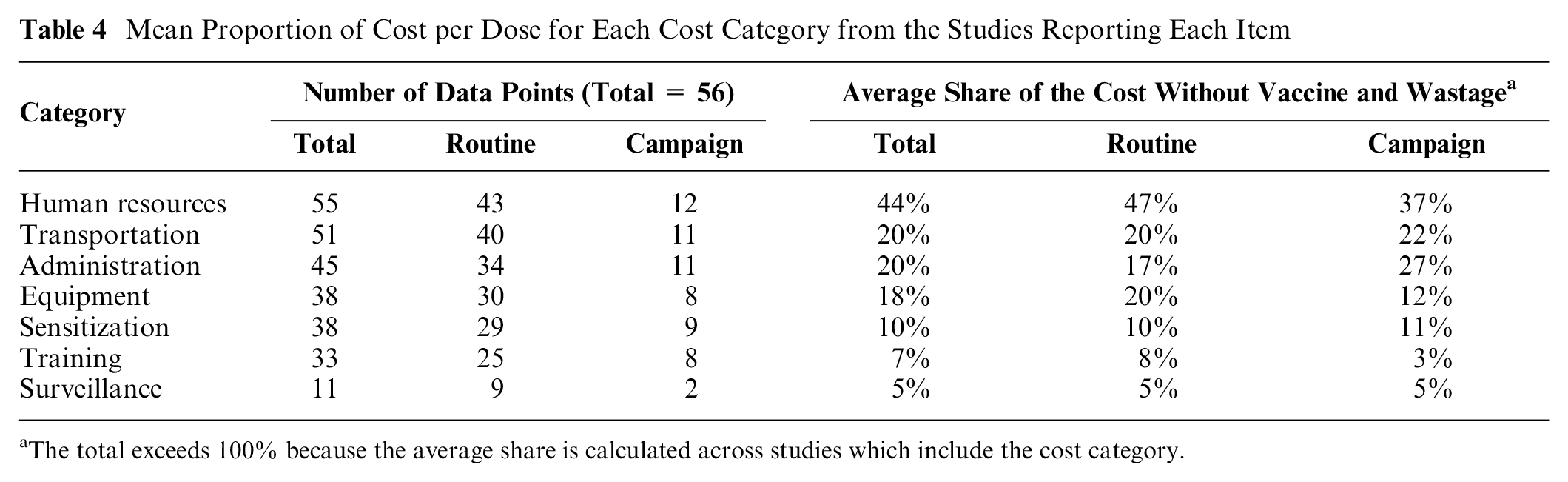
The total exceeds 100% because the average share is calculated across studies which include the cost category.
Discussion
This review and summary analysis of vaccination costing studies performed in sub-Saharan African countries shows that the estimated cost per dose (excluding vaccine and wastage costs) varies substantially across studies. Even though the costing methods used are fairly consistent, predominantly using an ingredients approach with data collection by questionnaires, interviews, or reports, the studies lack standardization with respect to which cost items are reported and how these are reported, so their findings are not easily comparable. We suspect that a large part of the variation in the estimated costs reflects differences in what is reported under each cost category.
It should be noted, however, that the largest study in our review using consistent methods and tools of data collection and cost estimation for approximately 50 health facilities in each of four sub-Saharan African countries also finds high variability between (and within) the countries with a more than threefold difference between the minimal and maximal cost per dose, that is, US$0.77 in Benin and US$2.67 in Ghana. The study authors consider that the estimated variation reflects real differences between the countries in unit prices, characteristics of the health systems, and in the practical organization of the vaccination programs. 20 Of note, the per capita income in Ghana is more than double that in Benin, the countries in that study with the highest and lowest cost per dose, respectively, so the unit costs of labor and other resources are much higher in Ghana. 51
Across the studies, personnel costs amounted to approximately half the cost per dose. Labor time is a shared resource, which requires estimation of both the proportion of the time of each type of personnel to vaccination activities and within vaccination the allocation of time to different activities. Allocation of labor time is probably one of the cost categories most difficult to estimate reliably without direct observation, because respondents may be motivated to report a particular allocation of effort. Interestingly, the above-mentioned four-country study reports a substantial reduction of the labor cost per dose with an increasing vaccination activity (number of doses administered per time period), which suggests improving efficiency through economies of scale in the use of labor with rising vaccination activity. 20 However, such a possible relationship has not been investigated in other studies in this review.
Our findings are similar to results reported by Portnoy et al., 52 which estimate the cost of vaccination programs in 94 low- and middle-income countries using model-based costs and cMYP planned budgets. The reported average cost per dose for routine and campaign delivery approaches in low income countries is (2010)US$1.75 when the vaccine cost is excluded, similar to our average of (2015)US$1.68 across the GAVI countries. Human resources cost categories are also identified as the most important but with a higher proportion (82%) than in our study, although this figure includes both low- and middle-income countries. Another recent review reports a range of the incremental economic cost for newly introduced vaccines (PCV and rotavirus vaccines) between (2016)US$0.48 to US$1.38 in Benin, Rwanda, and Uganda. These results are also close to our findings with means of (2015)US$0.81 and US$1.57 for these two vaccines. 53
Among the specific difficulties encountered in allocating costs into one of the categories we had predefined, were that some papers disclosed more details than the specified categories, for instance, reporting the cost of personnel time devoted to training; in such cases we had to choose the higher level category in which to allocate the cost. The reverse problem also occurred, where papers reported aggregated categories (e.g., transportation and equipment combined). In such cases we allocated the cost to the category expected to represent the highest cost. Another type of situation was that a category used in a study did not match the ones we selected. An example would be “cold chain,” which could either be part of the equipment (fridges) in a health facility or related to transportation of vaccines. In such cases we qualitatively based the cost allocation on elements of the text description in the paper.
We found that routine vaccination programs generally have higher reported costs than introduction campaigns even though some campaigns have very high human resources costs. The likely reasons for the lower cost of vaccination campaigns is that they require less capital investment in health facilities than routine vaccination, and/or capital investments are not considered into these studies. A further hypothesis is that this finding might be explained by a tendency for costing studies of routine vaccination programs to be thorough and comprehensive, whereas costing of campaigns or introduction programs perhaps tend to focus most on those costs that differed from routine programs. This somewhat counterintuitive finding could also be explained by the fact that studies did not generally report or describe differences in financial versus economic costs. Accordingly, we were unable to perform separate financial versus economic analyses. This, unfortunately, limits the generalizability of our study.
Authors more frequently reported incremental costs instead of average economic costs. Though this is helpful in determining the marginal cost of a hypothetical program to be rolled out, this also poses an important limitation to the applicability of our study: since incremental costs is highly contingent on local capacity and infrastructure, its variability is high, and its generalizability is low. Additionally, itemized summary statistics should be interpreted with caution, since our inclusion criteria was fairly broad, and because of the incompatibility of cross-study categorizations.
We included several studies with “multiple” vaccines because they were integration campaigns (i.e., integrating a new vaccine into an existent multi-vaccine program, or rolling out an intervention with multiple vaccines). This may lead to some cost inflation that would not have occurred were we to have limited our study pool to only those programs which administered one vaccine in isolation. However, we chose not to adopt such a restriction because 1) it would have reduced our sample significantly, ignoring otherwise useful information from multiple programs, and 2) it would have imposed a condition on costs which hypothetically might not even correspond to the rollout of an RTS,S campaign in the future (i.e., there is no reason to suggest at this point that RTS,S would not be rolled out in the framework of an “integrated” program).
Although some studies make a clear distinction between the cost of existing programs and the cost of introducing a new vaccine, most studies do not systematically separate capital costs and recurrent costs or average versus incremental costs. Very few studies make it clear how discounting of capital costs (including training costs as investments in human capital with an expected depreciation period of some years) has been handled. Capital cost is in general annualized based on the life expectancy of equipment but with limited information.
The studies generally apply a provider or health care system perspective and few studies consider the wider societal perspective by, for example, taking into account the costs for families in terms of transport and opportunity costs such as time lost for other activities when accompanying their child for vaccination. However, the wider societal perspective is mostly relevant for a comprehensive evaluation of the economic value of vaccination and less relevant if the purpose of the assessment is more specifically to understand the cost structure to possibly improve efficiency and reduce costs.
Our aim was to support the design of a field study to estimate the cost of RTS,S rollout. Also by generalizing to vaccines as a whole, our results may be generalizable to vaccine campaigns at large, rather than just RTS,S.
A limitation of this review is that we used one database (PubMed) for our search, which may have limited the numbers of studies identified. There may be additional studies published in peer-reviewed journals, and it is likely there are numerous small specific studies in the gray literature that have been overlooked. Nevertheless, this review of 58 articles provides an indication of the likely cost estimates and potential budget required for introducing a new vaccine. To conclude, given the wide variation in the cost per dose (between and within countries) even in studies using consistent data collection tools and analysis methods across a large number of health care facilities in several countries, it would not be reasonable to try to fix a point estimate for the costs per dose. When considering inclusion of a new vaccine in countries targeted by this review, perhaps the overall interquartile range of US$0.54 to US$2.31 estimated here could serve as a reasonably precise baseline estimate but at the country level it would be useful to perform cost estimations strictly following the guidelines already available.
It would thus be commendable in future studies to adopt the method of the EPIC studies with a distinction between resource items (such as personnel, equipment, vehicles, buildings, etc.) and the various functions or activities each of these are used for. For example, personnel are participating in training, so the cost item is “personnel cost” but the actual activity is indeed “training.” In this review, we have observed that studies use inconsistent approaches with regard to reporting costs by item or by activity, making aggregation and comparison difficult. Combining cost items and activities in a matrix for cost calculation as proposed by Brenzel et al. 54 and illustrated in Figure 3 could greatly increase transparency and improve the understanding of the cost structure and its determinants in order to increase efficiency and help planning resource requirements and financing needs. Additionally, aggregate studies such as this one would be of greater accuracy and applicability were the component costs categorized more consistently and transparently.

Matrix of cost items and activities for structuring vaccination cost calculations. 54
Supplemental Material
Focus_On_Patient_REVISED.rjf_online_supp – Supplemental material for A Systematic Review of the Incremental Costs of Implementing a New Vaccine in the Expanded Program of Immunization in Sub-Saharan Africa
Supplemental material, Focus_On_Patient_REVISED.rjf_online_supp for A Systematic Review of the Incremental Costs of Implementing a New Vaccine in the Expanded Program of Immunization in Sub-Saharan Africa by Joe Brew and Christophe Sauboin in MDM Policy & Practice
Supplemental Material
Supplemental_-_PRISMA.rjf_online_supp – Supplemental material for A Systematic Review of the Incremental Costs of Implementing a New Vaccine in the Expanded Program of Immunization in Sub-Saharan Africa
Supplemental material, Supplemental_-_PRISMA.rjf_online_supp for A Systematic Review of the Incremental Costs of Implementing a New Vaccine in the Expanded Program of Immunization in Sub-Saharan Africa by Joe Brew and Christophe Sauboin in MDM Policy & Practice
Footnotes
Acknowledgements
The authors would like to thank Elisa Sicuri (Barcelona Institute for Global Health) for her input and guidance. The authors would also like to thank Niels Neymark (independent scientific writer, on behalf of GSK) for medical writing assistance and Business & Decision Life Sciences platform for editorial assistance and manuscript coordination, on behalf of GSK. Fabien Debailleul coordinated publication development and editorial support.
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Joe Brew has nothing to disclose. Christophe Sauboin is an employee of the GSK group of companies and holds shares in this company.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Financial support for this study was provided entirely by GlaxoSmithKline Biologicals SA (TrackHO Number: HO-14-14725). The funding agreement ensured the authors’ independence in designing the study, interpreting the data, writing, and publishing the report. The following author is employed by the sponsor: Christophe Sauboin. Joe Brew is a recipient of an Erasmus Mundus Joint Doctorate Fellowship, Specific Grant Agreement 2015-1595.
Author contributions
All authors comply with the ICMJE criteria for authorship. J. Brew and C. Sauboin were involved in the conception and/or the design of the study. J. Brew and C. Sauboin participated in the collection or generation of the study data. J. Brew and C. Sauboin conducted the study. C. Sauboin contributed to the analysis tools. J. Brew and C. Sauboin were involved in the analyses and/or the interpretation of the data. All authors read and approved the present article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
