Abstract
Localized prostate cancer is the most common cancer among men in the United States. In 2018, there were 164,690 new cases of prostate cancer and 29,430 prostate cancer–related deaths. 1 Treatment choice for localized prostate cancer is preference sensitive given that several medically viable and effective treatment options are available, each with specific risks and benefits. The patients need to be aware of all treatment options to assess which options fit best with their outcome goals. For a treatment-naïve patient, this process is not intuitive. Despite the guidelines about providing patients with sufficient information, doing so to facilitate patient-centered decision making is complex.2,3
Patient-centered care is “providing care that is respectful of and responsive to individual patient preferences, needs and values, and ensuring that patient values guide all decisions.”4,5 Respecting and responding to patient preferences is the hallmark of patient-centered care and requires accurately eliciting preferences and aiding patients in constructing them. Incorporating patients’ values and preferences into health care decision making is essential for patient-centered care. 6 Values clarification is a term used to describe the process where patients evaluate the desirability or attributes (i.e., features) of options in the context of a specific decision to identify the preferred option.7–9 Of the available prostate cancer decision aid tools, only few focus on the complex process that patients undergo to assess the pros and cons of a decision.10–15 Tools that integrate input from multiple sources—patients, providers, and stakeholders who have experienced treatment decision—can help localized prostate cancer patients understand their preferences and facilitate patient-centered decision making.4,6 There are no established methods for helping patients understand the tradeoffs between different treatments in clinical settings. Moreover, patients are often hesitant or unable to reveal their preferences,13,16–19 which may be, in part, due to the difficulty in evaluating multiple aspects of treatment concomitantly. 20
Conjoint analysis (CA) is a method for assessing stated preferences. The CA is a de-compositional preference assessment technique developed in the fields of economics, marketing, and psychology and can enhance patients’ awareness of what they value about a given treatment.8,11,16,21,22 The CA has been used to study a wide range of health care–related decisions.23–29 Adaptive conjoint analysis (ACA) is a type of CA that includes a component where values clarification is made more explicit, through a ranking exercise that precedes the hypothetical treatment option questions. 30 The ACA has a strong theoretical basis and is customized for each respondent.8,16,21,30,31
While many have focused on the construction of decision aid tools using CA and experimental design, 29 relatively little attention has been paid to the crucial process of identifying the appropriate treatment attributes. Preference assessment is helpful if it addresses what is most significant to the patient. While patients’ perspectives have been used to develop CA-based decision aid tools, 32 there is less guidance on balancing the attributes identified by patients, clinicians, and researchers. The objective of this article was to describe the development of a patient-centered preference assessment tool, the Patient Preferences for Prostate Cancer Care or PreProCare tool, for localized prostate cancer.
Methods
Identifying attributes or features of treatment options and their levels that are relevant to patients for decision making is the critical component of PreProCare tool development. Here we describe our comprehensive approach of integrating systematic literature review, patient interviews, provider focus groups, and stakeholders’ feedback to determine the attributes and their levels that are relevant to localized prostate cancer. As presented in Figure 1, five steps in the development of PreProCare tool11,29,33 are the following: 1) systematic literature review and meta-analysis, 2) semistructured patient interviews, 3) provider focus groups, 4) finalizing attributes and levels, and 5) tool development and pilot testing. In the following sections, we discuss each step in detail. The local institutional review board approved the study.
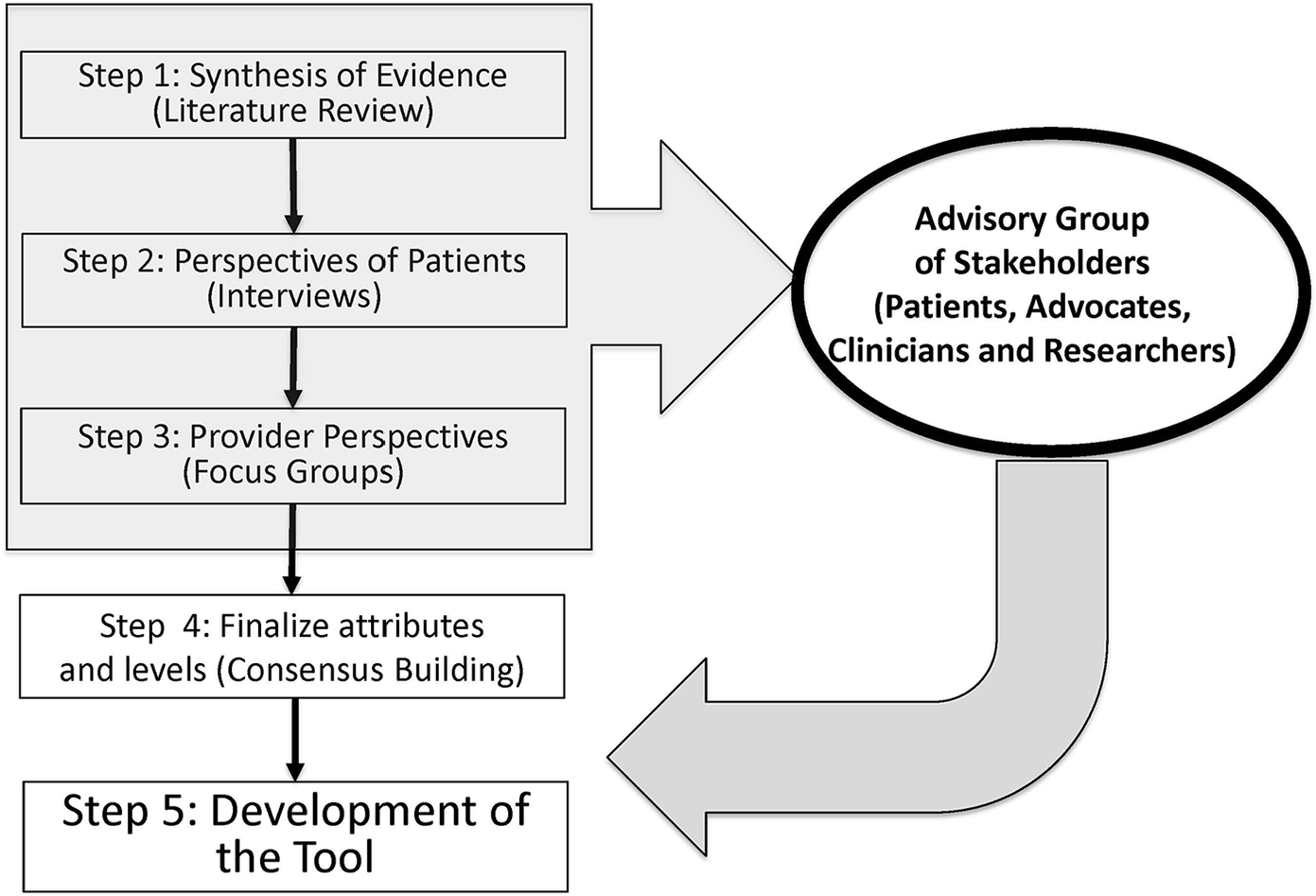
Steps in PreProCare tool development. Schematic representation of the steps leading to the development of PreProCare tool.
Systematic Literature Review and Meta-Analysis
The objective of the systematic literature review and meta-analysis was to ascertain available information regarding patient-centered attributes that are associated with prostate cancer treatment options. 34 A review of literature published in English was conducted using Medline, PubMed, CINAHL, EMBASE, and Cochrane Library. The search strategy used terms such as “prostate cancer,”“localized,”“outcomes,”“mortality,”“health related quality of life,” and “complications” to identify relevant studies. Prostate cancer patients of all ages were included. We excluded studies if the intent of treatment was salvage therapy, if it included patients with clinical stage >T3a, 35 or if patient-centered outcomes were not reported. When multiple articles resulted from the same study, the selection favored longer follow-up, larger sample size, and completeness of information. For prospective and retrospective studies, those adjusting for selection bias using propensity score or instrumental variable approaches were included. The literature review yielded attributes and helped develop questions for the semistructured interviews.
Semistructured Patient Interviews
One issue with many decision aids is that they use terminology and outcomes deemed important by the medical community. However, these outcomes may not necessarily align with the way patients think about treatment options or the terms they use. We therefore set out to capture the attributes driven by the experiences of a wide range of prostate cancer patients. We sampled prostate cancer survivors who had undergone different treatment modalities—surgery, hormone therapy, radiation therapy, or active surveillance. Our literature review informed the open-ended questions that were used in the semistructured interviews (Online Appendix). We included questions to probe prostate cancer survivors about the attributes of prostate cancer care that were commonly cited in the literature, as well as to understand their experiences. The questions focused on what patients understood about treatment options, how they decided on a treatment, their experience of side effects, impact on health-related quality of life (HRQoL), and anything they wish they had known before making a treatment decision. Patients also completed a survey that asked them about entities that were important sources of information, and for decision making, and finally a list of attributes to rate. Patients rated each attribute on a scale from 1 (“very important”) to 5 (“not at all important”) for decision making, assuming they were making a treatment decision. We evaluated the answers to determine the internal validity of the responses or the agreement of the responses with the overall discussion during the one-on-one interviews. 23
Provider Focus Groups
Providers are likely to see different sequelae and have different perspectives on treatment. Therefore, the third step in our PreProCare tool development consisted of focus groups with providers from a range of settings (academic practice, cancer center, and Veterans Administration–affiliated practice) and backgrounds (urologists, radiation oncologists, primary care providers, geriatricians, and urologic nurse practitioners). A facilitator conducted the provider focus groups in a semistructured interview format, and audio recorded the meetings. The discussions focused on providers’ experience in treating localized prostate cancer, clinical information deemed necessary for effective decision making, important attributes of treatment options and their levels, and barriers to optimum treatment choice.
Finalizing Attributes and Levels
We pooled the attributes from systematic literature review, patient interviews, and provider focus groups. This pool of attributes and their levels was reviewed by investigators, internal and external collaborators, and stakeholders’ advisory group, which included four prostate cancer survivors (patient stakeholders), physicians (urologists, oncologists, radiation oncologists, primary care providers), and nurse practitioners and researchers. We achieved consensus through deliberations during an in-person advisory board meeting that was about 90 minutes long. Our team of researchers, physicians, and the advisory board members discussed each attribute and associated levels to determine its importance in treatment decision making and developed a list of final attributes. The total number of attributes were decided to make sure that the instrument was not too long and did not overburden the participant.
PreProCare Tool
Sawtooth software 30 was used to develop the PreProCare tool. First, the respondent was asked how important the difference in a level of a particular attribute was (not important, somewhat important, very important, extremely important) while comparing two hypothetical treatment options (A and B) that were otherwise similar. Next, based on these responses, personalized, hypothetical choice scenarios were developed that present bundle of features for two hypothetical treatment options. The respondents were required to state the preference as follows: strongly prefer Treatment A, somewhat prefer Treatment A, no preference, somewhat prefer treatment B, or strongly prefer treatment B. These choice scenarios elicited the conjoint tradeoffs.
Pilot Testing
We piloted the PreProCare tool to test its feasibility and acceptability. From the urology practices of one of the study sites, a large urban academic health system, we obtained a convenience sample of newly diagnosed prostate cancer patients who were treatment naive. Those who agreed to participate gave informed consent. A participant could complete the tool in the office or was given a URL link, individualized user-ID, and password to access it at home. Each participant received a $20 gift card. We assessed the time needed to complete the PreProCare tool and asked the participants to rate the ease of completion, and the tool’s usefulness in clarifying values.
Patient Involvement
To ensure that research findings are relevant to the users, we designed our study carefully and obtained strong and substantial input from prostate cancer patients for all phases of the study. Three categories of prostate cancer patients were involved in the study: prostate cancer survivors (patient stakeholders) who were members of our advisory board; prostate cancer survivors who underwent one-on-one interviews as part of the development of our preference assessment tool; and newly diagnosed prostate cancer patients who participated in pilot testing of our tool. We engaged patient stakeholders (advisory board members) at the beginning of the study to help clarify assumptions, explore completing explanations, and develop consensus around the issues of prostate cancer treatment attributes. These patient stakeholders also provided input toward interview guide and surveys, PreProCare tool design and pilot testing of the tool to assure that most essential and appropriate information was collected.
Analysis
Analysis was consistent with the steps described above. The literature review was conducted by three independent reviewers per the Preferred Reporting Items for Systematic Reviews and Meta Analyses (PRISMA) criteria. 36 Patient-centered outcomes were summarized and tabulated for various treatments to facilitate identification of attributes of the treatment options and their levels. Next, thematic analysis was chosen as the methodological framework within which to analyze the interview and focus group data. 37 Thematic analysis is a method of “identifying, analyzing, and reporting patterns (themes) within data.” 38 It is a descriptive method that enables data reduction in a flexible way. 39 Our thematic analysis was performed in the following five steps: data organization and familiarization, initial broad code generation, applying broad codes to data, identifying attributes within coded text, and naming attributes. This thematic analysis was conducted by four members (SC, JG, RJ, and MW), who also verified the intercoder agreement. Discrepancies in coding or attribute definitions were resolved via discussion. Ongoing analysis led to refinement of each attributes, and generating clear definitions and names.
Results and Discussion
Systematic Literature Review and Meta-Analysis
We reviewed 56 articles that met our criteria. Details of the literature review have been reported earlier. 34 Table 1 presents a synthesis of comparative evidence for survival, cancer recurrence, side effects and HRQoL, satisfaction with care, and decision regrets across treatment groups, 34 including conservative management strategies such as watchful waiting or active surveillance. Watchful waiting is an unstructured follow-up, mostly among patients with an actuarial survival of ≤10 years. On the other hand, active surveillance represents a structured program involving PSA (prostrate-specific antigen) monitoring, physician exam, imaging, and pathological evaluation with biopsy. The literature synthesis informed the subsequent semistructured patient interviews (Online Appendix) and provider focus groups. The following patient-centered attributes were revealed from the literature review (Figure 2): survival (overall and disease-specific), cancer recurrence (metastasis), complications (blood loss, recurrence, incontinence, and erectile dysfunction), side effects (fatigue, nausea, loss of fertility), and HRQoL (functional status, generic and prostate cancer–specific HRQoL).
Synthesis of Patient-Centered Outcomes Evidence for Localized Prostate Cancer 33
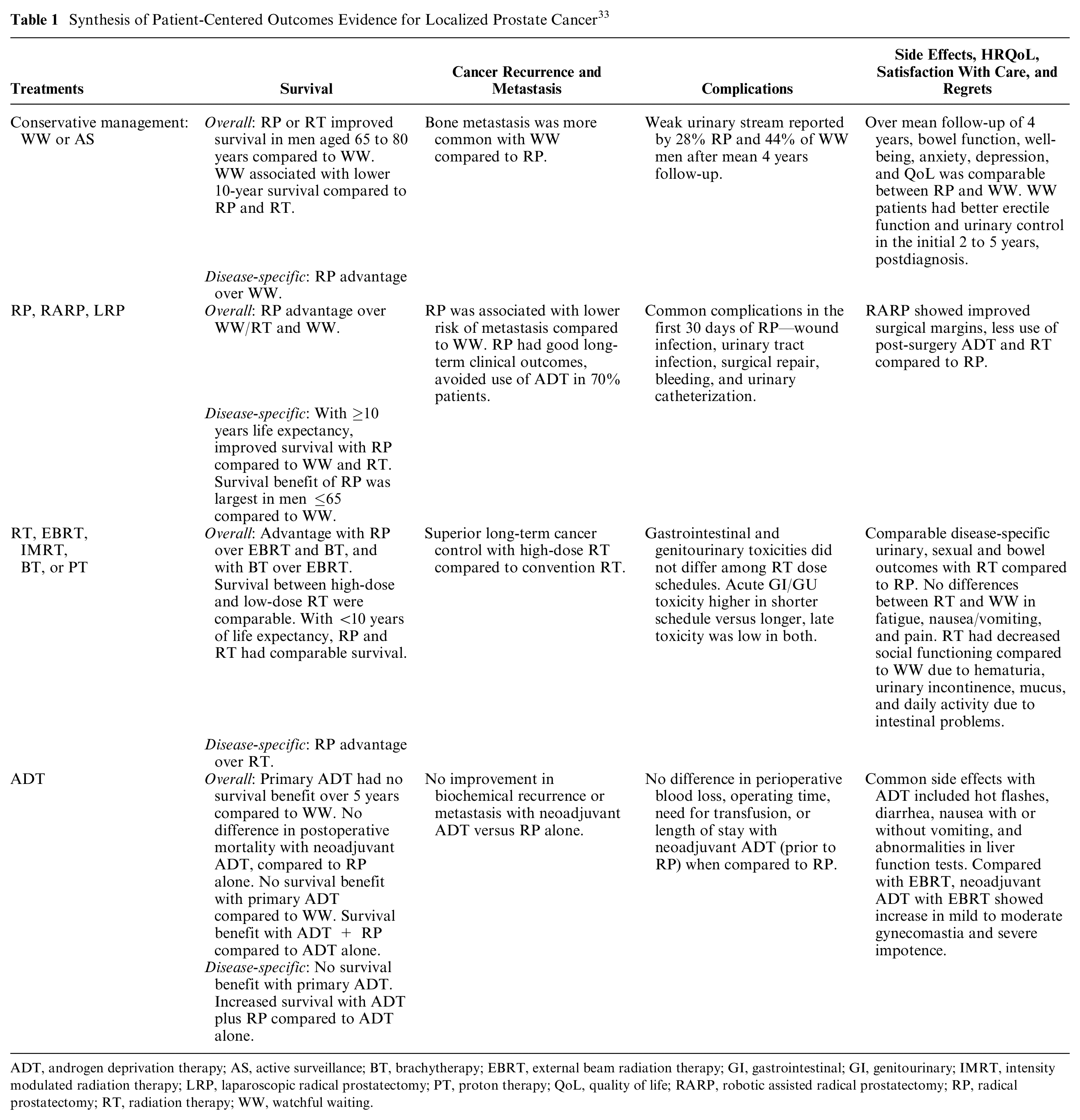
ADT, androgen deprivation therapy; AS, active surveillance; BT, brachytherapy; EBRT, external beam radiation therapy; GI, gastrointestinal; GI, genitourinary; IMRT, intensity modulated radiation therapy; LRP, laparoscopic radical prostatectomy; PT, proton therapy; QoL, quality of life; RARP, robotic assisted radical prostatectomy; RP, radical prostatectomy; RT, radiation therapy; WW, watchful waiting.

Sources of attributes. Various sources used for determining attributes and finalization of attributes.
Semistructured Patient Interviews
We conducted one-on-one interviews with prostate cancer survivors recruited from our study sites. Saturation was reached after 50 interviews and no new themes were identified. These interviews revealed the complexity of treatment decision, beyond clinical characteristics. Mean age of the participants was 65.8 years (SD = 7.9), and about two thirds (67%) were white. Sixty-five percent had surgery, 25% received radiation therapy, and 10% were on active surveillance. Mean time since diagnosis was 3.8 years (SD = 3.7). On average, each interview was 30 minutes long. In addition to the attributes identified from the systematic literature review (survival and side effects), participants identified following attributes (Figure 2): impact of treatment, impact on social life, recovery pattern, emotional concerns (depression and anxiety), caregiver burden, and out-of-pocket costs. Participants noted that burden on caregivers varied by type of treatment. For example, compared to radiation, surgery requires more support from family; therefore, patients with less family support may avoid surgery. One-on-one patient interviews were critical in identifying unique patient experiences and perspectives.
Provider Focus Groups
We organized five structured focus group meetings for providers—two with urologists (n = 19), and one each with radiation oncologists (n = 6), primary care physicians and geriatricians (n = 24), and nurse practitioners (n = 19). A facilitator conducted the focus group meetings that were 2 hours long on average. Overall, the following attributes emerged from the provider focus group meetings (Figure 2): treatment side effects, complications, survival, age, functional status, and clinical factors such as PSA level, stage, positive margin, comorbidity, and short and long-term prostate cancer–related HRQoL. Some of the attributes that the providers acknowledged and discussed were same as those that we had identified from the systematic literature review, including survival, side effects, and complications. Similar to prostate cancer survivors, providers noted that certain treatments could cause more (or less) caregiver burden. In addition, providers identified nuances in the timing of complications. Urologists and radiation oncologists made an important distinction between short-term and long-term outcomes in terms of severity and importance. Geriatricians and primary care physicians saw their role as patient advocates, and noted that erectile dysfunction and urinary incontinence affects men differently: men accept erectile dysfunction more easily than urinary incontinence, which affects everyday function. Nurse practitioners and registered nurses discussed the effects of treatment on quality of life. Provider focus group participants also determined the face and content validity of the attributes obtained from the systematic literature review.
Finalizing Attributes and Levels
We narrowed the patient-centered features into five broad categories: survival, cancer recurrence, prostate cancer treatment consequences and treatment specifics, emotional concerns and impact on social life, and burden of prostate cancer. Attributes belonging to these categories and their ideal wordings were discussed during the stakeholders’ advisory group (Figure 2). In case of disagreements, consensus was reached by deliberation. Validation was achieved using data triangulation. We developed a list of final attributes via discussion among investigators, internal and external collaborators, and the stakeholders’ advisory group (Table 2). The levels of the attribute were obtained from the literature as well as deliberated upon during provider focus group meetings and stakeholders advisory board meetings. We employed best practices to ensure that each attribute: 1) did not exceed a sixth-grade reading level, as measured by the Flesch–Kincaid Grade Level standard; 2) minimized ambiguity or cognitive difficulty; 3) were concise and simply worded; and 4) were easy to translate into other languages.11,40,41
Attributes, Definitions, and Levels

Development of the PreProCare Tool
We used the Sawtooth software’s Adaptive Conjoint Analysis (ACA) program to create computer based PreProCare tool consisting of 15 attributes. All possible resulting “pair comparisons” were reviewed for language and clarity. Our final version of the tool consists of three parts. An example of each part is shown in Figure 3.
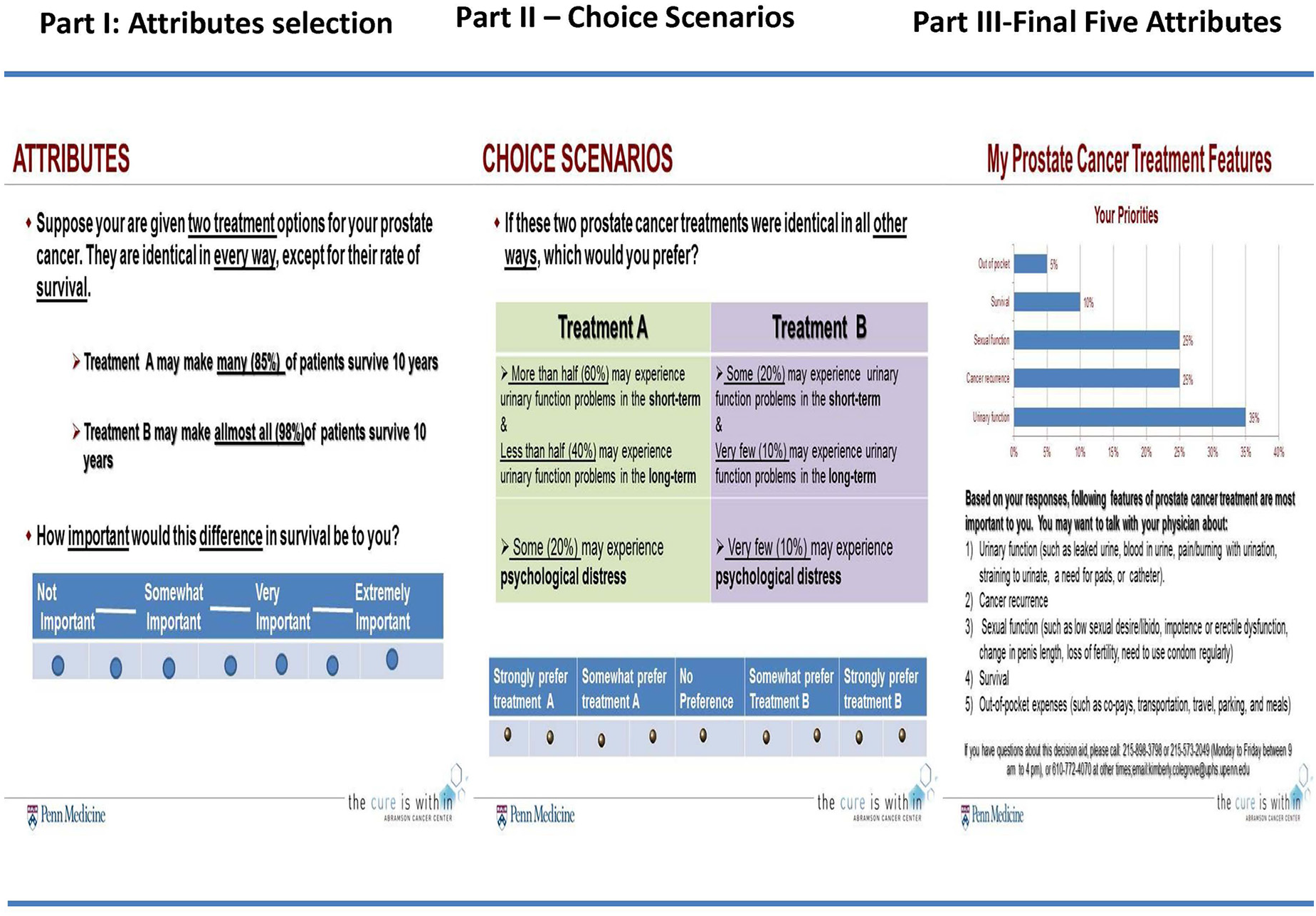
Adaptive conjoint analysis preference assessment, PreProCare tool. A snapshot of PreProCare tool depicting three parts of the tool: rating of attributes, choice scenarios, and list of attributes most preferred by the participant.
The PreProCare tool begins with a brief introduction and instructions. Description and definitions for important terms and levels were available. Here the participant was presented with a hypothetical choice of two treatments (treatment A and treatment B) with varying levels of an attribute, but who are otherwise similar. The participant was asked how important the difference in the levels was: not important, somewhat important, very important, or extremely important. The participant went through 15 such choices, and the answers formed the basis for Part 2. In Part 2, personalized, hypothetical choice scenarios were developed that presented bundle of attributes (features) for two hypothetical treatment options. For example, bundle of two attributes presented two attributes whose level differed between treatment A and treatment B. The participants stated the preference for one of the bundles (for treatment A or for treatment B) by selecting one of the following: strongly prefer treatment A, somewhat prefer treatment A, no preference, somewhat prefer treatment B, or strongly prefer treatment B. There were 15 bundled scenarios in total: five each with bundle of two attributes, three attributes, and four attributes. These choice scenarios elicited the conjoint tradeoffs. Finally, in Part 3, the PreProCare tool generated a list of five attributes that were deemed to be most important by the participant. This list is in a printable format and participants are encouraged to share it with their physicians.
Pilot Testing of the PreProCare Tool
The final version PreProCare tool was first reviewed during the stakeholder advisory board meeting and feedback was obtained about the comprehensiveness and flow, adopting think aloud technique. Based on this feedback, the content was fine-tuned, including simplifying the language, adding images, and presenting results as a printable list of top five attributes. Next, to determine the usability and acceptability of the PreProCare tool, 52 newly diagnosed prostate cancer patients who were treatment naïve completed the tool. There was no overlap between these patients and members of our stakeholder advisory board. On average, it took 30 minutes to complete the tool. The pilot test participants also completed a one-page survey about its usability and feasibility. As shown in Table 3, of the 52 pilot test participants, 49 (94%) said that the definitions were easy to understand, 45 (86%) felt that the tool was helpful in deciding treatment, and 46 (88%) said they would discuss the results with their physicians.
Pilot Testing of PreProCare Tool (N = 52)

Shared decision making is a collaborative process that allows patients and providers to make health care decisions together, taking into account the best scientific evidence available and the patients’ values and preferences.32,42,43 The significance of patients’ preferences to decision making is highlighted by the definition of a “high-quality decision” as one that is consistent with a patient’s underlying values and preferences.7,42,44,45 Localized prostate cancer patients have health-related preferences that go beyond cancer cure. Thus, patient involvement, as reflected in patient-centered decision making, is highly relevant to prostate cancer care. Although clinicians believe that they consider patient preferences, they may often be indifferent about these preferences. 44 Physician recommendations for treatments are strong predictors of treatment choice, so it is important to understand how well physicians’ views of preferences actually reflect patients’ preferences.19,32,46,47 The goal of our interactive, web-based, prostate cancer preference assessment tool, PreProCare, is to help newly diagnosed localized prostate cancer patients understand their preferences, and thus facilitate informed treatment decision making.
To address the limitation associated with using only one source to identify attributes (or features) of treatment options, we used a comprehensive approach consisting of extensive systematic literature review, one-on-one patient interviews, and provider focus groups to identify the attributes. Of treatment options, our patient-centered approach also engaged key stakeholders in the entire process of tool development. The pilot test results suggest that the PreProCare preference assessment tool is feasible and acceptable among localized prostate cancer and may help in value clarification. There are several potential ways our PreProCare tool may be implemented in a clinical setting. For example, it can be made available on a patient portal, and the patient can be directed to complete it prior to their visit. Alternatively, the patient can complete the tool in the waiting room.
Limitations
We note the following limitations. We did not capture certain clinical characteristics such as risk group, positive margins, and comorbidity. Also, the PreProCare tool based values clarification exercise is limited by the attributes selected. Determining the most appropriate way to integrate decision aids into the fast-paced nature of clinical decision making is an area in need of further research.
Conclusions
In this study, we have described the comprehensive process of development of our robust PreProCare tool. Decision aid intervention for localized prostate cancer was shown to reduce decision conflict, improve elements of shared decision making, patient knowledge, and satisfaction with decision.12,14,15,48,49 At the same time, a systematic review and meta-analysis of randomized controlled trials for localized prostate cancer decision aids highlights some challenges of these studies. 15 Incorporating patient-centered clinical decision making in real clinical settings is a challenge, as it requires the decision aid tool to quantify the utility levels of attributes (features) of treatment option. Such data are necessary to facilitate a patient-centered choice that integrates patient values and preferences. Thus, the PreProCare tool may offer an opportunity to quantify treatment preferences of localized prostate cancer patients in a clinical setting.
Our web-based PreProCare tool offers a feasible and acceptable approach to measure stated preference in patients with localized prostate cancer to facilitate patient-centered treatment decision. Our recent multicentered randomized controlled trial assessed the comparative effectiveness of PreProCare intervention on outcomes. 50 Future studies will address the concordance of preference with treatment choice on outcomes among localized prostate cancer patients.
Supplemental Material
Appendix_-online_only.rjf._online_supp – Supplemental material for Patient-Centered Approach to Develop the Patient’s Preferences for Prostate Cancer Care (PreProCare) Tool
Supplemental material, Appendix_-online_only.rjf._online_supp for Patient-Centered Approach to Develop the Patient’s Preferences for Prostate Cancer Care (PreProCare) Tool by Ravishankar Jayadevappa, Sumedha Chhatre, Joseph J. Gallo, S. Bruce Malkowicz, J. Sanford Schwartz and Marsha N. Wittink in MDM Policy & Practice
Footnotes
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
This study was supported by the Patient Centered Outcomes Research Institute (CE-12-11-4973). We would like to acknowledge patient and provider stakeholders who participated in our patient-centered prostate cancer grant.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
