Abstract
Introduction:
Periodontal disease is a multifactorial chronic inflammatory condition that progressively destroys the periodontal supportive tissues. While periodontal surgical therapy is a successful treatment for periodontal disease, patients often experience postoperative complications such as pain, swelling, and discomfort, which affect their quality of life. This systematic review evaluated the efficacy and safety of perioperative steroidal and nonsteroidal anti-inflammatory drugs (SAIDs and NSAIDs) on postoperative pain and swelling and rescue medication consumption in adults undergoing periodontal surgeries.
Methods:
A protocol guided by the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines was followed. Multiple online databases were searched up to April 2025 for randomized clinical trials (RCTs) comparing perioperative SAIDs and NSAIDs after periodontal surgery. The primary outcome measure was postoperative pain, while the secondary outcome measures were swelling and rescue medication consumption using a random-effects model. The GRADE system assessed the quality of the evidence for each included study.
Results:
Of 2,354 articles screened, 6 RCTs with 262 participants were included. Perioperative NSAIDs showed a statistically significant reduction in postoperative pain for the first 6 h postoperatively compared with perioperative SAIDs (mean difference [MD]: 1.00 to 2.23; 95% confidence interval [CI]: 0.21, 3.77). Perioperative SAIDs yielded a statistically significantly greater reduction in postoperative pain 48 h postoperatively than perioperative NSAIDs did (MD: −0.23, 95% CI: −0.31, −0.14). Perioperative SAIDs and NSAIDs had comparable outcomes in terms of postoperative swelling and rescue medication consumption.
Discussion:
Perioperative NSAIDs may be more effective in reducing early postoperative pain while SAIDs might offer better longer-term pain management postoperatively after periodontal surgery. However, these findings should be viewed with caution because of the clinically insignificant differences and the evidence based on few studies, with moderate-to-low certainty of evidence, due to methodological biases and discrepancies in drugs used and regimens. Therefore, further optimized RCTs confirming the efficacy and safety of perioperative SAIDs and NSAIDs in periodontal surgical procedures are still needed.
Knowledge Transfer Statement:
This systematic review and analysis found moderate-to-low-certainty evidence and limitations in the studies assessing the effectiveness of perioperative anti-inflammatory drugs to improve postoperative recovery after periodontal surgical therapies. This emphasizes the need for individualized perioperative therapeutic protocols based on surgical complexity and the need for tension-free wound healing pending further high-quality trials.
Keywords
Introduction
Periodontal disease (periodontitis) is a multifactorial chronic inflammatory disorder that progressively destroys the periodontal supportive tissues including the bone surrounding teeth (Hajishengallis and Chavakis 2021). Periodontitis is epidemiologically linked to various chronic diseases, such as cardiovascular disease, type 2 diabetes mellitus, rheumatoid arthritis, inflammatory bowel disease, Alzheimer’s disease, nonalcoholic fatty liver disease, and cancer (Genco and Sanz 2020). Severe periodontitis is considered the sixth most prevalent health condition, affecting 10.8% of the population worldwide (Peres et al. 2019). According to the National Health and Nutrition Examination Survey (2009 to 2014), more than 42% of the US population aged 30 y or older is considered to have periodontitis (Eke et al. 2018). Untreated periodontitis eventually results in tooth loss that often leads to nutritional deficits, impaired speech, low self-esteem, and poor overall quality of life (QoL), thus representing a significant burden to health care and socioeconomics (Genco and Sanz 2020).
Periodontitis treatment is categorized into initial nonsurgical and surgical therapies. Initial nonsurgical therapy mainly consists of oral hygiene homecare and professional mechanical plaque removal (scaling and root planing) (Sanz et al. 2020). After the initial therapy response is evaluated, the remaining sites with active periodontitis are treated by regenerative or resective surgical therapy (Sanz et al. 2020). While surgical therapy is often successful (Kaldahl et al. 1996), patients frequently experience postoperative complications such as pain, swelling, and discomfort that affect their QoL (Mei et al. 2016). Therefore, the adequate management of postoperative complications is not only crucial for proper healing and recovery but also improves patients’ QoL (Tan et al. 2015). Furthermore, there are several factors reported in the literature that increase the severity of postoperative complications, namely, surgery complexity, surgeon experience, surgery duration, surgical site extension, number of local anesthesia carpules used, type of pain medication prescribed, and having periosteal fenestration/dissection after surgery (Mei et al. 2016). Indeed, complex and plastic periodontal surgeries such as soft-tissue and bone augmentation surgery, periodontal regenerative surgery, and gingivectomy, have been associated with more perceived pain and swelling postoperatively when compared with simple surgeries such as open flap debridement and single implant placement (Mei et al. 2016). Thus, perioperative pain medications have been suggested to control postoperative complications (Pyati and Gan 2007).
Both steroidal (glucocorticoids) and nonsteroidal anti-inflammatory drugs (SAIDs and NSAIDs) have been widely used during the perioperative period to manage postoperative complications after oral surgical procedures (Yamaguchi and Sano 2013), including periodontal surgical therapies (Caporossi et al. 2020; Wagner et al. 2022; Isiordia-Espinoza et al. 2023; Santos et al. 2023; Lages et al. 2024). Dexamethasone, a commonly prescribed SAID, is a long-acting SAID that can be administered intravenously, intramuscularly, submucosally, or orally. Dexamethasone inhibits phospholipase A2 enzyme, in turn blocking the release of arachidonic acid from phospholipids that is used in both the cyclooxygenase and lipoxygenase pathways (Franchimont 2004). Inhibiting these pathways decreases proinflammatory mediators, such as prostaglandins and thromboxanes, formed in response to cellular damage (Gordon et al. 2002). These proinflammatory mediators activate nociceptors in peripheral tissues, causing postoperative pain perception (Hersh et al. 2020). Prostaglandins also result in postoperative swelling by influencing the vasodilation and permeability of peripheral venules adjacent to the injury site (Funk 2001). The long-term intake of glucocorticoids can cause several side effects, such as adrenal insufficiency, increased risk of infection, hyperglycemia, hypertension, osteoporosis, and diabetes mellitus (Polderman et al. 2018). In contrast, NSAIDs act by reversibly inhibiting the cyclooxygenase (COX) 1 and 2 enzymes, affecting arachidonic acid metabolism and suppressing the production of specific metabolites, including prostaglandins that have important roles in inflammatory and nociceptive processes (Halvey et al. 2023). For instance, COX-1 is essential for the integrity of both renal tract and gastrointestinal (GI) tissues; thus, the long-term intake of NSAIDs can also cause several side effects such as GI bleeding, peptic ulcers, and kidney disease (Halvey et al. 2023). However, due to the short duration and low doses of SAIDs and NSAIDs prescribed for oral surgical procedures, these adverse events are unlikely (Polderman et al. 2018; Halvey et al. 2023).
Both SAIDs (e.g., dexamethasone) and NSAIDs (e.g., ibuprofen) are frequently prescribed, perioperatively, after surgical extraction of third molars to reduce postoperative pain, swelling, and trismus (Pimenta et al. 2024). In addition, several systematic reviews were conducted to investigate the efficacy of various pharmacologic agents and analgesia protocols after periodontal surgeries (Caporossi et al. 2020; Brigola et al. 2022; Wagner et al. 2022; Isiordia-Espinoza et al. 2023; Santos et al. 2023; Lages et al. 2024). However, the findings presented from these reviews are inconclusive. Furthermore, to date, the evidence obtained from direct comparisons of the efficacy and safety of perioperative SAIDs and NSAIDs on postoperative complications after periodontal surgeries is still lacking, as the previous reviews mainly compared against placebo only. Therefore, this systematic review and meta-analysis aimed to directly compare the efficacy and safety of perioperative SAIDs and NSAIDs in managing postoperative complications after periodontal surgery, ultimately improving postoperative recovery.
Materials and Methods
Study Design
This systematic review and meta-analysis was conducted according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. The protocol was registered on the International Prospective Register of Systematic Reviews (PROSPERO, reference No. CRD42024568891).
Objectives and Research Question
This systematic review and meta-analysis aimed to compare the efficacy and safety of perioperative SAIDs and NSAIDs in managing postoperative complications after periodontal surgery. The research question in PICOS format was as follows: among patients undergoing periodontal surgery (P), do perioperative SAIDs (I) improve postoperative recovery such as postoperative pain, swelling, rescue medication consumption, and adverse events (O) in comparison with NSAIDs (C) in randomized clinical trial studies (S)?
Search Strategy and Selection Criteria
A systematic search was conducted using Medical Subject Headings (MeSH), keywords, and Boolean operators (OR/AND) on the following databases: EMBASE (via Ovid), MEDLINE (via PubMed), Scopus (via Elsevier), CINAHL, Cochrane Library, ProQuest, and EBSCOhost on August 1, 2024, and updated on April 9, 2025 (Appendix Table 1). Additional studies were identified through manual searches of the reference lists of relevant articles and systematic reviews.
The selection process of the included studies in this systematic review and meta-analysis is outlined in the PRISMA flowchart (Fig. 1). References were exported from the online databases, and duplicates were subsequently removed using EndNote 21 citation management software. Subsequently, 2 independent reviewers (A.E. and I.S.) conducted a blinded screening of articles on Rayyan, beginning with title and abstract, followed by full-text screening where available. Any disagreements between the reviewers were adjudicated by a third reviewer (M.A.). The calculated Cohen’s kappa score was 0.8, which indicates a substantial agreement between the reviewers.
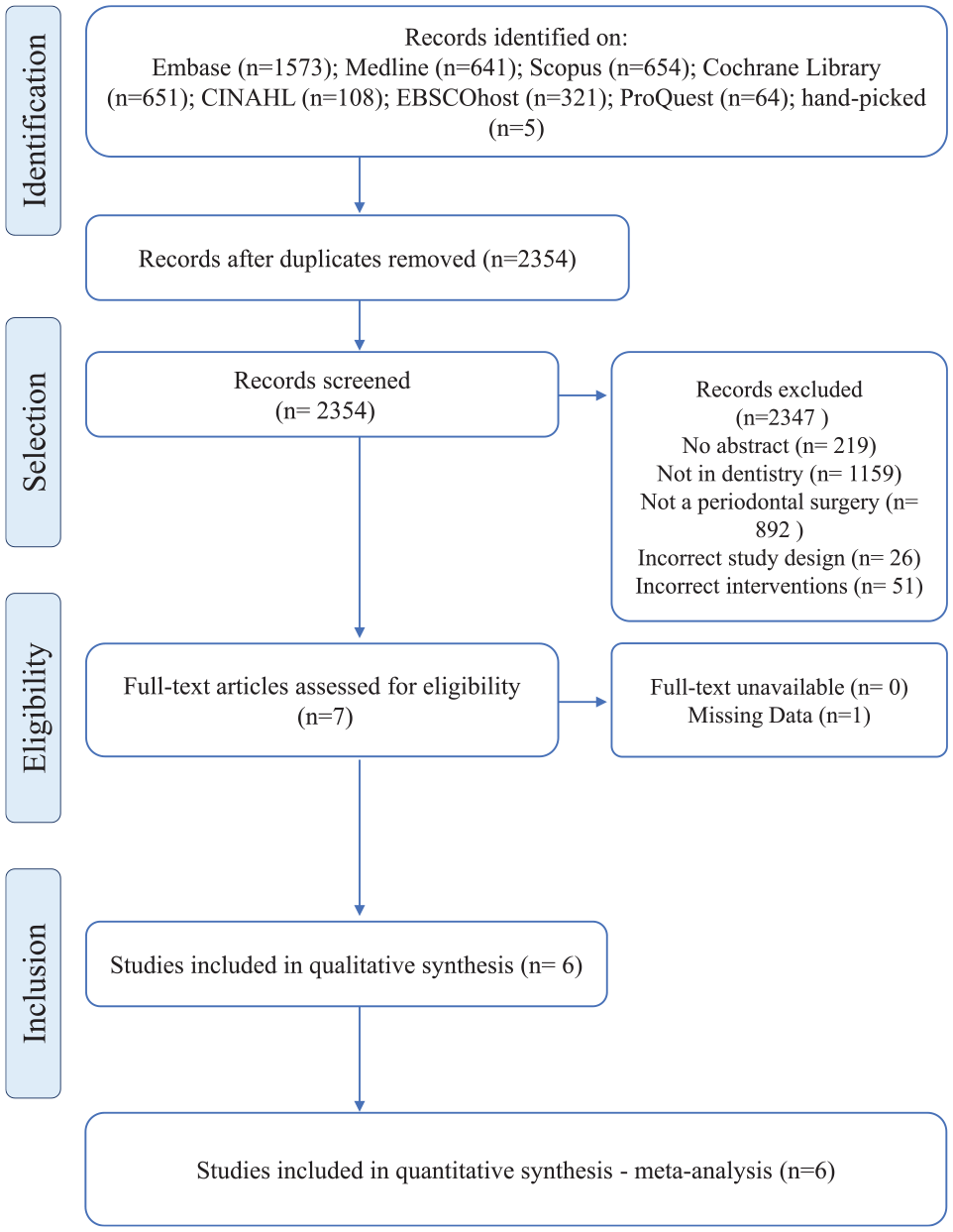
PRISMA flow chart of included studies.
This review included only (1) randomized controlled trials (RCTs), crossover or parallel design, that (2) directly evaluated the efficacy and safety of SAIDs and NSAIDs (3) using any postoperative recovery outcome or endpoint (short and long term) (4) after any type of periodontal surgery (5) with at least 1 follow-up assessment. On the other hand, we excluded the following studies if they (1) had no abstract, (2) were not in the field of dentistry, (3) used a surgical model other than a periodontal surgical model, (4) had an incorrect study design, or (5) had incorrect interventions (i.e., only SAID vs. placebo or NSAIDs vs. placebo)
Data Charting and Data Synthesis
Two reviewers (A.E. and I.S.) extracted and charted the data from included studies using a standardized spreadsheet in Microsoft Excel (Microsoft Excel, 2019, Microsoft Corporation). The extracted data were then summarized comprising each study’s authors, title, publication year, country, study design, number of participants, mean age, sex, sample size, type of surgical procedure, interventions, administration route, dosage, use of any placebos, outcomes, and results. In addition, information on the anesthetic agent, vasoconstrictor dosage and type, rescue medication utilization, pain assessment methodology, and evaluation frequency were documented. The same reviewers also conducted an independent quality appraisal of the included studies using the Cochrane Risk of Bias 2 (RoB 2) tool, applying the assessment criteria appropriate to each study design (Sterne et al. 2019). In instances in which essential data were unavailable, the corresponding authors were contacted via email. Studies with incomplete data were retained within the systematic review framework but were excluded from the quantitative synthesis.
Risk of Bias and Quality of Evidence Assessment
The risk of bias for each included study was assessed using the RoB 2 tool for both parallel and crossover design, as recommended by Cochrane for evaluating bias in RCTs. Each selected study underwent independent evaluation by 2 reviewers (A.E. and I.S.), with any discrepancies resolved through discussions with a third reviewer (M.A.). Based on the RoB 2 criteria, studies with adequate information were rated as low risk of bias. Conversely, studies lacking sufficient data were classified as high risk of bias. When information was limited and a clear risk assessment could not be determined, the item was classified as having some concerns. Quality of evidence for each meta-analysis was assessed (M.A.) using the GRADE approach (grading of recommendations assessment, development and evaluation).
Outcome Measures and Statistical Analysis
Continuous outcomes (primary: pain visual analog scale [VAS] at 1, 2, 3, 4, 5, 6, 7, 8, 24, 48, and 72 h; secondary: swelling VAS at 24 h and overall rescue medication consumption) were expressed as the mean difference (MD) and 95% confidence intervals (CIs). When applicable, the mean and/or standard deviation was indirectly obtained if authors did not report them directly selecting appropriate conversion equations (Higgins et al. 2024). Clinically significant differences in outcome measure scores using the VAS were set at 13%. Since various studies used different scales to quantify postoperative pain and swelling, all postoperative measures were standardized to the VAS scale for consistency using validated equations (Thorlund et al. 2011). If a study reported the same outcome measure with different scales, VAS was selected. Finally, we calculated the average outcome measure scores when multiple scores recorded throughout the same day after the first 8 h postoperatively.
A random-effects model (inverse variance method) was used to pool effect sizes, and Knapp–Hartung adjustment was used to calculate the CI around the pooled effect (Knapp and Hartung 2003). The restricted maximum likelihood estimator was used to calculate the heterogeneity variance τ2 and the Q-profile method to estimate the CI of the variance (Viechtbauer 2005). Also, Higgins and Thompson’s Ι2 and Cochran’s Q tests were performed to assess heterogeneity among included studies (χ2 = P > 0.10; I2 < 25%) (Cochran 1954; Higgins and Thompson 2002). Further, the prediction interval was calculated to present the intervention effects that could be evident in futures studies. If applicable, publication bias was assessed using funnel plots, followed by the trim-and-fill method for asymmetry adjustment (n > 10). Finally, R Studio, version 4.4.1, was used to perform all analyses (namely, meta [Balduzzi et al. 2019] and metafor [Viechtbauer 2010] packages) and create forest plot figures.
Results
Overall, our search identified 2,354 records that were screened by title and abstract. Of those, 2,347 articles did not meet our inclusion criteria and were therefore excluded. After full-text review, 1 research work was excluded due to missing data, and thus, 6 articles were finally included in our review for both qualitative and quantitative analyses (Pilatti et al. 2006; Steffens et al. 2010; Peres et al. 2012; Zardo et al. 2013; Bahammam et al. 2017; Giorgetti et al. 2018). The quality of the included studies was adequate, with 3 studies (50%) exhibiting a low risk of bias, while the other half showed some concerns (Appendix Table 2). However, none of them had substantial risk of bias (see the appendix, page 13). Publication years ranged from 2006 to 2018, and all included studies, except one, were conducted in Brazil.
This systematic review included 262 participants from 6 RCTs (4 parallel; 2 crossover) evaluating perioperative SAIDs versus NSAIDs on postoperative recovery following periodontal surgery (Table 1). There were notable differences in the included clinical studies in terms of study design, intervention, doses, and periodontal surgery. For instance, 2 RCTs included open flap debridement only (Pilatti et al. 2006; Steffens et al. 2010), 2 studies used soft-tissue graft surgical models (Zardo et al. 2013; Giorgetti et al. 2018), while Peres et al. (2012) and Bahamman et al. (2017) included crown-lengthening procedures and dental implants, respectively (Peres et al. 2012; Bahammam et al. 2017). Dexamethasone (SAIDs) dose varied among included RCTs, with 2 studies using 8 mg (Pilatti et al. 2006; Steffens et al. 2010), while the remaining trials used 4 mg (Peres et al. 2012; Zardo et al. 2013; Bahammam et al. 2017; Giorgetti et al. 2018). On the other hand, both nonselective COX-1 and -2 and COX-2–selective inhibitors (NSAIDs) were prescribed in different medications and doses (Table 1, Appendix Table 3). Furthermore, two-thirds of the included studies compared against a placebo intervention (Pilatti et al. 2006; Steffens et al. 2010; Zardo et al. 2013; Bahammam et al. 2017). Most clinical trials reported postoperative pain scores for the first 8 h and a few up to 7 d postoperatively. However, postoperative swelling was evaluated in only 2 RCTs (Peres et al. 2012; Giorgetti et al. 2018). As for rescue medications, all included studies prescribed acetaminophen (750 to 1,000 mg), whereas Peres et al. (2012) prescribed dipyrone 500 mg. Finally, only 3 studies reported no adverse events of any of the medications used (Pilatti et al. 2006; Zardo et al. 2013; Bahammam et al. 2017), while the remaining studies did not report whether any adverse events were observed or not (Steffens et al. 2010; Peres et al. 2012; Giorgetti et al. 2018).
Main Characteristics and Demographics of the Included Studies.
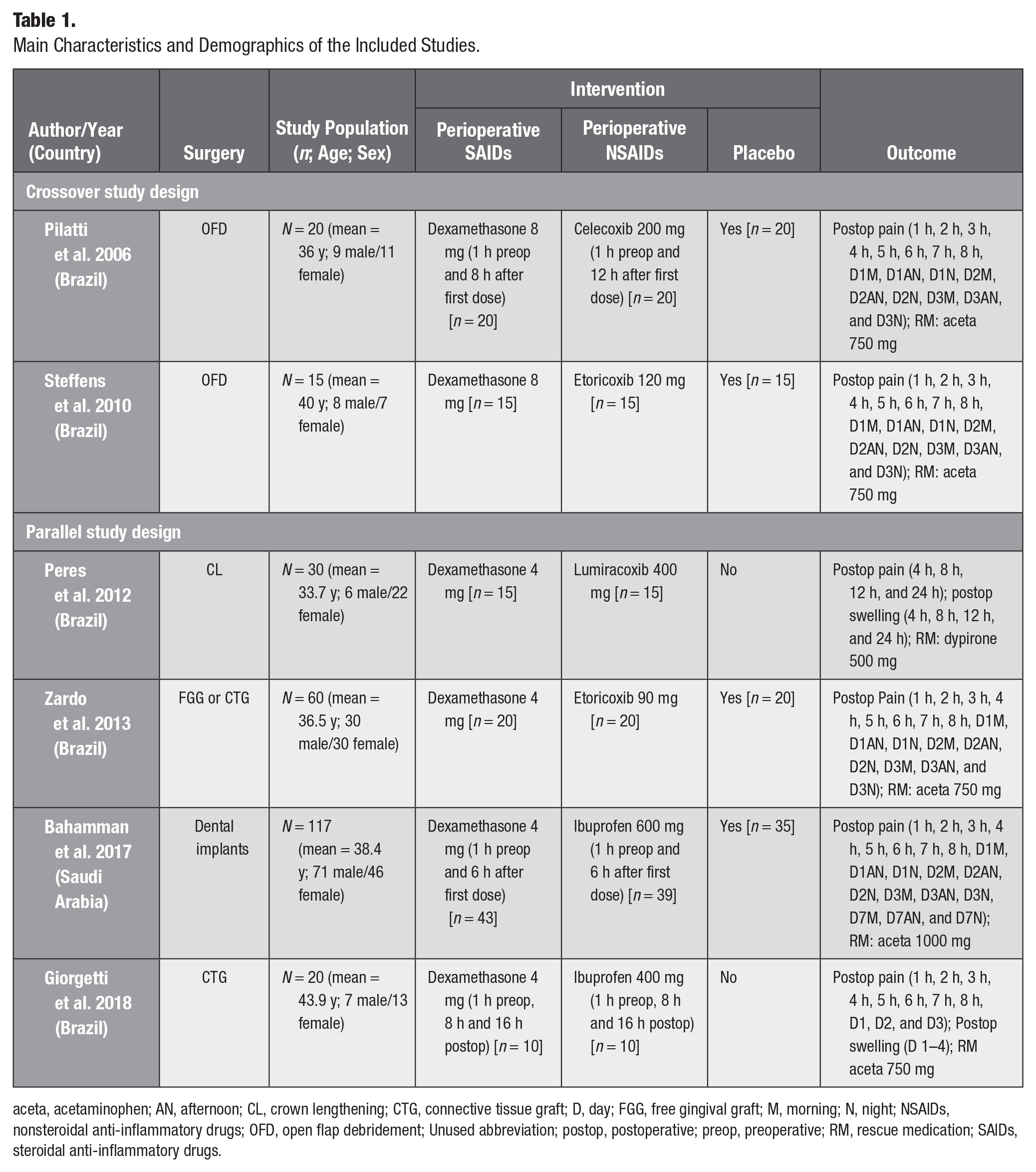
aceta, acetaminophen; AN, afternoon; CL, crown lengthening; CTG, connective tissue graft; D, day; FGG, free gingival graft; M, morning; N, night; NSAIDs, nonsteroidal anti-inflammatory drugs; OFD, open flap debridement; Unused abbreviation; postop, postoperative; preop, preoperative; RM, rescue medication; SAIDs, steroidal anti-inflammatory drugs.
Postoperative Pain after Perioperative SAIDs versus NSAIDs
Zardo et al. (2013) reported that perioperative NSAIDs had significantly improved postoperative pain relief in the first hour postoperatively compared with perioperative SAIDs (Zardo et al. 2013), while Giorgetti et al. (2018) showed that perioperative SAIDs had significantly superior pain control after 3 h postoperatively compared with perioperative NSAIDs (Giorgetti et al. 2018). However, both perioperative SAIDs and NSAIDs demonstrated significantly better postoperative pain management when compared with placebo (Pilatti et al. 2006; Steffens et al. 2010; Zardo et al. 2013; Bahammam et al. 2017).
The overall estimated variance of between-study heterogeneity was τ2 = 1.00 (95% CI: 0.38 to 1.55), with an Ι2 value of 94.9% (95% CI: 93.8% to 95.7%), and the prediction interval ranged between −1.39 and 2.74. The overall effect size estimate of perioperative SAIDs versus NSAIDs on postoperative pain was statistically significant, with conclusive CIs favoring NSAIDs; however, this effect size was clinically insignificant (MD: 0.673, 95% CI: 0.28 to 1.06, P < 0.05). Furthermore, subgroups of postoperative pain scores showed that perioperative NSAIDs significantly reduced postoperative pain for the first 6 h postoperatively compared with perioperative SAIDs (MD: 1.00 to 2.23; 95% CI: 0.21 to 3.77; Ι2: 0%–19%) (Fig. 2). On the other hand, perioperative SAIDs significantly reduced postoperative pain 48 h postoperatively as compared with perioperative NSAIDs (MD: −0.23, 95% CI: −0.31 to −0.14, I2: 0%, P < 0.05). It is important to note that while the CIs were conclusive, the pooled effect size was clinically insignificant.
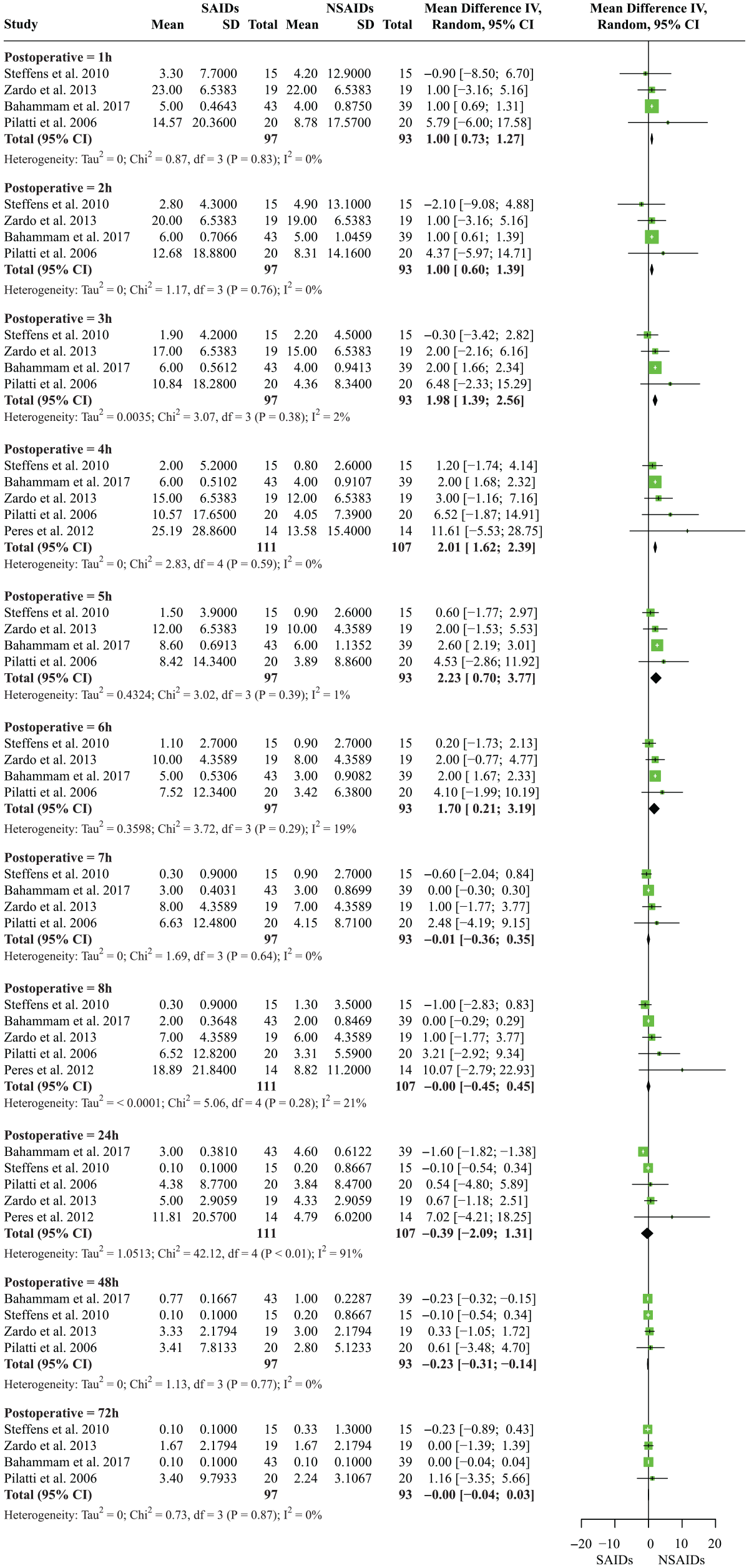
Postoperative pain intensity (visual analog scale) using perioperative steroidal anti-inflammatory drugs (SAIDs) versus nonsteroidal anti-inflammatory drugs (NSAIDs) after periodontal surgery. CI, confidence interval; SD: standard deviation.
Postoperative Swelling after Perioperative SAIDs versus NSAIDs
Only 2 RCTs evaluated postoperative swelling after periodontal surgeries (Peres et al. 2012; Giorgetti et al. 2018), and only 1 point of comparison was obtained (24 h postoperatively). Giorgetti et al. (2018) demonstrated significantly reduced postoperative swelling on the second day postoperatively in the group prescribed perioperative SAIDs (Giorgetti et al. 2018). The estimated variance of between-study heterogeneity was τ2 = 114.0 with an Ι2 value of 46.7%. As shown in Figure 3A, the effect size estimate of perioperative SAIDs versus NSAIDs on postoperative swelling was statistically insignificant with inconclusive confidence intervals (MD: −3.38, 95% CI: −126 to 120, P > 0.05).
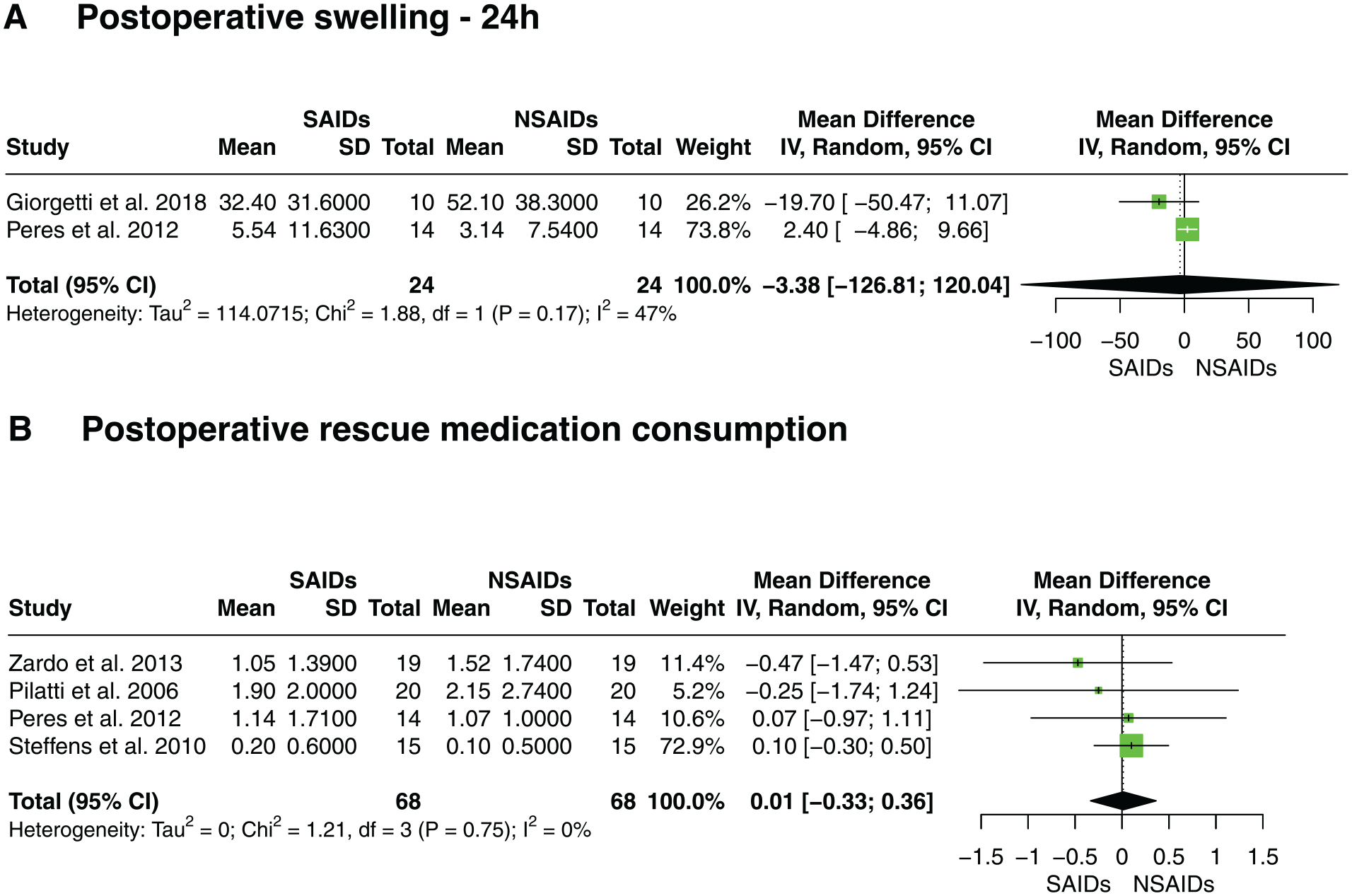
(
Postoperative Rescue Medication Consumption after Perioperative SAIDs versus NSAIDs
All included studies compared the number of rescue medication pills consumed after perioperative SAIDs versus NSAIDs following periodontal surgery and reported no statistically significant difference in terms of postoperative rescue medication consumption. When compared with placebo groups, 3 studies showed a significant reduction in the number of rescue medication pills in both perioperative SAIDs and NSAID (Zardo et al. 2013; Bahammam et al. 2017; Giorgetti et al. 2018). Conversely, Pilatti et al. (2006) observed no significant differences in the number of rescue medication pills among the perioperative SAID, NSAIDs, and placebo groups. The estimated variance of between-study heterogeneity was τ2 = 0.00 (95% CI: 0.00 to 0.29) with an Ι2 value of 0.00% (95% CI: 0.00% to 0.27%). As shown in Figure 3B, the effect size estimate of perioperative SAIDs versus NSAIDs on postoperative rescue medication consumption was statistically insignificant with inconclusive confidence intervals (MD: 0.01, 95% CI: −0.33 to 0.36, P > 0.05).
Quality of Evidence
The quality of evidence for each outcome is depicted in the GRADE evidence profile (Table 2). The certainty of evidence was rated as moderate for postoperative pain intensity (first 6 h and 48 h), while the remaining outcomes were rated as low and very low certainty of evidence. Lastly, publication bias could not be assessed due to the small number of included studies (n < 10).
Grading of Recommendations Assessment, Development and Evaluation Approach (GRADE).
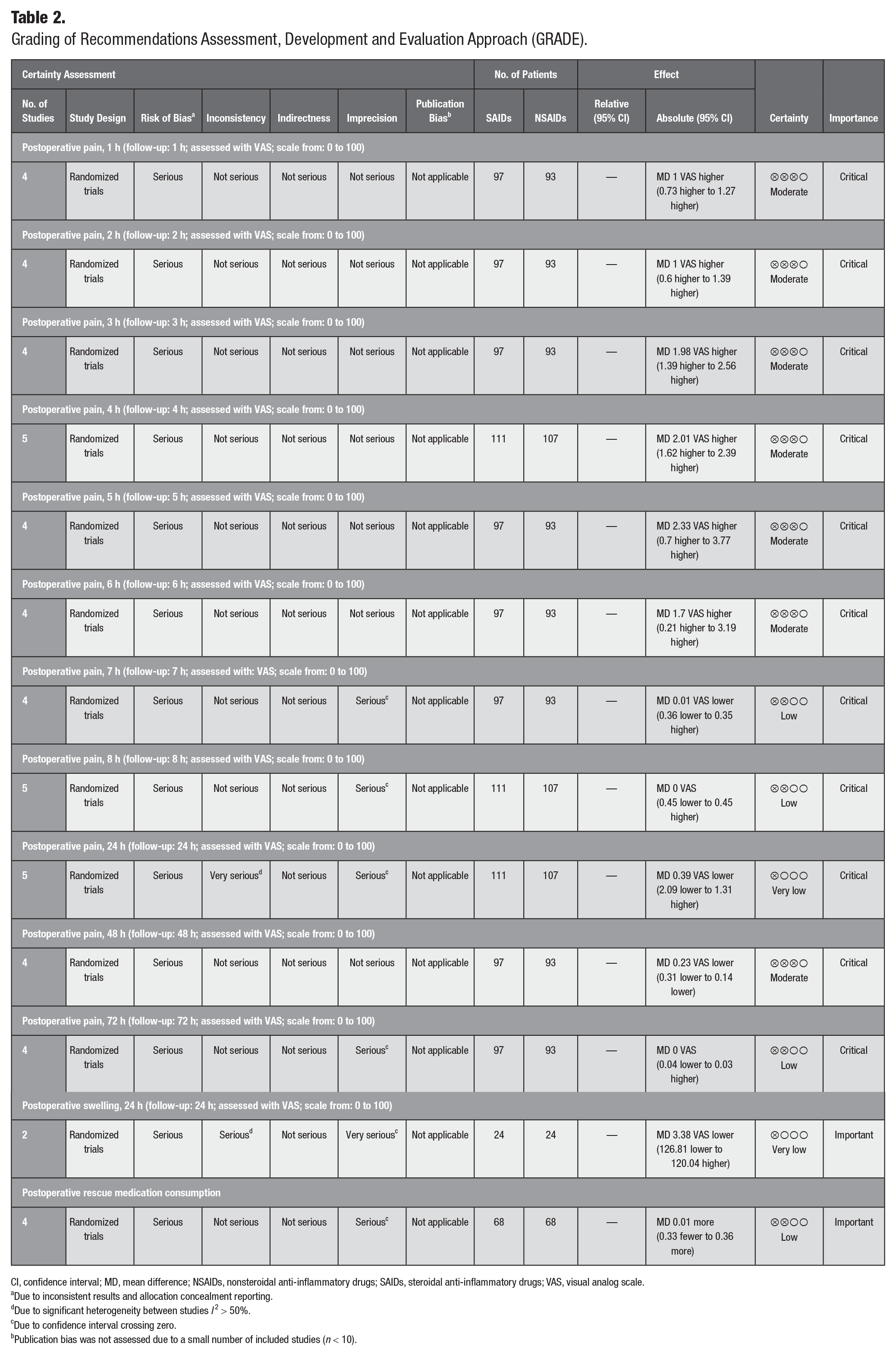
CI, confidence interval; MD, mean difference; NSAIDs, nonsteroidal anti-inflammatory drugs; SAIDs, steroidal anti-inflammatory drugs; VAS, visual analog scale.
Due to inconsistent results and allocation concealment reporting.
Publication bias was not assessed due to a small number of included studies (n < 10).
Due to confidence interval crossing zero.
Due to significant heterogeneity between studies I2 > 50%.
Discussion
This systematic review included 6 randomized clinical trials (2 crossover; 4 parallel) with a total of 262 participants. Overall, there were notable differences in the included clinical studies in terms of study design, intervention, doses, and type periodontal surgery. All included studies compared the effect of perioperative SAIDs and NSAIDs on postoperative pain management, while only 2 RCTs investigated postoperative swelling after periodontal surgeries. The surgical interventions included in this review ranged from simple surgeries (open flap debridement [n = 2], dental implants [n = 1]), to complex surgeries (osseous resective surgery [n = 1]), to periodontal plastic surgeries (free gingival/connective tissue grafts [n = 2]). To our knowledge, this is the first systematic review and meta-analysis to directly compare the effect of perioperative SAIDs and NSAIDs on postoperative recovery after periodontal surgeries. Recently, 6 reviews (systematic reviews and meta-analysis [n = 3; Caporossi et al. 2020; Brigola et al. 2022; Isiordia-Espinoza et al. 2023), systematic reviews [n = 2; Wagner et al. 2022; Lages et al. 2024], and an integrative review [n = 1; Santos et al. 2023) investigated different pharmacologic agents after periodontal surgeries. Caporossi et al. (2020) included 9 RCTs in their quantitative analysis (NSAIDs vs. placebo [n = 7] and SAIDs vs. NSAIDs [n = 2]) limited to open flap debridement surgery. They concluded that no statistical difference was found in postoperative pain management when comparing preemptive NSAIDs and SAIDs for open flap debridement (1, 2, 3, 4, and 8 h; Caporossi et al. 2020). However, this review included a wider range of periodontal surgeries and more studies with direct comparisons in the meta-analysis (n = 6) and evaluated several outcomes such as postoperative pain (1, 2, 3, 4, 5, 6, 7, 8, 24, 48, and 72 h), swelling (24 h), and rescue medication consumption. Brigola et al. (2022) included 11 RCTs in their meta-analysis to identify which analgesic protocol (preoperative vs. postoperative), compared with placebo, was most efficient in managing postoperative pain after periodontal surgery. They concluded that both analgesic protocols demonstrated at least a moderate effect on pain management compared with placebo (Brigola et al. 2022). Next, Isiordia-Espinoza et al. (2023) included 7 RCTs in which they evaluated preemptive COX-2 inhibitors versus placebo/active controls (i.e., acetaminophen and dexamethasone) on postoperative pain (1, 2, 3, 5, 6, 7, 8, 10, 12, and 24 h) and rescue medication consumption after periodontal surgeries. They reported that COX-2 inhibitors resulted in lower pain intensity at 4 h postoperatively and reduced rescue analgesic consumption compared with active controls (Isiordia-Espinoza et al. 2023). Our systematic review, on the other hand, directly compared perioperative SAIDs and NSAIDs; thus, the effect size estimated would not be diluted due to different active controls or placebo. The systematic review by Wagner et al. (2022) included 10 RCTs in their qualitative synthesis and concluded that practitioners must exercise caution when prescribing oral corticosteroids, considering the evidence supporting its use is limited, with a high risk of bias in the included studies. It is important to note that their inclusion criteria were not limited to periodontal surgeries, as several studies investigating SAIDs after third molar surgical extraction were included (Wagner et al. 2022). While Lages et al. (2024) used similar inclusion criteria to this systematic review, they compared only SAIDs and NSAIDs to placebo, with no direct comparisons between both pharmacologic agents. Lastly, the integrative review by Santos et al. (2023) mainly focused on the efficacy of local anesthesia, among other pharmacologic approaches, on preoperative, transoperative, and postoperative pain control after periodontal therapies (surgical and nonsurgical). They found that there was no standardized protocol, and the choice of pharmacologic approach depends on individual patient preferences and clinical situations (Santos et al. 2023). This systematic review and meta-analysis demonstrated that perioperative NSAIDs significantly, albeit clinically insignificant, reduced postoperative pain for the first 6 h postoperatively compared with perioperative SAIDs (MD: 1.00 to 2.23; 95% CI: 0.21 to 3.77; Ι2: 0%–19%, P < 0.05). However, perioperative SAIDs significantly, although clinically insignificant, reduced postoperative pain 48 h postoperatively as compared with perioperative NSAIDs (MD: −0.23, 95% CI: −0.31 to −0.14; I2: 0%, P < 0.05). Perioperative SAIDs and NSAIDs had similar outcomes in terms of postoperative swelling (24 h) and rescue medication consumption.
Interestingly, we found that NSAIDs were more effective in pain management in the first 6 h after surgery, while SAIDs showed superior pain control at 48 h postoperatively. When considering the half-life and plasma peak concentration of these medications, NSAIDs (i.e., ibuprofen) have a shorter half-life (1 to 2 h) and a faster plasma concentration peak/onset (30 to 60 min) (Bushra and Aslam 2010), while SAIDs (i.e., dexamethasone) have a longer half-life (6 to 12 h) and a slower plasma concentration peak/onset (3 to 4 h) (Loew et al. 1986). Thus, prescribing SAIDs 1 h before the surgery might not be as effective in managing postoperative pain after surgery, and perioperative NSAIDs might be better for postoperative pain control if given 1 h before surgery due to its fast plasma peak. Therefore, we suggest that clinicians may begin prescribing SAIDs 1 night before the surgery and 2 h before surgery to benefit from its perioperative benefits.
Our systematic review challenged several limitations. First, there were considerable differences between included RCTs in terms of study design (crossover vs. parallel), intervention, doses, and type of periodontal surgery. Indeed, combining different trial designs complicates data synthesis and may hinder external validity. Such heterogeneity between the included studies, although addressed using a random-effects model, may still affect the validity of the pooled results. In addition, subgroup analysis beyond postoperative pain endpoints could not be performed since only 2 studies had similar study design and periodontal surgery type (i.e., crossover and open flap debridement) (Pilatti et al. 2006; Steffens et al. 2010). Second, due to the small number of included studies published in the literature, we did not exclude from our meta-analysis RCTs with some concerns of risk of bias, and publication bias could not be assessed. Also, the small number of included studies limits the generalizability of our findings and might increase the risk of false negatives. However, the quality of all included studies was independently critically appraised, so our findings could be interpreted in a scientifically sound manner. Furthermore, various precautions were taken in our analyses to ensure that the pooled effect size estimates are accurate. Finally, most included studies were conducted in Brazil, and the genetic, cultural, and socioeconomic factors in this population may influence postoperative recovery outcomes, potentially limiting the external validity due to unaccounted epigenetic and genetic factors. However, the Brazilian population is highly diverse and multiethnic with various backgrounds including Europe, Africa, Asia, and South America; thus, the results may be considered representative of global populations. Nonetheless, multicentric and multiethnic clinical studies are still needed to evaluate the efficacy and safety of SAIDs and NSAIDs in managing postoperative pain and discomfort after periodontal surgery to obtain externally applicable findings. These abovementioned limitations highlight the need for standardized, multicentric RCTs with robust clinical endpoints and longer follow-up periods. Also, data on long-term outcomes such as wound healing and patient’s QoL should be also investigated in future studies.
Thus far, 14 RCTs (of which only 6 met our inclusion criteria) investigated the use of perioperative SAIDs and NSAIDs after periodontal surgeries between 1988 and 2022 (Minutello et al. 1988; Vogel et al. 1992; Pearlman et al. 1997; Pilatti et al. 2006; Karabuda et al. 2007; Steffens et al. 2010; Steffens et al. 2011; Peres et al. 2012; Zardo et al. 2013; Konuganti et al. 2015; Bahammam et al. 2017; Giorgetti et al. 2018; Vieth et al. 2022; Santos et al. 2022). These RCTs compared SAIDs to placebo, NSAIDs to placebo, NSAIDs to NSAIDs, or SAIDs to NSAIDs. While all studies reported significantly reduced pain and swelling measures as opposed to placebo, there was no clear evidence of any differences when comparing SAIDs to NSAIDs. There are several factors that must be considered due to methodological limitations. First, 8 RCTs included simple periodontal surgeries, namely, open flap debridement (Vogel et al. 1992; Pilatti et al. 2006; Steffens et al. 2010; Steffens et al. 2011; Konuganti et al. 2015; Santos et al. 2022) and studies with single implant placement (Karabuda et al. 2007; Bahammam et al. 2017), while 3 RCTs included complex and plastic periodontal surgeries such as crown lengthening (Peres et al. 2012) and soft-tissue augmentation (Zardo et al. 2013; Giorgetti et al. 2018), and 3 included a variety of different periodontal surgeries (Minutello et al. 1988; Pearlman et al. 1997; Vieth et al. 2022). As mentioned previously, the complexity of the surgeries influences the severity of postoperative complications (Mei et al. 2016). In fact, the median duration of pain and swelling after simple periodontal surgeries lasted for 1 d only (interquartile range = 2 d ) (Mei et al. 2016). Therefore, future RCTs should consider performing secondary analyses stratified by surgery type and complexity for postoperative pain and swelling outcomes. Second, only 1 RCT prescribed a standardized anti-inflammatory, analgesic regimen (i.e., ibuprofen and acetaminophen) to all their participants irrespective to treatment assignment (Vieth et al. 2022). Such a standardized pain management protocol is not only ethically needed and clinically appropriate, but it is also important to truly evaluate the effect of perioperative SAIDs or NSAIDs on postoperative recovery, as significant differences against placebo might be clinically irrelevant. Hence, researchers should adopt a standardized anti-inflammatory, analgesic regimen for 3 d postoperatively regardless of assigned interventions. Further, postoperative swelling after periodontal surgeries was subjectively measured using patient-reported outcome scales. Thus, future clinical trials should record objective measures of postoperative swelling (e.g., Laskin method) at baseline and during follow-up visits. Finally, only 3 studies evaluated short-term adverse events of perioperative interventions. While adverse events are unlikely when prescribing SAIDs and NSAIDs in low doses and for short duration, their prescription requires an individualized risk–benefit analysis for each patient (Polderman et al. 2018; Halvey et al. 2023). For example, the risk of renal injury, due to NSAID consumption, increases in certain subsets of patients (hypoperfused states, preexisting renal dysfunction, and drug interactions) (Halvey et al. 2023). On the other hand, SAIDs might delay wound healing and increase glucose levels in patients with diabetes undergoing surgery (Polderman et al. 2018). Because these conclusions are drawn from limited evidence, future studies should still report any safety concerns or adverse events of using perioperative anti-inflammatory drugs after periodontal surgeries.
Moreover, clinicians should be aware of the indications of perioperative analgesia needed for certain periodontal surgeries. For example, proper wound healing with tension-free closure is paramount in complex and plastic periodontal surgeries such as guided bone/tissue regeneration and soft-tissue augmentation (Sanz-Sánchez et al. 2022; Urban et al. 2023). Therefore, perioperative SAIDs might be preferable to control postoperative swelling to prevent wound dehiscence and/or membrane exposure causing potential complications and suboptimal therapeutic outcomes. Also, the authors recommend perioperative dose of SAIDs to begin 1 night and 1 to 2 h preoperatively, then 6 h postoperatively. On the other hand, perioperative NSAIDs might be considered for enhanced postoperative pain management after simple and complex periodontal surgeries in which tension-free wound healing is not as crucial for a successful therapy. Finally, it is important to note that despite the statistically significant findings, the overall certainty of evidence ranged from moderate to very low, limiting the strength of clinical recommendations.
Recommendations for Future Clinical Trials
Based on the findings and thorough analysis of the available literature, the following recommendations for researchers/clinicians investigating perioperative inflammatory drugs and postoperative recovery following periodontal surgery are suggested:
Both objective and patient-reported outcomes should be measured and recorded, when possible, to provide a comprehensive understanding of the efficacy and safety of perioperative SAIDs and NSAIDs on postoperative recovery after periodontal surgeries.
While investigating the efficacy of perioperative SAIDs and NSAIDs is clinically relevant to improve patients’ QoL, it is equally important to record and report the safety and adverse events.
The efficacy of perioperative SAIDs and NSAIDs on long-term outcomes such as wound healing, postoperative infection, and clinical therapeutic outcomes (e.g., clinical attachment level) should also be considered in future studies.
Multicentric and multiethnic clinical trials are still needed to evaluate the efficacy and safety of SAIDs and NSAIDs in managing short- and long-term postoperative complications after periodontal surgery.
Researchers should adopt a standardized postoperative anti-inflammatory, analgesic regimen for 3 d postoperatively regardless of assigned perioperative interventions, as significant differences against placebo might be clinically irrelevant.
Patients undergoing any type of periodontal surgery should be considered when recruiting for RCTs, as restricted inclusion criteria limit the external validity and the generalizability of the findings. However, researchers should perform secondary analyses stratified by surgery type and complexity to assess postoperative recovery outcomes.
Conclusion
Perioperative NSAIDs may be more effective in reducing postoperative pain in the first 6 h, while SAIDs might offer better pain management after 48 h postoperatively in patients undergoing periodontal surgeries, yet these differences were clinically insignificant. However, based on moderate-to-low certainty of evidence, and due to a lack of studies, methodological biases, and discrepancies in the drugs and regimens used, these findings might not be applicable to all periodontal patients and thus should be interpreted with caution. Therefore, further optimized multicentric and multiethnic RCTs confirming the efficacy and safety of perioperative SAIDs and NSAIDs for postoperative recovery after periodontal surgical procedures are needed, and robust evidence is still lacking.
Author Contributions
M. Abusamak, contributed to conception and design, data acquisition, analysis, and interpretation, drafted and critically revised the manuscript; A. Elananza, contributed to data acquisition and analysis, drafted and critically revised the manuscript; I. Alshdaifat, contributed to data acquisition, analysis, and interpretation; drafted and critically revised the manuscript; F.S. Al-Hamed, contributed to data interpretation, critically revised the manuscript; H. Al-Waeli, contributed to conception, data interpretation, critically revised the manuscript. All authors gave their final approval and agreed to be accountable for all aspects of the work.
Supplemental Material
sj-docx-1-jct-10.1177_23800844251365539 – Supplemental material for Perioperative Anti-inflammatory Drugs and Recovery after Periodontal Surgery: A Systematic Review and Meta-Analysis
Supplemental material, sj-docx-1-jct-10.1177_23800844251365539 for Perioperative Anti-inflammatory Drugs and Recovery after Periodontal Surgery: A Systematic Review and Meta-Analysis by M. Abusamak, A. Elananza, I. Alshdaifat, F.S. Al-Hamed and H. Al-Waeli in JDR Clinical & Translational Research
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Data Availability Statement
The data and R script are available upon reasonable request from the corresponding authors.
A supplemental appendix to this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
