Abstract
Background:
Dental caries remains a significant oral health burden globally. Scientific evidence has demonstrated the dose-dependent anticaries action of fluoride; however, more effective, comprehensive, and alternative prevention strategies are required.
Methods:
A 2-y, phase 3, randomized controlled trial based on a double-blind, 3-arm, parallel-group design was conducted from April 15, 2019, through March 12, 2022 across 3 centers in China. Six thousand children aged 10 to 14 y with ≥2 active caries lesions were assigned 1 of 3 study dentifrices: 8.0% arginine, 1.5% arginine, and 0.32% sodium fluoride (NaF). The primary efficacy outcomes were incremental DMFS (decayed, missing, and filled surfaces) and DMFT (decayed, missing, and filled teeth) caries indices scores after 2 y of product use. The secondary efficacy outcomes were the incremental caries indices scores after 1 y and 6 mo of product use. Noninferiority was achieved if the 95% CI of the mean difference in scores was below the noninferiority margin of 0.2545 after 2 y, 1 y, and 6 mo of product use.
Results:
After 2 y, the 8.0% arginine–containing dentifrice demonstrated a statistically significant reduction of 26.0% in DMFS scores (−0.16; 95% CI, −0.22 to −0.10; P < .001) and 25.3% in DMFT scores (−0.17; 95% CI, −0.24 to −0.11; P < .001) versus control. No statistical difference was measured between the 1.5% arginine–containing dentifrice and control in DMFS (−0.01; 95% CI, −0.07 to 0.05; P = .819) and DMFT (−0.01; 95% CI, −0.07 to 0.05; P = .739).
Conclusions:
Dentifrice containing 8.0% arginine showed a statistically significant reduction in caries incidence versus the NaF control, while the 1.5% arginine dentifrice showed equivalence to the NaF control regarding caries reduction. This clinical study confirms that arginine dentifrices are effective alternatives to fluoride in providing anticaries protection.
Knowledge Transfer Statement:
This study demonstrates that arginine is an efficacious anticaries agent at the examined doses of 1.5% and 8%. Clinicians and consumers can consider this a new caries preventive agent providing choice to people seeking fluoride-free alternatives. Policy makers could leverage these findings to guide oral health initiatives and inform regulations on dentifrice composition, promoting broader access to effective caries prevention methods.
Keywords
Introduction
Dental caries, a largely preventable disease, is a widespread problem leading to pain, discomfort, and severe health complications when left untreated (Pitts et al. 2017). Globally, its estimated prevalence in permanent teeth is nearly 30%, while the number of cases is >2 billion (World Health Organization 2022). Despite significant dental health advancements, caries remains a pervasive global health challenge affecting individuals across all age groups and socioeconomic strata (World Health Organization 2022). Dental caries persistence signals an urgent call for the development/identification of innovative and multifaceted approaches for addressing this disease.
Fluoride is widely recognized as safe and effective for its role in inhibiting demineralization and enhancing remineralization of enamel for caries prevention (Nassar and Brizuela 2023). In spite of its anticaries benefit, there are populations and individuals for whom fluoride use is contraindicated or who prefer nonfluoride alternatives (Aoun et al. 2018). In response to the limitations of fluoride-based therapies and diverse population needs, several potential nonfluoride alternatives for caries management have been investigated. These include antimicrobial peptides targeting cariogenic bacteria (Sullivan et al. 2011), bioactive glass application and nanohydroxyapatite that promote enamel remineralization (Huang et al. 2011; Bakry et al. 2014), remineralizing peptides to manage early carious lesions (Alkilzy et al. 2018), sugar alcohols (e.g., erythritol and xylitol) to reduce cariogenic potential (Honkala et al. 2014), and probiotics that can alter the oral microbiome in favor of a nonpathogenic biofilm (Cagetti et al. 2013; López-López et al. 2017).
Arginine, the most promising nonfluoride alternative, is a natural amino acid and prebiotic found in high levels within the saliva of individuals who are caries resistant and in low levels in persons prone to caries (Nascimento and Burne 2014). Arginine is metabolized by arginolytic bacteria to produce ammonia, thereby neutralizing the acidic oral environment and reducing enamel demineralization risk and caries progression (Cantore et al. 2013; Nascimento et al. 2013; Santarpia et al. 2014). Several trials have shown that toothpaste containing arginine and fluoride is more effective at preventing caries than fluoride toothpaste alone by enhancing enamel remineralization, modulating the cariogenic biofilm, and inhibiting cariogenic bacterial adhesion to tooth surfaces, thereby limiting biofilm formation (Kraivaphan et al. 2013; Souza et al. 2013; Li et al. 2015; Petersen et al. 2015; He et al. 2016; Nascimento 2018; Carda-Diéguez et al. 2022). The most significant impact of arginine on oral health is its potential to maintain a healthy microbiome and to enhance dental enamel remineralization and repair. Arginine can elevate the oral pH, increasing the degree of saturation with respect to hydroxyapatite, the primary inorganic component of enamel. This pH-modulating ability is a distinctive feature that differentiates arginine from other remineralizing agents (Wolff et al. 2013). Additionally, arginine is one of the few nonfluoride alternatives that have demonstrated an anticaries benefit in multiple 2-y caries trials (Acevedo et al. 2005; Li et al. 2021).
This clinical trial aimed to assess the anticaries efficacy of 2 arginine-containing, fluoride-free dentifrices—8.0% arginine in a calcium carbonate base and 1.5% arginine in a calcium carbonate base—relative to a positive control containing 0.32% sodium fluoride (NaF) in a silica base over a 24-month period.
Methods
Study Design
This phase 3 investigation was a randomized double-blind study based on a 3-cell parallel-group design that was conducted in China at 3 clinical sites: 1) West China College of Stomatology, Sichuan University, Chengdu; 2) School of Stomatology, Chongqing Medical University, Chongqing; and 3) College of Stomatology, Xi’an Jiaotong University, Xi’an. The study took place from April 15, 2019 (first patient visit), to March 12, 2022 (last patient visit). The study was planned and executed in compliance with the International Council for Harmonization and good clinical practice. This report followed the CONSORT guidelines (Consolidated Standards of Reporting Trials) for this randomized clinical trial’s design, analysis, and interpretation of results (Fig. 1). The study was approved by the institutional review board of the China Oral Health Foundation, Beijing, China. Colgate-Palmolive Company was the study sponsor. Written informed consent was obtained from parents/guardians. The study’s design included a detailed protocol for the delivery and use of the study dentifrices, brushing instructions, and monitoring oversight to ensure adherence and safety. A data safety monitoring board met regularly to ensure the safety and integrity of the study, which is registered at ClinicalTrials.gov (NCT05560945). The study protocol is Appendix 1.
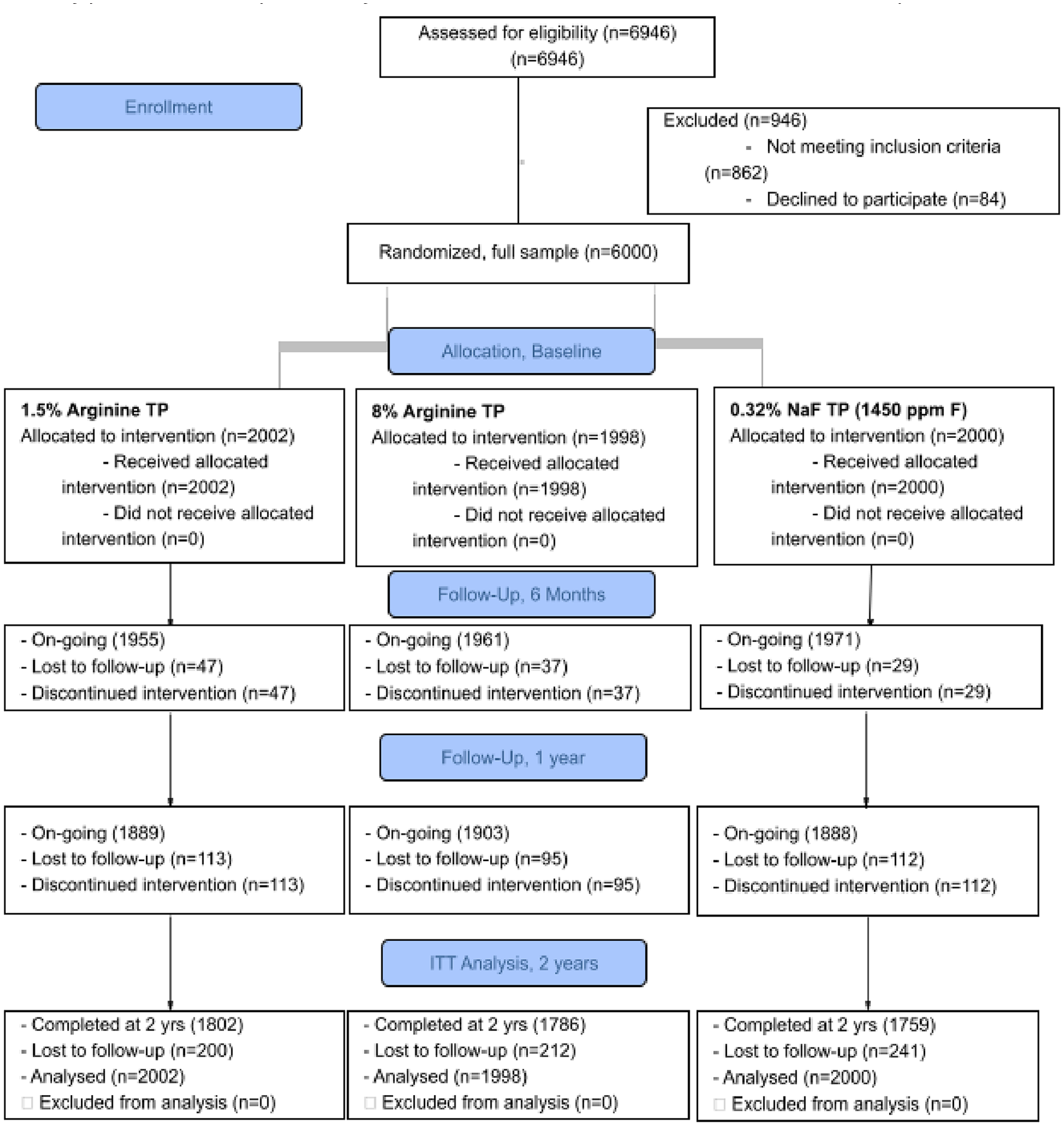
CONSORT diagram. The CONSORT flow diagram shows the number of participants assessed for eligibility; randomized; and allocated to the study products, follow-up, and analysis. ITT, intention to treat; NaF, sodium fluoride; TP, toothpaste.
Participant Eligibility Criteria
Participants were enrolled from 3 major areas within China: the greater areas of Chengdu, Chongqing, and Xi’an. Eligible participants included healthy male and female children aged 10 to 14 y. At this age, caries activity increases considerably due to an increase in susceptible tooth surfaces, immature permanent tooth enamel, defective oral hygiene, and poor diet (Li et al. 2021). Other inclusion criteria were as follows: ≥2 active caries lesions with scores ≥2 according to the International Caries Detection and Assessment System (ICDAS; Acevedo et al. 2008); previous caries experience with a DMFS score ≥2 (decayed, missing, and filled surface; Ekstrand et al. 2018); willingness to use the assigned products as directed; at least 1 second molar or evidence of its eruption; availability for all appointments; and a parent or guardian willing to sign an informed consent form. Parents and guardians were provided with a copy of the informed consent/assent and privacy notice forms to ensure transparency and compliance with data protection regulations.
Participants were excluded from the study for any of the following reasons: fixed or removable prosthetic appliances or orthodontic treatment involving ≥4 permanent teeth; medication use that could increase caries risk, such as medication-induced xerostomia; long-term antibiotic therapy; cognitive and/or motor impairment; severe malocclusion; severe caries (ICDAS, 5 or 6) on ≥5 teeth; moderate or severe periodontal disease; participation in other clinical study within 30 days preceding this trial; history of allergies or other adverse reactions to arginine, oral care products, or their ingredients; and pregnant or lactating females (self-reported).
Examiner Training and Calibration
Prior to the trial initiation, the 6 clinical examiners (2 per site) were trained and calibrated on caries detection methods on-site in Chengdu, China, in February 2019. Dr. Domenick Zero, cariology professor at Indiana University, Indianapolis, served as the gold standard trainer. This process involved 1) discussion on diagnostic criteria, examination methodologies, and procedures and 2) practical exercises to ensure inter- and intraexaminer reliability. These exercises utilized a representative sample to achieve a kappa statistic >0.70 for intraexaminers and >0.61 for interexaminers, signifying substantial agreement beyond chance. This interexaminer kappa value is consistent with acceptable reliability reported in other dental caries studies utilizing ICDAS, where values of 0.60 to 0.61 have been demonstrated as good reliability for severity and activity assessments (Cotta et al. 2015). Furthermore, general guidelines for kappa interpretation classify values between 0.60 and 0.80 as indicating moderate agreement, which is considered substantial for clinical research (Madadizadeh et al. 2023). This calibration ensured consistent and reliable caries assessments across all study sites. Examiners underwent initial calibration prior to baseline measurements, and all exceeded the kappa statistic targets.
Study Interventions
Participants were randomized to receive 1 of the 3 dentifrices: 1) 8.0% arginine in a calcium carbonate base, 2) 1.5% arginine in a calcium carbonate base, and 3) 0.32% NaF in a silica base as control (all from Colgate-Palmolive). The recommended fluoride concentration in toothpaste for caries prevention is 1,000 to 1,500 ppm (Walsh et al. 2019); therefore, a 0.32% NaF toothpaste (1,450 ppm) was selected as control.
Randomization was conducted via a computer-generated random number list by a clinical site staff member, ensuring equitable distribution among treatment groups. To maintain the study’s double-blind nature, products were overwrapped and coded at a separate facility by personnel with no participation in any other aspect of the trial, concealing their identities from examiners and participants. The toothpastes provided to all groups were identical in flavor and aroma and were presented in the same size and type of white overwrapped tubes. The study products were distributed to participants by staff members who were not involved in conducting clinical examinations. Participants were instructed to brush twice a day with their assigned products. To increase compliance, participants were provided with new toothpaste tubes at each visit, and they were asked to bring the leftover product, which was weighted by study personnel. Each toothpaste tube had a net weight of 120 g, and the participants were resupplied every 3 months, starting at baseline and again at months 3, 6, 9, 12, 15, 18, and 21.
Assessments
Qualified participants received a comprehensive baseline examination (oral soft/hard tissue assessment and a caries examination) performed by trained and calibrated examiners. Examiners adhered to established criteria, conducting caries examinations after meticulous tooth cleaning and drying. A halogen light source paired with a dental mirror (Miltex Inc.) and a World Health Organization Community Periodontal Index probe (Shanghai Medical Instruments Co. Ltd.) was used to facilitate visual-tactile assessments of caries status, verifying the enamel surface’s continuity. The assessment included all tooth surfaces of each permanent tooth, excluding third molars. Caries severity assessments were carried out with ICDAS (Ekstrand et al. 2018). Assessments of missing and filled teeth at the tooth surface and tooth level were conducted via the DMFS and DMFT (decayed, missing, and filled teeth) caries indices (Acevedo et al. 2008). For the decayed component of the DMFS and DMFT indices, only cavitated carious lesions were tallied. Specifically, ICDAS scores of 3, 4, 5, and 6 were considered to represent cavitated lesions for this purpose, consistent with their definition as localized enamel breakdown, underlying dark shadow from dentin, distinct cavity with visible dentin, and extensive distinct cavity with visible dentin, respectively (Dikmen 2015; Melgar et al. 2016; Zaazou et al. 2023). This approach aligns with the traditional clinical interpretation of “decayed” for the DMFT/DMFS indices. No radiographs were taken for several reasons: early enamel caries lesions are undetectable (Wenzel 2014), to avoid unnecessary risks (Alzen and Benz-Bohm 2011), and due to feasibility.
Data were directly entered (chairside) into the Microsoft Access database by trained recorders. Subsequent comprehensive examinations were conducted at 6, 12, and 24 mo. Additionally, oral soft and hard tissue safety examinations were performed at 3, 9, 15, 18, and 21 mo. At each follow-up appointment, adverse events were recorded. Adverse event data were obtained from interviews with each participant and from safety examinations.
Study End Points
The primary end point was assessed by determining the incremental subject-wise DMFS and DMFT scores after 2 y of product use. Secondary efficacy measures included incremental DMFS and DMFT scores after 6 mo and 1 y of product use.
Power
The study sample size (N = 6,000) was determined for the secondary efficacy outcome based on previous clinical studies, assuming an annual dropout rate of 10%, a significance level of α = 0.05, an estimated coefficient of variation ≤1.50 units, a noninferiority margin of 10% (upper interval margin based on the ratio of means), and a power level of 80%. After 9 mo of the ongoing study, the Food and Drug Administration (FDA) provided input on the noninferiority method for a proposed phase 3 study. The noninferiority method and a new specific noninferiority margin (0.2545) were proposed by experienced collaborators following substantial investigation using historical data from prior caries clinical trials (including a Cochrane review) and FDA communications (2019 to 2020).
Statistical Analyses
Participants were randomized to receive 1 of the 3 dentifrices. Randomization was conducted via a computer-generated random number list by a clinical site staff member, ensuring unbiased allocation to treatment groups. While study identifications were allocated sequentially to each research site (1 to 2,000 to Chengdu, 2,001 to 4,000 to Chongqing, and 4,001 to 6,000 to Xi’an) primarily for logistical and administrative purposes, it is acknowledged that simple randomization, while ensuring unbiased distribution on average, does not guarantee perfect balance of all baseline characteristics across treatment arms within each site in any single trial instance. As observed, DMFS and DMFT scores varied across arms at baseline. To rigorously address any potential baseline imbalances and minimize bias, our statistical analysis employed baseline-adjusted models using propensity score weighting. This robust approach is a standard method in clinical trials to account for observed differences at baseline and ensure the validity of treatment effect estimates.
An offline electronic data capture system (Microsoft Access 2010) developed in partnership with Zimmerman Software, Inc. was provided to each clinical site. Following ICDAS examinations and assessments of missing and filled teeth for each participant, ICDAS codes were converted into the components of the DMFS and DMFT indices to calculate caries experience (Ismail et al. 2007; Melgar et al. 2016). These scores were aggregated to compute the mean DMFT and DMFS scores for each dentifrice group. An intention-to-treat analysis was performed on all study participants based on the treatment group to which they were assigned.
Missing DMFS and DMFT scores at 3 follow-up periods were imputed via multivariate imputation by chained equations (van Buuren and Groothuis-Oudshoorn 2011). Predictive mean matching, a nonparametric imputation technique, was used where missing values were replaced with observed scores with similar predictive characteristics. Statistical analyses were conducted separately for the incremental DMFS and DMFT scores. Treatment group comparisons with respect to baseline scores were performed by bootstrapping incomplete data 200 times and then imputing each bootstrap 2 times, yielding 400 imputed datasets, to account for violation of the normal distribution and additional variance resulting from imputation (Bartlett 2019). Comparisons among the treatment groups with respect to baseline-adjusted incremental DMFS and DMFT caries scores at each examination were executed with the same bootstrapping and imputation method.
Pairwise comparisons of the treatments were carried out by 1) computing standardized mean differences after accounting for baseline caries scores via propensity score weighting and 2) deriving 95% CIs and corresponding P values from the bootstrapping and imputation method (8.0% arginine vs. 0.32% NaF and 1.5% arginine vs. 0.32% NaF; Bartlett 2019). All statistical tests of hypotheses were 2-sided and employed a significance level of α = 0.05.
Noninferiority assessments were conducted for the 2 arginine dentifrices (8.0% or 1.5% arginine) as compared with the positive control dentifrice for DMFS and DMFT increments. The clinical interpretation of noninferiority was determined through agreement following discussions between the FDA and Colgate-Palmolive for an ongoing US phase 2 study (NCT04750902), with assessments of comparative efficacy based on average incremental caries scores. The noninferiority margin 0.2545 used in this study was established per FDA guidance and historical data, including findings from Cochrane reviews on fluoride toothpastes of different concentrations for preventing dental caries (Ekstrand et al. 2018; Walsh et al. 2019). The comparative efficacy was assessed by expressing the mean incremental caries score for the test dentifrice as a difference from the control.
Results
A total of 6,000 participants with a mean age of 12.35 y (range, 10 to 14; 3,409 females, 56.8%) were randomized to 8.0% arginine, 1.5% arginine, or 0.32% NaF dentifrices (Table 1). A total of 5,347 (89.1%) participants completed the trial in full (Fig. 1).
Age and Gender of Participants Who Were Recruited for Clinical Study.
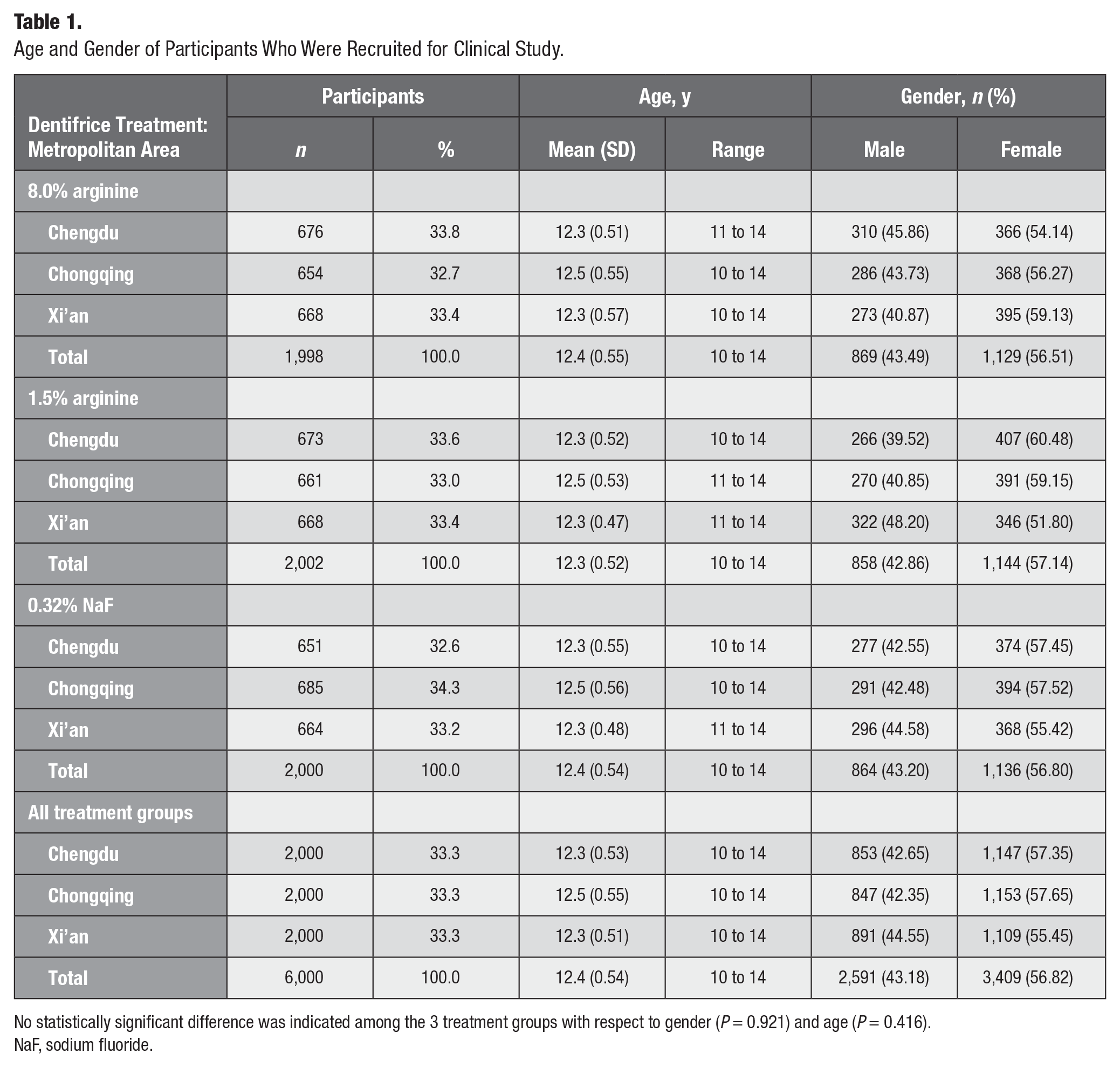
No statistically significant difference was indicated among the 3 treatment groups with respect to gender (P = 0.921) and age (P = 0.416).
NaF, sodium fluoride.
Incremental DMFS and DMFT Caries Scores and Noninferiority Assessment
DMFS and DMFT caries scores were measured at baseline, 6-mo, 1-y, and 2-y evaluations for all participants according to the intention-to-treat analysis (Table 2). Statistical differences at baseline were indicated among the 3 treatment groups: 8.0% arginine, 1.5% arginine, and NaF (mean [SE] DMFS: 4.26 [0.07], 4.13 [0.07], 3.91 [0.06], P < 0.001; DMFT: 3.08 [0.04], 3.15 [0.04], 2.92 [0.03], P < 0.001).
DMFS and DMFT Caries Scores at the Baseline, 6-mo, 1-y, and 2-y Examinations.
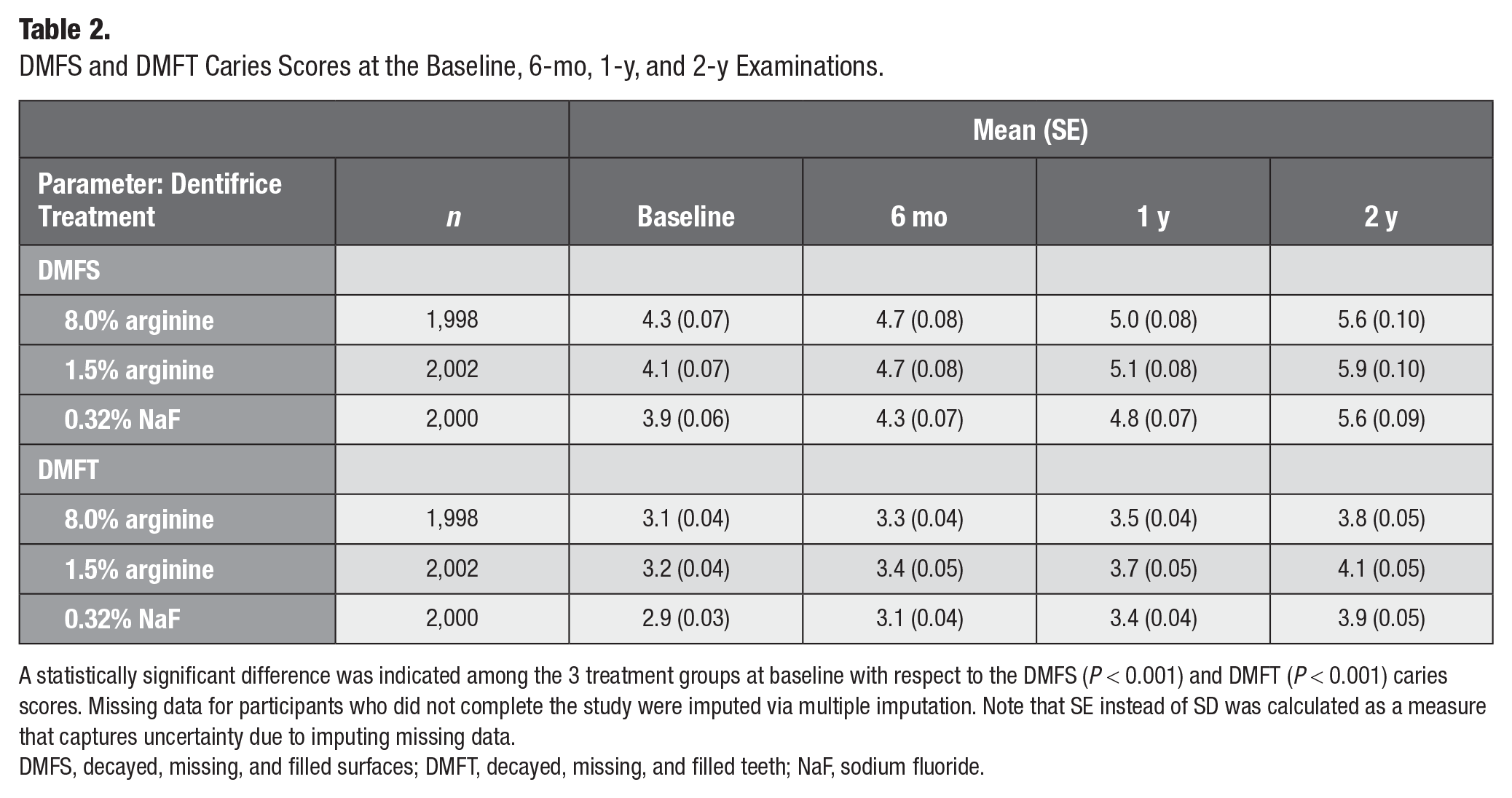
A statistically significant difference was indicated among the 3 treatment groups at baseline with respect to the DMFS (P < 0.001) and DMFT (P < 0.001) caries scores. Missing data for participants who did not complete the study were imputed via multiple imputation. Note that SE instead of SD was calculated as a measure that captures uncertainty due to imputing missing data.
DMFS, decayed, missing, and filled surfaces; DMFT, decayed, missing, and filled teeth; NaF, sodium fluoride.
At the 6-mo evaluation, there was a significant but noninferior difference for both arginine dentifrice groups as compared with the control group (Appendix Table 1, Appendix Fig. 1).
At the 1-y evaluation, the 8.0% arginine dentifrice was considered to have superior anticaries efficacy with a greater decrease in mean incremental DMFS and DMFT caries scores as compared with the control group (Appendix Fig. 2A, B; Appendix Table 2). The 1.5% arginine dentifrice showed parity to the NaF control dentifrice at this time point (Appendix Fig. 2C, D
At the 2-y evaluation, the 8.0% arginine dentifrice group exhibited a statistically significant reduction of 26.0% (−0.16; 95% CI, −0.22 to −0.10; P < 0.001) in mean incremental DMFS scores and superior anticaries effectiveness as compared with the control group (Table 3, Fig. 2A). The 1.5% arginine dentifrice exhibited a nonsignificant decrease of 0.7% (−0.01; 95% CI, −0.07 to −0.05; P = 0.819) and noninferior parity in incremental DMFS scores as compared with the control (Table 3, Fig. 2B).
Baseline-Adjusted Incremental DMFS and DMFT Caries Scores at the 2-y Examination.
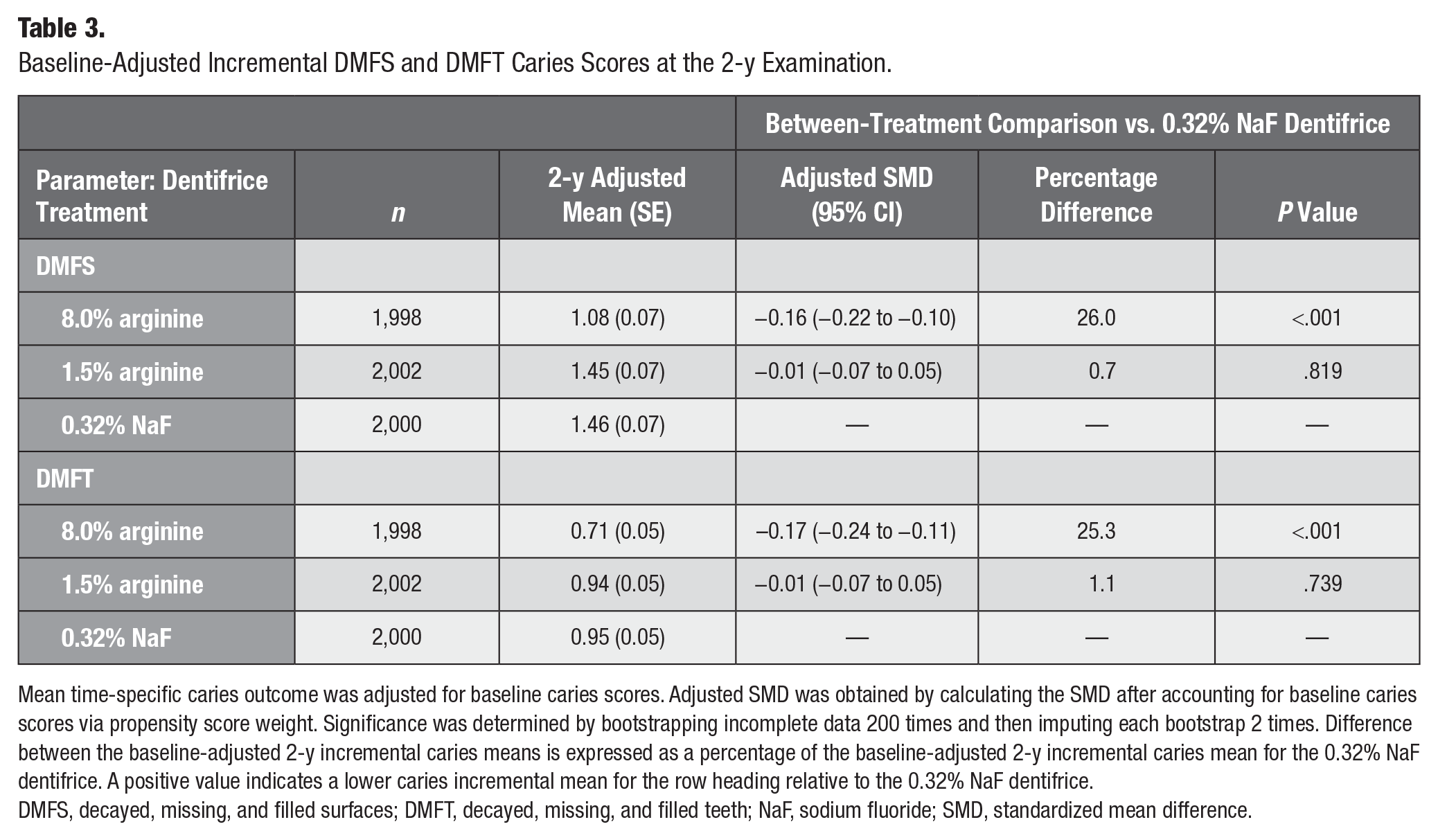
Mean time-specific caries outcome was adjusted for baseline caries scores. Adjusted SMD was obtained by calculating the SMD after accounting for baseline caries scores via propensity score weight. Significance was determined by bootstrapping incomplete data 200 times and then imputing each bootstrap 2 times. Difference between the baseline-adjusted 2-y incremental caries means is expressed as a percentage of the baseline-adjusted 2-y incremental caries mean for the 0.32% NaF dentifrice. A positive value indicates a lower caries incremental mean for the row heading relative to the 0.32% NaF dentifrice.
DMFS, decayed, missing, and filled surfaces; DMFT, decayed, missing, and filled teeth; NaF, sodium fluoride; SMD, standardized mean difference.
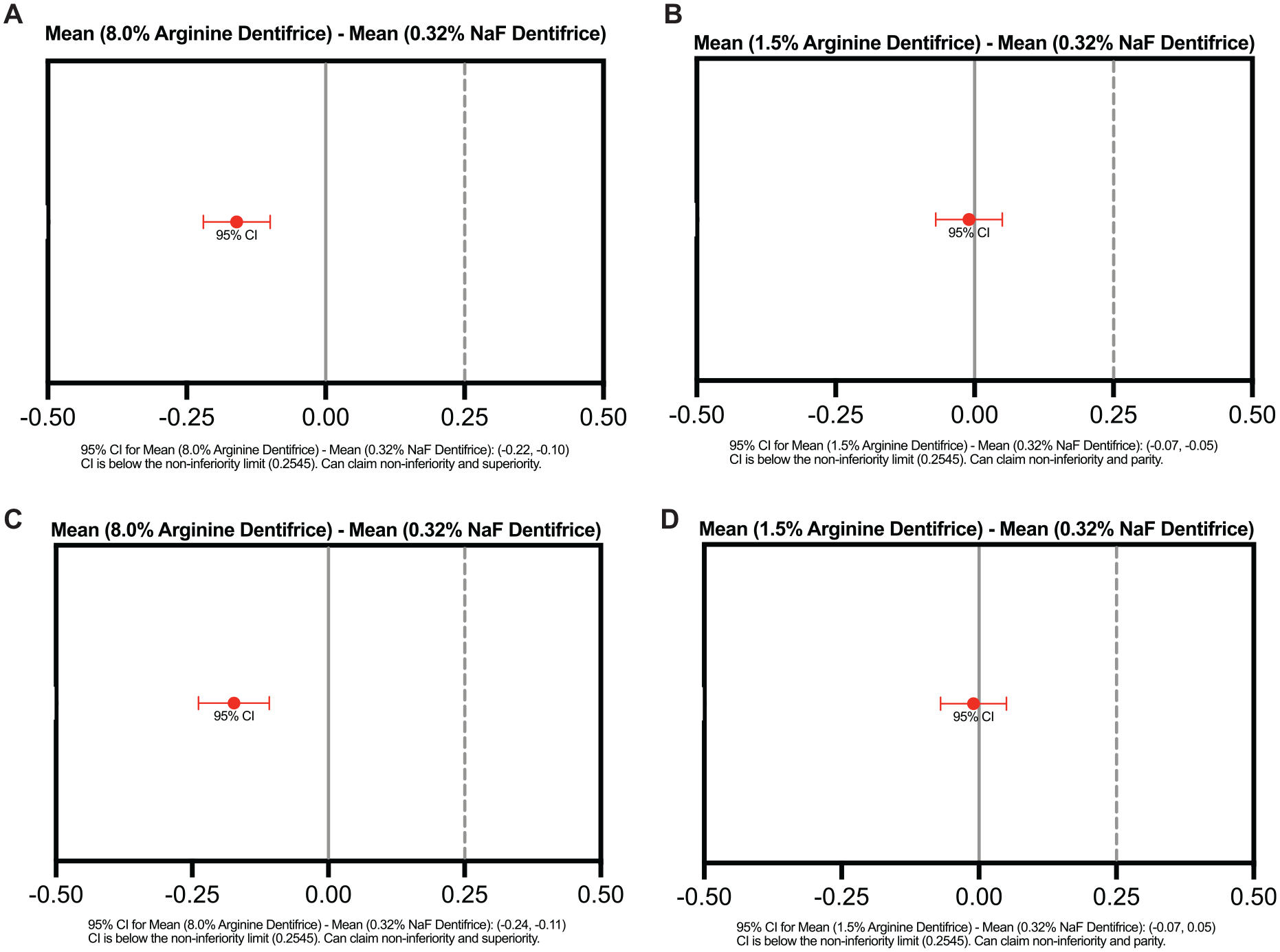
Noninferiority assessment of incremental DMFS and DMFT caries at the 2-y evaluation of 8.0% and 1.5% arginine dentifrice vs. 0.32% NaF dentifrice. DMFS caries index: (
At the 2-y evaluation, the 8.0% arginine dentifrice group exhibited a statistically significant reduction of 25.3% (−0.17; 95% CI, −0.24 to −0.11; P < 0.001) in mean incremental DMFT scores and superior anticaries effectiveness as compared with the control group (Table 3, Fig. 2C). The 1.5% arginine group exhibited a nonsignificant decrease of 1.1% (−0.01; 95% CI, −0.07 to −0.05; P = 0.739) and noninferior parity in incremental DMFT scores as compared with control (Table 3, Fig. 2D).
There were no serious adverse events. Participants (n = 37) reported nonserious side effects; 75.7% were nonrelated to the dentifrice. Related and possibly related nonserious side effects included unspecified occasional toothache (n = 4), dry mouth (n = 2), unpalatability (n = 2), and acne (n = 1).
Discussion
This 2-y study illustrated superior caries reduction with an arginine-containing dentifrice in a dose-dependent manner as compared with fluoride-based treatment, as evidenced by a 26.0% greater decrease in incremental DMFS scores and a 25.3% greater decrease in incremental DMFT scores after 2 y of brushing with an 8.0% arginine dentifrice as compared with the control NaF dentifrice. In contrast, a 1.5% arginine dentifrice maintained parity with the NaF control, demonstrating no significant additional caries protection benefit relative to fluoride alone. These results suggest that the anticaries effect of arginine is dose dependent and that a threshold concentration may be necessary to achieve a clinically meaningful benefit. This underscores the potential for arginine dentifrices at varying doses as effective alternatives to fluoride-based caries prevention strategies.
Previous trials report that fluoride dentifrices containing arginine have superior anticaries efficacy as compared with those containing fluoride alone, indicating the potential of a new active, arginine, to have an additive beneficial effect to fluoride (Cantore et al. 2013; Yin et al. 2013; Santarpia et al. 2014). An earlier 2-y clinical study in nearly 600 adolescents also shows that the anticaries effects of 1.5% arginine and 0.24% NaF dentifrices are comparable (Acevedo et al. 2005). To our knowledge, the current study represents the largest long-term trial to date on the dose-dependent anticaries efficacy of arginine-containing dentifrices in the absence of fluoride.
The efficacy design of the study is particularly relevant when considering alternative and adjunctive therapies to fluoride. Excessive levels of fluoride have been associated with dental fluorosis, a disorder characterized by enamel hypomineralization (World Health Organization 2022), and cases of fluoride allergy have been documented, with stannous fluoride dentifrice as a notable example (Center for Drug Evaluation and Research and Center for Biologics Evaluation and Research 2016). Confirmation of noninferiority assures clinicians and participants that the efficacy of arginine dentifrices is at least comparable to that of fluoride-based products, the cornerstone of caries prevention for decades. Beyond the exploration of new actives, such as arginine, to help alleviate the primary global oral health issue, factors such as sugar consumption, poor oral hygiene, and access need to be addressed in our fight against caries.
This trial boasts a large and diverse population of caries-risk children, the use of stringent ICDAS diagnostic criteria by trained and calibrated examiners, and low attrition. However, potential study confounders include diet, socioeconomic status, dental care access, fluoride exposure from other sources, and individual oral hygiene practices; all of which can influence caries development (Walsh et al. 2019). Consequently, further research in diverse populations may be necessary to generalize the results. It is important to acknowledge that DMFS and DMFT scores varied across arms at baseline. Yet, even with stratified or permuted block randomization by location, perfect balance in baseline data is not always achievable, especially in multicenter trials with inherent population differences. While this study used a simple randomization method, a baseline-adjusted statistical model was employed in the analysis to account for these differences and minimize bias. This adjustment is a standard and robust approach that addresses any potential imbalance across groups at baseline. Furthermore, to ensure the robustness of our conclusions, sensitivity analyses were performed by incorporating additional covariates, such as participant sex, age, and study site into our statistical models. These comprehensive adjustments consistently yielded similar treatment arm estimates and maintained the same significant differences or nondifferences among arms as reported in the primary analyses, underscoring the reliability and generalizability of our findings.
The significant caries reduction observed with the 8.0% arginine dentifrice provides evidence that high-concentration arginine formulations may be more efficacious than currently available levels of fluoride in caries prevention. Therefore, the results of this study are particularly relevant in the context of fluoride-free caries prevention. The superiority of 8.0% arginine and parity of 1.5% arginine seen at the 1- and 2-y evaluations across DMFS and DMFT caries scores suggests that while the lower concentration of arginine is adequate to maintain a caries prevention level comparable to NaF, higher concentrations may be necessary to achieve enhanced benefits. These consistent results across different caries measures strengthen the evidence for the effectiveness of arginine in caries prevention.
Conclusions
The findings of this study suggest that arginine-containing dentifrices are effective in preventing caries, offering caries protection comparable to that of a 0.32% NaF dentifrice at a 1.5% arginine concentration and superior protection at an 8.0% arginine concentration. Both concentrations of arginine-containing dentifrices present viable alternatives to traditional fluoride dentifrices, with the potential for enhanced caries prevention at higher arginine levels. The dose-dependent efficacy of arginine emphasizes the importance of optimizing concentration levels in oral care product formulation. Arginine emerges as a promising anticaries agent, particularly for individuals seeking an alternative to fluoride, as well as for regions or populations where dental fluorosis is a concern.
Author Contributions
W. Yin, contributed to acquisition, critically revised the manuscript; Z. Zhou, contributed to acquisition, critically revised the manuscript; R.-Z. Huang, contributed to acquisition, critically revised the manuscript; G. Sun, contributed to acquisition, critically revised the manuscript; Y. Zhong, contributed to acquisition, critically revised the manuscript; Z. Yang, contributed to acquisition, critically revised the manuscript; Y. Li, contributed to acquisition, critically revised the manuscript; Y. Zhang, contributed to acquisition, critically revised the manuscript; P. Zhang, contributed to acquisition, critically revised the manuscript; D. Hu, contributed to acquisition and interpretation, critically revised the manuscript; L.R. Mateo, contributed to analysis and interpretation, critically revised the manuscript; G. Gao, contributed to analysis, critically revised the manuscript; S. Lim, contributed to analysis and interpretation, critically revised the manuscript; A.I. Ismail, contributed to analysis and interpretation, critically revised the manuscript; J. Zimmerman, contributed to acquisition, critically revised the manuscript; B. Garcia-Godoy, contributed to conception and design, data interpretation, critically revised the manuscript; M. Ryan, contributed to conception and design, data interpretation, drafted and critically revised the manuscript; Y.-P. Zhang, contributed to conception and design, data interpretation, drafted and critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of the work.
Supplemental Material
sj-docx-1-jct-10.1177_23800844251361471 – Supplemental material for Arginine Dentifrices and Childhood Caries Prevention: A Randomized Clinical Trial
Supplemental material, sj-docx-1-jct-10.1177_23800844251361471 for Arginine Dentifrices and Childhood Caries Prevention: A Randomized Clinical Trial by W. Yin, Z. Zhou, R.-Z. Huang, G. Sun, Y. Zhong, Z. Yang, Y. Li, Y. Zhang, P. Zhang, D. Hu, L.R. Mateo, G. Gao, S. Lim, A.I. Ismail, J. Zimmerman, B. Garcia-Godoy, M. Ryan and Y.-P. Zhang in JDR Clinical & Translational Research
Supplemental Material
sj-docx-2-jct-10.1177_23800844251361471 – Supplemental material for Arginine Dentifrices and Childhood Caries Prevention: A Randomized Clinical Trial
Supplemental material, sj-docx-2-jct-10.1177_23800844251361471 for Arginine Dentifrices and Childhood Caries Prevention: A Randomized Clinical Trial by W. Yin, Z. Zhou, R.-Z. Huang, G. Sun, Y. Zhong, Z. Yang, Y. Li, Y. Zhang, P. Zhang, D. Hu, L.R. Mateo, G. Gao, S. Lim, A.I. Ismail, J. Zimmerman, B. Garcia-Godoy, M. Ryan and Y.-P. Zhang in JDR Clinical & Translational Research
Supplemental Material
sj-pdf-3-jct-10.1177_23800844251361471 – Supplemental material for Arginine Dentifrices and Childhood Caries Prevention: A Randomized Clinical Trial
Supplemental material, sj-pdf-3-jct-10.1177_23800844251361471 for Arginine Dentifrices and Childhood Caries Prevention: A Randomized Clinical Trial by W. Yin, Z. Zhou, R.-Z. Huang, G. Sun, Y. Zhong, Z. Yang, Y. Li, Y. Zhang, P. Zhang, D. Hu, L.R. Mateo, G. Gao, S. Lim, A.I. Ismail, J. Zimmerman, B. Garcia-Godoy, M. Ryan and Y.-P. Zhang in JDR Clinical & Translational Research
Footnotes
Acknowledgements
Technical writing assistance was provided by Meghan A. Berryman, PhD. W. Yin had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis. We thank Dr. Domenick Zero (Indiana University, Indianapolis) for serving as a gold standard trainer. He provided training and calibration to the 6 dental examiners to ensure intra- and interexaminer reliability, which was crucial for the accuracy and reliability of our data collection process. We also acknowledge Dr. Wensheng Rong (Peking University, Beijing, China) and Dr. Jiyao Li (Sichuan University, Chengdu, China) for their roles as board members on the data safety monitoring board. Their expertise in caries research and pediatric caries research provided significant insights and guidance, contributing to the safety and integrity of our clinical trial. Their efforts were instrumental in the successful completion of this research. We would like to express our gratitude for their invaluable contributions.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: W. Yin, G. Sun, Y. Zhong, D. Hu, Z. Zhou, Z. Yang, Y. Li, R.-Z. Huang, Y. Zhang, and P. Zhang are independent clinical dental researchers. These authors declare nonfinancial conflicts of interest. Independent statisticians A.I. Ismail, S. Lim, L.R. Mateo, and G. Gao also declare nonfinancial conflicts of interest. B. Garcia-Godoy, J. Zimmerman, M. Ryan, and Y.-P. Zhang are employees of Colgate-Palmolive. All authors, including those employed by Colgate-Palmolive, declare no competing interests.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported and funded by Colgate-Palmolive.
Role of the Funder/Sponsor
Colgate-Palmolive provided the study products and was involved in several aspects of the research process. Specifically, Colgate-Palmolive contributed to the design and conduct of the study, collection, management, analysis, and interpretation of the data. Additionally, Colgate-Palmolive was involved in the preparation, review, and approval of the manuscript.
Clinical Trial Registration
Calibration exercises for the 6 dental examiners were successfully completed prior to the commencement of the clinical trial in 2019. However, the COVID-19 pandemic disrupted subsequent planned calibration activities and in-person monitoring at the 3 study sites. Travel restrictions and limited access to facilities due to the pandemic also hindered the originally planned on-site monitoring activities in China. However, remote monitoring was deployed during the course of this study.
Furthermore, the COVID-19 pandemic had an impact on the additional cohort examinations that were planned as part of the study. These examinations were intended to utilize the Trios 3D Scanner and Diagnocam, as outlined at the end of the study protocol. However, due to the challenges posed by the pandemic, including restrictions on in-person interactions and access to clinical settings, these examinations were not completed as initially intended.
A supplemental appendix to this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
