Abstract
Introduction:
Concentrated growth factor (CGF), a novel autologous platelet concentrate, has gained substantial interest for its potential role in managing oral diseases. The objective of this study is to evaluate the effectiveness of CGF in regenerative dentistry.
Methods:
A systematic search was performed across MEDLINE, OVID, Scopus, Cochrane Library, Google Scholar, Web of Science, and Elsevier for publications between July 1, 2013, and July 1, 2023. Only randomized clinical trials were included. The quality of the methodology was assessed using the Cochrane Risk of Bias Tool. Data were analyzed using RevMan 5.4 software.
Results:
In 3 trials treating periodontal intrabony defects, CGF combined with bone grafts showed significant superiority over bone grafts alone (P < .00001), with a mean reduction of 1.41 mm in intrabony defect depth and a mean gain of 0.55 mm in clinical attachment level (P = 0.002). For gingival recession in 5 trials, CGF combined with coronally advanced flap (CAF) was more effective than CAF alone, resulting in a mean increase in keratinized tissue width of 0.41 mm and a mean increase in gingival thickness of 0.26 mm (P < 0.00001). However, CGF combined with CAF was less effective than connective tissue graft (CTG) combined with CAF, showing a mean difference in root coverage of −15.09% and a mean difference in gingival thickness of −0.50 mm (P < 0.0001). In alveolar ridge preservation, sinus elevation, guided bone regeneration, dental implant procedures, and postextraction healing, CGF gave better values in clinical practice.
Conclusions:
CGF enhances regenerative surgery outcomes for periodontal intrabony defects. Combined with CAF, CGF improves therapeutic efficacy for gingival recession, although less so than CTG with CAF. CGF shows promise in various regenerative dentistry areas. However, the absence of meta-analysis and low-quality assessments in many studies necessitates further high-quality research.
Knowledge Transfer Statement:
The use of concentrated growth factors (CGFs) in regenerative dentistry, within the limitation of low-quality studies, shows promising benefits in treating periodontitis infrabony defects, gingival recession, guided bone regeneration, dental implant alveolar ridge preservation, sinus elevation, and third molar extraction. Future research should focus on high-quality studies and explore combining CGF with grafting materials to enhance therapeutic outcomes in oral surgery.
Keywords
Introduction
Regenerative therapy in dentistry aims to restore or regenerate oral tissues affected by disease or injury. The complexity of oral tissues, including hard and soft tissues, presents significant challenges due to their diverse cell populations and intricate extracellular matrices (Dangaria et al. 2009).
Autologous platelet concentrates (APCs) collect platelets and growth factors from the patient’s plasma to facilitate localized healing at surgical sites. APCs are effective in soft tissue restoration due to their ability to release cytokines (Anfossi et al. 1989). Three generations of APCs have been developed: platelet-rich plasma (PRP), platelet-rich fibrin (PRF), and concentrated growth factor (CGF).
APCs, particularly Leukocyte-Platelet Rich Fibrin (L-PRF), are extensively utilized in clinical applications for their role in enhancing tissue healing and regeneration. Their controlled release of growth factors makes them effective in alveolar ridge preservation, sinus floor elevation, periodontal plastic surgery, and guided tissue and bone regeneration (Quirynen et al. 2025).
PRP, the first generation, concentrates platelets through centrifugation and requires chemical additives for fibrin polymerization. PRF, the second generation, simplifies preparation and eliminates the need for additional chemicals (Choukroun et al. 2001). CGF, a second-generation platelet concentrate similar to PRF, is created using the Medifuge MF200 (Silfradent, Srl) by centrifuging blood at speeds of 2,400 rpm (400 g) to 3,000 rpm (600 g) (Rodella et al. 2011).
CGF shows promising regenerative potential and versatility. It enhances sinus elevation and alveolar ridge structures; supports mesenchymal stem cell proliferation, osteogenic maturation, and bone defects remediation; and stimulates periodontal ligament stem proliferation. It also aids in treating chronic venous ulcers (Sohn et al. 2011).
CGF offers enhanced regenerative capacity and adaptability. Its higher fibrinogen concentration, factor XIII, and thrombin improve fibrin clot quality. Activated factor XIIIa strengthens the clot, increasing tensile strength and adhesive potency while reducing hemostatic duration (Mansour and Kim 2010).
Despite its potential, the clinical effectiveness of CGF remains uncertain. This systematic review aims to evaluate CGF’s clinical utility in regenerative dental procedures by comparing it with conventional biomaterials based on human clinical trials.
Numerous studies, including case reports, case series, and randomized controlled trials (RCTs), have highlighted CGF’s benefits in regenerative therapy. While 3 systematic reviews have explored CGF’s efficacy in intrabony defects, gingival recession, and dental implantology, there is a lack of systematic reviews specifically focused on CGF’s benefits in regenerative dentistry through RCTs. This review aims to provide the highest level of evidence from secondary studies on the efficacy of the CGF in regenerative dentistry.
Methods
Protocol Development and Registration
The study follows Preferred Reporting Items for Systematic Reviews and Meta-Analyses standards and is registered on the PROSPERO database (CRD42024546802). A detailed protocol was developed before the review, covering all aspects of systematic review methodology: defining the research questions, creating a PICOS framework (elaborated in Appendix Table 3), developing a search strategy, establishing inclusion criteria (elucidated in Appendix Table 4), and determining outcome measures, methods of screening, data extraction, analysis, and synthesis.
Assessment of Outcome Measures
The study assessed CGF’s regenerative potential in regenerative dentistry and analyzed it using Review Manager v5.4. Meta-analysis incorporated mean differences with 95% confidence intervals (CIs). Heterogeneity was tested with χ2 and I2 statistics (significant if P < 0.05). Fixed-effect models were used for I2 < 50%, and random-effect models were used for I2 > 50%. Studies are summarized in Table 1.
Characteristics of the Included Studies.
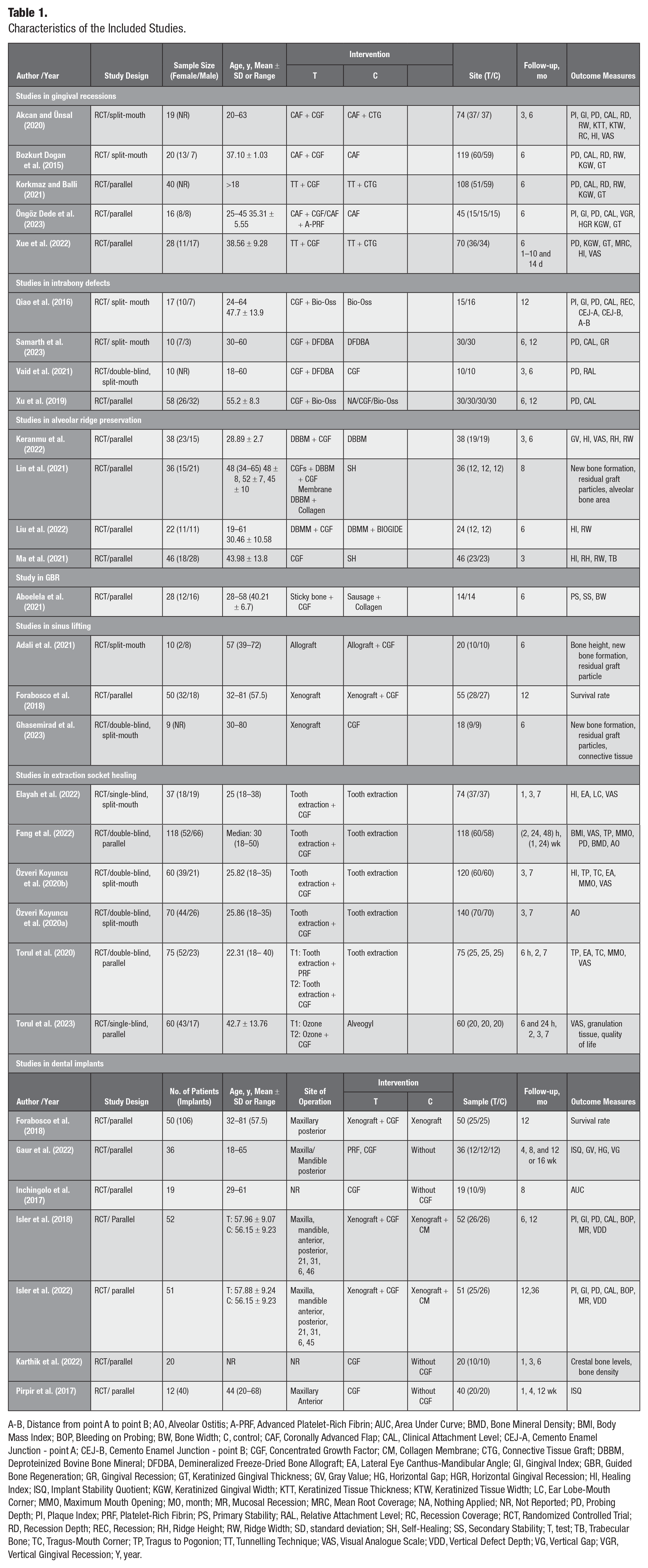
A-B, Distance from point A to point B; AO, Alveolar Ostitis; A-PRF, Advanced Platelet-Rich Fibrin; AUC, Area Under Curve; BMD, Bone Mineral Density; BMI, Body Mass Index; BOP, Bleeding on Probing; BW, Bone Width; C, control; CAF, Coronally Advanced Flap; CAL, Clinical Attachment Level; CEJ-A, Cemento Enamel Junction - point A; CEJ-B, Cemento Enamel Junction - point B; CGF, Concentrated Growth Factor; CM, Collagen Membrane; CTG, Connective Tissue Graft; DBBM, Deproteinized Bovine Bone Mineral; DFDBA, Demineralized Freeze-Dried Bone Allograft; EA, Lateral Eye Canthus-Mandibular Angle; GI, Gingival Index; GBR, Guided Bone Regeneration; GR, Gingival Recession; GT, Keratinized Gingival Thickness; GV, Gray Value; HG, Horizontal Gap; HGR, Horizontal Gingival Recession; HI, Healing Index; ISQ, Implant Stability Quotient; KGW, Keratinized Gingival Width; KTT, Keratinized Tissue Thickness; KTW, Keratinized Tissue Width; LC, Ear Lobe-Mouth Corner; MMO, Maximum Mouth Opening; MO, month; MR, Mucosal Recession; MRC, Mean Root Coverage; NA, Nothing Applied; NR, Not Reported; PD, Probing Depth; PI, Plaque Index; PRF, Platelet-Rich Fibrin; PS, Primary Stability; RAL, Relative Attachment Level; RC, Recession Coverage; RCT, Randomized Controlled Trial; RD, Recession Depth; REC, Recession; RH, Ridge Height; RW, Ridge Width; SD, standard deviation; SH, Self-Healing; SS, Secondary Stability; T, test; TB, Trabecular Bone; TC, Tragus-Mouth Corner; TP, Tragus to Pogonion; TT, Tunnelling Technique; VAS, Visual Analogue Scale; VDD, Vertical Defect Depth; VG, Vertical Gap; VGR, Vertical Gingival Recession; Y, year.
Search Strategy
The literature search includes electronic and manual methods across multiple databases: PubMed, EMBASE, Scopus, Cochrane Central Register of Controlled Trials, Cochrane Oral Health Group Trials Register, Google Scholar, Web of Science, and SciVerse. Searches focus on articles in English published between July 1, 2013, and July 1, 2023, using terms related to CGF effects on regenerative dentistry. Specific journals are manually searched for the same period.
Search Screening
The initial search yielded 1,054 records, narrowed to 498 abstracts after removing duplicates. Thirty full-text articles were examined for CGF’s effectiveness in dental treatments, such as intrabony defects, gingival recession, guided bone regeneration (GBR), alveolar bone preservation, implants, third molar extraction, and sinus lift procedures, as depicted in Figure 1.
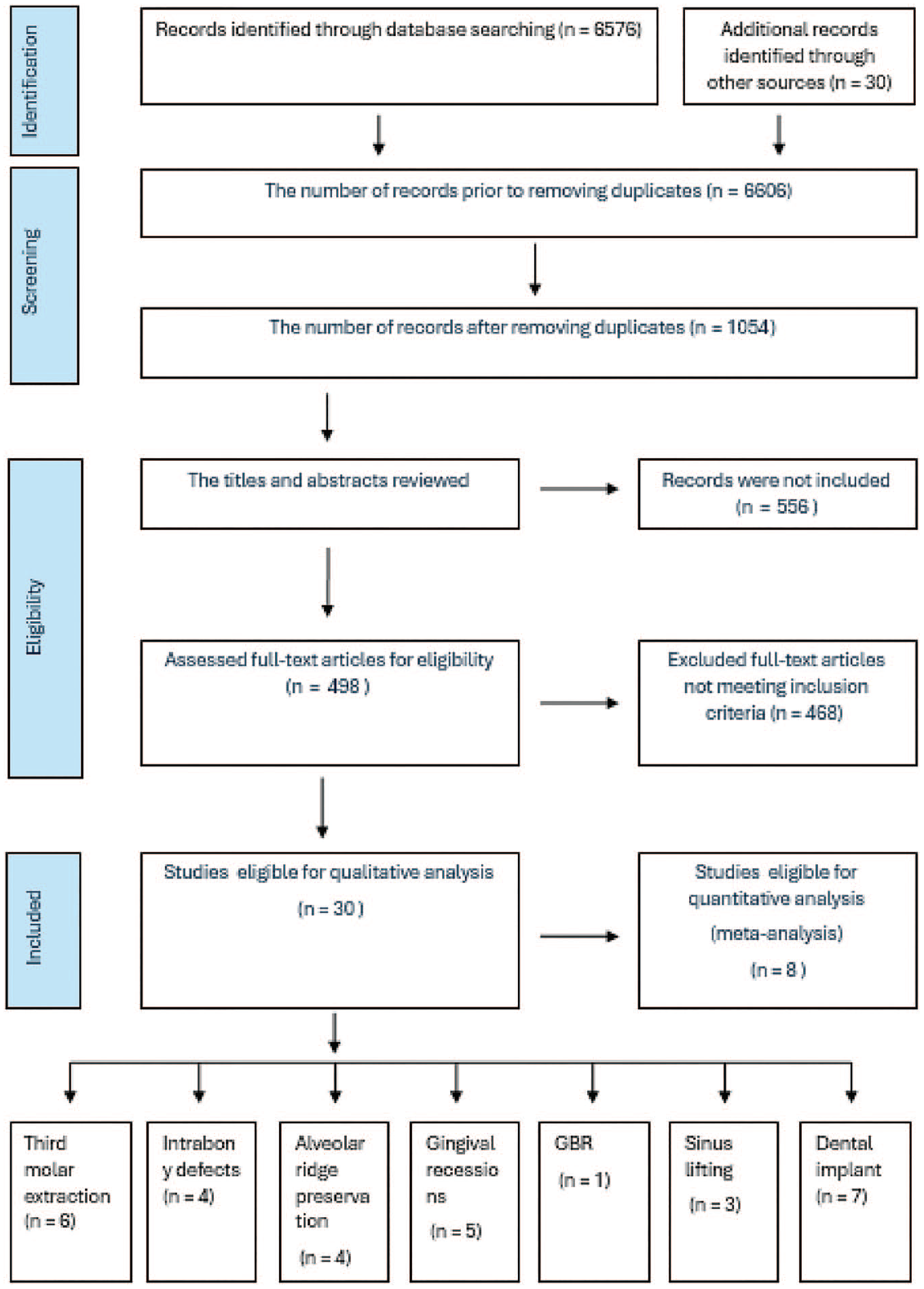
Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) 2020 flowchart for study selection. This flowchart illustrates the study selection process according to the PRISMA 2020 guidelines. The diagram shows the number of records identified, screened, excluded, and included in the systematic review and meta-analysis. Each step of the selection process is depicted, highlighting how the final set of studies was determined from the initial pool.
Method of Screening
The titles and abstracts of the selected studies were independently assessed by 2 reviewers (A.A. and F.A.) to determine the impact of CGF on critical outcomes in various dental treatments. Full-text articles were obtained for positive screenings. Reviewer concordance was measured using κ values, and disputes were resolved through discussions with Z.A. If essential data were missing, the corresponding authors were contacted for clarification.
Data Validity Analysis
Data extraction followed inclusion criteria, categorized by study design and outcomes, and the data were verified for accuracy. Quality assessment using the Cochrane Risk of Bias Tool identified 1 low-risk, 5 unclear-risk, and 24 high-risk RCTs, with common biases in performance, attrition, detection, and selection, as shown in Appendix Table 5 and Appendix Figure 4.
Results
Intrabony defects, gingival recession, GBR, alveolar ridge preservation, dental implants, third molar extraction, and sinus lifts were studied. A quantitative meta-analysis included 8 out of 30 relevant studies, while the remaining 22 were in qualitative systematic review analysis as the following:
Intrabony Defects
CGF targets periodontal intrabony defects for regeneration. Four RCTs assessed CGF for this purpose, often comparing it with bone graft materials. Vaid et al. (2021) found no significant differences between CGF and demineralized freeze-dried bone allograft (DFDBA) after 6 mo.
Three studies (Qiao et al. 2016; Xu et al. 2019; Samarth et al. 2023), with a 12-mo follow-up, were included in the meta-analysis, which examined reduced probing pocket depth (PPD) and clinical attachment level (CAL). Qiao et al. (2016) and Samarth et al. (2023) also recorded intrabony defect depth (IDD), while the study by Vaid et al. (2021), with a 6-mo follow-up, was excluded from the meta-analysis.
These studies included 151 intrabony defects in 85 patients, with 75 experimental and 76 control sites. Meta-analysis compared CGF + bone grafts to bone grafts alone. There was no significant difference in PPD reduction (P = 0.15), though CGF + bone grafts showed a slight advantage (0.49 mm). CAL improvement was significant (P = 0.002), favoring CGF + bone grafts by 0.55 mm. Qiao et al. (2016) and Samarth et al. (2023) reported a significant IDD reduction (P < 0.00001), favoring the CGF group by 1.41 mm, as shown in Figure 2.
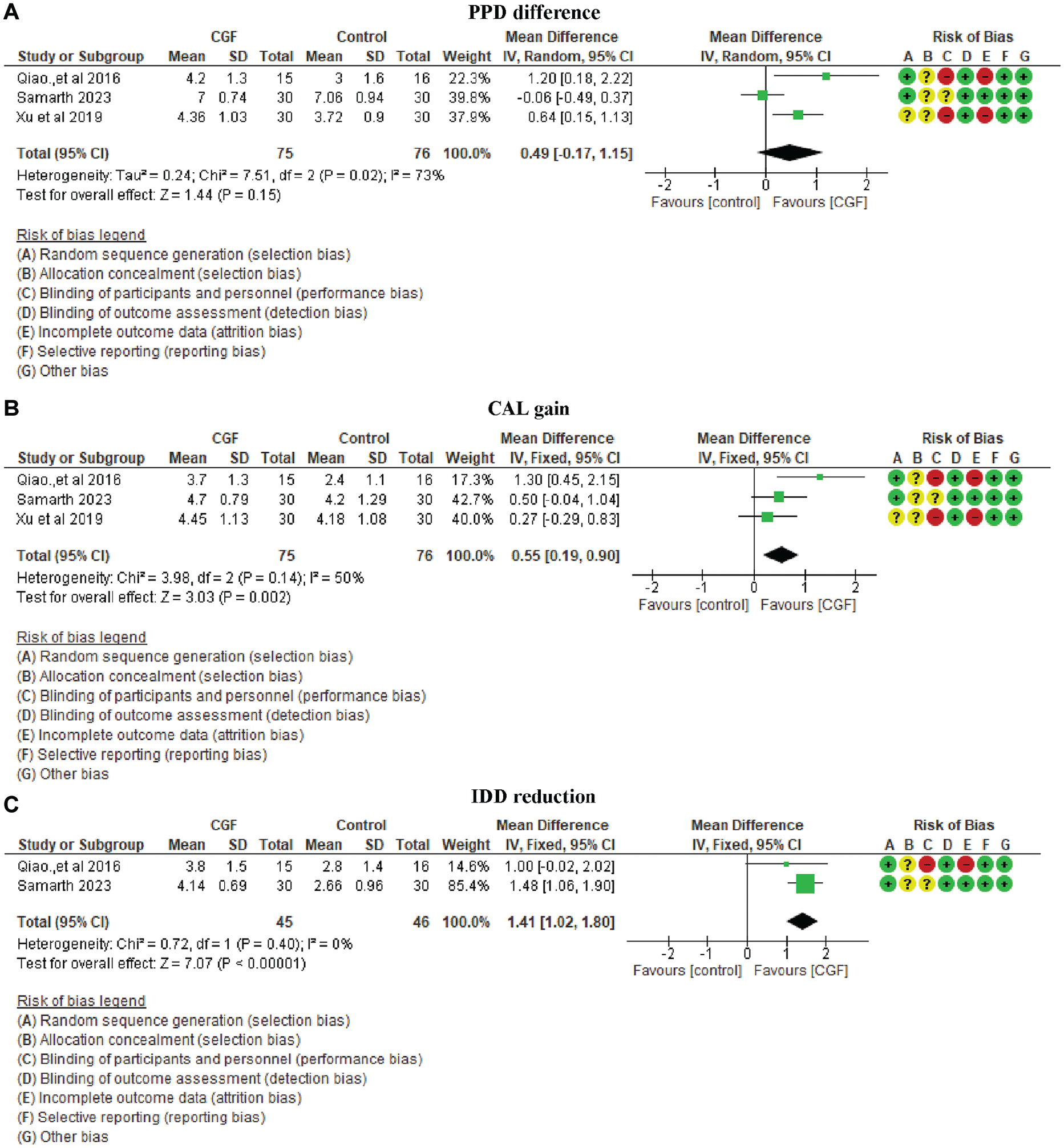
Forest plots of intrabony defect studies. This figure displays forest plots of intrabony defect studies representing the meta-analysis results for 3 key clinical outcomes: (
Gingival Recession
Two studies investigated CGF for root coverage in gingival recessions, comparing the effectiveness of coronally advanced flap (CAF) alone versus CAF + CGF. Öngöz Dede et al. (2023) and Bozkurt Doğan et al. (2015) included 149 sites (74 CAF alone, 75 CAF + CGF).
Three studies (Akcan and Ünsal 2020; Korkmaz and Balli 2021; Xue et al. 2022) compared connective tissue graft (CTG) with CGF, with a total sample size of 252 sites (128 CGF, 124 CTG). For CAL measurement, 2 studies (Akcan and Ünsal 2020; Korkmaz and Balli 2021) assessed 182 gingival recession sites (94 CGF, 88 CTG).
The meta-analysis evaluated 5 studies to compare the following outcomes: for PPD, 2 studies with 149 sites showed no statistically significant difference (mean: 0.11 mm; P = 0.18). Similarly, 3 studies with 252 sites found no significant advantage of CGF over CTG (mean: 0.07 mm; P = 0.19; Fig. 3A). Regarding CAL, 2 studies with 182 gingival recession sites favored CTG (mean: –0.38 mm; P = 0.0002), and 2 studies with 149 sites showed slight improvement with CAF + CGF over CAF alone (mean: 0.23 mm; P = 0.03; Fig. 3B). For keratinized gingival width (KGW), 2 studies with 149 sites showed a significant increase with CAF + CGF (mean: 0.41 mm; P < 0.0001), although the 3 studies found no statistically significant difference (mean: –0.89 mm; P = 0.29; Fig. 3C). For gingival thickness (GT), 2 studies reported gain with CAF + CGF (mean: 0.26 mm; P < 0.00001), while 3 studies favored CTG (mean: –0.50 mm; P < 0.0001; Fig. 3D). Finally, 3 studies indicated that CTG was more effective for mean root coverage (MRC), with a mean increase of 15.09% (P < 0.00001; Fig. 3E).
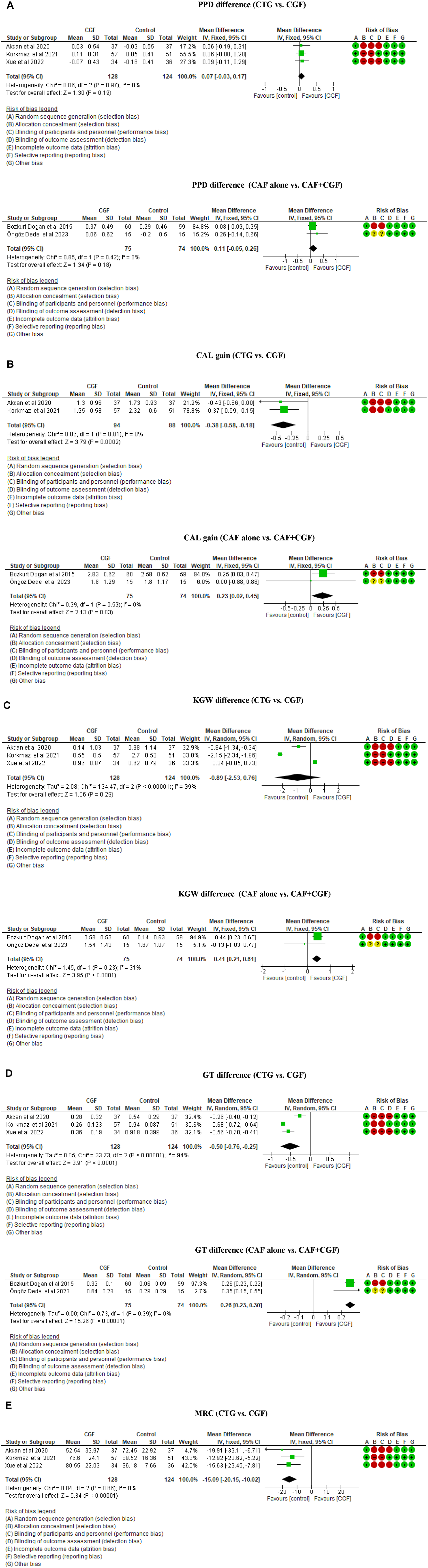
Forest plots of gingival recession studies. This figure presents forest plots of gingival recession studies showing clinical outcomes assessed in the meta-analysis: (
Guided Bone Regeneration
A single study (Aboelela et al. 2021) compared GBR using collagen membranes versus CGF membranes in 28 patients. Both groups used a mix of autogenous bone particles and Anorganic Bovine Bone Mineral (ABBM), with the experimental group adding autologous fibrin glue and CGF membranes. The study found no significant differences in graft consolidation or implant stability between the 2 groups.
Sinus Elevation
Studies on CGF in sinus elevation (Adalı et al. 2021; Ghasemirad et al. 2023), bone production and resorption (with Ghasemirad et al. 2023 also assessing connective tissue), and implant survival (Forabosco et al. 2018) did not meet meta-analysis criteria. Adalı et al. (2021) conducted a split-mouth randomized study with 10 patients, comparing allograft with CGF to allograft alone. After 6 months, cone beam computed tomography scans showed significantly lower bone height resorption in the test group compared to the control group. New bone formation was higher in the test group but not statistically significant. Ghasemirad et al. (2023) conducted a double-blind, split-mouth study on 18 maxillary sinuses. They found significantly higher bone formation in the CGF group than in the control group, but the amount of connective tissue was similar. Forabosco et al. (2018) evaluated 50 individuals undergoing maxillary sinus augmentation. The implant survival rate was almost the same in both the CGF and control groups. Using CGF improved predictability and reduced complications.
Alveolar Ridge Preservation
The studies differed in healing index (HI), soft tissue healing, and bone width. Three studies (Ma et al. 2021; Keranmu et al. 2022; Liu et al. 2022) evaluated ridge width at 6 mo. Keranmu et al. (2022) compared Deproteinized Bovine Bone Mineral (DBBM) + CGF to DBBM alone, Liu et al. (2022) compared DBBM + CGF to DBBM + BIOGIDE, and Ma et al. (2021) compared CGF to self-healing. Lin et al. (2021) studied groups after 8 mo. Keranmu et al. (2022) had 38 patients. Significant ridge height variations at 3 and 6 mo favored the experimental group. Ridge width differences at 1 mm and 2 mm were significant at 3 and 6 mo and at 3 mm after 3 mo. Gray value changes favored the experimental group at 3 months. Healing index was better at 1 wk, with less pain on the first day. Lin et al. (2021) compared CGF + DBBM to DBBM with a collagen membrane and self-healing control in 36 patients. After 8 mo, there were no significant differences in ridge area reductions, but the control group had significantly more resorption. CGF + DBBM showed significantly less residual material (P = 0.005) and more new bone (P = 0.031). Liu et al. (2022) studied 22 patients, finding no significant ridge width differences at 1 mm, 3 mm, and 5 mm after 6 mo. Soft tissue healing improved at 7 and 14 d, but keratinized gingiva width did not change. Ma et al. (2021) studied 46 patients, finding significant differences in ridge height and width at 1 mm and 2 mm after 3 mo. Significant HI variation occurred at 10 days. Micro–computed tomography (CT) showed significant differences in Bone Mineral Density (BMD), Bone Volume Fraction (BV/TV), Trabecular Number (Tb.N), Trabecular Separation (Tb.Sp), and Tb.Sp but not in trabecular thickness.
Dental Implants
This study examines CGF use in dental implants, highlighting differences in methods, sites, and follow-up. Alveolar bone levels, bone density, and Implant Stability Quotient (ISQ) varied, prohibiting meta-analysis and emphasizing the necessity for consistent methods.
Four studies (Gaur et al. 2022; Isler et al. 2018; Isler et al. 2022; Karthik et al. 2022) confirmed alveolar ridge preservation using radiographic assessments. Two studies (Isler et al. 2018; Isler et al. 2022) tracked vertical defect depth (VDD) reductions over 6, 12, and 36 mo in 52 patients. The CGF group showed a significant VDD decrease, while the CM group showed greater improvement in VDD and other clinical measures at 12 mo. Gaur et al. (2022) studied 36 patients, finding no significant improvement in bone gaps. Karthik et al. (2022) assessed 20 implants and found no significant changes in crestal bone levels over 6 mo. Methodological differences hinder meta-analysis.
Two studies checked bone density around dental implants. Inchingolo et al. (2017) observed significant bone quality improvement in CGF-treated patients, with better Area Under the Curve (AUC) values at 8 mo compared to the control group. Gaur et al. (2022) found no statistically significant differences in grayscale values among control, PRF, and CGF groups, despite initially higher values in PRF and CGF groups.
One study (Forabosco et al. 2018) reported survival rates in dental implant patients after sinus augmentation. The CGF + xenograft group had a higher survival rate, while the xenograft-only group had a slightly lower rate. Both groups showed similar outcomes with no statistical difference.
In ISQ, 2 studies reported changes. Gaur et al. (2022) found that ISQ increased in all therapy groups except the control. Significant improvements were seen in PRF and CGF by weeks 12/16 (P = 0.04, P = 0.02). Pirpir et al. (2017) observed a significant ISQ increase in the CGF group compared to a decrease in the control group (P < 0.001).
Healing after Teeth Extraction
Six studies (Özveri Koyuncu et al. 2020a; Özveri Koyuncu et al. 2020b; Torul et al. 2020; Elayah et al. 2022; Fang et al. 2022; Torul et al. 2023), assessed CGF’s short-term effects after tooth extraction. Five studies focused on mandibular third molar recovery, while Torul et al. (2023) examined alveolar osteitis treatment. CGF showed improved healing, reduced pain, and reduced swelling in 4 studies. Two studies (Torul et al. 2020; Fang et al. 2022) found no significant benefits for edema or trismus, as elaborated in Table 2.
Summary of Randomized Controlled Trials Investigating the Effects of CGF in Third Molar Extraction Procedures.
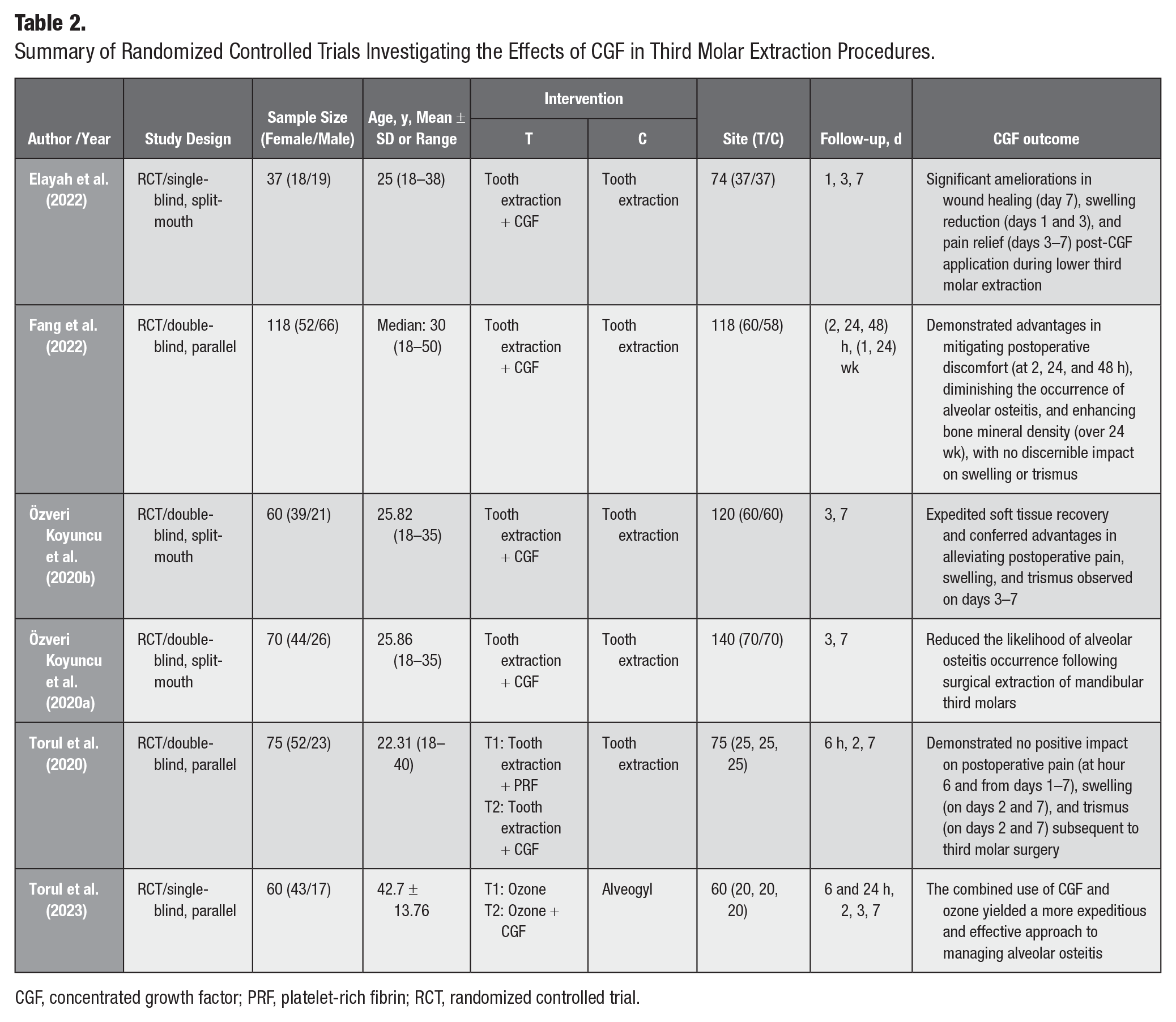
CGF, concentrated growth factor; PRF, platelet-rich fibrin; RCT, randomized controlled trial.
Discussion
This study conducted a systematic review and meta-analysis to evaluate the supplementary benefits of CGF in improving the healing of both hard and soft tissues in regenerative dentistry. By classifying the research into different groups according to regeneration interventions, it was possible to decrease the variability between trials, therefore making the meta-analysis easier. However, a meta-analysis was possible for periodontal intrabony defects and gingival recession due to the homogeneity found in a limited number of studies. However, the presence of substantial variation in other oral surgery groups made it impossible to carry out a meta-analysis. This systematic review emphasizes the potential beneficial effects of CGF in various dental treatments such as GBR, sinus elevation, alveolar ridge preservation, dental implant–related procedures, and postoperative healing of tooth extractions. However, it is crucial to acknowledge that there are currently insufficient meta-analytic data to statistically validate these findings. While CGF shows promise, limited meta-analysis data highlight the need for further empirical inquiry.
Treating periodontitis-related intrabony defects is challenging. CGF promotes osteoblast and gingival fibroblast growth more effectively than second-generation PRF. However, some studies question its impact on periodontal tissue healing (Isler et al. 2018). This meta-analysis compares CGF to grafting materials for periodontal intrabony defects, focusing on 12-mo outcomes in PPD, CAL, and IDD.
Meta-analysis of 151 defects in 85 patients showed no statistically significant difference in PPD reduction between groups, but CGF + bone graft had slight advantages in CAL and IDD reduction. No histological evidence supports full periodontal regeneration using CGF. Yao et al. (2023) reviewed 8 RCTs, finding significant improvements in PPD, CAL, and bone fill (BF) with CGF, although moderate RCT quality calls for cautious interpretation. Clinically, CGF is used as a membrane or mixed with grafts to enhance tissue regeneration. Further research is needed to confirm CGF’s efficacy and differentiate between histological regeneration and repair. Limitations include patient costs and variability in study designs and follow-up durations. Including CGF in gingival recession surgeries, like the CAF procedure, has increased. CGF is believed to enhance healing and reduce infection risk, but evidence of its efficacy remains limited. This meta-analysis evaluates CGF’s clinical impact when combined with CAF or tunneling techniques for Miller class I and II or Cairo class RT1 gingival recessions. Analysis of 6-mo studies on PPD, CAL, KGW, GT, and MRC showed similar PPD and KGW compared to CTG, but CTG offered better CAL, GT, and MRC improvements. It is noted that there are mild benefits in PPD, KGW, and GT with CGF and CAF. Li et al. (2019) found that APCs with CAF, especially PRF, improved root coverage, recession depth, CAL, KTW, and GT. Despite limitations like outcome variability and study bias, CGF with CAF shows potential benefits for CAL, KTW, and GT. Future high-quality, unbiased research is needed to validate these findings and refine clinical guidelines. The previous meta-analysis plots indicate that overall, the improvements in the measured outcomes were approximately half of those achieved with CTG. Additionally, the rapid degradation of CGF resulted in it lasting no more than 14 d. It has been confirmed that the concentrated PRF (C-PRF) protocol involves targeted harvesting of 0.3 to 0.5 mL of the buffy coat layer of L-PRF to achieve a 10-fold increase in platelet and leukocyte concentrations. These improvements enhance PRF’s regenerative efficacy, especially for applications requiring high concentrations of cells and growth factors (Miron et al. 2020). Therefore, advancements in CGF technology, such as the development of albumin-CGF with a concentration of 0.5 mL of the buffy coat layer, are recommended to enhance outcomes and reduce patient morbidity. A systematic review highlighted that albumin preparation from the PPP layer through heating, requiring approximately 10 min, enables the production of Extended Platelet-Rich Fibrin (e-PRF), which offers extended resorption properties, serving as a promising replacement for collagen membranes or as a biological filler (Miron et al. 2024).
Moreover, findings underscore the benefits of horizontal PRF (H-PRF), which achieves higher yields and more even distribution of platelets and leukocytes in PRF matrices. Unlike fixed-angle centrifuges that cause angled separations and compact cells along tube walls, horizontal centrifugation permits a direct flow of cells, reducing cellular trauma and improving layer uniformity. This method also increases the Relative Centrifugal Force (RCF) differential across the tube, facilitating better separation based on cell density (Miron et al. 2019). So, including albumin with H-PRF is recommended as an advanced technique.
Furthermore, the crucial impact of tube materials on PRF quality was emphasized, especially when additives like silica and silicone are present. Although these substances aid coagulation, they introduce risks of cytotoxicity and cellular apoptosis. Silica-containing plastic tubes, in particular, release particles that disrupt cell growth within PRF matrices. This finding highlights the importance of using plain, additive-free glass tubes, which yield safer and larger PRF clots, thereby promoting superior outcomes in clinical applications (Miron et al. 2021). So, checking the tube type in CGF preparation is recommended to optimize the outcomes.
One RCT found CGF beneficial in ridge augmentation. However, CGF membranes, with a short resorption period of less than 2 wk, are not reliable barriers to GBR (Aboelela et al. 2021). The study highlighted the need for more high-quality, large-scale RCTs and research on advanced CGF types, like albumin-enriched CGF, for extended periods. This study corroborates Miron’s systematic review of PRF in regenerative dentistry, affirming its efficacy in soft tissue healing. However, robust evidence for PRF’s role in bone repair is lacking, highlighting the need for further human randomized clinical trials on its effects on bone formation. Clinically, while PRF enhances soft tissue regeneration and prevents postextraction dimensional changes, limited evidence supports its use in GBR (Miron et al. 2017).
The use of CGF for sinus elevation is growing due to its high success rates. Studies (Adalı et al. 2021; Forabosco et al. 2018; Ghasemirad et al. 2023) compared CGF with allograft or xenograft versus graft alone, assessing bone height (BH), new bone formation (NB), residual material (RM), resorption percentage, connective tissue (CT), and survival rate. Adalı et al. (2021) found CGF improved BH and reduced resorption, Ghasemirad et al. (2023) noted better NB and RM with CGF and xenograft, and Forabosco et al. (2018) found no significant difference in survival rates. Some researchers argue that CGF alone could be effective for sinus lift cases (Sohn et al. 2011), although limitations in control and research persist. Comparisons with PRF (Zhang et al. 2012) showed increased bone volume but no significant differences. Miron et al. (2017) noted the need for better trials, but CGF’s simplicity, low cost, and success rates suggest it may be effective for sinus lifts.
Research on alveolar bone changes after tooth extraction aims to address impacts from periodontal ligament loss. The resorption, evident within 8 wk postextraction, results from reduced blood flow. This systematic review explored CGF’s role in preventing bone height and width. While CGF effectively maintained bone dimensions and early healing advantages (Ma et al. 2021; Keranmu et al. 2022; Liu et al. 2022), these effects were not sustained long term. PRF studies also support CGF’s potential to reduce dimensional changes and infection rates, enhance bone healing, and maintain ridge quality (Hauser et al. 2013). Despite promising results, further high-quality studies are needed to confirm CGF’s benefits in various clinical scenarios.
Research on CGF in implant dentistry indicated potential benefits for enhancing osseointegration, being economical, and promoting wound healing. Four studies assessed CGF’s impact on alveolar bone levels around implants. Two studies (Gaur et al. 2022; Karthik et al. 2022) found no significant differences in bone levels between CGF-treated and control groups. In contrast, 2 studies (Isler et al. 2018; Isler et al. 2022) observed no initial differences between CGF and collagen for GBR but noted that collagen outperformed CGF after 3 y. Conversely, 3 studies (Sohn et al. 2009; Sohn et al. 2011; Kim et al. 2012) demonstrated that CGF facilitated rapid bone regeneration in sinus augmentation procedures, while Manoj et al. (2018) reported improvements in buccal bone width and vertical bone growth with CGF.
Regarding bone density, 2 studies (Inchingolo et al. 2017; Karthik et al. 2022) found significant bone density elevation with CGF, while Gaur et al. (2022) observed no significant differences after 8 wk. Kim et al. (2014) noted that CGF improved bone quality compared to control. For implant survival rates, Forabosco et al. (2018) found comparable rates for CGF and control, and Masuki et al. (2016) showed that PRF and CGF promote periosteal cell proliferation, acting as growth factor reservoirs. ISQ measures implant stability, with a value below 49 indicating loading should be avoided. Pirpir et al. (2017) reported increased ISQ values with CGF at 1 and 4 wk, suggesting enhanced osseointegration, although Gaur et al. (2022) found minimal improvement at 4 and 8 wk. Dohan Ehrenfest et al. (2009) proposed that L-PRF might enhance wound healing and early implant stability. Overall, further rigorous, large-scale studies are needed.
Following third molar extraction, discomfort such as edema, trismus, and pain is common. PRF has been shown to reduce these symptoms (Uyanık et al. 2015). Advanced platelet-rich fibrin (A-PRF) and CGF enhance PRF’s benefits further, with A-PRF increasing white blood cells and softening fibrin clots and CGF offering higher platelet concentration and firmer fibrin structure (Masuki et al. 2016).
Despite advancements, research on CGF’s effects on postoperative symptoms after lower third molar surgery is limited. Six RCTs on CGF showed mixed results in reducing discomfort and complications. Similarly, PRF studies have inconsistent findings, with some showing reduced swelling and pain and others finding no significant benefits due to varying g-force reporting at different tube positions depending on the inaccurate setting of rotation per minute. It was advocated to standardize g-force reporting at RCF-max to improve accuracy (Miron et al. 2018). To ensure consistency, CGF was prepared using the Medifuge MF200 with controlled angulation, time intervals, and radius, but tube types were not specified.
Bias and varying outcome measurements affect reliability. Future research should involve larger, well-designed trials better to assess CGF’s effectiveness in postoperative recovery.
Limitations and Future Suggestions
This review examined 30 RCTs. Appendix Figure 4 summarizes the 30 trials’ bias evaluation. One RCT had a low bias risk, 5 had an unclear risk, and 24 had a high risk, according to the Cochrane Collaboration. Performance bias, attrition bias, detection bias, and allocation concealment indicated selection bias. Performance bias was equivocal or elevated in over 75% of trials, and allocation concealment was similar. Over 40% of studies had an uncertain or considerable risk of detection bias, and over 50% had a high risk of attrition bias.
To increase research quality, future studies should follow strict protocols for patient selection, randomization, allocation concealment, participant and assessor blinding, and outcome measurement. Due to restricted RCTs, inconsistent outcome assessments, and insufficient data, the meta-analysis omitted implant-related diseases, alveolar ridge preservation, sinus elevation, guided bone regeneration, and postoperative recovery.
Comprehensively assessing CGF’s significance in oral surgery requires well-designed trials, and CGF and CTG need to be compared in clinical settings in future studies. CGF with albumin to withstand quick degradation may reduce morbidity and simplify procedures compared to CTG. These discoveries will improve periodontal care by updating clinical standards.
Conclusions
This systematic review and meta-analysis examines the role of CGF in oral regenerative applications, although the limited quality of available evidence constrains the findings. Key areas identified are as follows.
In treating intrabony defects, CGF shows promise in enhancing bone graft outcomes and supporting periodontal tissue repair. Adding CGF to CAF demonstrates improved coverage for gingival recession, although it remains less effective than the established combination of CTG and CAF. High-quality studies are required to substantiate these findings for intrabony defects and recession management.
CGF appears beneficial for postextraction recovery, including pain reduction, ridge preservation, and sinus augmentation; however, heterogeneity among studies prevents meta-analysis. CGF may contribute to increased gingival thickness in soft tissue applications, although its reliability as a barrier membrane compared to collagen in GTR and GBR is variable.
For bone height preservation and implant success, CGF exhibits potential; nonetheless, it is not recognized as an equivalent to collagen membranes, with insufficient meta-analytic support.
The field needs methodologically rigorous studies with minimized bias to delineate CGF’s precise utility in regenerative dentistry. Standardized protocols and outcome metrics are essential to facilitate consistent assessment across applications. Future investigations should prioritize larger, well-defined samples to generate robust evidence of CGF’s clinical relevance.
Author Contributions
A.A. Alshirah, contributed to conception, design, data analysis, drafted and critically revised the manuscript; M.H. Elnaem, Z. Al-Ani, M. Almasri, and P.A. McCarron, contributed to conception, design, critically revised the manuscript; F. Alzahrani, contributed to analysis, critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of the work.
Supplemental Material
sj-docx-1-jct-10.1177_23800844251325532 – Supplemental material for Effect of Autologous Concentrated Growth Factor in Regenerative Dentistry: A Systematic Review and Meta-Analysis
Supplemental material, sj-docx-1-jct-10.1177_23800844251325532 for Effect of Autologous Concentrated Growth Factor in Regenerative Dentistry: A Systematic Review and Meta-Analysis by A.A. Alshirah, M.H. Elnaem, Z. Al-Ani, F. Alzahrani, M. Almasri and P.A. McCarron in JDR Clinical & Translational Research
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Supported by Ulster University, School of Pharmacy and Pharmaceutical Sciences and College of Medicine & Dentistry.
A supplemental appendix to this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
