Abstract
Introduction:
Cost-utility analysis (CUA)—a method to evaluate intervention cost-effectiveness—transforms benefits of alternatives into a measure of quantity and quality of life, such as quality-adjusted life year (QALY), to enable comparison across heterogeneous programs. Measurement challenges prevent directly estimating utilities and calculating QALYs for caries in primary dentition. Proxy disease QALYs are often used as a substitute; however, there lacks quantitative evidence that these proxy diseases are comparable to caries.
Objective:
To employ a discrete choice experiment (DCE) to quantitatively determine the most comparable proxy disease for different levels of caries in primary dentition.
Methods:
A cross-sectional DCE survey was administered to respondents (N = 461) who resided in California, were aged ≥18 y, and were primary caretakers for ≥1 child aged 3 to 12 y. Four attributes were included: pain level, disease duration, treatment cost, and family life impacts. Mixed effects logistic regression and conditional logistic regression were used to analyze the survey data.
Results:
Respondents from the overall sample preferred no pain over mild (odds ratio [OR] = 0.50, P < 0.05), moderate (OR = 0.57, P < 0.05), and severe pain (OR = 0.48, P < 0.05). Acute gastritis (OR = 0.44, P < 0.05), chronic gastritis (OR = 0.31, P < 0.01), and cold sore (OR = 0.38, P < 0.05) were less preferred than stage 1 caries. Acute tonsilitis (OR = 0.43, P < 0.05), acute gastritis (OR = 0.38, P < 0.05), chronic gastritis (OR = 0.26, P < 0.01), and cold sore (OR = 0.33, P < 0.01) were less preferred than stage 2 caries. Chronic gastritis (OR = 0.42,P < 0.05) was less preferred than stage 4 caries.
Conclusions:
Parents viewed the characteristics of many diseases with similar QALYs differently. Findings suggest that otitis media and its QALY—as commonly used in CUAs—may be a suitable proxy disease and substitute. However, other disease states with slightly different QALYs may be suitable. As such, the recommendation is to consider a range of proxy diseases and their QALYs when conducting a CUA for child caries interventions.
Knowledge Transfer Statement:
This study reviews and systematically compares pediatric diseases that are comparable to caries in primary dentition. The findings may inform future research using cost-utility analysis to examine the incremental cost-effectiveness ratio of interventions to prevent and treat caries as compared with an alternative.
Keywords
Introduction
Childhood caries is a highly preventable disease yet remains the most common chronic disease in children (Dye et al. 2007; National Institute of Dental and Craniofacial Research 2021). To reduce childhood caries prevalence, it is important to evaluate not only the effect but also the intervention costs to prevent and treat childhood caries, especially caries in primary dentition. Cost-effectiveness analysis (CEA) and cost-utility analysis (CUA) are 2 common methodologies for determining intervention cost-effectiveness (Stone et al. 2002). CEA measures incremental costs of alternatives relative to incremental benefits, measuring benefits with a single consistent effectiveness metric, such as decrease in the number of people with caries. CUA, which is a specific type of CEA, transforms benefits of alternatives into a measure of the quantity and quality of life, such as quality-adjusted life year (QALY) or disability-adjusted life year. These measures allow comparison across heterogeneous programs and technologies.
QALY is a widely accepted measure for disease burden and is obtained through multiplying the utility value associated with a given state of health by the years lived in that state (Drummond et al. 2015). This information is collected typically through surveying respondents with a questionnaire. For example, the utility estimates can be collected with a standard gamble or time trade-off (TTO) method (Drummond et al. 2015), which asks respondents to indicate how much time they would be willing to spend in a hypothetical health state. The TTO questions vary the time spent in the specific health state until respondents are indifferent.
Methodologies such as TTO provide valuable information for adult health states; however, these methods are less applicable for collecting information for pediatric health states. A child’s ability to complete a long survey—specifically, one that requires the child to imagine hypothetical situations and report a preference for time spent in situations—is hampered by cognitive and age limitations. As such, various approaches to pediatric health state valuation were considered to circumvent the challenge. One approach is to ask adult respondents to imagine that they are children and to answer survey questions accordingly (e.g., for a TTO survey). Another approach is to use the adapted instrument for children, Child Health Utility (Stevens 2012; Koh et al. 2015; Petersen et al. 2019), and survey children repeatedly for a duration of time.
While these novel approaches provide usable measurements for pediatric health states, the measurement challenges and threats to validity persist (Ungar 2011). To date, there are no published studies that provide directly calculated QALYs for caries in primary dentition; QALYs for caries are derived from the QALYs of proxy diseases. That is, the literature relies on utility estimates and resulting QALYs from heuristic arguments (e.g., Espinoza-Espinoza et al. 2019) or childhood diseases similar to caries in terms of duration, pain, and impacts, such as otitis media (e.g., Kay et al. 2018), when conducting a CUA for caries intervention for primary dentition (Oh et al. 1996).
An accurate QALY is critical for CUAs. Nevertheless, quantitative evidence is lacking to support that QALYs for otitis media and other similar disease states can be comparable to tooth decay or a filling. Furthermore, whether a disease is a suitable proxy for various severity levels of caries has yet to be evaluated quantitatively.
The objective of this study was to employ a discrete choice experiment (DCE) to identify comparable diseases that can provide QALYs for different levels of caries in primary dentition. The DCE survey design allows for eliciting relative preferences in the absence of directly revealed preference data and analyzing the collected data quantitatively.
Methods
Discrete Choice Experiment
We conducted a DCE to determine quantitatively more appropriate proxies—otitis media or other diseases—for caries of different stages. DCE is a widely employed, robust methodology for evaluating patient preferences in health care decision making (Ryan and Farrar 2000; Hauber et al. 2016; Soekhai et al. 2019). The literature suggests that DCEs are a valuable tool to quantify preferences related to a health care priority setting. DCE involves 1) identification of attributes and attribute levels, 2) experimental design of the choice sets (typically as pairs), 3) survey development, 4) sample selection and survey administration, and 5) data analysis.
Utilizing the features of DCE to evaluate proxy diseases is an innovative approach to quantitatively determine disease comparability. In this case, the DCE quantified the relative preferences and risk acceptance for disease state attributes (i.e., pain level, duration, treatment cost, and impact on family life) and systematically elicited risk benefit trade-offs. The DCE allowed for analyzing characteristics of disease conditions and provided information on which characteristics parents prioritized (i.e., valued). Furthermore, the responses allowed the study to compare disease states and identify which were more similar to each other.
Understanding and weighing disease state options with varied risks and benefits can be understandably difficult for parents, particularly when the disease process and treatment options may affect the quality of life of their children. In this study, we hypothesized that the process of the DCE would decrease respondents’ decisional conflict through presenting repeated disease profiles and soliciting implicit preferences.
DCE Survey Instrument
We employed a DCE to evaluate 5 stages of caries and 15 pediatric disease states. The caries severity levels for comparison were denoted as follows:
Stage 1: white spots
Stage 2: enamel decay and dentin decay
Stage 3: pulp involvement
Stage 4: abscess formation, allergic rhinitis, wrist fracture, acute gastritis, chronic gastritis, conjunctivitis, epilepsy, lost thumb, cold, cold sore
Stage 5: tooth loss. The noncaries health conditions were: acute otitis media, chronic otitis media, acute tonsilitis, chronic tonsilitis, acute eczema, chronic eczema
The 15 diseases were identified through the study team conducting a literature review. The review identified 2 studies (Janssen et al. 2008; Salomon et al. 2012) that outlined diseases with a similar QALY (within 0.03 range) to otitis media: a commonly used proxy disease for caries. Experts reviewed these diseases and selected those that are relatively comparable to caries as the main source of proxy diseases. Given the study’s aim to identify the most suitable proxy disease for different stages of caries, we selected the following attributes and levels for comparison by identifying the major differences among these disease states through expert review and feedback: pain level (none, mild, moderate, severe), disease state duration (≤1 wk, 1 mo, 4 mo, 1 y), treatment cost (≤$100, $1,000, $10,000), and impact on family life (≤28 d, >28 d). Impact on family is characterized by disruption in normal work and sleep schedule due to the need to care for a child experiencing the disease. Respondents were probed on these elements during the cognitive interview stage, and most interviewees (all of whom were parents with children aged <12 y) reported that these elements captured the top concerns regarding the quality of family life. While many potential attributes were considered for inclusion, we chose these 4 attributes to 1) allow for evaluation of the differences among these diseases and 2) ensure that we captured the characteristics of disease comprehensively while not overburdening respondents with the number of attributes and levels.
Survey Design
After respondents signed the electronic consent form, they were presented first with 10 sociodemographic questions on their age, gender, education, income, number of children, age of children, residential setting (urban, suburban, rural), household size, and whether they were participants of the Special Supplemental Nutrition Program for WIC (Women, Infants, and Children) or the Early Head Start program. We stipulated a logical branch in the survey platform to automatically calculate whether the respondent’s household was below or above the federal poverty level (FPL) based on household size and income. Next, respondents were presented with a PDF educational pamphlet (Appendix 1) with a picture and an explanation of each disease state; information included disease hallmarks, characteristics, and symptoms. Respondents were encouraged to download the educational pamphlet for questions regarding any of the disease states. Respondents were then presented with 18 DCE choice sets (pairs of disease states). Each choice set was accompanied by the following survey question: “Which of the 2 health problems do you think is worse for your child?” Each question was followed by a different combination of 1 randomly selected caries severity level and 1 randomly selected proxy disease state or caries severity level. The reason for asking respondents to compare 1 caries severity level with another was to evaluate and ensure consistency of selecting caries severity level. In addition, we included 2 test questions that asked respondents to compare caries with a very high-severity condition (e.g., brain tumor) and a very low-severity condition (e.g., a sneeze); these questions allowed the study team to ensure that respondents were answering logically and that the data remained logically consistent. In cases of illogical responses, study protocol dictated dropping those respondents from the analysis. In our sample, no respondents answered illogically (i.e., that low-severity caries is worse than a brain tumor). Each respondent was presented with a randomly selected subset of 10 possible choice sets to compare. Different profile combinations were presented to ensure that all comparisons were being offered to enough respondents without each respondent being overwhelmed by all possible profile combinations.
The survey was available in English and Spanish. Cognitive interviews with 12 respondents were conducted in English. During the cognitive interviews, many respondents commented on how the survey made them feel worried about their children’s health and how it was emotional because it prompted them to imagine their children being sick. As such, changes in survey wording were made in English and Spanish to acknowledge that it may be difficult to imagine their children suffering from an illness, to highlight that the survey presented only hypothetical scenarios, and to restate that it is okay for respondents to discontinue the survey at any time. Prior to launching the survey, pilot testing was performed with 6 respondents who completed the survey in English and 5 who completed it in Spanish. No changes were made to the survey after pilot testing, as respondents did not report concerns and there were no notable issues with it.
Study Setting and Population
The cross-sectional anonymous DCE survey was administered online to a validated Qualtrics panel; respondents and their sociodemographic information were verified by Qualtrics. These respondents also consented to participating in survey research via Qualtrics. The survey was constructed on Qualtrics, and a pilot study was conducted and followed by a soft survey launch before the full survey launched. Recruitment occurred during a 45-day period between December 1, 2020, and January 14, 2021. Individuals who 1) were vetted by Qualtrics and included in the Qualtrics panel, 2) resided in California, and 3) were at least 18 y old and primary caretakers to at least 1 child between 3 and 12 y old were eligible to participate in the survey. We specified the recruitment to include a sample representative of California’s racial distribution. To inform disparity in health care, we also specified that the sample comprise a sufficient number of people who were below the FPL for household income for a subgroup analysis on their preferences. Individuals who met the inclusion criteria were sent an electronic informed consent form prior to being presented the main survey.
Ethics
The study and research design received an exempt status from the University of California, San Francisco, Institutional Review Board and the University of California, Los Angeles, Institutional Review Board. Adhering to global protocol (Campus et al. 2020), all eligible individuals received an informed consent question embedded on the first page of the survey, and they could deny or approve being involved in the study. Once they agreed to the informed consent, the main survey launched.
Sample Size Calculation
We employed the sample size formula for DCEs suggested by de Bekker-Grob et al. (2012): N > 500c/(t × a), where t is the number of “choice tasks,” or sets of alternative combinations of disease attributes with specified levels of each attribute; a is the number of alternative scenarios per choice task; and c is the largest product of levels for any 2 attributes, which determines the difficulty of estimating interactions between attributes. We specified that t = 18 to keep the number of questions manageable. We also specified that a = 2, a common number of scenarios per task used in prior research (Kruk et al. 2009; Abiiro et al. 2014; Allaire et al. 2016). Finally, we specified that c = 16 (4 × 4), assuming that the largest number of levels will be 4. Based on these specifications and by rounding up to a whole number, the minimum sample size for the DCE was 223. This study included 461 respondents overall and 223 from households below the FPL for income for the subgroup analysis that focused on families with fewer resources.
Statistical Analysis
To evaluate the attributes and determine which ones parents prioritized (i.e., valued more than others), we analyzed the collected DCE survey responses using a mixed effects logistic regression model (StataCorp 2013; Wilson et al. 2017). The model was chosen to account for the inter- and intraperson variability due to each respondent answering 18 multiple-choice pairs. We included 3 mixed effects logistic regression models: the overall sample, a subsample of respondents from households with income below the FPL, and a subsample of respondents from households with income above the FPL.
We analyzed the data using a conditional logistic regression model (Aizaki and Nishimura 2008; Hauber et al. 2016). This analysis provided the odds ratio (OR) of each caries stage as compared with other stages and all 15 noncaries disease conditions. The results allowed for quantitative evaluation of all caries stages and disease states and which disease states were more similar to each level of caries severity. Additionally, linear and nonlinear (quadratic, cubic, quartic) trends for preferences among the 5 caries stages were assessed to confirm that parents perceived the 5 stages as monotonically decreasing with increasing severity.
Results
Descriptive statistics are presented in Table 1. Our sample included 461 respondents; 10 respondents completed the survey in Spanish. The median age was 35 y (IQR, 28 to 41). In the sample, 41.9% identified as male, 57.7% as female, and <0.4% as other. Those who identified as White accounted for 59.7% of the sample, followed by 15.6% Asian, 10.6% other, 9.8% Black or African American, 3.7% American Indian or Alaskan Native, and 0.7% Native Hawaiian or Pacific Islander. Most respondents’ highest education was high school diploma or less (39.5%), followed by bachelor degree (18.7%), graduate degree (17.6%), or some college (6.3%); 18% did not report their education. Most resided in urban settings (60.0%), with 33.5% in suburban settings and 6.7% in rural settings.
Sociodemographic Characteristics of Respondents (N = 461).
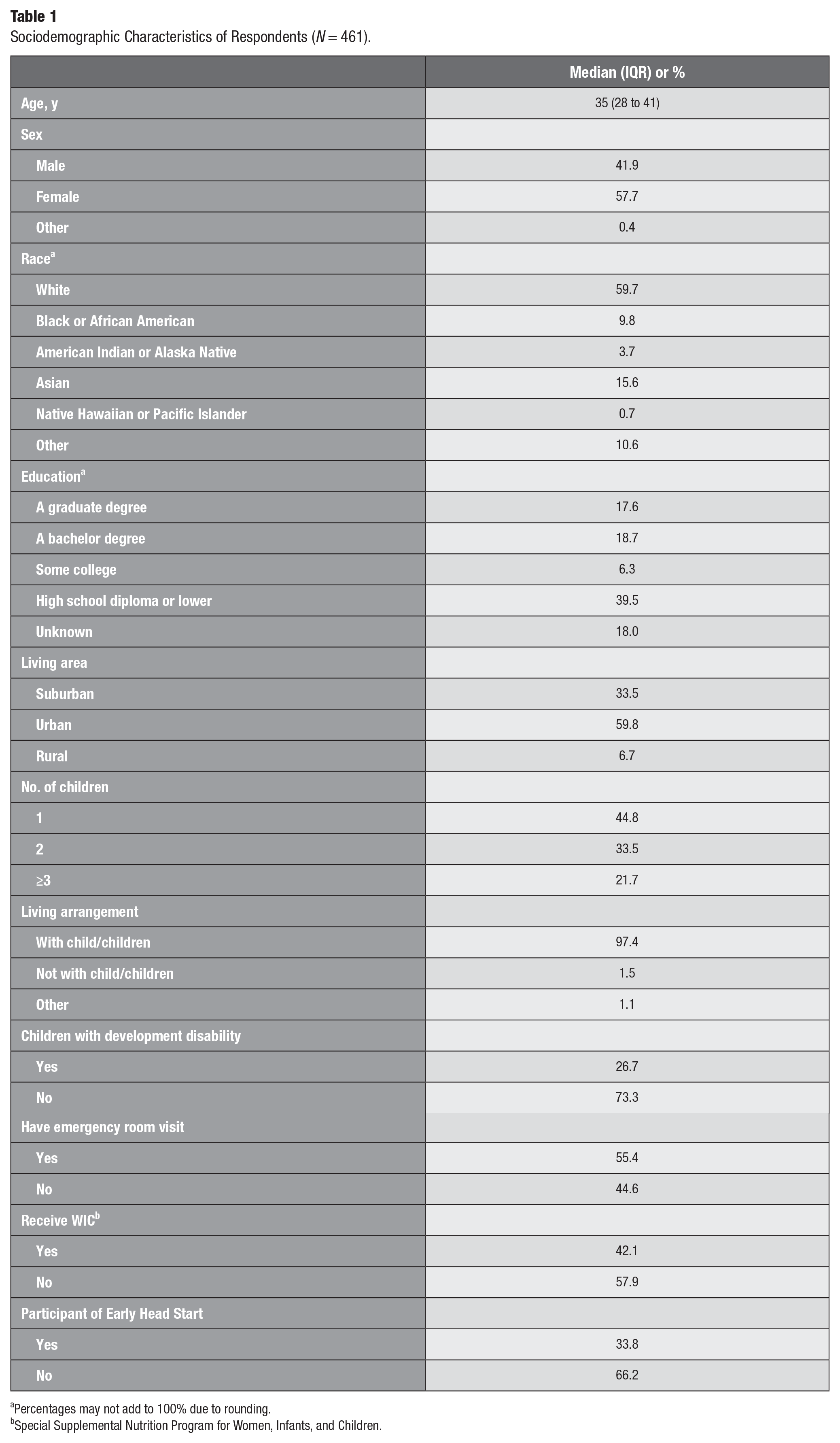
Percentages may not add to 100% due to rounding.
Special Supplemental Nutrition Program for Women, Infants, and Children.
All respondents were not only parents but also primary caretakers: 44.8% for 1 child, 33.5% for 2 children, and 21.7% for ≥3 children. Most (97.4%) lived with their children. Parents reported the following about their children: 26.7% had developmental disabilities; 55.4% had been to an emergency room for care; 42.1% were WIC participants; and 33.8% were Early Head Start participants.
Results from the mixed effects logistic regression model are presented in Table 2. Model 1 analyzed the overall sample (N = 461), finding that respondents prefer no pain over mild (OR = 0.50, P < 0.05), moderate (OR = 0.57, P < 0.05), and severe pain (OR = 0.48, P < 0.05). Preferences regarding duration of disease state, cost, and impact of family were not statistically significant in the overall sample.
Mixed Effects Logistic Regression Results for the Overall Sample and Income-Based Subsamples.
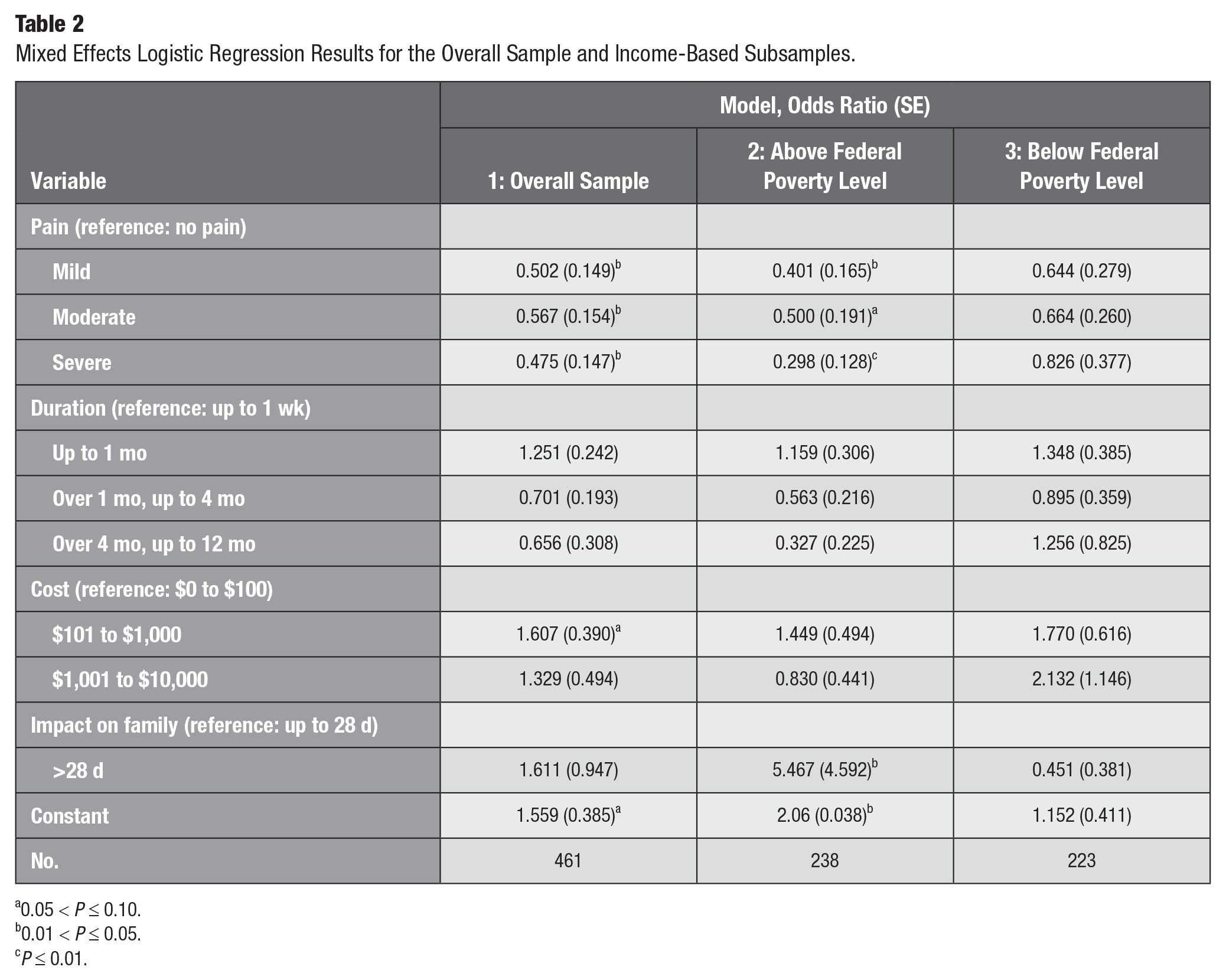
0.05 < P ≤ 0.10.
0.01 < P ≤ 0.05.
P ≤ 0.01.
To examine health care disparities, we conducted subanalyses. We analyzed separate samples of parents from households above and below the FPL. Model 2 focused on a subsample of respondents (n = 238) from households above the FPL, finding that they preferred no pain over mild (OR = 0.40, P < 0.05) and severe pain (OR = 0.30, P < 0.01). Unlike the overall sample, these respondents preferred >28 family impact days (OR = 5.47, P < 0.05) over ≤28 family impact days. Preferences regarding disease state duration and cost were not statistically significant. Model 3 focused on the subsample (n = 223) from households below the FPL, finding no statistically significant preferences among attributes.
Results from the conditional logistic regression models are presented in Table 3. Model 1 indicated that acute gastritis (OR = 0.44, P < 0.05), chronic gastritis (OR = 0.31, P < 0.01), and cold sore (OR = 0.38, P < 0.05) were statistically significant and less preferred than stage 1 caries. Model 2 revealed that acute tonsilitis (OR = 0.43, P < 0.05), acute gastritis (OR = 0.38, P < 0.05), chronic gastritis (OR = 0.26, P < 0.01), and cold sore (OR = 0.33, P < 0.01) were less preferred than stage 2 caries. Model 3 indicated no statistical significance among all 15 disease states and comparator caries stages. Model 4 indicated that chronic gastritis (OR = 0.42, P < 0.05) was the only disease state less preferred than stage 4 caries. Model 5 indicated that no disease state had a significant OR when compared with stage 5 caries.
Conditional Logistic Regression Results (N = 461).
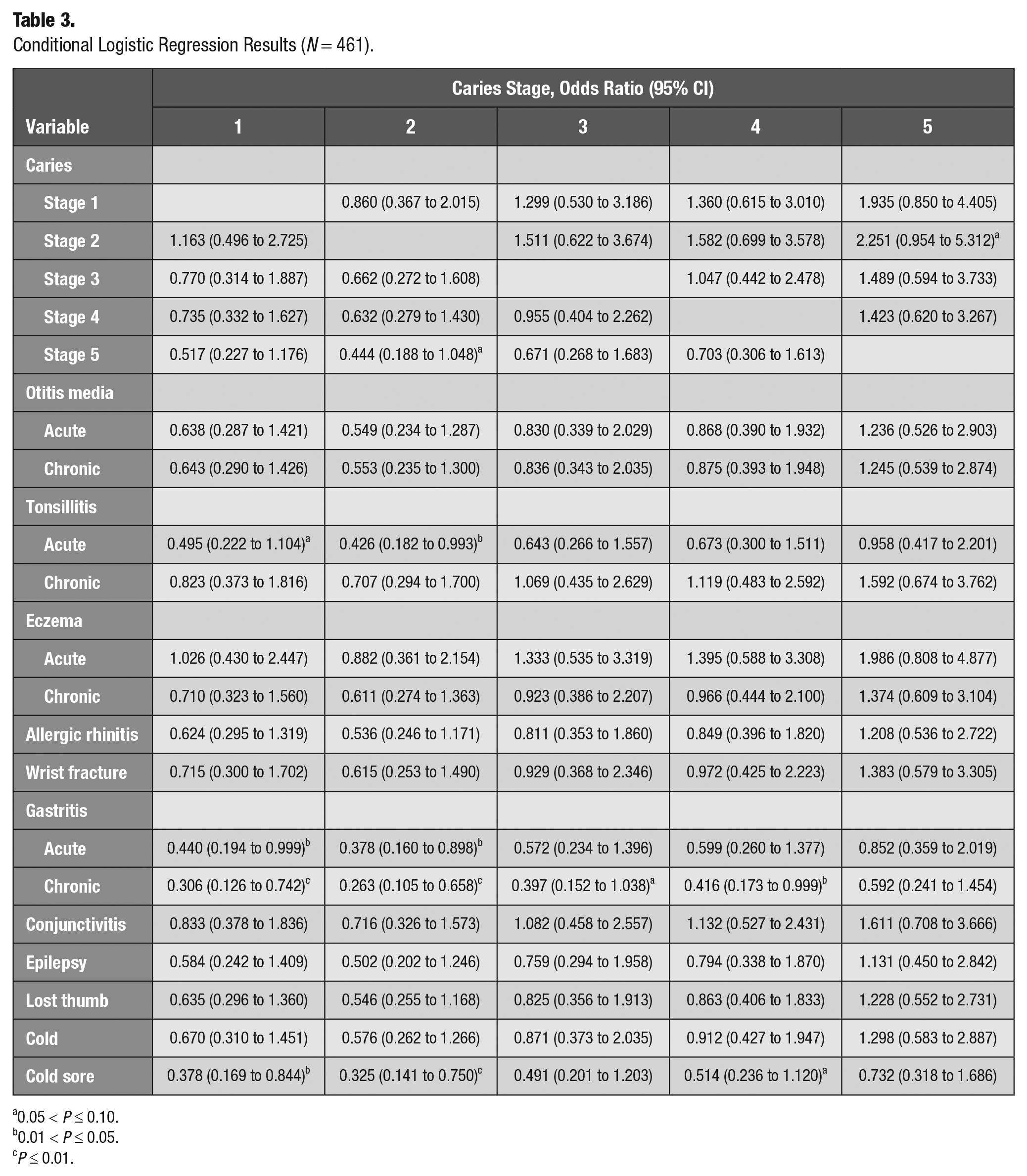
0.05 < P ≤ 0.10.
0.01 < P ≤ 0.05.
P ≤ 0.01.
We tested the linear trend for caries stages 1 to 5, which was statistically significant. Quadratic, cubic, and quartic effects were not significant. This finding suggests that parents perceived increasing caries severity with linear decreasing preference, which provides a face validity confirmation.
In all 5 models, no caries stage resulted in a statistically significant odd ratio. Though, notably, the ORs were logically consistent for caries stages 3 to 5, where respondents preferred stage 5 less than stage 4 and stage 4 less than stage 3. The ORs for stage 1 and 2 caries were inconsistent in models 1, 3, 4, and 5.
Discussion
This study’s findings suggest that pain level is the main attribute that parents value and prioritize when comparing disease states. In overall sample analyses, the other levels in each attribute—which included disease state duration, treatment cost, and days of impact on family—were not driving parents’ choices when selecting a disease from each choice set. This result is not surprising. Parents are risk adverse when it comes to their children’s health and well-being; thus, pain being the sole attribute reaching statistical significance is reasonable. Notably, the ORs for mild and moderate pain were logically inconsistent: parents were 50% less likely to choose mild pain but only 43% less likely to choose moderate pain. We reason that this pattern may be from respondents having difficulty clearly delineating between mild and moderate pain.
To inform disparity in health care, we conducted subanalyses examining preference differences of parents from households above and below the FPL for income. Similar to the overall sample, respondents from households above the FPL preferred no pain over mild and severe pain. Contrary to the overall sample, respondents from the above-FPL subsample preferred a disease state that exerted a longer impact on family life. This counterintuitive finding may be attributed to families with higher incomes being able to absorb a disease state’s impact on family life more easily.
The subanalysis on respondents from households below the FPL showed no clear preference pattern. This finding suggests that other characteristics and concerns drove the preferences of respondents from lower-income households. In summary, the subsample analysis adds to the evidence that individuals from different economic backgrounds may have different preferences. Future research should include more detailed analysis on the impact of individuals’ financial situations on their preferences for the health care of their children.
The conditional logistic regression models evaluated how differently respondents viewed the 5 caries severity levels and 15 noncaries childhood health states in our study. In these models, a statistically significant OR suggested that the disease state significantly differed from the comparator (i.e., 1 of the caries stages). A disease state with an OR not significantly different from another disease was consistent with the disease state being a suitable proxy disease, with known utility estimates, and could therefore have its QALY used as a substitute for caries. We found that acute gastritis, chronic gastritis, and cold sore were significantly less preferred than stage 1 caries; thus, these 3 disease states appear to be unsuitable proxies for stage 1 caries. Acute tonsilitis, acute gastritis, chronic gastritis, and cold sore were significantly less preferred than stage 2 caries, suggesting that they should not be used as proxies for stage 2 caries. None of the 15 diseases had statistically significant ORs versus stage 3 caries, thus suggesting that they may be suitable proxies for stage 3 caries. Chronic gastritis was the only disease state significantly different from stage 4 caries. Last, in the model comparing the 15 disease states with stage 5 caries, no disease statistically significantly differed, again suggesting that all these diseases may be suitable proxies for stage 5 caries.
One limitation of DCEs is how each respondent interpreted the different attributes. For example, the attribute characterizing the impact on family can be interpreted differently by families with different structures and available resources. During the cognitive interview process, we found that parents did indeed focus on disruption of work and sleep schedule when they considered this attribute; nevertheless, we recognize that a subset of respondents may interpret this element differently. This study included a sample of respondents with lower education levels (40% with high school diploma or less) as compared with California overall (17% with high school diploma or less; US Census Bureau n.d.); this pattern is reflective of our choice to ensure an adequate sample of respondents from households below the FPL. To ensure that the respondents had adequate opportunity to interpret the attributes that the study defined, we provided them with an educational pamphlet. The pamphlet contained detailed information in lay terms regarding the disease states and attributes.
Another limitation is that this study relied on parents, instead of children, to answer questions regarding pediatric disease states; such practice may lead to biases as compared with directly querying children. Given the challenges in asking young children to complete a survey that requires understanding disease states and comparing hypothetical scenarios, a sample of parents is the most suitable respondent group, as compared with either children or adults who are not primary caretakers of 3- to 12-y-old children.
To leverage the DCE to evaluate proxy diseases, this study included actual characteristics of disease states as the levels within each attribute, instead of hypothetical disease states with random levels. The consequence of using actual disease state levels instead of randomized hypothetical levels was that the analyses may not have balanced option levels. We found that the impact on family was one that faced such issues and ameliorated the sample size issue in the analytic steps. Furthermore, it is reasonable to suspect that hypothetical levels that are more refined or different may lead to statistically significant differences that do not exist in our study; yet, the aim of this study was to determine the similarities and differences among the diseases and not at which level would the difference be statistically significant. Nevertheless, this design allowed us to achieve our main aim to compare disease states in addition to evaluating attributes.
Despite the limitations, the findings suggest that while diseases may have similar utility estimates and QALYs, parents view characteristics of these diseases, as well as their children’s experience in these disease states, differently. As such, proxy diseases require careful evaluation before being used as a substitute in CUAs. The findings here provide evidence that otitis media and its QALY—which is commonly used in CUAs evaluating programs and interventions for childhood caries prevention and treatment—may be one of the most suitable proxies. However, there are other disease states with slightly different QALYs that may be suitable. As such, the recommendation is to consider a range of proxy diseases and their QALYs when conducting a CUA for interventions on childhood caries. Outside of CUAs, CEAs based on caries averted as a measure of effectiveness may yield more accurate incremental cost-effectiveness ratios; however, the shortcoming of a CEA is that the results would not be comparable across disease states outside of caries—rather, it would be under a CUA, which uses QALY or disability-adjusted life year as a measurement of effectiveness.
Conclusions
This study employed a DCE approach to determine characteristics of childhood disease states that parents value, and it quantitatively evaluated the most comparable disease proxy that can provide a QALY for different caries stages in primary dentition. We focused on 4 attributes with varying levels: pain (none, mild, moderate, severe), disease state duration (≤1 wk, 1 mo, 4 mo, 1 y), treatment cost (≤$100, $1,000, $10,000), and family life impact duration (≤28 d, >28 d).
Results from the mixed effects logistic regressions indicated that pain was the attribute that parents prioritized; they preferred disease states that led to the least pain for their children. We also found that parents from different economic backgrounds may have different preferences; this pattern underlines the need to consider respondents’ financial situations in detail in future studies.
Findings from conditional logistic regressions suggested that parents viewed many diseases with similar QALYs similarly. Moreover, they viewed these disease states similarly to caries. The findings suggested that these diseases may be suitable substitutes for use in CUAs examining interventions for caries. We also identified some diseases that were not suitable substitutes. Acute gastritis, chronic gastritis, and cold sore were less preferred than and not suitable substitutes for stage 1 caries. Acute tonsilitis, acute gastritis, chronic gastritis, and cold sore were also less preferred than and not suitable substitutes for stage 2 caries. Chronic gastritis was less preferred than and not a suitable substitute for stage 4 caries. Acute otitis media and chronic otitis media, which have been used as proxy utilities and QALYs for dental caries, do appear to be reasonable proxies for dental caries.
Given the findings, we recommend that future studies consider a range of QALYs derived from proxy diseases (excluding chronic gastritis) for sensitivity analysis when conducting CUAs for interventions on childhood caries. Furthermore, we recommend innovating a study design that can accurately and directly measure QALYs in pediatric caries.
Author Contributions
T.K. Lin, S.A. Gansky, contributed to conception and design, data acquisition, analysis, or interpretation, drafted and revised the manuscript; D.E. Arriola Zarate, contributed to acquisition, data analysis, or interpretation, revised the manuscript; H. Lindau, MPH, N. Iribarren, contributed to conception, data acquisition, revised the manuscript; F. Ramos-Gomez, contributed to data conception, revised the manuscript. All authors gave final approval and agreed to be accountable for all aspects of the work.
Supplemental Material
sj-pdf-1-jct-10.1177_23800844221149337 – Supplemental material for Quality-Adjusted Life Year Proxies for Caries in Primary Dentition: A Discrete Choice Experiment
Supplemental material, sj-pdf-1-jct-10.1177_23800844221149337 for Quality-Adjusted Life Year Proxies for Caries in Primary Dentition: A Discrete Choice Experiment by T.K. Lin, D.E. Arriola Zarate, N. Iribarren, F. Ramos-Gomez and S.A. Gansky in JDR Clinical & Translational Research
Footnotes
Acknowledgements
We thank Janelle Urata, RDH, MS, for her insight on setting up a DCE on the Qualtrics platform.
A supplemental appendix to this article is available online.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: US National Institutes of Health/National Institute of Dental and Craniofacial Research (grant UH3-DE025514) and Coordinating Center (grant U01-DE025507).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
