Abstract
Introduction:
Children and adolescents living with HIV (CALHIV) have a higher risk of hard and soft oral tissue diseases as compared with their healthy peers. It is important to increase awareness regarding the need to integrate oral health within medical care among pediatric HIV populations. Studies on associations of oral diseases with oral health–related quality of life (OHRQoL) in CALHIV are lacking. This study examined the association between oral diseases and OHRQoL in Kenyan CALHIV.
Methods:
This cross-sectional analysis was nested in a longitudinal cohort study of CALHIV in Nairobi. CALHIV received oral examinations, and the World Health Organization’s Oral Health Surveys and Record Form was administered. OHRQoL was measured with the Parental-Caregiver Perceptions Questionnaire, with the subdomains of global, oral symptoms, function limitations, and emotional and social well-being, with higher scores indicating poorer OHRQoL. Linear regression was used to model associations between OHRQoL and oral diseases, adjusting for age at the time of oral examination, CD4 counts, and caregiver’s education.
Results:
Among 71 CALHIV, the mean age was 12.6 y (SD, 2.9; range, 10 to <21), and the mean composite OHRQoL score was 12.6 (SD, 11.2). Ulcers (not herpes simplex virus or aphthous) were associated with the worst overall OHRQoL (mean, 21.8; SD, 11.1;
Conclusions:
Oral ulcers, dry mouth, and untreated caries were associated with poorer OHRQoL in CALHIV. Integrating oral health into the primary care of CALHIV may improve their OHRQoL.
Knowledge Transfer Statement:
This study aimed to determine the association of oral diseases with the oral health–related quality of life of children and adolescents living with HIV (CALHIV). The findings will form part of the evidence to incorporate oral health protocols into care programs for CALHIV. Oral health monitoring has the potential to increase the surveillance of HIV clinical status, monitor the effectiveness of antiretroviral therapy, and improve the oral health–related quality of life of CALHIV.
Background
Effective antiretroviral therapy (ART) has changed the lives of children and adolescents living with HIV (CALHIV) in low- and middle-income countries. Due to significant progress in diagnosis and treatment (Dollfus et al. 2010; Ferguson et al. 2012), HIV infection has evolved from a disease with a high mortality rate to a chronic morbidity condition (Deeks et al. 2013), thus increasing the risk for noncommunicable diseases (El-Sadr and Goosby 2018). Even though ART has undoubtedly decreased oral manifestations of HIV (Nittayananta et al. 2010; Kalanzi et al. 2018), recent studies show evidence of increasing oral diseases and conditions among individuals with well-controlled HIV infection (Rwenyonyi et al. 2011; Oliscovicz et al. 2015).
CALHIV experience more oral diseases than healthy peers due to their compromised immune system (Ramos-Gomez and Folayan 2013; Lauritano et al. 2020). Additionally, the high prevalence of dental caries has been associated with HIV infection in children (Masiga and M’Imunya 2013; Rajonson et al. 2017). CALHIV have been found to have a higher prevalence of dental caries and salivary gland disease (Schiødt 1992; Dos Santos Pinheiro et al. 2009; El Howati and Tappuni 2018). Salivary gland disease results in a dramatic reduction of salivary flow, leading to significant morbidity in CALHIV (Walsh 2007; Gennaro et al. 2008; Ramos-Gomez and Folayan 2013). ART-associated enamel hypoplasia is common in the permanent dentition of CALHIV, possibly linked to protease inhibitors or efavirenz (Pontes et al. 2017).
The increased level of oral diseases among patients with HIV on long-term ART has been linked to poor oral health–related quality of life (OHRQoL; da Costa Vieira et al. 2018; Kikuchi et al. 2018; Greenspan and Challacombe 2020). OHRQoL is a sociodental indicator that explains the association between clinical outcomes and self-reported subjective evaluations of oral health–related experiences on physical, mental, and social well-being (Yengopal et al. 2016). To measure OHRQoL among children, common questionnaires include the Child Perception Questionnaire (Gilchrist et al. 2014), Early Childhood Oral Health Impact Scale (Buczynski et al. 2011), Oral Health Impact Profile (Gilchrist et al. 2014), and Child Oral Impact on Daily Performance (Raymundo de Andrade et al. 2011). However, there is limited research on the effects of oral diseases on OHRQoL among CALHIV (Raymundo de Andrade et al. 2011; Birungi et al. 2020). Outcomes from analyzing OHRQoL have been used successfully in planning oral health programs in populations highly affected by oral diseases (Massarente et al. 2011). With CALHIV being a high-risk population for oral diseases, assessment of OHRQoL could provide insights into their oral health care priorities.
Even though the number of CALHIV in Kenya decreased from 180,000 in 2010 to 111,500 in 2020 (UNICEF 2021), Kenya still registered approximately 8,000 new HIV pediatric infections in 2018 (prevalence, 4.9%; Kenya Ministry of Health 2015; UNICEF 2021). While Kenya has been successful in implementing national public health interventions for the medical care of CALHIV, oral health care remains limited despite the existence of universal health care coverage for children 0 to 5 y of age (Masiga and M’Imunya 2013; Hussein and Opinya 2017). The aim of this article was to assess the association between oral diseases and quality of life in Kenyan CALHIV participating in a >10-y-long longitudinal study. We hypothesized that oral diseases such as dental caries, enamel hypoplasia, and dry mouth will be associated with poorer OHRQoL. By providing a better understanding of the impact of oral diseases on the quality of life of CALHIV, we hope to increase awareness for integration of oral health within medical care of pediatric HIV populations.
Methods
This cross-sectional study was approved by the Institutional Review Board at the University of Washington (STUDY00003298) and by the Ethics and Research Committee of the Kenyatta National Hospital/University of Nairobi (KNH/ERC/R/133). The reporting in this article conforms to the STROBE guidelines.
Study Population
This study recruited CALHIV from an ongoing cohort study in Nairobi, Kenya. These children had originally been recruited for 2 randomized controlled trials from 2017 to 2019; after completion of the trials, the children were reenrolled into an ongoing cohort study to monitor long-term HIV reservoir dynamics. Both studies recruited, enrolled, and followed up with participants at the same study clinic at Kenyatta National Hospital. In the Optimizing Pediatric HIV Therapy (OPH) study (NCT00428116), all children were recruited before their first birthday from 2007 to 2010. In the OPH study, all children started ART immediately, regardless of their CD4 counts or HIV stage. At 24 months post-ART, all children had a scheduled treatment interruption and resumed ART within 6 months. The Pediatric Adherence Diary study enrolled children who were >1 y old from 2004 to 2005. The ART initiation criterion was moderate to severe HIV-1 disease per the World Health Organization (clinical stage 2 with CD4 <15% to clinical stage 3 or 4, respectively). More details about the 2 cohorts can be found elsewhere (Wamalwa et al. 2016; Pankau et al. 2017). Children in both cohorts received ART regimens that followed contemporaneous guidelines. Study staff caring for participants in the Pediatric Adherence Diary and OPH studies recruited families into the current study. All parents/legal guardians provided written informed consent for participation, and all children >8 y old also assented to study procedures. Consent information was provided in English and Kiswahili; staff providing consent were fluent in both languages.
Data Collection
We used standardized clinical assessments and questionnaires to collect data. Demographic and clinical information (CD4 count and plasma HIV RNA viral load data) was extracted from the primary study database. The oral examinations, conducted during routine HIV care visits, were performed by local community oral health officers who had been trained in the diagnosis of dental diseases and oral manifestations of HIV: angular cheilitis, oral hairy leukoplakia, necrotizing ulcerative gingivitis/periodontitis, necrotizing stomatitis, herpetic stomatitis/gingivitis and/or labial lesions, herpes simplex virus (HSV) ulcer, herpes zoster, molluscum contagiosum, cytomegalovirus, oral warts (papillomavirus), Kaposi sarcoma lesion, aphthous ulcer, other ulcerations, dry mouth due to decreased salivary flow, unilateral or bilateral swelling of major salivary glands, and other diagnoses—such as gum disease, calculus (tartar), xerostomia (sensation of dry mouth), impacted tooth/teeth, abscess, enamel hypoplasia, fluorosis, and potential malignancy. All diagnoses were recorded as present/absent by these examiners, who had been calibrated by following validated training modules provided by the University of Washington and the University of California, San Francisco (Shiboski et al. 2015). The inter- and intrarater reliability scores were 0.78 and 0.85, respectively. The Oral Health Surveys and Record Form for Oral Manifestations of HIV/AIDS (World Health Organization 2013) was used to evaluate oral diseases and HIV-associated oral mucosal lesions. It was not feasible to take radiographs due to the lack of infrastructure at the care center. Dental caries diagnosis was based on visual examination under natural light, as augmented with a headlight, gauze for cleaning and drying teeth, a probe (PCP12/11.5B; Hu-Friedy), and dental mirrors. The carious lesions were recorded (cavitated and white spots; Carney 2019). Each tooth was evaluated for caries, fillings/crowns, missing status due to caries/other reasons, white spots, sealants, and enamel developmental defects. Dental caries was summarized by presence (yes/no of any tooth). Dry mouth was defined as xerostomia, a subjective sensation. In addition to individual oral disease and paired oral diseases, we generated the burden of oral conditions using the count of oral conditions per child. The variable is defined across 4 conditions: untreated caries, dry mouth, dental hypoplasia, and ulcers, ranging from 0 (if they have no condition) to 4 (if they have all 4 conditions).
If an oral lesion was identified during the examination, children were referred directly to the University of Nairobi dental clinic, located near the HIV Comprehensive Care Clinic where the examination took place.
Oral Health Quality of Life Questionnaire
The Oral Health Quality of Life Questionnaire assessed the impact of oral and orofacial conditions on the functional, emotional and social well-being of children and their families. The questionnaire was in English, as all participating children and their parents understood the English language (help was available if any needed help), and included 5 components: Parental-Caregiver Perceptions Questionnaire (P-CPQ), Family Impact Scale, and Child Perception Questionnaire for 3 age groups (6 to 7, 8 to 10, and 11 to 14 y). Each response was measured by a Likert scale (never, once/twice, sometimes, often, every day/almost every day) and coded from 0 to 4 in the past 3 months. In the study, overall OHRQoL was measured by the total score of the P-CPQ (n = 31 subdomain items + 2 global items), which can vary from 0 to 132. Higher scores indicate worse OHRQoL. The score consisted of a global domain and 4 subdomains: oral symptoms (n = 6; range, 0 to 24), functional limitations (n = 7; range, 0 to 28), emotional well-being (n = 8; range, 0 to 32), and social well-being (n = 10; range, 0 to 40; Jokovic et al. 2006; Dawoodbhoy et al. 2013). The global domain was assessed by 2 questions: “How would you rate the health of your child’s teeth, lips, jaws, and mouth?” and “How much is your child’s overall well-being affected by the condition of his/her teeth, lips, jaws, and mouth?” All scores in this article were treated as continuous variables. In the available literature, there was no cutoff value to define the status of good OHRQoL (Opondo et al. 2017; Kemoli et al. 2018).
Statistical Analysis
The characteristics of children and parents were reported in the univariate analysis. Whereas for continuous variables the mean and standard deviation were calculated, for the categorical variables we reported frequency and percentage. The P-CPQ scores and burden of oral diseases were also characterized for the overall sample. Mean OHRQoL scores were compared by the presence and absence of oral diseases with 2-sample t tests or analysis of variance if >2 groups. To measure the association between oral diseases and OHRQoL and its domains, multivariable linear regression models were used with adjustment for age, sex, CD4 counts at oral examination, and number of years of parent education. For each model with OHRQoL and its domains as outcomes, we reported the coefficient and 95% CI of oral diseases and R2, which measured the percentage of variance in the OHRQoL scores that can be explained by the model. SAS 9.4 (SAS Institute) was utilized for all analyses.
Results
Our study population (N = 71) was 52%male with a mean age of 12.6 y (SD, 2.9; range, 9.9 to 20.5; Table 1). Approximately 68% of children between the ages of 6 and 12 y were at the transition from primary teeth to permanent teeth. Most children (91%) had a CD4 count ≥500 cells/mm3 at the time of oral examination, with only 1 child having severe immunosuppression (<200 cells/mm3). About three-quarters of children (74%) had ART initiated within the first year of life, with the mean age of initiation at 1.5 y. Most caregivers were female (92%), and about half (45%) of mothers accessed prevention of mother-to-child transmission interventions during pregnancy. The mean number of years of education for the parent was 10 (SD, 2.9). Most (96%) had at least primary education.
Characteristics of the CALHIV and Their Caregivers.
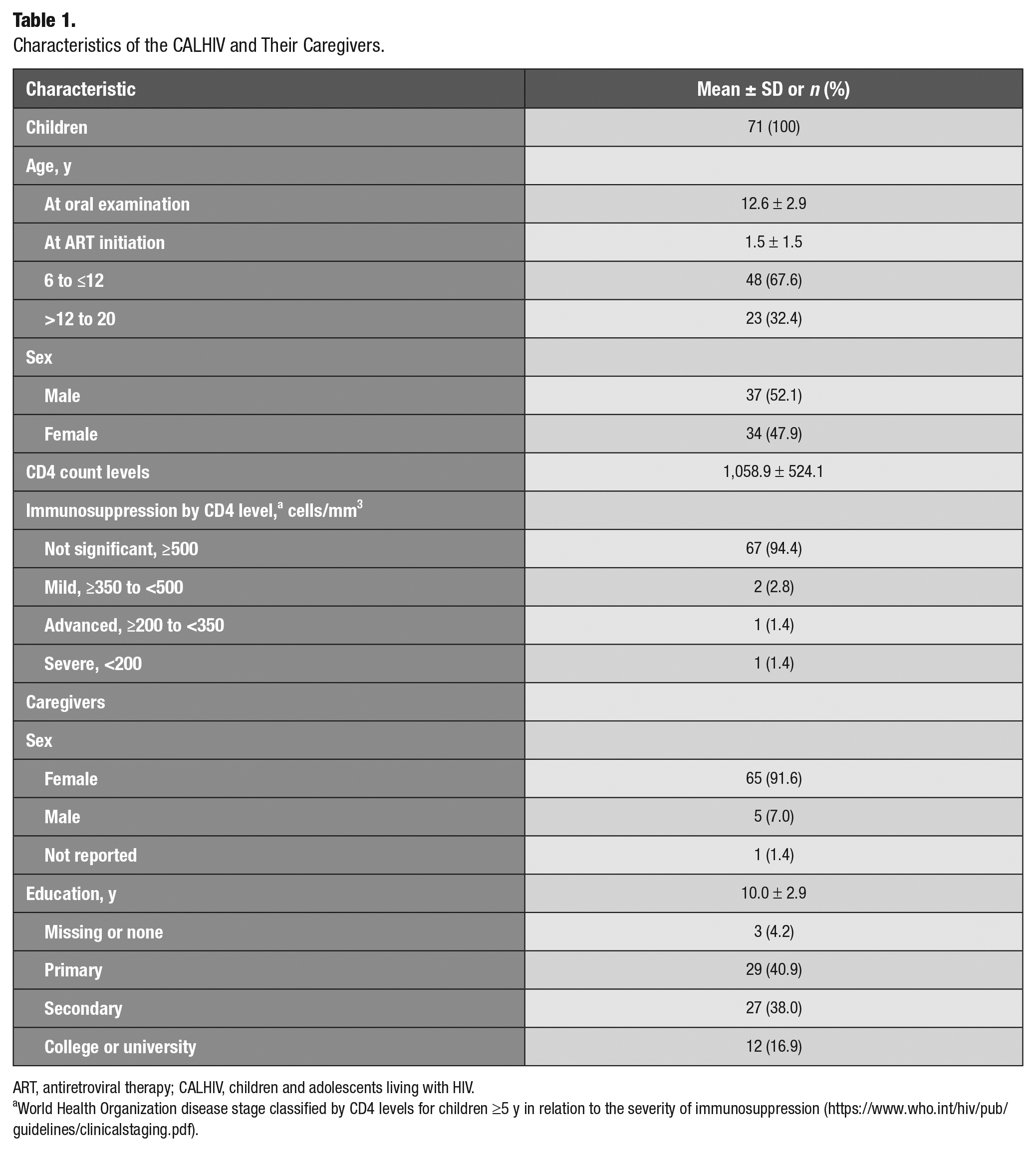
ART, antiretroviral therapy; CALHIV, children and adolescents living with HIV.
World Health Organization disease stage classified by CD4 levels for children ≥5 y in relation to the severity of immunosuppression (https://www.who.int/hiv/pub/guidelines/clinicalstaging.pdf).
Most children (83%) had at least 1 current oral disease: untreated dental caries (66%), dry mouth (28%), enamel hypoplasia (10%), and ulcers other than HSV and aphthous ulcer (7%; Table 2). Among participants aged 6 to 12 y, current oral disease included untreated dental caries (73%), dry mouth (35%), enamel hypoplasia (10%), and ulcers other than HSV and aphthous ulcer (8%). No cases of the following were detected: angular cheilitis, oral hairy leukoplakia, necrotizing ulcerative gingivitis/periodontitis, necrotizing stomatitis, HSV ulcers, herpes zoster, molluscum contagiosum, cytomegalovirus, Kaposi sarcoma lesions, or swelling of major salivary glands. The mean ± SD scores for each subdomain were as follows: global, 2.9 ± 1.7; oral symptoms, 3.7 ± 2.7; function limitations, 2.8 ± 3.2; emotional well-being, 1.8 ± 3.4; and social well-being, 1.4 ± 3.5. There were some differences in OHRQoL and its domains between the younger and older groups, but these differences were not significant. For example, the social domain of OHRQoL for those aged 6 to 12 y was better (0.9 ± 2.7; lower score) than those aged 13 to 20 y, but the difference was not statistically significant. The burden of oral conditions ranged from 0 to 3, with 1.1 ± 0.9 conditions.
Oral Diseases of CALHIV and Caregiver-Reported Quality of Life Scores.
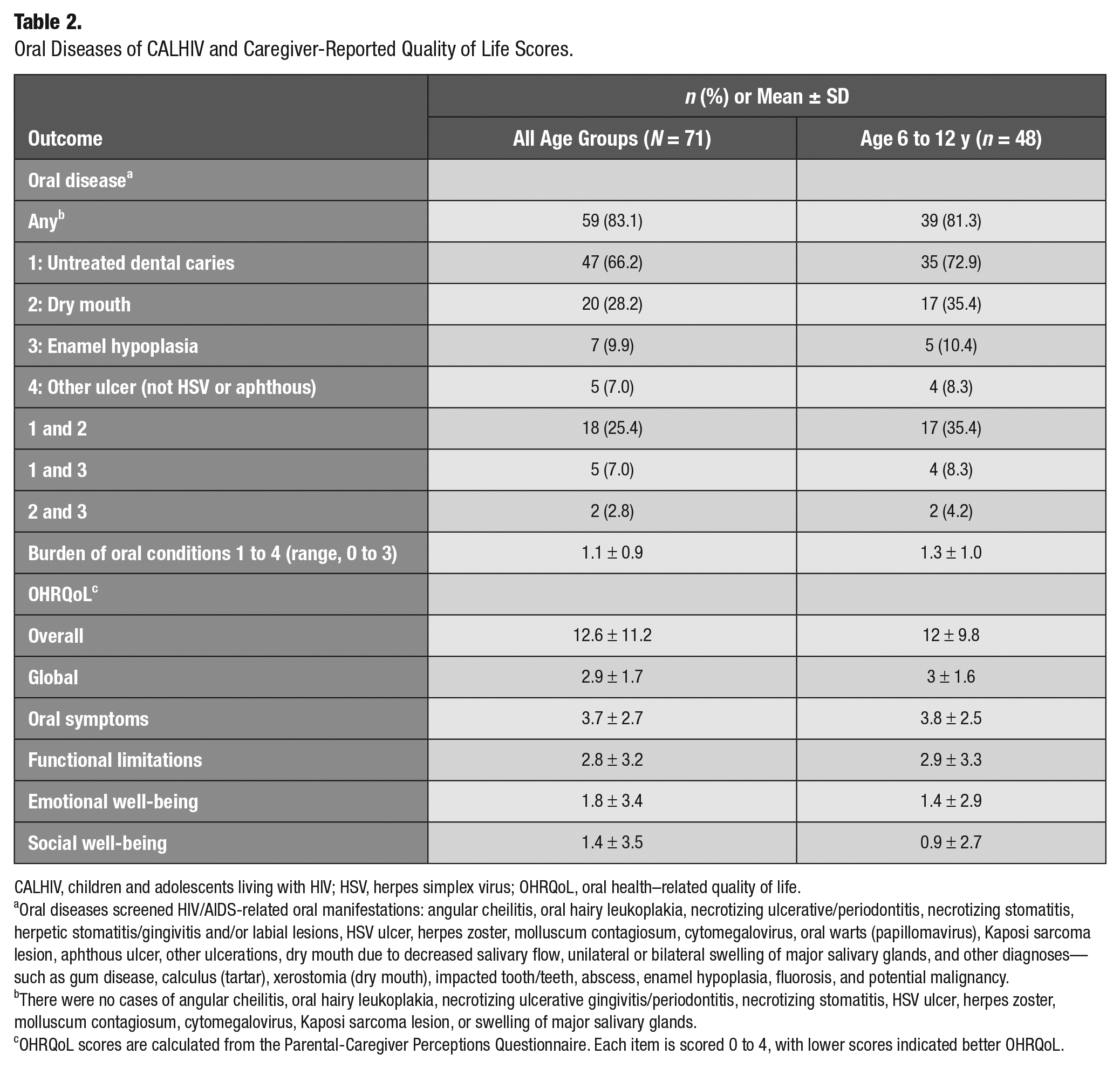
CALHIV, children and adolescents living with HIV; HSV, herpes simplex virus; OHRQoL, oral health–related quality of life.
Oral diseases screened HIV/AIDS-related oral manifestations: angular cheilitis, oral hairy leukoplakia, necrotizing ulcerative/periodontitis, necrotizing stomatitis, herpetic stomatitis/gingivitis and/or labial lesions, HSV ulcer, herpes zoster, molluscum contagiosum, cytomegalovirus, oral warts (papillomavirus), Kaposi sarcoma lesion, aphthous ulcer, other ulcerations, dry mouth due to decreased salivary flow, unilateral or bilateral swelling of major salivary glands, and other diagnoses—such as gum disease, calculus (tartar), xerostomia (dry mouth), impacted tooth/teeth, abscess, enamel hypoplasia, fluorosis, and potential malignancy.
There were no cases of angular cheilitis, oral hairy leukoplakia, necrotizing ulcerative gingivitis/periodontitis, necrotizing stomatitis, HSV ulcer, herpes zoster, molluscum contagiosum, cytomegalovirus, Kaposi sarcoma lesion, or swelling of major salivary glands.
OHRQoL scores are calculated from the Parental-Caregiver Perceptions Questionnaire. Each item is scored 0 to 4, with lower scores indicated better OHRQoL.
Children with oral diseases had worse OHRQoL than children without (Table 3). The overall mean of OHRQoL among children with any oral disease was 13.7 ± 11.5, as compared with 7.0 ± 6.9 (P = .056, not significant [NS]) among those without any oral diseases. This was also true across all subdomains when CALHIV with and without oral diseases were compared: global, 3.1 ± 1.7 vs 1.8 ± 1.3 (P = .02); oral symptoms, 3.9 ± 2.7 vs 2.9 ± 1.9 (NS); function limitations, 2.1 ± 3.2 vs 1.5 ± 2.9 (NS); emotional well-being, 2 ± 3.7 vs 0.8 ± 1.6 (NS); and socialwell-being, 1.5 ± 3.7 vs 0.2 ± 0.8 (P = .01). When compared with children without oral diseases, patients with dry mouth and dental caries were significantly associated with a worse OHRQoL score in the global domain (4.4, P < 0.0001). When analyzed individually, ulcers (other than HSV or aphthous) were associated with the worst overall OHRQoL (21.8, P = .05) and with worse OHRQoL in the oral symptoms domain (7.0, P = .003). The higher number of oral conditions was associated with worse global OHRQoL (P < 0.0001) and worse oral symptoms (P = .004).
Comparison of Mean Oral Health Quality of Life Scores among CALHIV by the Presence and Absence of Oral Diseases.
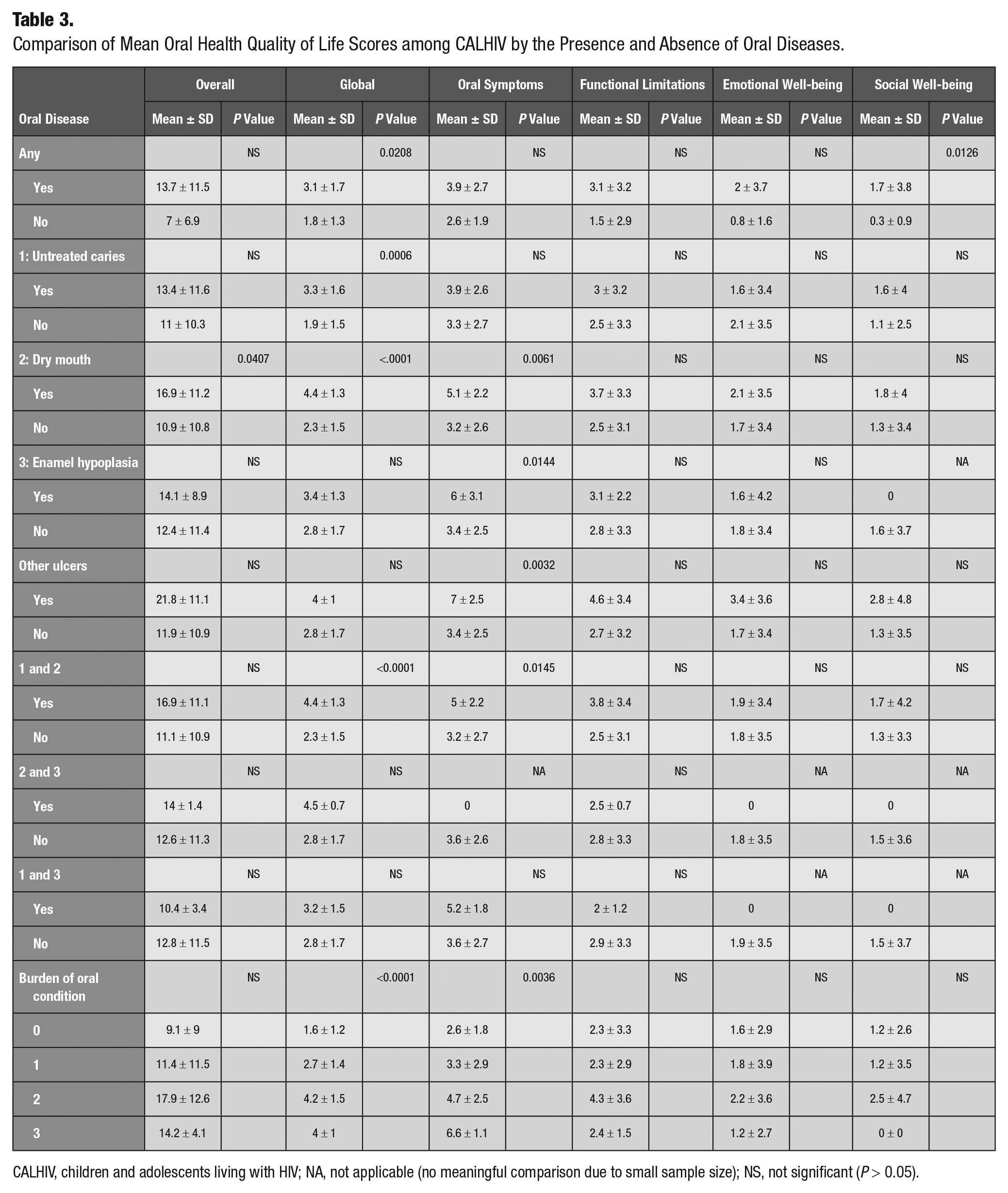
CALHIV, children and adolescents living with HIV; NA, not applicable (no meaningful comparison due to small sample size); NS, not significant (P > 0.05).
We hypothesized that the presence of >1 oral disease would have a strong effect to predict worse OHRQoL scores, and we tested this using linear regression (Table 4) adjusting for age at oral examination, CD4 count, and years of caregiver education. We found that children with any oral disease had a 6.7-unit (95% CI, –0.6 to 13.9) higher OHRQoL score as compared with those without oral disease. The presence and absence of oral disease explained 6.1% of the variability in OHRQoL in the model, while dry mouth and untreated dental caries together explained 7.3% of the variation of OHRQoL. Those with dental caries had a 4.2-unit (−2.0 to 10.4) higher global OHRQoL than those without oral disease, and those who had dry mouth and dental caries had a 6.8-unit (0.3 to 13.3) higher global OHRQoL score than those without oral disease. Conversely, those who had dental caries had an 8.3-unit (0.5 to 16.1) worse overall OHRQoL, and those who had dental caries and dry mouth had a 6.9-unit (0.5 to 13.3) worse overall OHRQoL as compared with those without oral disease. In the regression model, oral diseases significantly affect the global and oral symptoms domains. For example, the regression model with dry mouth explained 30% of the variation in the OHRQoL global domain and 13% of the variation in the oral symptoms domain. The burden of oral conditions was significantly associated with global OHRQoL (P < 0.0001), accounting for 32.6% of variations of global OHRQoL, as compared with dry mouth itself, which accounted for 31.9% of variations of global OHRQoL. The higher burden of oral conditions (larger number of conditions) was significantly associated with lower OHRQoL in the domain of oral symptoms (P < 0.01). In the regression model, the model including burden of oral conditions accounted for 18.8% of the variation of the oral symptom OHRQoL score, as compared with other ulcers, which accounted for 15.4% variation of this score.
Association Between the Presence of Oral Diseases and OHRQoL Scores in Children and Adolescents Living with HIV.
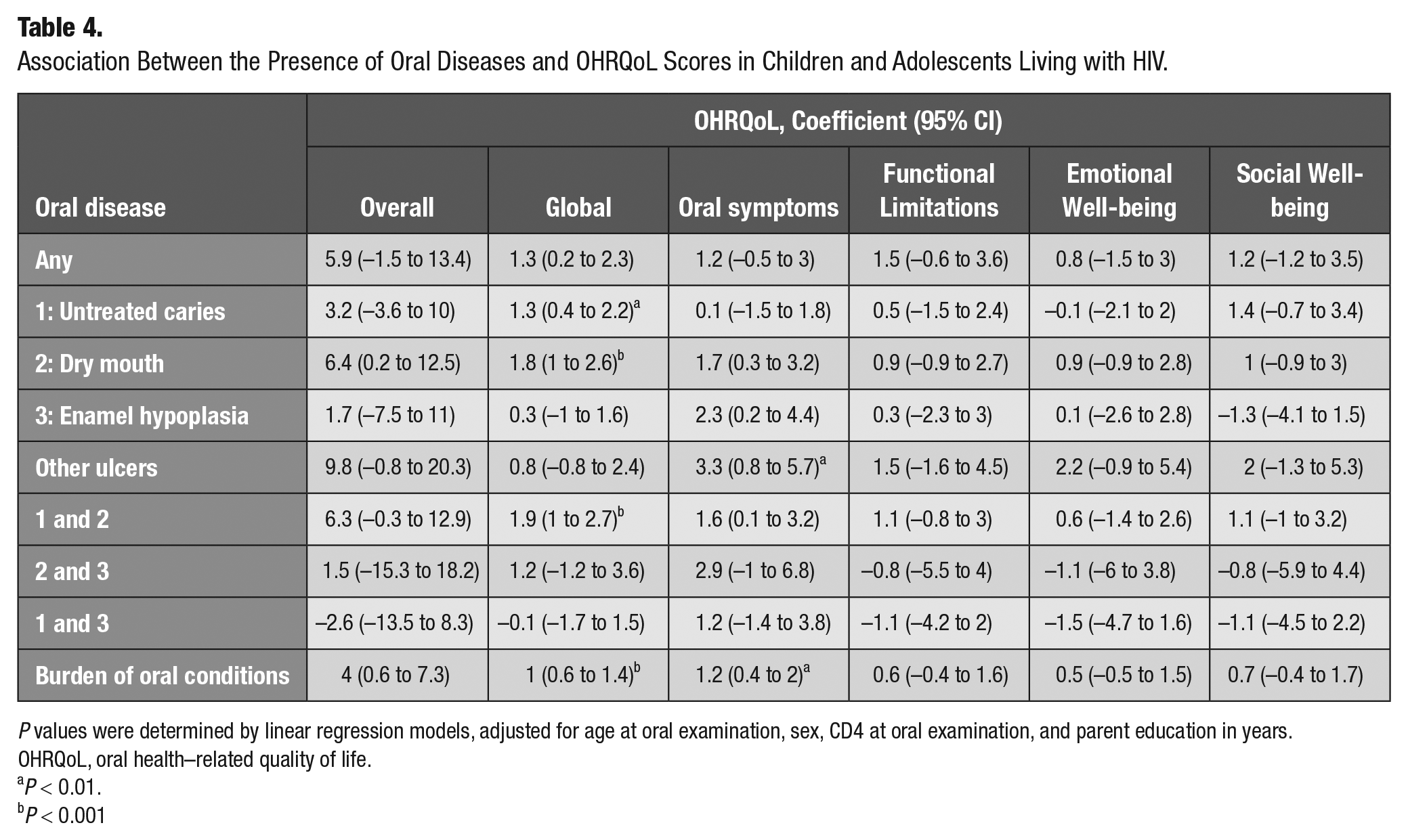
P values were determined by linear regression models, adjusted for age at oral examination, sex, CD4 at oral examination, and parent education in years.
OHRQoL, oral health–related quality of life.
P < 0.01.
P < 0.001
At the end of the study, 29 children had been identified as having urgent care needs and were referred to the University of Nairobi’s dental clinic for treatment. Of these children, 15 (52%) had sought treatment, and 14 were fully treated at the University of Nairobi.
Discussion
Our study is among a small number of studies to date examining the association between oral diseases and the quality of life of CALHIV. While the mean composite OHRQoL score (11.5) was similar to previous studies of the general population in other counties (Goursand et al. 2009; Albites et al. 2014), our results suggest that 1) oral diseases are associated with compromised quality of life in CALHIV, with multiple concurrent oral morbidities predicting the poorest quality of life, and 2) these associations are independent of age and immune status. CALHIV with dry mouth and dental caries had the worst OHRQoL composite and global domain scores. The single oral condition that was associated with the poorest OHRQoL was ulcers (not HSV or aphthous), and across all oral diseases, the OHRQoL oral symptoms subdomain was associated with having dry mouth, enamel hypoplasia, or other ulcer (Massarente et al. 2011). All children with oral diseases and treatment needs were referred to the dental clinic and treated. These findings provide evidence supporting the need to integrate oral health care into the primary care of CALHIV.
Among children 6 to 12 y of age, children with dry mouth and dental caries had the worst overall OHRQoL and functional limitations subdomain. This was consistent with the findings from a cross-sectional study in Cambodia that compared CALHIV and HIV-negative children aged 3 to 15 y (Kikuchi et al. 2021). CALHIV had a higher prevalence of dental caries and a lower salivary flow that significantly affected their OHRQoL but not the HIV infection itself. Children who had dry mouth and enamel hypoplasia had the worst OHRQoL in the global domain and oral symptoms subdomain. Similar results were found in other studies among children with mixed dentition (age, 6 to 12 y; Buczynski et al. 2011; Ivanova et al. 2017). When compared with other studies on the OHRQoL among children and adolescents without HIV infection in Kenya, CALHIV’s OHRQoL score was lower (Kemoli et al. 2018).
CALHIV have a higher prevalence of dental caries and oral diseases as compared with their HIV-negative peers (Kikuchi et al. 2021). While our study showed 68% of children with dental caries and 83% with at least 1 oral disease, a study among 2- to 5-y-old children in Nairobi and Mombasa found an 84% prevalence of untreated dental caries (Hussein and Opinya 2017). Although dry mouth has been underreported and diagnosed late, especially among children (Walsh 2007; Ivanova et al. 2017), it was associated with worse OHRQoL among children, especially those aged 6 to 12 y. For CALHIV, dry mouth might be due to HIV-associated salivary gland disease or the side effects of ART (Schiødt 1992). There were 5 children in the study with ulcers other than HSV or aphthous ulcers.
Our study had many strengths: assessment of a large cohort of children with well-managed HIV infection, inclusion of clinical and laboratory data to assess confounding, detailed assessment of oral conditions, and tracing of linkage to care outside the study clinic. Because this select cohort received comprehensive high-quality HIV care since early childhood, our results may not be generalizable to more heterogenous populations or settings of CALHIV. Because our study was cross-sectional, we cannot infer the direction of causality in an association between oral diseases and OHRQoL; however, it is reasonable to assume a causal link. Other contributors to oral health and potential confounders were not assessed, such as dietary exposures, oral hygiene habits, and fluoride exposure. The number of oral conditions in the linear regression model were treated as continuous variables due to the small sample size. Because the oral health examinations were nested into the children’s routine study visits, their oral assessments were short and conducted under natural light. Still, this initial study supports feasibility for further studies aimed at integrating oral health into the primary care of CALHIV. The study was a snapshot of the participants at different stages, at primary, mixed, or permanent dentition. All findings were associations with the oral health outcomes at the time of dental examination. Because the study was cross-sectional, we cannot estimate the effects of oral diseases over time on the OHRQoL.
There is an urgent need for longitudinal follow-up studies on the impact of poor oral health within the quality of life of CALHIV. Access to oral health care for CALHIV could be significantly improved by integrating it within the ongoing comprehensive medical care received in HIV treatment programs (Walsh 2007; Gennaro et al. 2008; Ramos-Gomez and Folayan 2013). In our setting, all children with oral diseases were referred for care and successfully treated. Because dental care is included in the National Health Insurance (National Hospital Insurance Fund), integration of oral care into HIV care could be efficient, cost-effective, and highly acceptable. Incorporating oral health protocols into children’s HIV care programs has the potential to increase surveillance of HIV disease trajectory, monitor the effectiveness of ART, and improve the quality of life for CALHIV (Ramos-Gomez and Folayan 2013; Lauritano et al. 2020).
Author Contributions
Y. Wang, F. Ramos-Gomez, contributed to conception, data acquisition, analysis, and interpretation, drafted and critically revised the manuscript; A. Kemoli, contributed to conception, design, data analysis and interpretation, drafted and critically revised the manuscript; G. John-Stewart, D. Wamalwa, S. Benki-Nugent, contributed to design and data interpretation, critically revised the manuscript; J. Slyker, A.L. Seminario, contributed to conception, design, data acquisition, analysis, and interpretation, drafted and critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of the work.
Footnotes
Acknowledgements
We thank the patients, parents, and staff of the Kenyan Pediatric Studies for their generosity and willingness to participate in this study. We greatly thank Hu-Friedy Mfg.Co., LLC for its generosity at donating dental instruments for our oral examinations.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Field site and biostatistical support were provided by the University of Washington/Fred Hutch Center for AIDS Research (CFAR), a National Institutes of Health (NIH)–funded program (P30 AI027757), and UW School of Dentistry, SunStar Award. Y. Wang was supported by the National Institute of Mental Health (NIH award T32MH080634). The content is solely the responsibility of the authors and does not necessarily represent the official views of the NIH.
