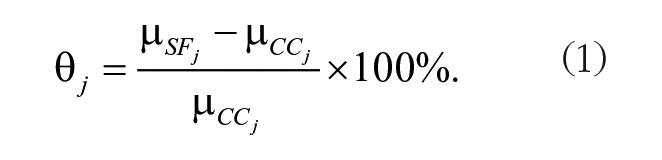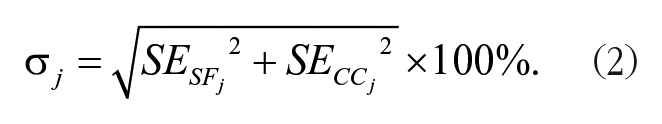Abstract
Introduction:
Estimating the risk of dental problems in long-duration space missions to the Moon and Mars is critical for avoiding dental emergencies in an environment that does not support proper treatment. Previous risk estimates were constructed based on the experience in short-duration space missions and isolated environments on Earth. However, previous estimates did not account for potential changes in dental structures due to space travel, even though bone loss is a known problem for long-duration spaceflights. The objective of this study was to systematically analyze the changes in hard tissues of the craniofacial complex during spaceflights.
Methods:
Comprehensive search of Medline, Embase, Scopus, the NASA Technical Report Server, and other sources identified 1,585 potentially relevant studies. After screening, 32 articles that presented quantitative data for skull in humans (6/32) and for calvariae, mandible, and lower incisors in rats (20/32) and mice (6/32) were selected.
Results:
Skull bone mineral density showed a significant increase in spacefaring humans. In spacefaring rodents, calvariae bone volume to tissue volume (BV/TV) demonstrated a trend toward increasing that did not reach statistical significance, while in mandibles, there was a significant decrease in BV/TV. Dentin thickness and incisor volume of rodent incisors were not significantly different between spaceflight and ground controls.
Discussion:
Our study demonstrates significant knowledge gaps regarding many structures of the craniofacial complex such as the maxilla, molar, premolar, and canine teeth, as well as small sample sizes for the studies of mandible and incisors. Understanding the effects of microgravity on craniofacial structures is important for estimating risks during long-duration spaceflight and for formulating proper protocols to prevent dental emergencies.
Knowledge Transfer Statement:
Avoiding dental emergencies in long-duration spaceflights is critical since this environment does not support proper treatment. Prior risk estimates did not account for changes in dental structures due to space travel. We reviewed and synthesized the literature for changes in craniofacial complex associated with spaceflight. The results of our study will help clinicians and scientists to better prepare to mitigate potential oral health issues in space travelers on long-duration missions.
Introduction
Microgravity-induced bone loss is a well-known and still unmitigated effect of long-term spaceflight on the human and rodent skeleton (Stavnichuk et al. 2020; Fu et al. 2021), but the effect of space travel on the oral cavity and jaw bones is far less studied. Hard tissues of the oral cavity are constantly used for vital tasks such as mastication, speech, respiration, and deglutition. Understanding the effect of spaceflight on the overall health of the oral cavity and its supporting structure is critical for avoiding dental emergencies in an environment that does not support proper treatment. This becomes especially important when planning for long-duration missions to the Moon and Mars.
Dental issues reported in space travel include dental caries, crown displacement, and lost fillings (Menon 2012). However, when preflight and postflight dental events in astronaut corps were taken into account, several cases of pulpitis and dental abscesses were recorded, which may result in severe health consequences when untreated (Menon 2012). Documented dental emergencies accounted for 1% of all medical events aboard the Mir space station in a 3-y period (Gontcharov et al. 2005), as well as a case of a cosmonaut who spent 2 wk of the 96 d in space in incapacitating pain (Ball et al. 2001). Based on the estimates developed for isolated Earth-based environments such as expeditions (Brown et al. 1977; Kupper et al. 2014), the frequency of potential dental emergencies will increase to substantial levels in longer-term spaceflights. Taking into account the potential length of the space mission to Mars of 9 to 12 mo and a team of 10 space travelers, these risk estimates translate into at least 1 certain event resulting in significant discomfort to crew members. However, none of the previous estimates accounted for potential changes in dental structures due to space travel that may affect the risk estimates.
Understanding how microgravity influences craniofacial hard structures is essential for estimating risks during long-duration spaceflight and for formulating proper protocols to prevent dental emergencies. The objective of this study is to systematically review the literature for changes in hard tissues of the craniofacial complex during spaceflights and use meta-analytic approaches to quantify these changes.
Methods
This study was performed in compliance with the Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) statement (PRISMA checklist is provided in Appendix Note 1).
Search Strategy, Inclusion, and Exclusion
A systematic search strategy (Appendix Note 2) containing relevant terms for spaceflight, space missions, teeth, and all cranial bones was constructed and reviewed by a medical librarian. The search was performed in Medline, Embase, and Scopus on June 9, 2021, and was complemented by a manual search of the NASA technical report server. Title and abstract screenings were carried out by 2 independent reviewers (MG and MSM). Articles in any language were included for full-text screening if they alluded to any craniofacial structure in any species that has experienced spaceflight. Articles describing simulated microgravity were excluded. Included studies were scored for reporting quality (Appendix Note 3).
Data Extraction
From studies with quantitative parameters for craniofacial structures, we recorded authors, year of publication, mission, duration of spaceflight, species, craniofacial structures analyzed, type of quantitative measurement, and control groups involved in study. For studies included in the meta-analysis, we also extracted sample sizes for spaceflight and comparison group(s), as well as craniofacial structure and its region being assessed, and for each parameter, we extracted mean spaceflight (SF) group and the mean comparison control (CC) group values (preflight measurements for humans or ground control [GC] and vivarium control [VC] for animals), along with corresponding measure of variance (standard errors, standard deviations, or interquartile ranges). When articles presented similar data for 2 identical populations, we selected the one with higher-quality score.
Measurement-Level Outcomes
Two types of CC groups were used: GC, where some aspects of the spaceflight environment, excluding microgravity, were mimicked and VC, where animals lived in a standard laboratory environment. When possible, GC was used as a comparison to SF animals, with additional analysis conducted when VC data were presented. Data were processed as previously described (Mikolajewicz and Komarova 2019; Fu et al. 2021). Briefly, percentage difference between SF and CC for an individual measurement θj was calculated from the mean SF values, µSFj, and the mean CC value, µCCj, using equation (1).
Standard errors sej for each measurement in the SF or CC group were extracted or calculated based on the provided data (Mikolajewicz and Komarova 2019) and then normalized as SEj = sej / µCCj. To calculate the standard error for the percentage difference of a measurement σj, SF and CC groups were assumed to be independent, allowing to use equation (2).
For tissue mineral density (TMD)measurements, different bone regions in the same group of animals were combined as unweighted averages.
Meta-analytic Model, Global Outcome, and Heterogeneity
We used the random-effects model to calculate global effect size
Heterogeneity and Bias
To quantify heterogeneity, we calculated H2 and I2 as described previously (Mikolajewicz and Komarova 2019). Potential bias was assessed using the largest bone volume/tissue volume (BV/TV) data set, in which we preformed single data set exclusion analysis and funnel plot for publication bias (Appendix Fig. 1).
Additional Analysis
Subgroup analysis was performed for species (humans, rats, mice) and bone type (calvariae, mandible) by combining mission-level outcomes and standard error within each category.
Results
Overview of Relevant Literature
Systematic search conducted in Medline, Embase, and Scopus retrieved 1,577 candidate articles. Eight additional sources were from the NASA Technical Report Server. Following the screening, 30 articles that discuss any hard structure of the skull that has experienced spaceflight were included in the systematic review (Fig. 1A). Included articles described studies in rats (20/32) (Prokhonchukov et al. 1977; Prokhonchukov, Kolesnik, et al. 1978; Prokhonchukov, Zaitsev, et al. 1978; Savostin-Asling 1978; Simmons et al. 1980; Roberts and Mozsary 1981; Roberts et al. 1981; Simmons, Russell, and Winter 1981; Simmons, Russell, Winter, Rosenberg, et al. 1981; Tran Van et al. 1981; Simmons et al. 1983; Rosenberg et al. 1984; Roberts et al. 1987; Kleber et al. 1989; Volozhin et al. 1989; Simmons, Grynpas, and Rosenberg 1990; Simmons, Grynpas, Rosenberg, and Durnova 1990; Davis et al. 1998; Hatton et al. 2002; Keune et al. 2015), mice (6/32) (Zhang et al. 2013; Ghosh et al. 2016; Macaulay et al. 2017; Dagdeviren et al. 2018; Dadwal et al. 2019; Maupin et al. 2019), and humans (6/32) (Oganov et al. 1992; Shigematsu et al. 1997; Grigoriev et al. 1998; Miyamoto et al. 1998; Oganov 2003; Oganov et al. 2005). The most studied hard structures within the skull were the mandible, calvariae, and the lower incisors (Fig. 1B), which were first studied in Cosmos missions in the 1970s and continued with STS, Bion, and CRS flights (Fig. 1C).
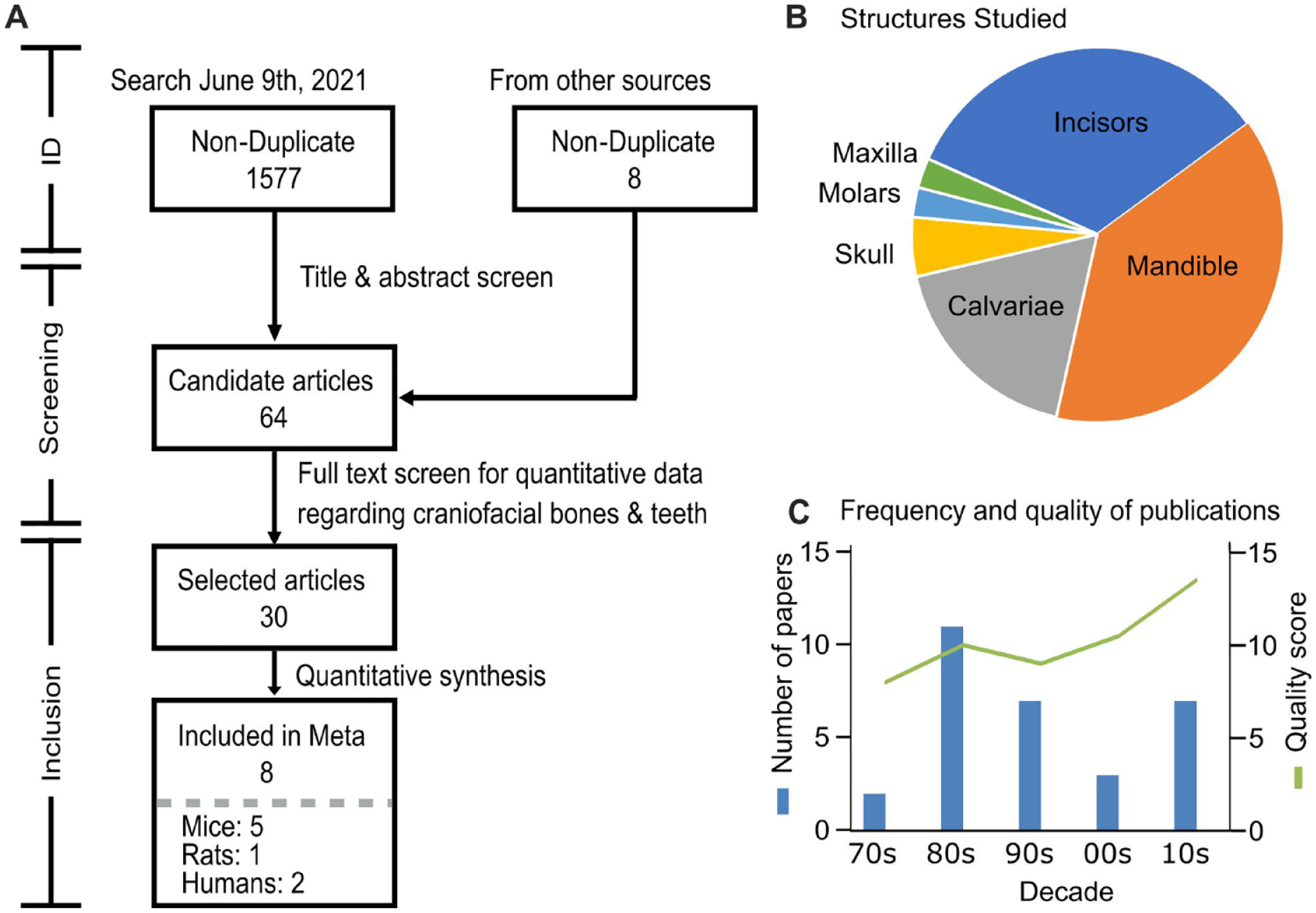
Systematic review information flow and Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) diagram. (
Thirty-two studies reported the changes in craniofacial structures from 15 missions that took to space rats (9 missions), mice (4 missions), and humans (multiple Mir and space shuttle missions) (Table). The earliest Cosmos missions, 605 and 782, studied incisors and mandible in rats. Unfortunately, the reporting of these mission was not sufficient to extract quantitative data, with some articles not presenting the measure of variance and others not distinguishing between craniofacial structures and other bones. Later Cosmos missions, 1129, 1667, and 1887, provided clearer reporting on calvaria, mandible, incisors, and, for the first time, molars in spacefaring rats. Our search retrieved multiple articles concerning Cosmos 1129, but there was a large overlap in presented data. We included 2 articles (Simmons et al. 1983; Rosenberg et al. 1984) that contained the most data and scored the highest in quality. Spacelab 3 (SL-3) was the only mission to investigate the maxilla, but reporting was limited to fibroblast-like cells in the periodontal ligaments. Although in missions STS-62, STS-70, and STS-80, craniofacial structures in rats were assessed, these missions included ovariectomized, pregnant, or hypertensive rats without the corresponding untreated controls; therefore, these missions were excluded from subsequent analysis. Since 2010, calvaria, mandible, and incisors were investigated in mice on 4 missions, STS-131, STS-135, Bion-M1, and CRS-10. Reporting for these missions was consistent and qualified all articles except for 1 for further analysis (Zhang et al. 2013; Ghosh et al. 2016; Macaulay et al. 2017; Dagdeviren et al. 2018; Maupin et al. 2019). The remaining study presented overlapping data (Dadwal et al. 2019). Four studies described changes in the skull in astronauts visiting the Mir space station in the 1990s. Since these studies provided updated information for the same population of space travelers, only the last, most complete study (Oganov et al. 2005) was used in the analysis. Two additional studies provided overlapping data for changes in the skull of 2 astronauts who flew aboard space shuttle missions; the higher-quality study (Miyamoto et al. 1998) was included in the analysis. Human studies generally reported on the skull with respect to the rest of the skeleton and did not provide specifics about individual bones within the skull.
We further assessed the potential for meta-analysis of different parameters for the 3 most studied craniofacial hard tissues in spaceflight. In mandibles (Appendix Table 1), spaceflight-induced changes in mineral content, tissue-level properties, and microarchitecture were analyzed. From these parameters, only BV/TV, TMD, and osteoclast surface/bone surface were studied in at least 3 independent missions and were included in the meta-analysis. In calvaria (Appendix Table 2), mineral content was assessed only in the Cosmos-1887 mission, and calvaria thickness and BV/TV were reported in 3 mouse missions and TMD in 2 missions. Human studies reported on the skull using the dual-energy X-ray absorptiometry whole-body imaging. While this region includes both calvaria and mandible, the contribution from calvaria is larger; therefore, we combined these data with calvaria measurements in rodents. Incisors were the only reasonably studied teeth (Appendix Table 3), with studies reporting mineral content, dentin, and enamel properties, of which we were able to find sufficient data sets for analysis of incisor volume, dentin thickness, and cemento-enamel junction to apical crest distance (CEJ-AC). Thus, although data scarcity was very noticeable, after the analysis of quality and overlap in presented data, we identified several parameters, for which data reported by 8 studies describing 7 missions were included in the meta-analysis (Simmons et al. 1983; Miyamoto et al. 1998; Oganov et al. 2005; Zhang et al. 2013; Ghosh et al. 2016; Macaulay et al. 2017; Dagdeviren et al. 2018; Maupin et al. 2019).
Overview of Studies That Reported Spaceflight-Induced Changes in Craniofacial Bones.
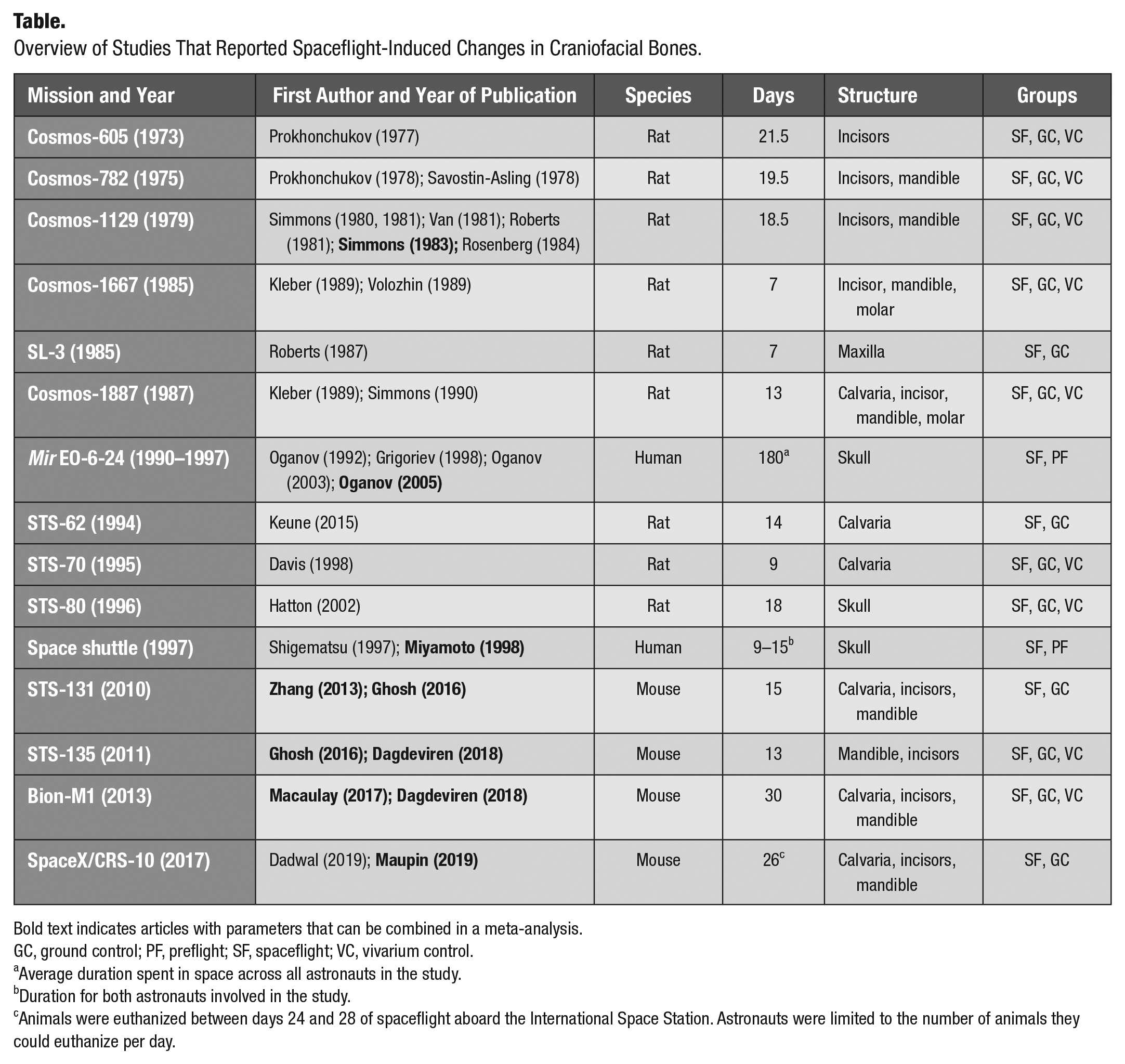
Bold text indicates articles with parameters that can be combined in a meta-analysis.
GC, ground control; PF, preflight; SF, spaceflight; VC, vivarium control.
Average duration spent in space across all astronauts in the study.
Duration for both astronauts involved in the study.
Animals were euthanized between days 24 and 28 of spaceflight aboard the International Space Station. Astronauts were limited to the number of animals they could euthanize per day.
Spaceflight-Induced Changes in Calvaria and Mandible
Previously, it was shown that humans experiencing spaceflight exhibit an increase in bone mineral density in the skull region (Oganov et al. 2005; Stavnichuk et al. 2020). We meta-analyzed available data for spaceflight-induced changes in BV/TV in calvaria and mandible of rodents (Fig. 2A). While the calvariae demonstrated a trend toward increasing BV/TV, in mandibles, there was a significant decrease in BV/TV in SF animals compared to GC. TMD in rodent calvaria tended to increase, and in humans, it was significantly increased due to spaceflight, while TMD in rodent mandible was not significantly affected by the spaceflight (Fig. 2B). Calvarium thickness was not significantly affected by spaceflight (Fig. 2C). Heterogeneity was low among BV/TV and TMD and moderate in calvarium thickness. Other parameters measured only once or twice and not included in the meta-analysis were calcium, phosphorus, and magnesium content, which were not significantly different between SF and CC rats (Simmons, Grynpas, and Rosenberg 1990). Taken together, these data suggest that spaceflight affects the bones of the skull differently, resulting in bone gain in calvaria and bone loss in mandible.
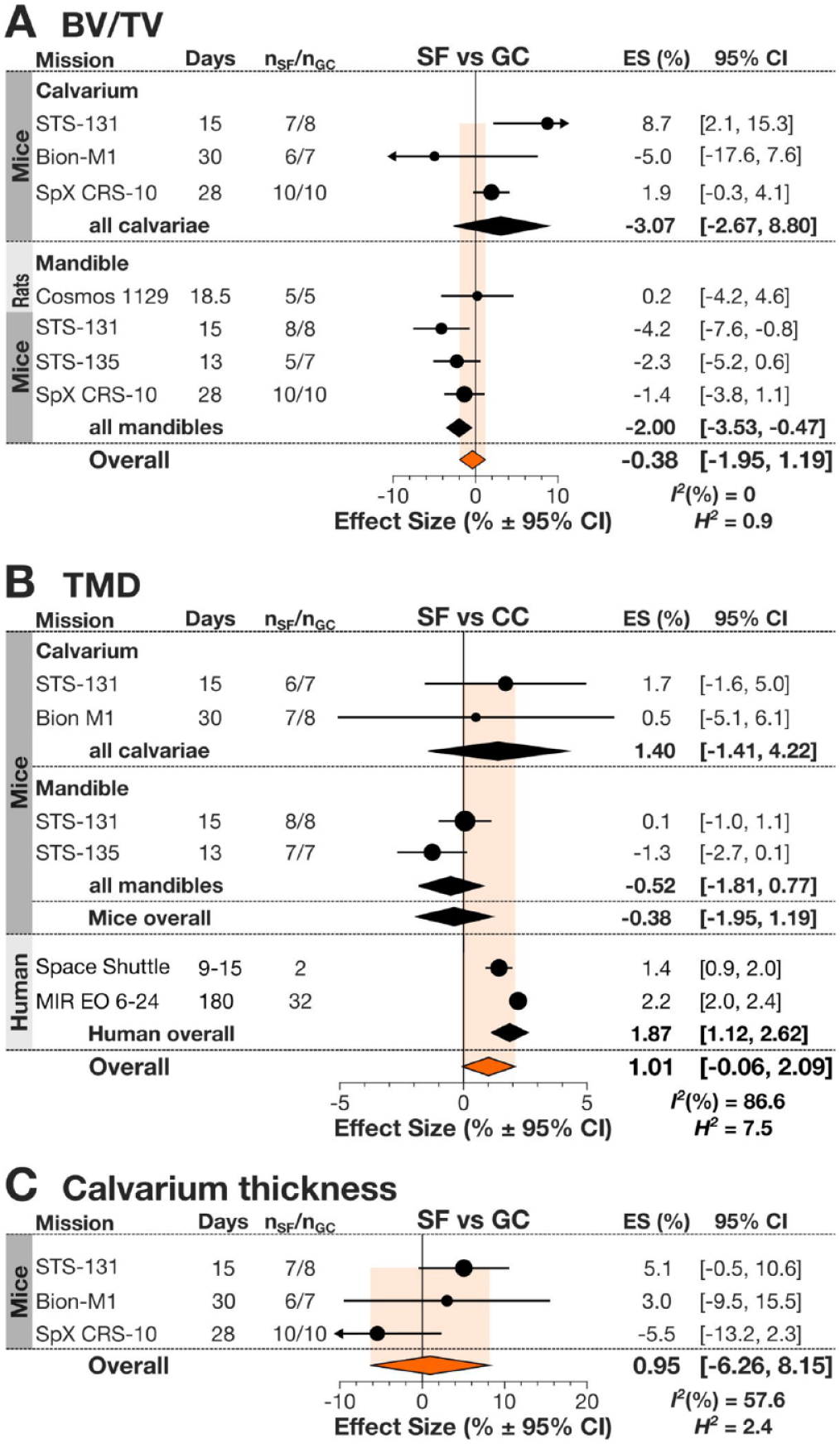
Spaceflight-related changes in bones of the skull. (
Spaceflight-Induced Changes in Incisors of Spacefaring Rodents
Incisor volume, dentin thickness, and CEJ-AC were not significantly different between SF and GC rodents (Fig. 3). Heterogeneity was low for dentin thickness, moderate for incisor volume data, and high for the CEJ-AC data sets. Other parameters not included in the meta-analysis assessed incisor mineral content, length, and pulpal space. Incisor calcium (Ca) and phosphorus (P) content was examined in 2 studies: while it was found to be generally unchanged in SF animals on the Cosmos-1887 mission (Simmons, Grynpas, and Rosenberg 1990), the Cosmos-1129 study demonstrated a more complex relationship (Rosenberg et al. 1984). The later study demonstrated that while overall Ca and P content in the entire dentin was greater in SF compared to GC, Ca in the inner half of dentin (formed during flight) was relatively deficient (Rosenberg et al. 1984). This is consistent with the observation that in SF mice on CRS-10, the pulpal space area was significantly increased, also potentially suggesting deficient dentin formation in SF animals (Maupin et al. 2019). Incisor length was increased in SF animals in the STS-131 mission but not in Bion-M1. Thus, there are indications that SF induces specific changes in teeth, but more studies are needed to understand spaceflight-induced changes in and around dentition.
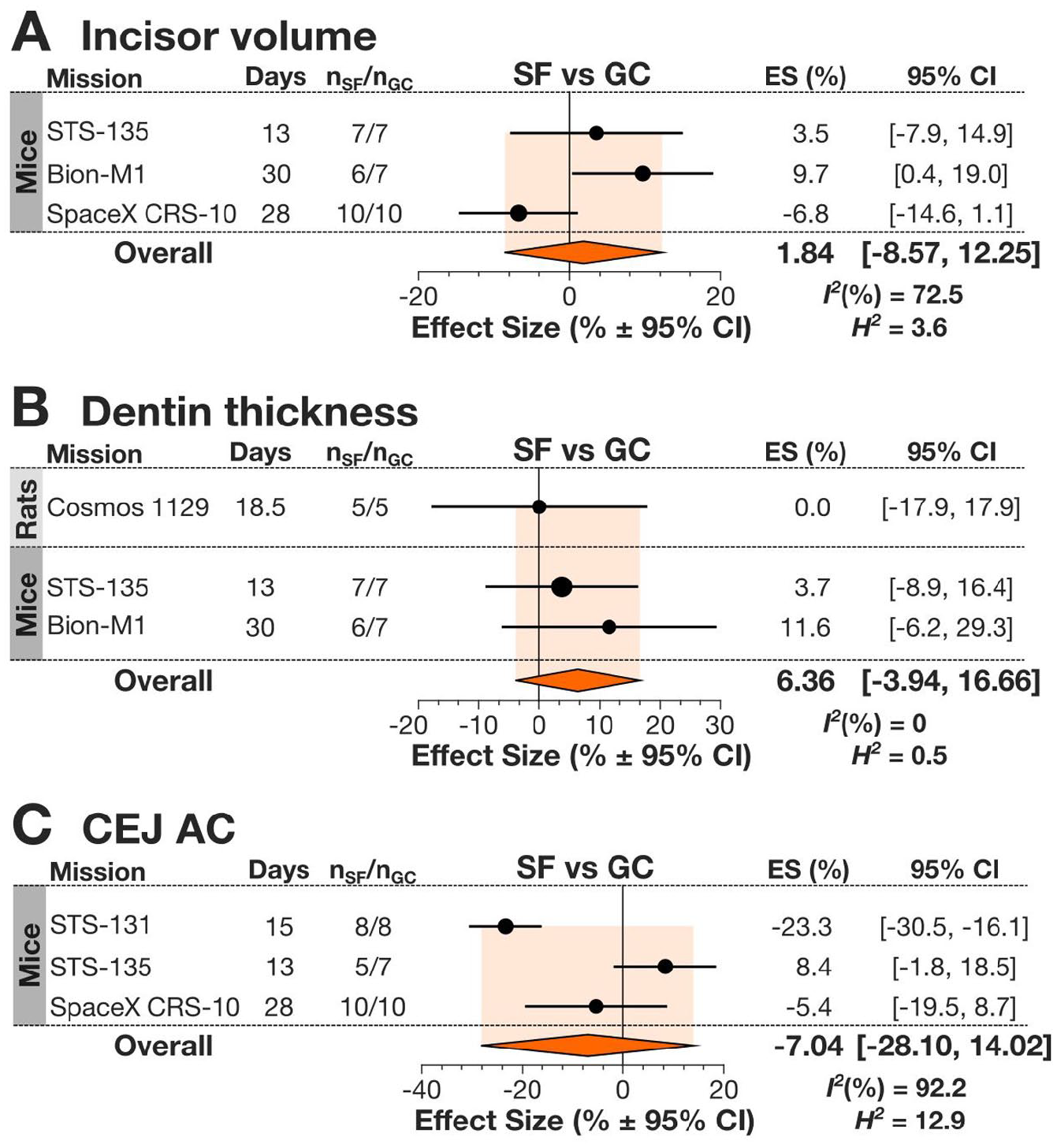
Spaceflight-induced changes to mandibular incisors in spacefaring rodents. (
Spaceflight-Induced Changes in Craniofacial Bone Turnover
The only bone turnover parameter suitable for the meta-analysis was the osteoclast surface per mandibular bone surface (Oc.S/B.S) reported on the septal bone of the first and second molars as well as the interradicular surface between the roots of the second molar in rodents. Overall Oc.S/B.S was not significantly different between SF and GC animals (Fig. 4). For this data set, we also analyzed the difference between GC and VC animals, which helps to account for factors other than microgravity that are associated with spaceflight. While overall GC and VC were not significantly different, at least in 1 mission (Bion-M1), Oc.S/B.S was significantly higher in GC animals compared to VC. Heterogeneity was low when comparing SF with GC but moderate when comparing GC with VC. A slight and nonsignificant decrease in other resorption parameters such as osteoclast number (Oc.N) and an increase in osteoid thickness on the formation side of tooth sockets were noted in flight group aboard Cosmos-1129, potentially suggesting decreased posterior drift of teeth (Simmons et al. 1983). Bone formation parameters were measured in rats in the Cosmos-1129 mission (Simmons et al. 1983), where mean calcification rates were decreased in the areas without muscle attachment. Thus, there are changes in mandibular bone remodeling in spaceflight, but more studies are needed to fully characterize them.
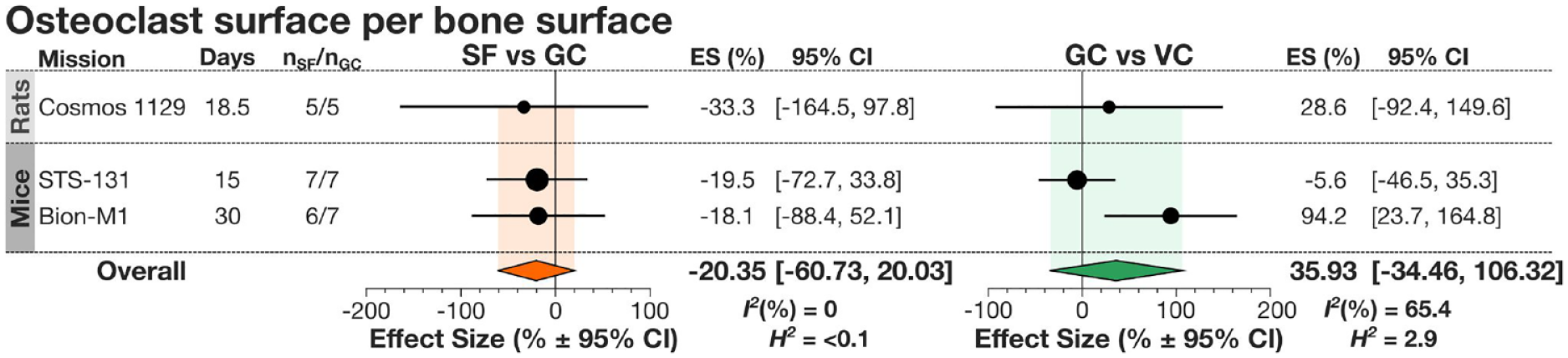
Changes in alveolar bone resorption in spacefaring rodents. Forest plot for changes in osteoclast surface per bone surface between spaceflight (SF) and ground control (GC) (left) and GC and vivarium control (VC) (right). Indicated are the species of spacefarers, missions, days spent in space, and number of spaceflight to ground control animals (nSF/nGC). Circles and lines represent the mission’s effect size (ES) (%) and 95% confidence interval (CI); the size of the circle is dependent on nSF. Overall effect sizes and CI are presented by diamonds. I2 and H2 were calculated for each parameter analyzed.
Discussion
We systematically reviewed and quantitatively synthesized available literature on craniofacial bone and teeth changes in spacefarers. We report that craniofacial bones react differently to spaceflight. We showed that in calvaria, there is a trend toward increased BV/TV and TMD in rodents and a significant increase in TMD in humans. In contrast, BV/TV significantly decreased compared to GC in mandibles of spacefaring rodents. Rodent incisors demonstrated no significant differences from GC in incisor volume or dentin thickness, but they were investigated in only a small number of missions. We identified a significant gap in knowledge regarding changes in mandible, maxilla, and dentition in humans, for which only the TMD for the whole skull was reported for a limited number of missions. Similarly, no data were found for changes in hard tissue of rodent maxilla or molars. Thus, we demonstrate that space travel may induce unexpected changes in craniofacial bones and identify a substantial gap in our knowledge of spaceflight-related changes in craniofacial hard tissues.
Previously, an association between bone loss and bone position relative to gravitational vector was identified in human space travelers (Oganov et al. 2005; Stavnichuk et al. 2020), where bone loss was most pronounced in lower limbs and bone gain was identified in the upper skeleton. We also demonstrated potential differences between the distal and axial regions in rodents (Fu et al. 2021). However, we now identify that 2 bones of the axial skeleton, calvaria and mandible, likely respond differently to spaceflight, with mandible significantly losing bone in space. The mandible, although nonweightbearing, is subject to mastication-induced mechanical loads, which may be negatively affected by spaceflight diet. Spaceflight diets evolved from the paste diets used early in missions to the rice-based solid diets and then to food bars developed by NASA to avoid crumbling while providing the consistency important for rodent teeth maintenance (Sun et al. 2014). It is known that on Earth, the use of soft diets for rats results in significant decreases in bone parameters in mandible but not maxilla (Shimizu et al. 2013). A decrease in masticatory force in the mandible has been shown to decrease mandibular growth (Bresin 2001). In our data sets, the mice aboard Bion-M1 were fed a paste diet, and those on STS-131 and STS-135 were fed the NASA food bar (Dagdeviren et al. 2018). If it is the softer food that results in lower mandibular mechanical loading, leading to the mandibular bone loss, then we should observe higher bone deficits in the Bion-M1 mission compared to the STS-135 mission. However, examination of data suggests that similar mandibular bone deficits were observed in Bion-M1 and STS-135 missions, suggesting that diet may not be the primary contributor to the mandibular bone loss. Another difference between the alveolar bone of the mandible and maxilla in craniofacial skeleton is in the remodeling rates. Rodent molars exhibit a natural physiological distal drift supported by high alveolar bone turnover rate (Vignery and Baron 1980), which potentially can make it more susceptible to environmental changes. Our analysis of bone turnover data indicates potential trends to decreased resorption and formation. This suggests a potential scenario where bone remodeling is suppressed in both mandible and calvaria in spaceflight, but in mandible, higher initial bone turnover rates result in its increased susceptibility to bone loss, while calvaria is either unaffected or exhibits anabolic response in space. More data directly comparing responses to spaceflight of different bones and bone regions in the craniofacial skeleton, while taking into account diet, age, and sex of the animals, are needed to answer this question.
Although we did not identify significant differences in incisor parameters in SF rodents, the data cannot be considered conclusive, since multiple confounding factors could not be taken into account. First, the diet was significantly different between the missions, as previously discussed. Second, age and sex of animals for which incisor parameters were reported were very different. If we consider the changes separately, we can see that incisor volume in the SF group was increasing in older males (Bion-M1), tended to decrease in younger males (CRS-10), and was unchanged in younger females (STS-135). To further complicate the interpretation, only the older male group was on the paste diet during the flight. Similarly, although no significant difference was found in the CEJ-AC, in 1 mission, which was also the only mission with older females (STS-131), CEJ-AC was significantly decreased (Ghosh et al. 2016). Thus, it is premature to conclude that teeth are not affected by the spaceflight, and more studies are needed to take into account all the potential confounding factors. In fact, several findings from individual studies suggest a potential detrimental effect of spaceflight on teeth. In the Cosmos-1129 mission, calcium content in dentin formed in space was demonstrated to be lower than in dentin formed on Earth (Rosenberg et al. 1984). In the CRS-10 mission, incisors of SF animals demonstrated an increase in pulp area with a corresponding decrease in hard tissue area in cross-sectional samples, which may be suggestive of altered morphology (Maupin et al. 2019). Moreover, changes in salivary glands have been described in spacefarers, which may also contribute to microgravity-induced changes in teeth, as saliva is well known for its protective function (Hand et al. 2020). Thus, more data are needed to fully understand the effect of spaceflight on teeth structure. It is also important to note that incisors in rodents are exceptionally different from humans—they are continuously erupting, and tooth enamel and dentin are constantly deposited by ameloblasts and odontoblasts, respectively (Goldberg et al. 2014). Unlike incisors, molar teeth in rodents demonstrate limited eruption and do not renew their dental tissues and thus are more similar to human teeth in their response to environmental changes (Goldberg et al. 2014). Thus, molars may present a more appropriate model for studying the effects of spaceflight on dental tissues.
The main limitation of this study was the lack of sufficient data reported on craniofacial hard structures in space missions for the past 50 y. Furthermore, no reporting was found for many craniofacial hard structures such as the maxilla; base of the skull; molar, premolar, and canine teeth; and the temporomandibular joint in any spacefarer. This scarcity in reporting prevented us from preforming analysis on many parameters measured in 1 or 2 missions. For animal studies, very few confounding factors could be investigated, since even for the very important ones, such as spaceflight duration, animal sex, and age, the data lacked sufficient spread. For example, no data were available for long-duration (>30 d) missions with rodents; the oldest animals in the data set were 23 wk old at the time of launch, which is barely reaching skeletal maturity, and no data for female rats were available (although some recent missions did include female mice). Thus, animal studies aiming to comprehensively assess the effects of spaceflight on the craniofacial complex are needed. For humans, even less data were available, with no information for specific craniofacial regions or for any teeth.
Our study suggests that to properly estimate the risk of dental events in space, in addition to mission duration and the number of travelers (Menon 2012), it may be important to account for spaceflight-induced changes in craniofacial structures. In similarly isolated Earth-based conditions, such as Antarctic and submarine missions (Lloro et al. 2019), as well as in trekkers in Nepal, where a dental event was calculated to occur once every 23.7 trekking days (Kupper et al. 2014), the most common dental issues were caries, tooth fractures, and periodontal health problems (Kupper et al. 2014; Lloro et al. 2019). Our data suggest that spaceflight-induced changes, including significant bone deficits in mandible and change in teeth morphology and composition observed in some studies, may act to increase the risks of fractures and caries progression.
We investigated the available literature on the effect of spaceflight on craniofacial skeleton. Our study demonstrates that this effect is complex, and the underlying mechanisms remain obscure. Based on the previous studies that implicated fluid shifts as a strong contributor to bone loss in spaceflight (Colleran et al. 2000; Oganov et al. 2005; Stavnichuk et al. 2020), we had expected to see similar changes in all bones in the skull. However, the mandible showed a tendency for bone loss, while calvariae demonstrated bone gain, suggesting an interplay between different driving forces. Importantly, our study demonstrates significant knowledge gaps regarding many structures of the craniofacial complex, such as the maxilla, molar, and premolar and canine teeth, as well as small sample sizes for the studies of mandible and incisors. Concrete understanding of the effects of microgravity on craniofacial structure is important for understanding the risks of space travel for oral health and for developing the strategies to mitigate these risks as humanity continues to explore the cosmos.
Author Contributions
M.S. Moussa, contributed to conception, design, data acquisition, analysis, and interpretation, drafted and critically revised the manuscript; M. Goldsmith, contributed to conception, design, data acquisition and analysis, drafted the manuscript; S.V. Komarova, contributed to conception, design, data analysis and interpretation, critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of the work.
Supplemental Material
sj-docx-1-jct-10.1177_23800844221084985 – Supplemental material for Craniofacial Bones and Teeth in Spacefarers: Systematic Review and Meta-analysis
Supplemental material, sj-docx-1-jct-10.1177_23800844221084985 for Craniofacial Bones and Teeth in Spacefarers: Systematic Review and Meta-analysis by M.S. Moussa, M. Goldsmith and S.V. Komarova in JDR Clinical & Translational Research
Footnotes
Acknowledgements
We thank an academic librarian, Martin Morris (Schulich Library of Physical Sciences, Life Sciences and Engineering, McGill University, Montréal, Canada), for help with developing the search strategy.
A supplemental appendix to this article is available online.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by operating grants from the Canadian Space Agency (19HLSRM04) and Natural Sciences and Engineering Research Council (NSERC, RGPIN-288253) to S.V. Komarova. M.S. Moussa and M. Goldsmith were supported by a graduate scholarship from McGill Faculty of Dentistry.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.