Abstract
The Inpatient Experience with Nursing Care Scale (IPENCS) is a valid and reliable instrument for measuring patients’ experiences with nursing care. This study aimed to develop and validate a short-form of this scale. To ensure the experiment was scientific and rigorous, the short-form IPENCS was developed and validated by both qualitative and quantitative methods in three phases. Phase one is to develop the short version of the instrument. Phase two is to evaluate the validity and reliability of the short-form IPENCS. Phase three is to conduct a cognitive interview (CI) to evaluate the comprehensibility of the short version. 1565 valid questionnaires were received and the response rate was 75.02%. Thirteen items were excluded under four methods of simplification. The 17-item IPENCS showed good reliability and validity. Less completion time and high acceptance about the scale length were also found in the CI. The short-form IPENCS showed acceptable measurement properties which was regarded as an alternative to the original scale to provide rapid assessment and quality improvement suggestions.
Introduction
Traditionally, healthcare providers have put more emphasis on professional skills and advanced technology, regarding them as crucial elements to improve healthcare service quality. 1 However, as medical patterns shift from “disease-centered” to “patient-centered,” patient experience has become a fundamental composition and a direct reflection of patient-centered care. 2 It focuses on patients’ actual experiences and aims to avoid subjective judgments (eg, how often did nurses give clear information about how to exercise appropriately). 3 Patient experience currently is one of the most common indicators used to evaluate the healthcare quality worldwide. 4 Some countries like USA and England even linked patient experience results to financial reimbursement from Medicare and other insurers for promoting quality improvement in hospitals. 5
Measuring patient experience with nursing care is also very important and necessary. Numerous studies have shown that nursing service is the most important predictor of patients’ overall satisfaction with their hospital experience.6,7 In 2022, a validated instrument measuring patient experience with nursing care, the Inpatient Experience with Nursing Care Scale (IPENCS), was developed under the guidance of the theoretical framework of “I accelerate.” 8 The “I Accelerate” framework emphasizes nurses’ multifaceted roles in meeting the holistic needs of patients and building the “therapeutic relationship” with patients. The IPENCS is composed of 30 items with seven domains. The original version of the IPENCS demonstrated strong psychometric properties, including high internal consistency (Cronbach's α = 0.95) and good content validity (item content validity index (I-CVI) = 0.96). Factor analysis confirmed its structural validity, with Exploratory factor analysis identifying a seven-factor model and Confirmatory factor analysis supporting this with acceptable fit indices (CFI = 0.965, TLI = 0.960, RMSEA = 0.043). 9 It has also been widely used in more than 20 medical institutions and became the specification for evaluating inpatient experience with nursing services (No.T/CRHA 064—2024) in China. 10
Ample evidences have shown that patients definitely have the capacity to provide reliable statements and valid information about their experience of service quality, efficiency, and outcomes. 2 The significant problem for patients is how to ensure their response quality. Scale length is a major barrier to its completion, often contributing to low-quality responses and survey fatigue.11–13 Shorter tools have been shown effective means to reduce fill-in burden and increase the accuracy of the results.14,15 For example, short-form Hong Kong Inpatient Experience Questionnaire (SF-HKIEQ) 2 and the Picker Patient Experience Questionnaire-15 (PPE-15) 16 were both created from longer instruments. These short forms required less completion time, reduced missing values, and ultimately enhanced feasibility for large-scale surveys. 17 Few scales were developed to measure patient experience with nursing care in China or globally, not to mention the development and validation of short-form versions. Hence, a validated short-form of IPENCS is needed to expand its applicability in fast-paced clinical settings.
In recent years, considerable attention has been devoted to advancing methods for simplifying scales to enhance their utility and efficiency in research practices. 18 The methods used for shortening questionnaires vary, primarily including factor analysis, item response theory (IRT), and machine learning techniques. 19 Some researchers also suggested that a shortening process must combine qualitative content analysis and quantitative psychometric analysis. 20 Since there are no standardized methods for questionnaire shortening, we built upon existing literature and guidelines to develop a set of criteria for item selection, incorporating classical test theory (CTT), IRT, and cognitive interviews (CIs).
Thus, the aim of this study was to develop and validate a short version of the IPENCS and test its comprehensibility among patients.
Methods
Study Design
The short-form IPENCS was developed and validated by both qualitative and quantitative methods in three consecutive phases.
Participants
Eligible study participants were adult inpatients admitted and discharged in one tertiary hospital in China from February 2023 till July 2023. Convenience sampling was used as the sampling method due to logistical constraints, including limited time and resources, and because the primary aim of this early psychometric work was preliminary validation rather than population representativeness. The inclusion criteria of the inpatients were as follows: (1) Age ≥ 18 years old; (2) Hospitalization time ≥ 2 days; (3) Clear awareness and stable condition, able to clearly express one's own views.
Patients with severe complications or mental health conditions were excluded.
In the study period, the IPENCS questionnaire was sent to 2086 patients. The response rate was 75.02%. Characteristics of the participants are presented in Table 1. Purposive sampling was used to include another 13 patients with different education levels, disease types, and ages in the CI to ensure that all individuals with varying health literacy could understand the short-form questionnaire.
Characteristics of the Participants (n = 1565).
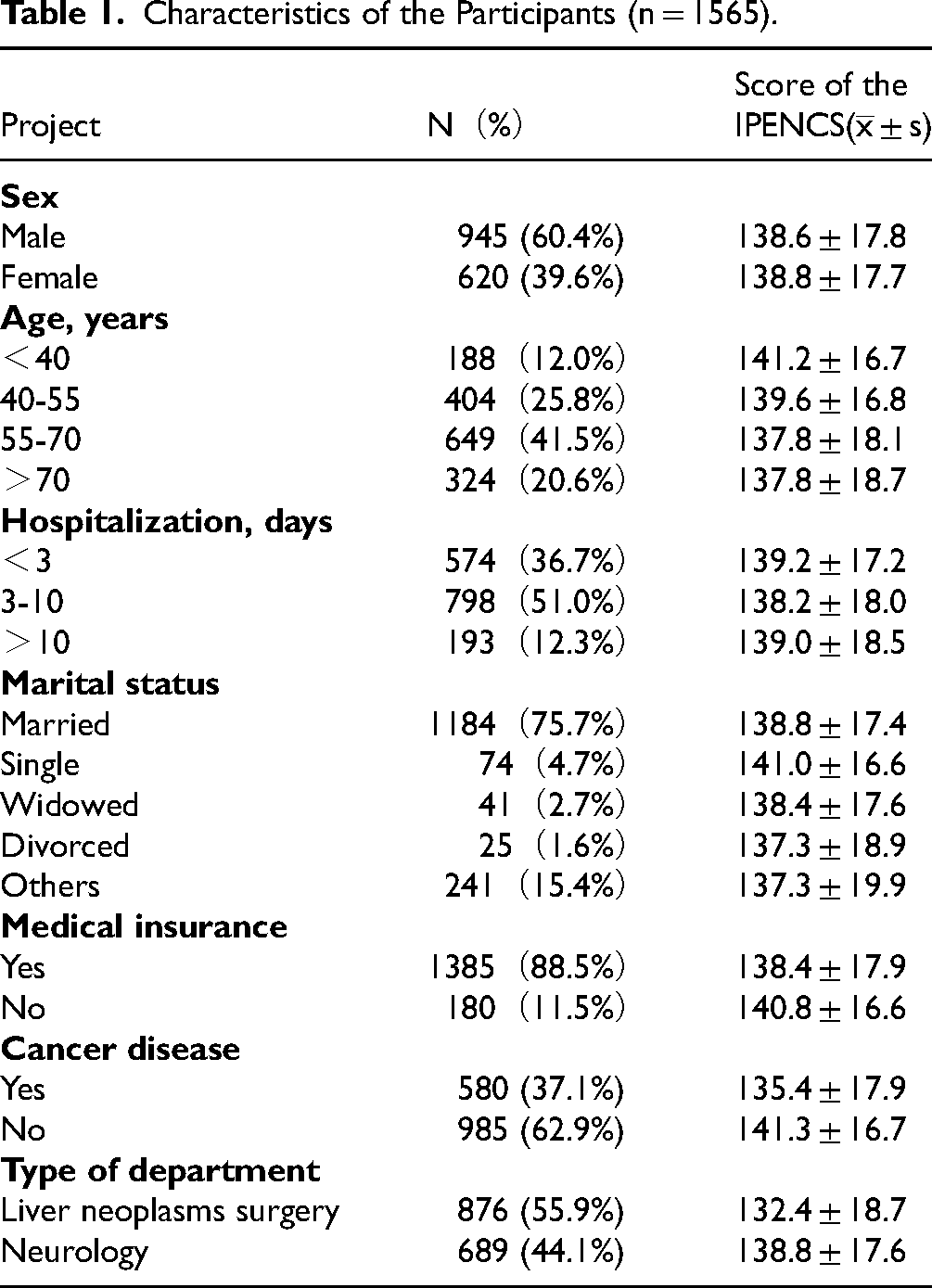
Experts with the criteria below were also invited in the phase one to develop the short-form of IPENCS: (1) engaged in clinical nursing, nursing education, and nursing management work for at least 10 years; (2) bachelor degree or above, senior professional, and technical positions; (3) be familiar with and had researched in patient experience area. Twelve experts, including patient experience research experts (six people), instrument developer experts (two people) and nurse managers (four people) were finally invited to participate in the consultation.
Short-Form Development and Validation
Phase One—Development of Short-Form Inpatient Experience with Nursing Care Scale
Reducing items from a validated instrument should be done cautiously. Several criteria need to be considered to maintain the reliability and validity, which implies that elimination must not lead to substantial loss of information. As a result, four methods were utilized simultaneously and independently in the simplification process.20,21 Preset criteria specified that the item would be eliminated if it failed to meet two or more criteria.
First, items with a score of >10% “not applicable” and/or “missing” were removed from analysis.
Second, multiple regression analysis was applied with the sum of the scores as outcome variables and the score of each item as individual predictor variables. The adjusted R2 was considered to provide information about the performances of predictor variables maintained in the regression model. The primary objective was to guarantee nonredundancy in the sense that the regression model of the individual items of the final version, as predictor variables, should explain at least 50% of the variance of the total score.
Third is the expert consultation. Twelve experts from the patient experience research area were invited to score the importance of each item in the original scale (1 being very unimportant, 2 being not important, 3 being neither important nor unimportant, 4 being important, 5 being very important). I-CVI, the number of experts giving a 4- or 5-point evaluation for each item divided by the total number of experts, was generally expected to exceed 0.78 22 which was regarded accepted or remained.
Last method is the IRT. According to the scoring characteristics of IPENCS scale, this study selected the Graded Response Model (GRM) in IRT and used Marginal Maximum-Likelihood Estimate to estimate the parameters of each item. The items were screened according to three properties: Item Discrimination (a), Item Difficulty (b), and information of each item.
Phase Two—Validation of Short-Form Inpatient Experience with Nursing Care Scale
Pearson correlation analysis between the original 30-item scale and the 17-item short form and Cronbach's alpha was used to validate the validity and reliability of the short-form IPENCS.20,23,24
Phase Three—Cognitive Interview
Cognitive interviewing is a formal method used to assess the clarity of questionnaire items and ensure they are comprehensible to respondents. It is a cost-effective technique to provide evidence for the content validity of items. It can help to detect issues related to clarity, comprehension, ambiguity, cognitive recall burden, timeframe, missing answer categories, inaccurate instructions, and relevance of questionnaire items. 25
The primary methods employed during CI include thinking aloud and verbal probing. 26 Thinking aloud involves the researcher taking on the role of observer and encouraging the participant to spontaneously verbalize their thoughts in response to a question. After the participant finished the whole scale, researcher would immediately calculate the questionnaire completion time. Then the interviewer asked the respondent probing questions based on the cognitive interviewing framework: Comprehension, Recall, Judgement, and Response. 27 Hence, probing in this study was conducted using the retrospective technique. 27 This entails asking patients to explain the meaning of specific terms in the items, prompting them to restate item stems in their own words and asking retrospective questions about how they made choices based on past experiences. Any words or problems which were found to be difficult and their overall impressions of the scale during the interview process were reported finally. Each interview outline was tailored based on related literature review.26,28–30 Take the item “How often did nurses give clear information about how to diet appropriately?” as an example, the interview outline is detailed in Supplemental material Table 1 .
Data Collection
For the quantitative study, investigators were trained centrally by the researcher for data collection. A total of 10 team members were involved in data collection, and they were blinded to the specific study hypotheses to minimize potential bias. Data collection was done on the discharge day. The questionnaire was administered after the data collector obtained informed consent. After completion, the researcher checked the questionnaire and collected it.
For the CI, a face-to face interview was used in a quiet and independent space. Each interview was in a duration between 40 and 50 min. Recording devices were used under the informed consent of patients. For each interview, one team member led the whole interview process while another team member calculated the survey completion time and took notes for subsequent data analysis. After the researcher explained the detailed information about the study aim and the IPENCS, participants finished the scale by reading the item aloud along with their chosen response. Then one team member measured the survey completion time and another launched the probing process to elucidate the participant's comprehension of each item.
Data Analysis
Quantitative data were recorded and examined in Excel and were analyzed by using IBM SPSS Statistics 25.0. Missing data were handled by using multiple imputation. The counting data is described using frequency and percentage, while the measurement data is described using mean ± standard deviation. R statistical environment, version 3.6.1 was used to perform the IRT analysis. CI data was recorded but not transcribed. Detailed notes were taken during each interview and organized by item in Excel. Two researchers independently coded the notes by using a content analysis approach. When uncertainties arose, the corresponding audio recordings were reviewed to clarify participants’ responses. Consensus was reached on the revisions for items among all the team members.
Criterion validity was analyzed by using Pearson's correlation. Coefficient of <0.1 indicated a negligible and >0.9 a very strong relationship. 31 Internal consistency reliability was evaluated by Cronbach's alpha. The level of internal consistency generally regarded as adequate is over 0.7. 24
I-CVI, the number of experts giving a four- or five-point evaluation for each item divided by the total number of experts, was used to assess content validity of the instrument. It was generally expected to exceed 0.78, assuming that as least 9 of the 12 experts agreed the importance of the item.
IRT was assessed by the item discrimination, item difficulty and information. Item Discrimination (a) indicates the ability to distinguish potential traits of each item, which should be between [0.3,5]. Item Difficulty (b) should be in the range of [-4,4] (Item Difficulty b consists of 4 values, b1 ∼ b4 should meet the above criteria, and should show a monotonically increasing trend in this interval). The amount of information provided by the entire scale is less than 16 indicating that the assessment item is poor, 16 to 25 indicating that the item needs to be improved, and more than 25 indicating that the quality of the assessment item is good. Therefore, the information content of an item >0.83 (25/30) was judged as excellent, < 0.53 (16/30) was judged as poor/to be improved, and 0.53 ∼ 0.83 was judged as good in this study.32–35
Ethics
This study was approved by the Medical Ethical Review Board of Zhongshan Hospital Fudan University. Signed informed consent was required and collected. The survey data and information obtained were kept strictly confidential and only limited to research use.
Results
Phase One—Development of Short-Form Inpatient Experience with Nursing Care Scale
During the study period, two items with a not applicable score of >10% (range 4%-13%) did not meet the criteria. For I-CVI, they ranged from 0.33 to 1. For multiple regression analysis, adjusted R2 ranged from 29% to 75.4%. The explained variances on each item and total score mostly were satisfactory, except for the item 2, 5, 9, 10, 19, 20, 24, 25, 26, and 28. Detailed information is shown in Table 2.
Results of “not applicable” Rate, I-CVI and Multiple Regression Analysis.

*means not meet for the criteria.
aItem content validity indexes.
bMultiple Regression Analysis measured the correlation of each item score with the total score.
I-CVI, item content validity index.
Prior to conducting the IRT analysis, confirmation whether items violated the basic unidimensional assumption of the GRM model needed to be done. It was suggested that items would be properly examined for unidimensionality if the ratio of the first and second eigenvalues was over three by applying the principal component analysis. The results of the unidimensionality test were 18.084/1.850 = 9.775>3, so the unidimensionality of the scale was suitable to conduct an IRT research. 36 Additionally, another assumption of local independence was automatically examined when unidimensionality was satisfied with these items. The GRM evaluated item discrimination (a) and item difficulty (b) based on participants’ response patterns regarding the IRT model ( Supplemental material Table 2 ). Overall, the discrimination (a) was high, ranging from 1.844 to 6.479. This indicates that the items were well differentiated at the trait level, but some of the differentiations exceeded 5, suggesting that these entries could be further improved. The item difficulty (b) ranged from −3.423 to −0.446, showing a monotonic increase with difficulty level (b1-b4), and there is no inverse threshold. The average information ranged from 0.3774 to 2.5218.
Combining the above four methods and screening items that did not meet two or more criteria, 13 items were eliminated and 17 items were retained. The final shortened form is shown in Supplemental material Table 3 .
Phase Two—Validation of Short-Form Inpatient Experience with Nursing Care Scale
Short-form version showed high correlations (0.994) with the full IPENCS for total score. Among seven domains, all correlations remained high while domain5: Responding to requests was slightly lower (0.824). This may be due to the exclusion of items that captured nuanced aspects of responsiveness. Cronbach's alpha of the short version exceeded 0.9 and a bit lower than the original one which all showed excellent reliability (Table 3).
Total, Domain Scores and Correlation Between Full and Short-Form IPENCS.
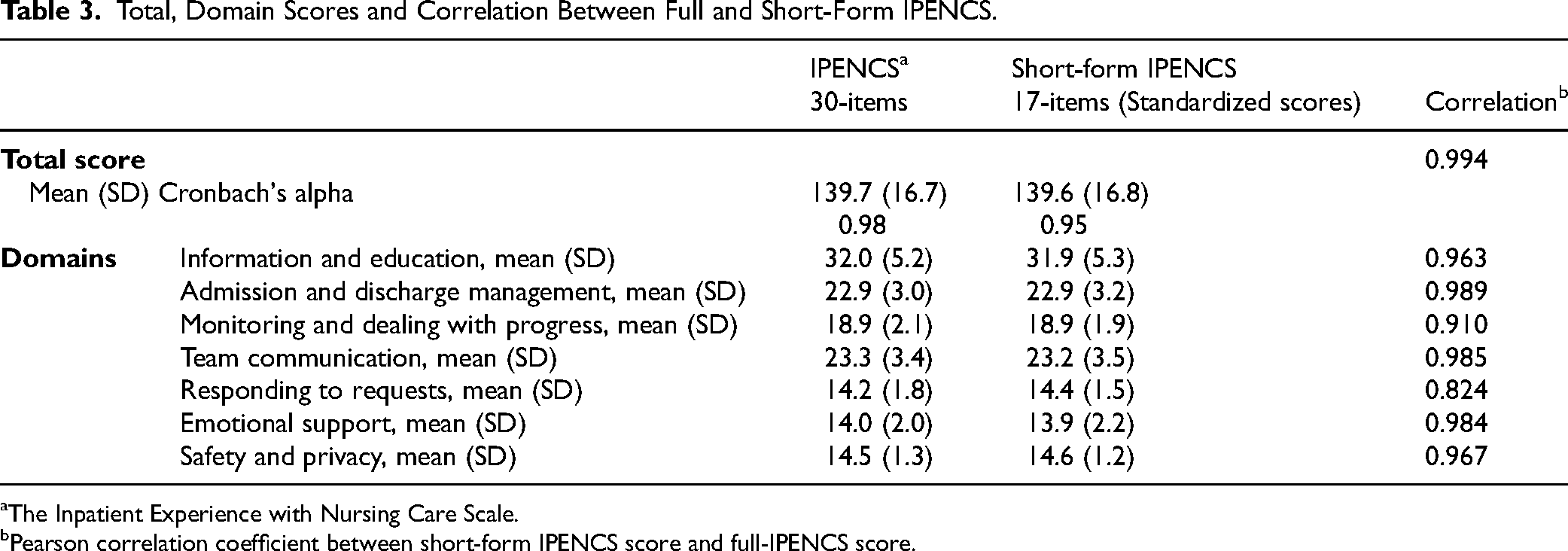
aThe Inpatient Experience with Nursing Care Scale.
bPearson correlation coefficient between short-form IPENCS score and full-IPENCS score.
Phase Three—Cognitive Interview
On average, the participants spent 11 min (SD: 2 min) completing the short version which was significantly shorter than the original scale completion time (22 min, SD: 4 min). This significant reduction represented a major improvement in the usability of the instrument.
Eight respondents were included in the first round of CI and most participants indicated that nearly all items were clearly expressed while five items had some problems in expression, order and meaning which need to be revised. Another five respondents were included to re-verify the comprehensibility of the scale. All five respondents indicated that they could totally understand the entire items and could select the option easily based on their own experience. The number of 17 items was also considered to be acceptable for most participants. Five items were discussed and revised through expert consensus based on the suggestions from the interviewees. The detailed information is reported in Table 4.
Issues Identified and Corrections of the Scale in the CI.

CI, cognitive interview.
Discussion
The study identified 17 core items from the original IPENCS to provide a shorter version for both patients and healthcare stakeholders. A 5-Likert-type scale ranging from 1(never) to 5(always) was offered to patients to answer the questionnaire. We demonstrated that the short-form IPENCS is a reliable and valid measure of patient experience with nursing care.
The original IPENCS has well-documented measurement properties, 9 so the choice of original scales for simplification is compelling. 20 Four methods were used to select the eliminated items to develop the short-form IPENCS, including the “not applicable” rate, expert consultation, multiple regression analysis, and IRT. A total of 17 items remained to form the short version. The shortened scale significantly reduced completion time and respondent burden, as confirmed by participant feedback during CIs. Additionally, short-form version demonstrated high correlations with the full IPENCS for total score as well as individual domains (Pearson's correlation coefficient ranged from 0.824-0.994). The short-form also maintained excellent internal consistency (Cronbach's alpha = 0.95), closely aligned with the original scale (0.98).
A large body of previous studies has shown that the need of using modern statistical techniques is highly recommended, such as IRT. It is a set of statistical models that describes the association between latent constructs (eg, a person or respondent's “ability” or “trait”) and observed outcomes (eg, responses to items). 37 The IRT results in our study provided a set of interpretive tools (item discrimination, item difficulty, and information of each item) that were useful for scale refinement. Simplification process also highlighted the complementary relationship between IRT and CTT methods. 38 Traditional descriptive statistics and multiple regression analysis combined with detailed item-level information from IRT provided researches many useful perspectives for scale simplification in this study.
Short forms can broaden the application of a measure by reducing assessment time, costs, and the risk of missing items. They are well suited for use in large-scale testing efforts to reduce respondent burden and improve the quality of test results. However, it's important to note that the original scale will not totally be replaced by the short scale. In certain studies requiring deep data analysis, longer and more comprehensive scales with more items might be a better choice. Short forms are more suitable for frequent or large-scale applications like routine audits, while the original scale may be preferable for detailed audits or research. Therefore, researchers must understand the pros and cons of both long- and short-form instruments and choose the appropriate tool based on their needs and study design. 23 In this study, short-form IPENCS was developed for large-scale application to reduce patient burden and completion time, whereas the original scale remains more suitable for small-sample studies requiring a comprehensive assessment of patient experience.
This research has some limitations. Firstly, the short-form scale development and validation research was conducted in a single tertiary hospital, which may restrict applicability across diverse healthcare settings. Secondly, no external or follow-up validation of the short form was conducted after finishing the phase three—CI, which may limit the robustness of the psychometric conclusions. In this study, same data collected with the original IPENCS was analyzed to develop and validate the short-form version which may lead to a possible halo effect. 20 Using the same dataset for both item reduction and validation may introduce a halo effect—potentially overestimating the scale's reliability due to lack of independent testing. Future research should conduct more rigorous psychometric testing and use the COnsensus-based Standards for the selection of health Measurement Instruments standards as a reference to ensure greater credibility of the instrument. Moreover, the researcher found the differences in how respondents used the 5-point Likert scale. For example, some people have higher expectations and requirements for hospital services, which lead to the low scores, so how to keep the questionnaire filling in the consistency of the questionnaire understanding is also a difficult but worthwhile problem for future study.
As a whole, 17-item IPENCS can be a time-efficient alternative to the original scale for both patients and nursing managers, especially in time-intensive survey and large-scale research. 39 And both instruments have the ability to measure the patient nursing care experience and guide the quality improvement initiatives.
Implications for the Future Study
Recently, some researchers have applied machine learning approaches to the study of simplifying psychometric tools as the development of artificial intelligence. 14 It offers a promising approach for simplifying scales with high administrative efficiency, score comparability, and good psychometric properties. 40 Additionally, further research should evaluate the generalizability of simplified scales across diverse clinical departments and patient demographics. 41 Cross-cultural applicability should be also assessed in other healthcare systems and linguistic contexts.
Conclusion
The short-form IPENCS was developed to measure the patient experience with nursing care with fewer items in large-scale use. It showed high acceptance among patients in CI, along with evaluated validity and reliability. The short-version instrument has the potential to be a rapid and accurate tool for health policy makers and healthcare providers; however, further investigations are needed to confirm its efficiency in practical use.
Supplemental Material
sj-docx-1-jpx-10.1177_23743735251415067 - Supplemental material for Development and Validation Study of a Short-Form Version of the Inpatient Experience with Nursing Care Scale
Supplemental material, sj-docx-1-jpx-10.1177_23743735251415067 for Development and Validation Study of a Short-Form Version of the Inpatient Experience with Nursing Care Scale by Yichen Kang, Xiao Chen, Yuhong Zhang, Meijuan Lan and Yuxia Zhang in Journal of Patient Experience
Footnotes
Abbreviations
Availability of Data and Materials
The data that support the findings of this study are available from the corresponding author, upon reasonable request.
Consent to Publication
Written informed consent was obtained from the patient for publication of this study.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Approval and Consent to Participate
This study was approved by the medical ethical review board of Zhongshan Hospital (clinical trial number B2022-613R). Signed informed consent were required and collected from individual or guardian participants. The survey data and information obtained were kept strictly confidential and only limited to research use.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by National Natural Science Foundation of China ( grant number 72204053) and the Science and Technology Innovation Plan Of Shanghai Science and Technology Commission (grant number 22692109400).
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
