Abstract
Keywords
Introduction
Approximately 23% of US adults suffer from degenerative knee conditions such as osteoarthritis (OA), 1 and prevalence is expected to rise by 49% by 2040. 2 Nonsurgical treatments for knee OA (KOA) include weight loss, exercise, physical therapy (PT), injections, and oral medications. 3 The American Academy of Orthopedic Surgeons guidelines describe PT and steroid injections as having strong to moderate evidence bases. 4 One randomized controlled trial (RCT) found that PT was superior to glucocorticoid injection in reducing pain at one-year follow-up. 5 A study comparing a placebo injection-then-PT versus corticosteroid injection-then-PT found no difference in pain scores at 14 weeks postbaseline. 6 Research comparing injection to PT to combined injection and PT is inconclusive.
The Choosing Optimal Physical Therapy or Injection Nonoperative Guidance (COPING) study was an RCT for patients with KOA aimed at contributing to this literature. Patients were randomized to (1) intraarticular triamcinolone injection, (2) PT, or (3) intraarticular triamcinolone injection followed by PT. We measured clinical outcomes at 6, 12, and 52 weeks postrandomization. Primary outcomes included pain, function, and strength measured with patient-reported outcome measures, musculoskeletal exams, and questionnaires. Patients in groups requiring PT were also asked to complete home exercise logs. Prescreening 581 patients over 5 months resulted in an enrollment rate of 7.5%. Low enrollment in clinical trials leads to concerns about the generalizability of study findings and selection bias. Many studies with low enrollment rates are never published. 7 Due to low enrollment, we suspended recruitment and focused instead on identifying reasons why otherwise eligible patients declined to enroll in the RCT.
Few studies have explored the willingness of KOA patients to enroll in an RCT of conservative treatment and the impact of past treatment on enrollment decisions. Factors influencing patient treatment preferences may include cost, impact on daily life, and perceived adverse effects of treatment. 8 A prospective preferences assessment among patients with degenerative meniscal tear and KOA found that patients with higher educational attainment and those with no treatment preferences were more willing to participate in an RCT. 9 To explore these dynamics further in our patient population, we conducted a secondary analysis of screening and prerandomization data to identify factors associated with patients’ decisions to enroll in an RCT of conservative KOA treatment. Specifically, we sought to (1) examine whether demographic characteristics, treatment preferences, and treatment history correlated with enrollment decisions, and (2) identify treatment preferences among those who declined study enrollment.
Methods
We conducted a secondary analysis of screening and prerandomization data over a 5-month recruitment period of the COPING trial. Eligible patients were English-speaking adults aged 45 to 85 years old who presented with knee pain and a Kellgren Lawrence (K-L) grade of 2 to 4 on weight-bearing anteroposterior view (AP) x-rays at a single university-affiliated orthopedics and sports medicine practice.
To identify factors associated with patients’ decisions to enroll in the COPING trial, we reviewed medical charts for demographics and past treatment history. At the time of RCT invitation, patients were asked why they declined enrollment. Data were extracted from the medical record, prescreening form, and from the screening call notes and were entered into the Research Electronic Data Capture (REDCap) tool hosted at our university (University at Buffalo). 10 Assessment of data accuracy was conducted primarily by a research team of 4 (1 research scientist, 1 research assistant, and 2 medical students), with input from clinical research team colleagues, including one orthopedic surgeon and one primary care sports medicine physician.
The total sample (n = 134) of patients was grouped by enrollment status, then compared by demographic and clinical characteristics. Patients who declined enrollment were grouped by treatment decision and then compared by treatment history. A P-value < .05 was considered statistically significant for the enrolled and declined comparison, and < .008 (.05 divided by 6 pairs) was used for the declined patient subanalyses by treatment decision. All analyses were performed using SPSS Version 29 (IBM Corp). The reporting of our study follows the Standards for Quality Improvement Reporting Excellence (SQUIRE) guidelines for reporting healthcare research. 11
Results
Of those who were eligible for the trial, 10 enrolled (7.5%) and 124 declined. Prescreened eligible patients were 52% female, on average 62 years old, primarily identified as White (89.6%), and had an average BMI of 30.6. There were no statistically significant differences in demographics or treatment history between the enrolled and declined groups (Table 1). Of the 10 enrolled, 7 were lost to follow-up and 3 completed all study activities.
Demographic and Clinical Characteristics Among Otherwise Eligible Participants by Enrollment Status Into the Choosing Optimal Physical Therapy or Injection Nonoperative Guidance (COPING) Trial, December 2022 to April 2023, n= 134.
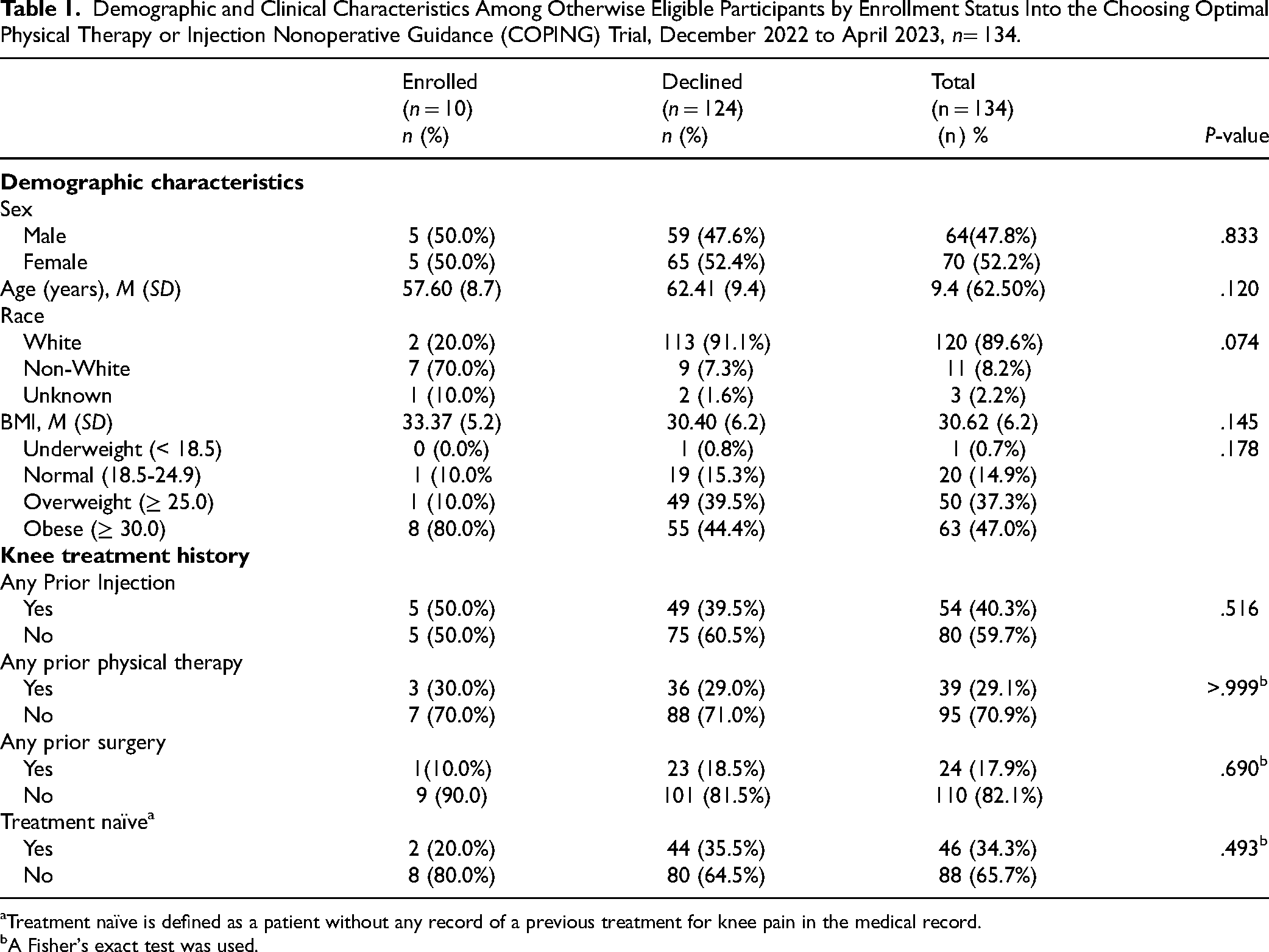
Treatment naïve is defined as a patient without any record of a previous treatment for knee pain in the medical record.
A Fisher's exact test was used.
Among those who declined enrollment (n = 124), 46% received an injection alone, 29% received a PT referral alone, 15% received an injection and PT referral, and 10% chose another treatment option (Table 2). Treatment history appeared to be associated with the patient's treatment decision at the time of their visit. Average BMI varied across treatment groups (P = .006). On pairwise comparison, the injection-only group had a higher average BMI when compared to the PT-only group (mean difference = 4.56, P = .003). Differences were also observed in treatment history by treatment groups, including history of injection (P < .001), history of surgery (P = .026), and treatment naive (P = .006; Table 2). Patients who received a PT referral were less likely to have had a prior injection (2.8% vs 68.4%, P < .001), were more likely to have had prior surgery, and were more likely to be treatment naïve (52.8% vs 19.3%, P = .006) than the injection-only group (Table 2). Additionally, patients with PT referrals only were less likely to have received a prior injection as compared to the injection and PT referral combination group (Table 2).
Demographic and Clinical Characteristics by Current Treatment Among Otherwise Eligible Participants Who Declined Participation in the Choosing Optimal Physical Therapy or Injection Nonoperative Guidance (COPING) Trial, N = 124.
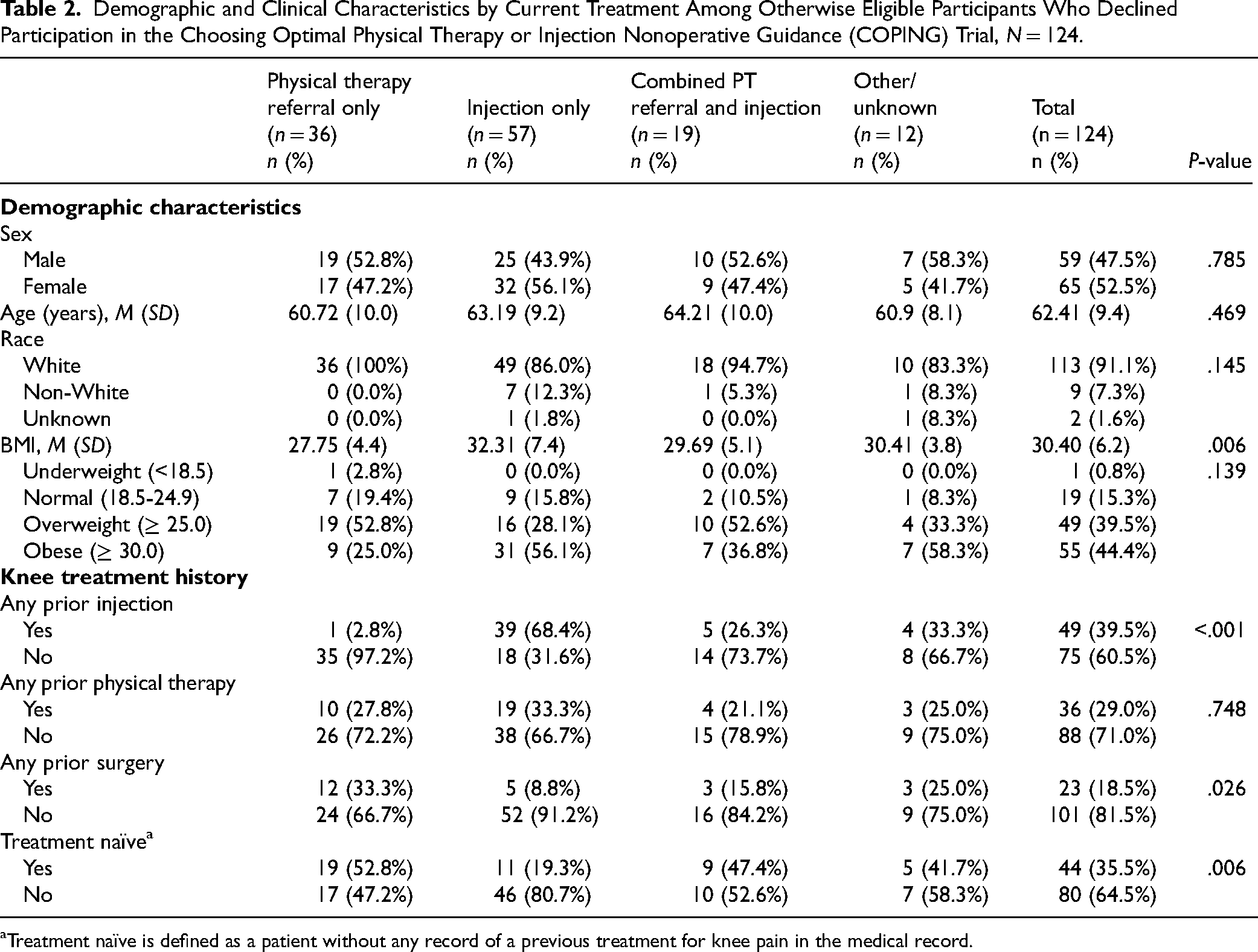
Treatment naïve is defined as a patient without any record of a previous treatment for knee pain in the medical record.
Patients who were approached for possible trial enrollment also provided study staff with multiple reasons for their decision to decline participation in the study. Some were related to treatment experiences. For example, a number of patients said they preferred injections because they had responded well to them in the past; others stated that they wanted to avoid injections altogether. Some patients expressed a preference for PT and therefore declined to enroll in the RCT. A handful of patients also declined due to logistical reasons (eg, travel).
Discussion
Our study found that many patients have treatment preferences and have low randomization tolerance. Among patients who declined study participation, a history of injection was associated with a higher likelihood of receiving another injection. Patients may perceive injections to be more immediately effective and convenient as compared to the demands of PT. 12 Posnett et al 13 found that patients were willing to pay a premium for treatments, often injections, that they deemed more effective, longer lasting for pain relief, and that could be administered in fewer office visits. Another study of correlates of the likelihood of RCT enrollment found that while demographic characteristics did not vary between enrolled and declined groups, psychosocial factors did; anxiety symptoms were associated with lower rates of enrollment, and higher perceived self-efficacy, curiosity, and social support were associated with higher rates of enrollment13. Future studies in the area of KOA could address these factors as well.
Limitations
Recruitment for our study came from a single suburban orthopedic clinic among predominately White, English speakers, which could limit the generalizability of its findings to the general KOA population. Future iterations of this work should include multiple clinical sites and recruit a more diverse patient population. Additionally, we urge caution when interpreting the findings related to our comparison between enrolled versus declined groups; unequal sample sizes may have limited our ability to draw statistical conclusions, and we may have missed differences that indeed exist. Further, the COPING trial was not designed to answer questions about study design and enrollment preferences; data reported in this manuscript came from a secondary analysis of screening data.
Conclusion
Our study suggests that KOA patients with an injection history want to continue that course of treatment, treatment naïve patients may prefer PT initially, and many patients are not open to relinquishing control over KOA treatment decisions. In our secondary analyses, we identified that these factors contributed to the difficulty we experienced in recruiting for the RCT. To improve this outcome, future researchers may consider a patient preferences trial to allow more enrollment flexibility than a traditional RCT. 14 Additionally, researchers could conduct a formal patient preferences assessment9,15 to investigate potential barriers, concerns, and patient skepticisms related to KOA treatments. Researchers and clinicians may be well-served by having conversations with patients about their concerns and questions regarding KOA treatment prior to study invitation so that these concerns can be creatively addressed to support successful study enrollment and completion. One way to do this would be by adding a patient advocate 16 or patient navigator 17 to the study team during its design and implementation phases. This individual could support patients throughout any treatment modality they are assigned to in the trial (eg, obtaining transportation or health coverage related to PT). Orthopedics researchers and clinicians can strengthen their practices by adopting strategies demonstrated in oncology research that have successfully integrated patient navigators into their care and clinical trials. 17 Integrating patient input, advocacy, and treatment choice may improve study design in orthopedics and other related fields.
Footnotes
Abbreviations
Acknowledgments
We would like to thank Cassandra Sherman, MPH, Kathleen Lafferty, AAS, Peter Tonsoline, PT, and Brian DeLuca, PT, for their contributions to this work.
Ethical Approval
This study received ethical approval from the University at Buffalo IRB (approval # STUDY00006620) on September 13, 2022.
Informed Consent to Participate
The ethics review committee of the University at Buffalo approved this protocol on September 13, 2022. Number: STUDY00006620. Date: September 13, 2022. Written informed consent for inclusion in this research was obtained from the patients prior to randomization and administration of the intervention.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets generated during and/or analyzed during the current study are not publicly available because data come from medical chart records and study screening forms; inquiries for data access can be made to the corresponding author and will be considered by the study team.
