Abstract
Introduction
Evaluating healthcare care services from the patient's point of view is a crucial aspect in ensuring high quality care. This approach helps healthcare care providers identify areas of improvement and make the necessary changes to improve patient experience and satisfaction. Previous efforts to improve quality of care have focused on patient satisfaction and broad surveys such as the Healthcare Consumer Assessment of Healthcare Providers and Systems (HCAHPS). Rather than focusing on patient satisfaction, there is a wide interest in patient experience, which aims to provide a broader and more objective measure that encompasses all patient interactions with healthcare providers and how they experience those interactions. It involves testing a defined quality standard of care and ensuring that what should happen happens. The patient experience includes aspects such as timely appointments, easy access to information, effective communication, and support of patient morale. 1 There are key differences in these for quantifying the outcome of healthcare services.
Measurement of patient experience is considered as a basis for improving and maintaining quality of care in modern healthcare organizations and hospital care.2,3 Indeed, it is a powerful way to enable facts to settle from complex to simple phenomena of quality of care and can indicate possible solutions and opportunities for future improvements. 2 Furthermore, the establishment of reliable and valid tools to measure patient experience will reflect the main aspects of care from the perspective of patients, as well as drive improvement in quality of care by increasing patient participation and raising their voices. 4 Studies have shown the correlation between positive patient experience and the results of some health aspects such as improved quality of healthcare care, patient safety, better health outcomes, greater employee satisfaction, reduction in resource utilization, reduction in readmission rate, increased physician participation, and improvement of cooperative culture. 5 Similarly, other studies have indicated 4 main drives for patient experience; excellent communication, wait time, staff empathy, and access to care. 5
Researchers and healthcare providers use patient experience and satisfaction scoring as a tool to improve patient-centered healthcare or as a resource to conduct patient surveys. 6 There are different tools to assess patient experience around the world. Patient-reported outcome measures and patient-reported experience measures are commonly used tools in the United Kingdom and other countries to assess the views and their perception of the received care. 6
Norway uses the scandalized patient experience questionnaire, which includes 35 questions. 6 The Netherlands implemented the consumer quality index as a standardized tool, which combines the HCAHPS questionnaire and the Dutch QUOTE tool (Quality of Care through Patient Eyes) tool. 6 The Picker Institute Europe has a standardized survey questionnaire and conducts national surveys. The Picker patient experience survey questionnaire includes 40 questions related to the patient's experience and a question about whether the patient would recommend the hospital to their family and friends. 6
Although patient satisfaction is subjective and based on personal expectations, patient experience is more objective and focused on quality of care provided. 7 Second, patient satisfaction is about whether patient expectations are met, whereas patient experience is about the quality of care delivered.
In Oman, the biomedical healthcare system has only recently emerged. In the initial phase, Oman has to face widespread communicable diseases, malnutrition, and improve the health of the mother and child. The country's universal free healthcare has been compartmentalized into primary, secondary, and tertiary. However, with increasing improvements in standard of living, Oman has begun to witness the “double sword” of communicable diseases and rapidly increasingly noncommunicable diseases. The current health services model in the country, which is top-down, professionally driven, and cure-oriented, is finding it increasingly challenging to address evolving health issues. In response, the Ministry of Health in Oman has decided to revamp the system to effectively address these new challenges. 8 As a result of this initiative, some studies have emerged to examine patient satisfaction.9,10
Patient satisfaction surveys have long been used by healthcare managers and decision-makers as a quality tool to improve overall organizational performance. 11 It should be noted that patient satisfaction is a complex concept that lacks a widely accepted definition and a standardized tool for its measurement. This makes it difficult to evaluate and compare patient satisfaction levels in healthcare settings and can affect quality of care delivery. 12 Furthermore, it is largely influenced by patient expectations and lacks an assessment of service provider performance. 13 Therefore, feedback tools to assess patient perceptions shifted from the perspective of patient satisfaction to inquire about “patient experience.” 14 Certainly, patient input is considered the main element of patient-centered care using the patient experience that highlights the details of what happened during a specific episode, rather than only focusing on the satisfaction rate using a general evaluation tool. 4 However, there is no single universally accepted definition of the patient experience, but it includes patient feedback as well as highlights of events that involve patients throughout the continuum of care. 15 Friedel et al 16 have recently simplified the definition of patient experience to include what happened during an episode of care and what happened from the patient's perspective. It is a common mistake to use the terms “patient satisfaction” and “patient experience” interchangeably, despite the fact that they have different meanings.
Although national strategies to record patient experience in healthcare services are relatively new in some countries, the United Kingdom and the United States are leaders in applying patient experience assessment and integrating it into their system-wide strategies. 6 The use of patient experience surveys was made mandatory at the national level by the National Health System (NHS) in the United Kingdom in 2001. 17 This was followed by the introduction of the HCAHPS survey in the United States in 2006, which was nationally used in 2008 and has been linked to hospital quality performance since 2012. 18 On the other hand, in Oman, a country in the Arabian Gulf with a universal free healthcare system, it has yet to invest in patient experience, with some exceptions. There is some anecdotal report indicating that a tertiary care hospital has instituted a hospital-based survey to assess patient experience. However, the instrument used has not yet been psychometrically evaluated. The objectives of this research are to validate the patient experience scale. It is crucial to have a set of standardized questions to evaluate the performance of an organization in relation to the patient experience. The instrument has the potential to help compare results over time and track trends that help improve the overall patient experience. 17 To provide a standardized tool to evaluate patient experience, allowing comparison of results over time and helping to identify areas for improvement in patient-centered care, this study has embarked to (i) develop a patient experience assessment scale (PXAS) tailored for use in tertiary care in Oman and (ii) ensure the reliability and validity of the PXAS through comprehensive item generation, language and grammar checks theoretical validity checks, and external validity and reliability assessments.
Methodology
The instrument validation process is carried out in stages.19,20 Validation is needed for both an existing (established) tool if used in different cultures and for a newly developed (empirical) tool in the same culture. The validation process of a scale involves checking the reliability and validity of the scale.
After building the scale based on conceptualization theory, the validity of content and face validity should be evaluated.19–21 The content validity of a scale is to assess to what extent the scale items/questions (the content of the scale) are relevant to the conceptualization of the construct and the aim of the scale.19–22 Content validity can be assessed qualitatively or quantitatively. 20 The content validity index (CVI) is a popular quantitative assessment of content validity of a scale, and it was used in this study.23,24 It is calculated by calculating the proportion of agreement among the panels.20,23
On the other hand, face validity is defined as the degree to which a measure appears to be related to a specific construct in the judgment of nonexperts. 25 It can be assessed by the same panel that is involved in CVI but this time focusing on the clarity of the questions on the scale. However, face validity is better checked by seeking the opinion of lay people who could use the service and who were not involved in the construction or content assessment of the scale.21,26 Face validity is calculated by calculating the proportion of agreement among raters. 26 If the overall agreement is <80%, this indicates that the scale items are of poor agreement and the questions to be restructured; if it is 80% to 90%, this indicates that there is a substantial agreement and the scale items need to be revised. Whereas, if the agreement is above 90%, this indicates the strength of the agreement, and hence all scale items to be retained.
The reliability of a tool is usually assessed by checking the internal consistency, test-retest reliability, and intra-rater reliability. 27 Inter-rater reliability is only in the case of third person rating, for example, parents or teachers rating a child or student's behavior and attitude. Internal consistency is generally assessed by measuring the Cronbach's alpha coefficient, which should be equal and greater than 0.7 to be satisfactory.27,28 The test-retest reliability is when the test (instrument) is applied twice in the same group of people over 3 to 4 weeks apart between the first and second tests. The total score of the instrument is checked at both times and the correlation between them is assessed. High correlation means good test-retest reliability.
The development of the PXAS was carried out in 4 stages to ensure reliability and validity: item generation, language and grammar check, theoretical validity checks, and external validity and reliability checks. The term “scale” will be used interchangeably with “tool” and “questionnaire.”
Stage 1: Item Generation
A team of 7 experts in patient-centered care formed the patient experience team (PXT), in Royal Hospital. They identified key domains for the PXAS through brainstorming and reviewing the relevant literature and existing scales. The PXAS consisted of 18 questions, the first 4 questions covered demographic data and the department the participant visited. The subsequent 12 questions covered the identified domains: physician communication, nurses communication, emotional support, cleanliness, quietness, smoothness of referrals (internal and external), response to patient needs, discharge information, medication-related information, catering services, and overall hospital experience. Each question had 6 response options: excellent (scores 5), very good (scores 4), good (scores 3), acceptable (scores 2), not acceptable (scores 1), and not applicable (scores 0) which were added to certain domains. Additionally, a net promoter score (NPS) question was included, and the last question was to encourage patients for their feedback, and suggestions. The team developed the PXAS in both English and Arabic, ensuring that it was concise and covered the identified domains. The total score of PXAS was calculated by summing the scores of the 12 questions covering the domains mentioned previously; excellent (49-60), very good (37-48), good (25-36), acceptable (3-24), and poor equal to and less than 12.
Stage 2: Language and Grammar Check
The PXAS was reviewed by a language expert proficient in Arabic and English for grammar, spelling, and clarity. The PXT accepted the suggested minor changes while keeping to the context of the scale.
Stage 3: Theoretical Validity Checks
The content and face validity of the PXAS were assessed. The validity of the content ensures the relevance of the scale items to the construct being measured. The CVI was used, where 6 content experts independently rated each question on a scale of 1 to 4, where 1 (not relevant), 2 (somewhat relevant), 3 (quite relevant), and 4 (highly relevant). Questions with scores of 2 or less were removed. The CVI was calculated for individual items (I-CVI) and the overall scale (S-CVI).
Face validity measures how well the scale appears to measure the intended construct from the perspective of nonexperts. This was assessed using the face validity index. A diverse group of educated individuals outside the medical field independently reviewed the scale and completed an evaluation survey. The survey evaluated different aspects of the scale including grammar, clarity, spelling, sentence structure, font size, legibility, instruction, and overall appropriateness. Six people evaluated the Arabic version and 6 evaluated the English version.
Stage 4: External Validity and Reliability Check
External validity and reliability were assessed by administering PXAS to end users. Calculation of sample size using Yamane's (1967) formula with a margin of error of 5%, a confidence interval of 95%, and a study power of 80% resulted in 292 participants. PXAS was distributed to outpatients, hospitalized, and day care in Royal Hospital after receiving ethical approval. Participants were informed of the study and consent was obtained. They were assured that their participation or opinions would not affect their treatment, and data confidentiality was maintained.
The collected data was entered into SPSS version 27 for analysis. Reliability was evaluated through internal consistency (Cronbach's alpha) and test-retest reliability over a period of 3 to 4 weeks. External validity was confirmed by participating in a diverse population of patients, ensuring the generalizability of the results.
Results
A total of 464 participants (196 male and 268 females) with 203 patients and 261 attendants participated in this study. Most (36%) of the participants fell into the age group (31-40 years) followed by (21-30 years) who represented 27% of the study sample. In 3 to 4 weeks, 69 participants completed the questionnaire again for the test-retest correlation. The data show that most of the participants had excellent experience with the services provided and 45% scored excellent and 41.2% scored very good (Figure 1). Furthermore, 173 (37%) scored 10 in the promotor score, where 10 means that they will always recommend the RH to their families and friends.
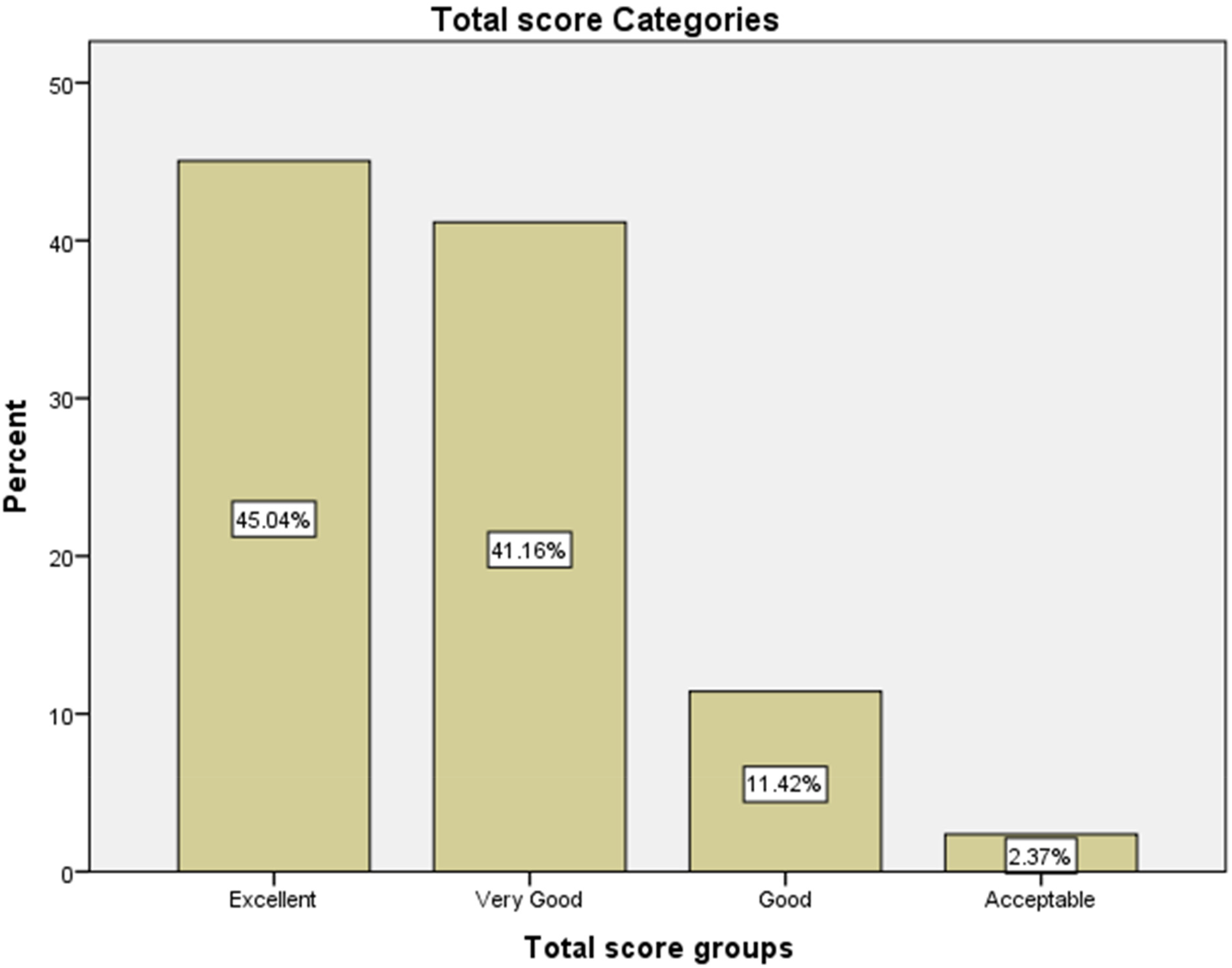
Total score groups.
Reliability
PXAS showed good internal consistency with Cronbach's alpha coefficient of 0.88. The correlation between the test and the retest was good (r = 0.72).
Content and Face Validity
The PXAS has excellent content and face validity (Tables 1 and 2). The CVI for items (I-CVI) ranges between 0.83 and 1.0 while for the scale (S-CVI) it is 0.93. The face validity of the PXAS is excellent with high overall agreement of the scale (98.3%) between raters, and hence all items of the scale will be retained.
Content Validity Index (CVI) of Patient Experience Assessment Scale.

S-CVI/Ave = sum I-CVI/number of items = [(0.83 × 5) + (1×7)]/12 = 11.15/12 =
NB: content grading: 1 = not relevant, 2 = somewhat relevant, 3 = quite relevant, and 4 = highly relevant.
Face Validity Index of Patient Experience Assessment Scale.

Overall agreement is calculated by the following equation:
Sum of % of all questions/total number of questions = 983.3/10 =
NB: Q1= appropriateness of grammar, Q2 = the clarity and unambiguity of items, Q3 = the correct spelling of words, Q4 = the correct structuring of the sentences, Q5=appropriateness of font size and space, Q6 = legible printout, Q7=adequacy of instruction on the instrument, Q8=the structure of the instrument in terms of construction and well-thought of out format, Q9=appropriateness of difficulty level of the instrument for the participants, and Q10=reasonableness of items concerning the supposed purpose of the instrument.
Discussion
PXAS has excellent reliability and validity, as shown in the Results section. The Cronbach's alpha in this study was 0.88 which indicates a very good internal consistency of the scale (PXAS). The correlation of the test-retest scores was 0.72 reflecting a good test-retest reliability. PXAS has excellent content validity with I-CVI ranging between 0.83 and 1.0 and S-CVI of 0.93. The cut-off points for I-CVI and S-CVI of 0.78 and 0.90, respectively, are required for a scale to be considered as having excellent content validity. PXAS, the overall agreement of the scale was 98.3% indicating excellent face validity.
Overall, the PXAS appears to show promising initial results, but its readiness for widespread implementation would require further scrutiny as its practical utility will depend on additional validations in diverse settings. Specifically, future research should focus on testing PXAS in different population groups, geography, and contexts to ensure its generalizability and reliability. Moreover, it is important to refine certain features of the PXAS to address. This involves broadening its applications and guaranteeing smooth incorporation into current systems. As such, more research is needed to optimize PXAS for broader applications, improve its user interface, and improve its predictive accuracy. Testing with larger sample sizes and longer study durations could also provide information on its long-term effectiveness and potential adaptations for specific sectors. In summary, although the PXAS is not yet fully ready for large-scale use, the present study provides a foundation for future research that will advance its development. In the future, it will be important to focus on expanding the size and diversity of study populations, tackling technical challenges related to scalability, and assessing its effectiveness in various real-world settings.
Limitations
This study has several limitations. The cultural and language parameters may have affected the precision of the response, despite the bilingual design. The validation sample size might not be generalized to all patient populations. Comparisons with Western-validated tools may not fully capture the unique aspects of patient experience in Oman. The short interval for the reliability of the test-retest may not reflect the long-term stability. Future research should focus on ongoing use, and refinement of the survey will help ensure that it remains relevant and effective in capturing patient experiences in an evolving healthcare landscape.
Conclusion
In conclusion, this study successfully developed and validated a patient experience survey adapted to the Omani healthcare context. PXAS is short (17 items with demographic data), simple, bilingual, easy to use, and has been shown to be a valid tool. The establishment of this survey addresses a significant gap in the region, providing healthcare providers with an instrument to measure and improve patient experience. This tool not only facilitates direct patient feedback, but also supports ongoing efforts to improve patient-centered care, drive quality improvements, and increase patient participation within the healthcare system. The authors recommend future studies to validate PXAS across diverse settings and populations in Oman.
Supplemental Material
sj-docx-1-jpx-10.1177_23743735241311090 - Supplemental material for Development and Validation of the Patient Experience Assessment Scale (PXAS) in Tertiary Care Hospital - Oman
Supplemental material, sj-docx-1-jpx-10.1177_23743735241311090 for Development and Validation of the Patient Experience Assessment Scale (PXAS) in Tertiary Care Hospital - Oman by Naima Khamis Al-Bulushi, Zainab Ali Al-Mukhaini, Jehan Al Fannah, Afaf Ali Al Zadjali, Huda Al Zidi, Zubaida Mahmood Al-Balushi, Aida Khalfan Al-Hadidi and Samir Al-Adawi in Journal of Patient Experience
Supplemental Material
sj-docx-2-jpx-10.1177_23743735241311090 - Supplemental material for Development and Validation of the Patient Experience Assessment Scale (PXAS) in Tertiary Care Hospital - Oman
Supplemental material, sj-docx-2-jpx-10.1177_23743735241311090 for Development and Validation of the Patient Experience Assessment Scale (PXAS) in Tertiary Care Hospital - Oman by Naima Khamis Al-Bulushi, Zainab Ali Al-Mukhaini, Jehan Al Fannah, Afaf Ali Al Zadjali, Huda Al Zidi, Zubaida Mahmood Al-Balushi, Aida Khalfan Al-Hadidi and Samir Al-Adawi in Journal of Patient Experience
Footnotes
Acknowledgments
The authors would like to thank all those who participated in this project and made it possible. Those are the expert panel; Ahmed Al-Mukhaini, Basma Al-Yazeedi, Arjun Balasingham, Aysha Al-Nadabi, Madiha Al-Anqoodi, Hasna Al-Abri, Ashraf Al-Balushi, Fatma Al-Ghaithi, Hasina Al-Harthi, Khalid Al-Busaidi, Khalid Al-Harthi, Moza Al-Abri, Mohammed Al-Zidjali, Hajer Al-Mukhaini, Waleed Elshoubary, and Reem Al-Zadjali. In addition, the authors would like to thank the Royal Hospital patient experience committee members who contributed in conducting the surveys; Salma Al-Sharji, Khalsa Al-Mushaiqari, Ibtisam Al-Siyabi, Sameeha Al-Habsi, and Aisha Al-Habsi.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
The study was approved by the scientific research committee in Royal Hospital (approval ID no: SRC#26621) on July 4, 2023.
Statement of Human and Animal Rights
All procedures in this study were conducted in accordance with the scientific research committee (approval ID no: SRC#26621) approved protocols.
Informed Consent
Verbal informed consent was obtained from the patients for their anonymized information to be published in this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
