Abstract
Involving patients as co-leaders and co-creators in research is key to reflecting the patient’s voice in decision-making. However, co-creation of patient-centered data to inform decisions is rare, especially in early drug development where patient input is critical to prioritizing patient-relevant outcomes and endpoints for use in clinical trials. Despite the industry's growing commitment to patient centricity, most patients are excluded from sharing their expertise in research; more inclusive methods of engaging patients as research partners are needed. We describe a collaboration between a pharmaceutical company and a patient organization in co-leading and co-creating a program to understand priorities of patients and caregivers for treatment features and outcomes in sickle cell disease to inform endpoint selection in clinical development. The results of this program will be used as a basis for continued interaction between patients and the sponsor and to inform ongoing clinical development and evidence-generation activities. This case study demonstrates an approach to meaningful collaborations between patient organizations and pharmaceutical companies aimed at including the patient's voice early in the medical product lifecycle.
Keywords
Key Points
Patient-centered co-creation of data can be conducted at any point in the medical product lifecycle from discovery and design through post-marketing.
Research partnerships that ensure patients participate as co-leaders in research and have an active role in decision-making can be used to overcome practices that exclude patients from sharing their expertise in a disease area and promote patient leadership in the co-creation of patient-centered data.
Building trust between patients and researchers requires that: (a) the content of the research is co-created so that it reflects what matters to patients, (b) the patient organization can determine the methods for outreach, communication, and enrollment that are most appropriate, (c) the results are communicated to the patient community in a form that is understandable and useful, and (d) sufficient resources are provided to the patient organization to execute the program.
Research partnerships can improve health equity for the patient community by driving partnerships with community-based organizations and utilizing community-health workers who are engaged with patients and families in healthcare.
Meaningful involvement and genuine commitment to listening to and incorporating diverse patient feedback and ideas into decision-making processes may improve patient outcomes, product innovation, and trust between patients and the healthcare industry.
Introduction
Patient-centered health research is necessary to meet patients’ needs, increase diversity in clinical trials, promote health equity in data collection, and inform decision-making throughout the medical product lifecycle.1,2,3 Effective patient-centered health research is built on principles of respect, equity, trust, and empowerment, 4 and requires continued engagement and co-creation of data with patients. 5 A case study of collaboration between a pharmaceutical company (Pfizer) and a patient group (Sick Cells) in developing, conducting, and disseminating the results of a patient preference evidence-generation program in the United States is presented as a model of effective patient engagement in health research during early clinical development.
Method
Although including patient insights in drug development is essential to developing therapies that meet sickle cell disease (SCD) patient needs, 6 no studies exist which quantify the importance of treatment features or outcomes to patients with SCD or caregivers of patients with SCD. The objective of the program was to generate evidence of the importance patients and caregivers place on treatment features and outcomes to inform the early development of novel SCD therapies.
To achieve this objective, patients and caregivers were asked to complete multiple online point allocation (PA) exercises.7,8 In each exercise, participants were presented with a set of SCD treatment features and asked to assign points to each feature indicating how important they thought it was that an SCD treatment address that feature. More points indicated stronger preferences and the total number of points across all features was equal to 100 (Figure 1, Supplemental Material). Twenty-seven features (Table 1) were evaluated across five PA exercises. A sixth exercise was used to weight the most important features in the five PA exercises so data could be combined across PA exercises.

An example of a PA exercise in the sickle cell disease survey.
Importance Weights for Sickle Cell Disease Treatment Features and Outcomes.
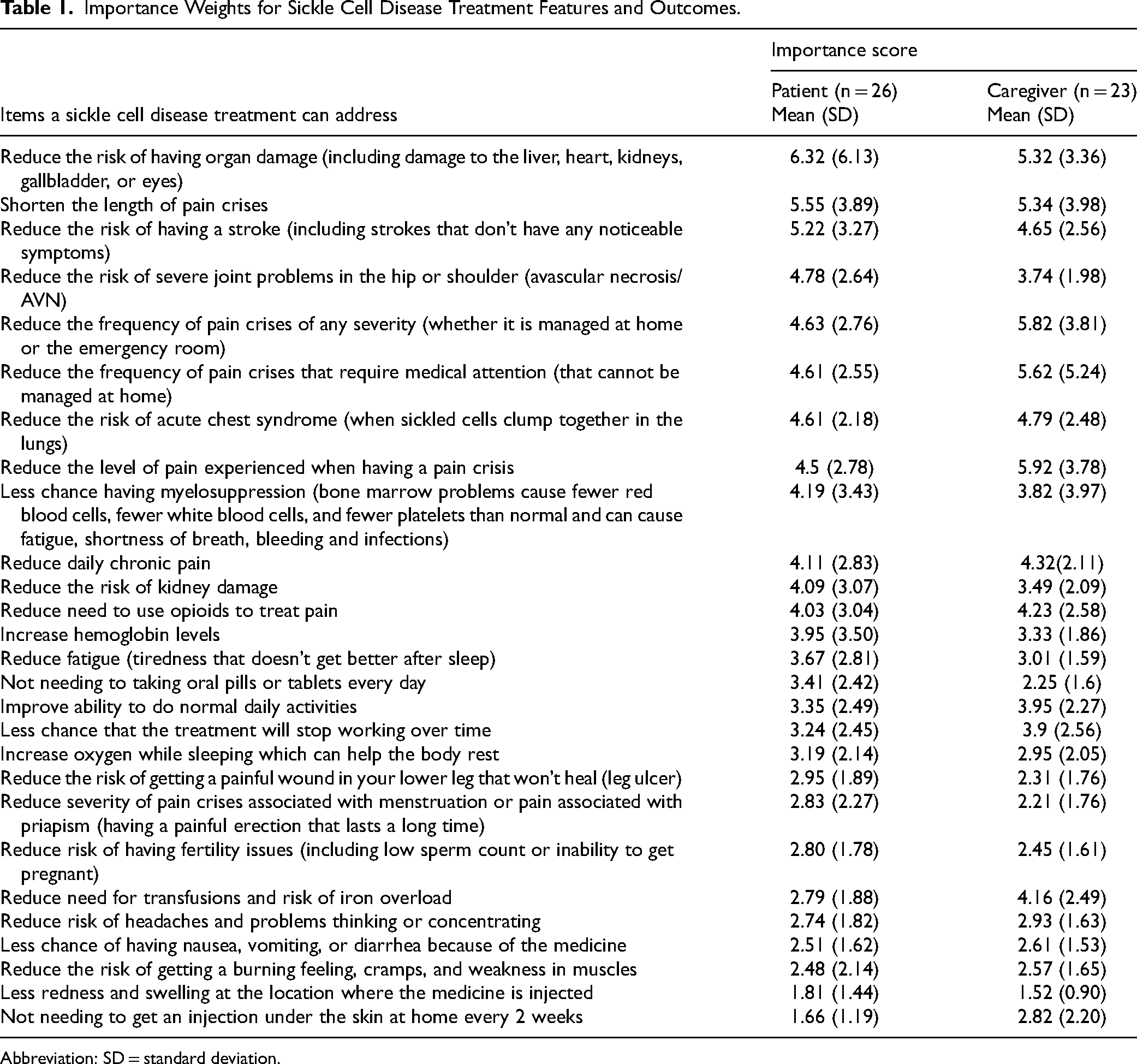
Abbreviation: SD = standard deviation.
An initial list of 22 features was developed by the sponsor to include candidate endpoints for clinical trials, modes of administration, and adverse events associated with SCD treatments. The list was modified by representatives from the patient group to ensure that all features in the list were meaningful to patients and caregivers and that all important features were included in the exercise. For example, daily chronic pain and fertility concerns were not included initially; however, the patient group indicated that addressing these concerns is important to patients. Therefore, reducing daily chronic pain and reducing the risk of fertility issues were included in the list of features. The final list was developed using an iterative process between the sponsor and the patient group. A training video, which was recommended by the patient group to improve participant understanding of the exercise, was also included.
The patient group recruited participants through their network of US community-based organizations (CBOs) that provide support services to families living with SCD (Figure 2). This community-based recruitment utilized the expertise of trained community health workers and social workers to act as a bridge between researchers and patients. Because the CBOs had existing relationships within their local communities, they could contact patients and caregivers who reflected the diversity of the population, paying particular attention to those often underrepresented in SCD research. The CBOs were trained by the patient group to screen interested participants and direct them to the online exercise. All participants provided online consent to participate after being informed of the sponsor's adverse-event reporting obligations, how data would be handled, and how confidentiality would be assured. Participants were informed of the identity of the survey sponsor; the sponsor was blinded to participants’ identities. The patient group provided an electronic gift card ($100) to each participant. After the data were analyzed, the sponsor and patient group jointly developed a plain-language summary for patients and caregivers, describing the results.

Recruitment process led by the patient group.
Results
Forty-nine people (26 patients and 23 caregivers) participated in the exercise. The majority (69%) of patient participants were female. The majority of caregivers (52%) were providing care for a male patient. Over 90% of patients and patients being cared for were Black or African American and 10% were Hispanic or Latino. The majority of patient participants (56%) were <35 years old and the majority of patients being cared for (79%) were <18 years old. Characteristics of participants are summarized in the Supplemental Material. Importance weights for all features are presented in Table 1.
For patients, the most important features were reducing the risk of end-organ damage and reducing the duration of vasoocclusive (ie, pain) crises. For caregivers, the most important features were reducing the severity of pain during pain crises and reducing the frequency of pain crises. For both groups, the least important features were reducing the risk of headaches and cognitive problems and reducing gastrointestinal side effects. In contrast to patients, caregivers placed much greater weight on reducing the need for transfusions.
Discussion
Quantitative data on treatment priorities of people with SCD is limited. In addition, many patient preference studies are conducted only after a medicinal product is developed and the potential outcomes and product features are well understood. At this point it is too late to inform clinical trial endpoint selection. This collaborative approach to understanding preferences for treatment features and outcomes in SCD provides a good example of how drug manufacturers and patient organizations can partner to ensure that the patient voice is incorporated in early development in a meaningful way. Partnerships between sponsors and patient groups and between patient groups and CBOs promote health equity, build trust and can increase participation in research by patients who may otherwise be excluded from sharing their expertise. This type of partnership demonstrates how to balance what is clinically feasible with what is necessary from a patient-centered perspective. The patient group was actively involved in the development of the patient-preference exercise, ensuring that it was understandable and accessible to all patients and included all treatment features and outcomes that matter to patients. The patient group determined how best to recruit and screen participants, ensuring that they had the opportunity to engage with multiple CBO partners and could speak directly to participants throughout the process while ensuring that no personally identifiable information was available to the sponsor. Finally, the sponsor and patient group developed a plain-language summary of the results that could be shared with CBOs, participants, and the broader SCD community, ensuring continued patient engagement.
Patient engagement can help pharmaceutical companies better understand patient priorities which can lead to the development of products that better serve patient needs. This, in turn, may lead to improved patient outcomes and satisfaction. Meaningful patient involvement in decision-making throughout the medical product lifecycle can benefit pharmaceutical companies in additional ways. Engaging with patients throughout the lifecycle provides new insights, leading to more innovative healthcare solutions. Regulatory agencies encourage increased patient engagement by industry throughout the medical product lifecycle. In addition, engaging patients earlier in clinical development may result in financial benefits to sponsors. 9
After the program was completed, the participating CBOs completed an 8-question survey. CBOs indicated that their reasons for participating in this process included providing community members with opportunities to contribute to research, building the CBO's research capacity, and building partnerships with other community organizations. CBOs indicated that the partnership with the patient group provided positive outcomes for their organizations, including connecting local organizations with the broader SCD community and helping them gain insight into the diversity of their clients through their participation in recruitment efforts. In addition, most CBOs agreed that they were able to address any fear, mistrust, or questions about participation in this program from their communities. These findings indicate that the collaboration of a large pharmaceutical company and a national patient organization can have local benefits if local organizations are involved.
Limitations
There are methodological limitations to interpreting the numeric results. This exploratory exercise involved a small number of patient and caregiver participants. The standard deviations surrounding the importance weights were large and may be a result of the small sample size, an artifact of the PA method, or reflect heterogeneity in preferences among participants. Finally, patient participants were ≥18 years of age and caregivers cared primarily for patients <18 years of age. If treatment priorities differ based on age, the results from patients and caregivers may not be directly comparable. From a patient engagement perspective, there are two primary limitations. First, a brief guide providing additional instructions or information about the exercise may have reduced the burden on CBOs who recruited participants. In addition, while the patient group was actively involved in the development of the survey, it may also have been helpful to have greater direct patient input into the process.
Conclusion
Meaningful collaborations between patient organizations and pharmaceutical companies to include the patient voice early in the medical product lifecycle is possible provided that: (a) the content of the research is co-created to ensure that it reflects what matters to patients, (b) the patient organization can determine which methods for outreach, communication, and recruitment are most appropriate, (c) the results are communicated to the patient community in a form that is understandable and useful, and (d) sufficient resources are provided to the patient organization to execute the work.
Supplemental Material
sj-docx-1-jpx-10.1177_23743735231213767 - Supplemental material for Priority Outcomes in Sickle Cell Disease Treatment: Co-Creation and Implementation of a Preference Exercise With Patients and Caregivers to Inform Drug Development
Supplemental material, sj-docx-1-jpx-10.1177_23743735231213767 for Priority Outcomes in Sickle Cell Disease Treatment: Co-Creation and Implementation of a Preference Exercise With Patients and Caregivers to Inform Drug Development by Maggie Jalowsky, Brett Hauber and Mariah Jacqueline Scott, Steven Arkin, Joshua R. Coulter, Stephen J Watt, L Mariah G Kelly, Ashley Valentine in Journal of Patient Experience
Supplemental Material
sj-docx-2-jpx-10.1177_23743735231213767 - Supplemental material for Priority Outcomes in Sickle Cell Disease Treatment: Co-Creation and Implementation of a Preference Exercise With Patients and Caregivers to Inform Drug Development
Supplemental material, sj-docx-2-jpx-10.1177_23743735231213767 for Priority Outcomes in Sickle Cell Disease Treatment: Co-Creation and Implementation of a Preference Exercise With Patients and Caregivers to Inform Drug Development by Maggie Jalowsky, Brett Hauber and Mariah Jacqueline Scott, Steven Arkin, Joshua R. Coulter, Stephen J Watt, L Mariah G Kelly, Ashley Valentine in Journal of Patient Experience
Footnotes
Acknowledgments
This work was funded by Pfizer under a consulting agreement. The funds were used to compensate Sick Cells staff for their time and materials in the development and execution of this project. Sick Cells also used funds to compensate participating patients and caregivers for their time in completing the survey. Pfizer provided funding for the programming and hosting of the online survey and for the preparation and formatting of the plain-language summary. Sick Cells also received support from the PhRMA Foundation.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Pfizer, Inc., Pharmaceutical Research and Manufacturers of America Foundation.
Ethical Statement
This collaboration was conducted as a market research program under Pfizer's internal procedures for Customer Engagement Programs. Each participant was provided with a description of the PA exercise (including the purpose), was informed about required adverse event reporting procedures and confidentiality, and was asked to provide consent before participating. During the engagement, personally identifying information was not collected and free-text boxes were not included in the exercise. Invitations to participate were sent to participants who indicated to the CBOs that they were interested in participating using a no-reply email. Participants were made aware of Pfizer's role as the sponsor of this engagement, but the identities of all participants were blinded to Pfizer. Copies of the introductory text regarding adverse-event reporting, confidentiality, and participant consent are available from the corresponding author upon request.
Supplemental Material
Supplemental material for this article is available online.
Author Biographies
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
