Abstract
Person-centered narrative interventions offer potential solutions to facilitate a connection between the person receiving care and the person delivering the care, to improve quality of care, and positively impact a patient's biopsychosocial well-being. This single-arm feasibility study investigates patient-reported outcomes and barriers/facilitators to the implementation of an all-virtually delivered person-centered narrative intervention into the person's electronic health record. Overall, electronic data collection for the patient-reported outcomes was feasible. All 15 participants felt participating in the study was “easy” and “enjoyable,” and “not a burden.” The facilitators of implementation included: “helpful to the clinician,” “appreciated looking at me as whole person,” “be seen and heard,” “had a connection and trust,” and “felt comfortable and relaxing.” The barriers to implementation included: “completing all the paperwork,” “being rushed for time to complete the PCNI,” and some “emotion” during collection of narrative. The use of person-centered narrative interventions is a way to deploy dedicated tools to shift dehumanized healthcare delivery to a more humanized person-centered care that treats people as experts in their own life narratives by incorporating their beliefs, values, and preferences into their plan of care.
Keywords
Key Points
Evaluating the barriers and facilitators to implementation for the adapted virtual delivery of the person-centered narrative interventions (PCNI) is essential for effective translation to practice.
For Aim 1, the findings provide a deeper understanding of the barriers/facilitators of PCNI, not just from the research team, but also from the patient participants’ perspectives.
For Aim 2, the study was feasible for conducting the virtual narrative interview and collecting pre-post intervention patient-reported outcomes.
Introduction
Person-centered narrative interventions (PCNI) that elicit a person's psychological, social, and spiritual experiences can contribute to person-centered care recognizing and bringing attention to the varied aspects of an individual's values, beliefs, and preferences through the expression of the person's narrative. According to narrative theory, when a clinician knows more of the person's (patient's) narrative, both direct and indirect positive effects occur. Narrative interventions have been used in many ways.1-4 Briefly, some PCNI have used a person's life review recorded on DVDs or use of digital storytelling uploaded to YouTube.5-8 However, in these interventions, the narrative was shared with families, not the clinical team members, and had limited interface with a patient's electronic health record (EHR). In this increasingly technology-rich health care environment, the EHR is one of the primary modes of communicating health care information. Our person-centered narrative intervention is a cocreated document based on the narrative interview with persons living with serious illnesses, written in first person (patient voice), and then after approval from the patient, the document is uploaded into that patient's EHR.9-11 Furthermore, in the population of persons living with serious illness, the use of narratives has been able to reduce psychological illness impact and positively contributes to the person’ quality of life.5-7,9-11 Therefore, the innovative use of the integration of a cocreated person-centered narrative into that person's EHR is a potential solution to facilitate a connection between persons receiving care (patients) and the persons delivering the care (clinicians) and positively impact a person's psychosocial well-being.9-11
Prior to the COVID-19 pandemic, our program of person-centered narrative intervention studies was delivered in-person and in acute care settings 9 ) or in a person's home in the community. 12 One of the main findings of our earlier studies was that PCNI provided opportunities for “connection” between the clinician and the person needing care. 9 The pandemic prompted an immediate shift to care models using digital technology. Our health care system reported an increase in portal activation from 3511 enrolled in January 2020 to 10 077 by May 2020. 13 Yet, this mode of care delivery and conducting research during the pandemic has influenced “connection”; thus, it was time to take the next step of transitioning from an in-person delivery of the PCNI to an all-virtual delivery. During the beginning of the pandemic, where available, outpatient clinical practices converted to all-virtual clinical visits—adapting and using all forms of technology to provide “connection” honoring infection risks for everyone: the person receiving care, their family, and the clinicians providing care. We also witnessed the social isolation brought about by the pandemic. Therefore, the impetus to use available technology to transition our person-centered narrative intervention took on increased importance.
Building on our prior work, this study focuses on the continued investigation of innovative ways to integrate person-centered narratives into the EHR. This single-arm feasibility study has 2 aims: (1) identify barriers to and facilitators of a virtually delivered person-centered narrative intervention and (2) determine feasibility of collecting virtually delivered person-reported outcome measurement data collection. These adaptations to PCNI for this study included: a new setting in the outpatient palliative care clinic, a new modality of collecting research e-consents and electronically delivered patient-reported outcomes, and cocreating the person-centered narrative using a virtual platform.
Methods
This study used a single-arm pretest/posttest design 14 to collect data from persons living with serious illness. In this design, all participants received the PCNI and then followed over time to observe response to the PCNI. All data collection and study procedures were conducted virtually, using electronic consent forms, secure video conference/phone, and REDCap surveys. The research protocol was approved by the Institutional Review Board. All participants signed informed consent.
Further details of the PCNI intervention, study design, recruitment process, and data collection can be found in our previously published work, “An Electronically Delivered Person-Centered Narrative Intervention for Persons Receiving Palliative Care: Protocol for a Mixed Methods Study”. 11
Aim 1: Barriers and Facilitators of PCNI
The research team conducted exit interviews with patient participants at the end of their study participation to collect information about their experiences of the PCNI. See Supplemental File A. Exit Interview Guide.
Aim 2: Patient-Reported Outcomes
Per protocol, patient-reported outcomes were completed at baseline (Time 1), within 48 h after the patient's narrative was uploaded to the patient's EHR (Time 2), and 1 week after the palliative care new patient visit (Time 3). Three time points of the patient-reported outcomes were collected to evaluate changes in the patient's biopsychosocial well-being15,16 over time. The first patient-reported outcome was the PROMIS 29 profile. 17 This measure consists of a total of 29 items, measured from 1 to 5, and assesses the following 8 subscales: physical function, anxiety, depression, fatigue, sleep, ability to participate in social activities, and pain interference and intensity. Each subscale has 4 items. The values for each subscale were calculated as the sum of 4 items, with a potential range of 4 to 20, where lower values reflect more favorable outcomes (eg, lower anxiety, lower depression) and higher values reflect less favorable outcomes (eg, higher anxiety, higher depression). The second patient-reported outcomes were the PROMIS Psychosocial Illness Impact measure. 17 This measure consists of 16 items and assesses negative and positive aspects of the illness experience. In this measure, participants are asked about psychosocial illness impact since their diagnosis and we estimated separate means for positive psychosocial illness impact and negative psychosocial illness impact (see Table 1 Study Activities with Time Analysis).
Study Activities With Time Analysis.
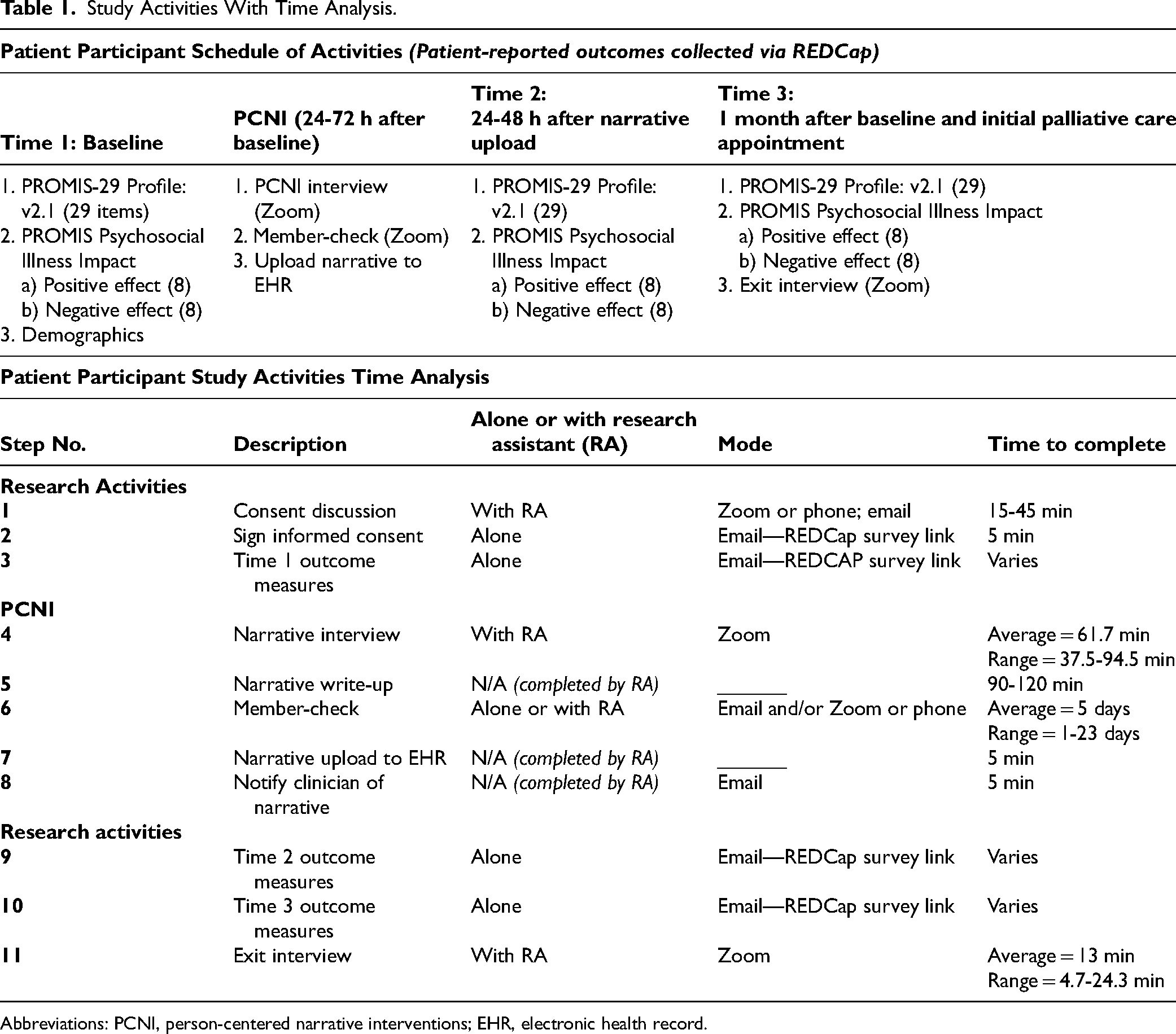
Abbreviations: PCNI, person-centered narrative interventions; EHR, electronic health record.
Aim 1: Barriers and Facilitators of PCNI
Using a qualitative descriptive approach, the participants’ exit interview transcripts were organized using Excel in a patient exit interview matrix. 18 This matrix was organized by each exit interview question. These matrices were used to examine the participant's responses. These responses were examined and discussed among the research team until reaching consensus about the final categories of barriers and facilitators.
Aim 2: Patient-Reported Outcomes
SAS Version 9.4 19 was used for all quantitative analyses of patient-reported data. Descriptive statistics were used to summarize responses to sociodemographic variables using counts/proportions for categorically measured variables and means/standard deviations (SDs) for continuously measured variables. Each of the patient-reported outcomes at each of the 3 time points was summarized using means, SD, and range.
Results
Over the course of the 2-year study, 189 persons were identified as eligible for the study. We enrolled 20 patient participants (planned N). Due to the steps involved in PCNI, we chose to enroll only those persons who had recently received an outpatient palliative care consult order. This was purposeful to be able to complete the steps of the PCNI which include: (1) a narrative interview collection; (2) a review and approval of the final cocreated narrative with the patient; and (3) the uploading of the person's narrative into the EHR prior to the patient's visit with a their palliative care clinician. Patient characteristics of the n = 19 who completed demographic data are presented in Table 2.
Description of Patient Participants.

Abbreviation: SD, standard deviation.
Aim 1: Facilitators and Barriers
Narrative Interviews. The average length of the narrative interviews was 61.7 min (range = 37.5-94.5 min). Writing the narratives took between 90 and 120 min, depending on the length of the audio recording. The cocreation step (member-checking) was conducted via email; first, the research assistant sent the Word document of the participant's narrative and instructions for member-checking. Fourteen of 19 who completed narratives (73.7%) made changes to the original narrative. For those who preferred (n = 4), an additional phone call or video conference was scheduled during which the participant directed the research assistant in making the requested changes to the document. The others (n = 15) completed the process on their own, sending their final version back to the research assistant, or confirming that they did not want changes. The average length of time for the member-check process (ie, from when the research assistant emailed the narrative to when the final approved version was uploaded to EHR) was 5 days, with a range of 1 to 23 days.
Exit Interview. Only 15 of the 20 enrolled participants chose to complete the exit interviews at Time 3. In these exit interviews, the patient experiences with the PCNI were “positive” overall. All patient participants except one (P7), confirmed they discussed their cocreated narrative with other individuals. These individuals included: “palliative care clinician, doctor, faith counselor, therapists, family (husband, wife, son, children), close friends, and persons who have the same diagnosis.” Only 2 of 15 patients reported their palliative care clinician was the one who initiated the conversation about their narrative.
Facilitators
All 15 participants felt participating in the study was “easy” and “enjoyable,” and “not a burden.” In response to inquiries about what they liked about participating in the study, the participants responses included: “helpful to talk through the tough aspects of disease,” “helpful to the clinician,” “appreciated looking at me as whole person,” “be seen and heard,” “had a connection and trust,” and “felt comfortable and relaxing.” When the patient participants were asked to describe what they didn’t like about participating in the study, all 15 participants said “nothing,” and all participants confirmed they would be willing to participate in the study again. Regardless of this initial “nothing” response, some patient participants did report tips to improve the processes of the PCNI and decline the barriers to their experience.
Barriers
Barriers reported by the participants were: “completing all the paperwork,” “being rushed for time to complete the PCNI” before their new patient palliative care visit, and some emotional responses during collection of narrative, that is, “we cried a little bit.” Despite this emotional response, participants described that this sharing of their narrative provided an opportunity to be “moving on” as this was helpful to process their story with another person, and the process was “gratifying.” In Table 3, we have included additional verbatim quotes from the patient exit interviews.
Barriers and Facilitators.

Aim 2: Patient-Reported Outcomes
Electronic e-mail recruitment was a challenge. Overall, 20 of 189 persons contacted for participation enrollment, there was only an uptake of 10.6%. However, once the participant enrolled, the narrative interview completion rate was 95% (n = 19) and patient-reported outcome data collection completion rate was 84.2% (n = 16 of 19 narrative interview completers) (see Figure 1) 20 and for CONSORT 2010 statement: extension to randomized pilot and feasibility trials checklist, see Supplementary File A. 10
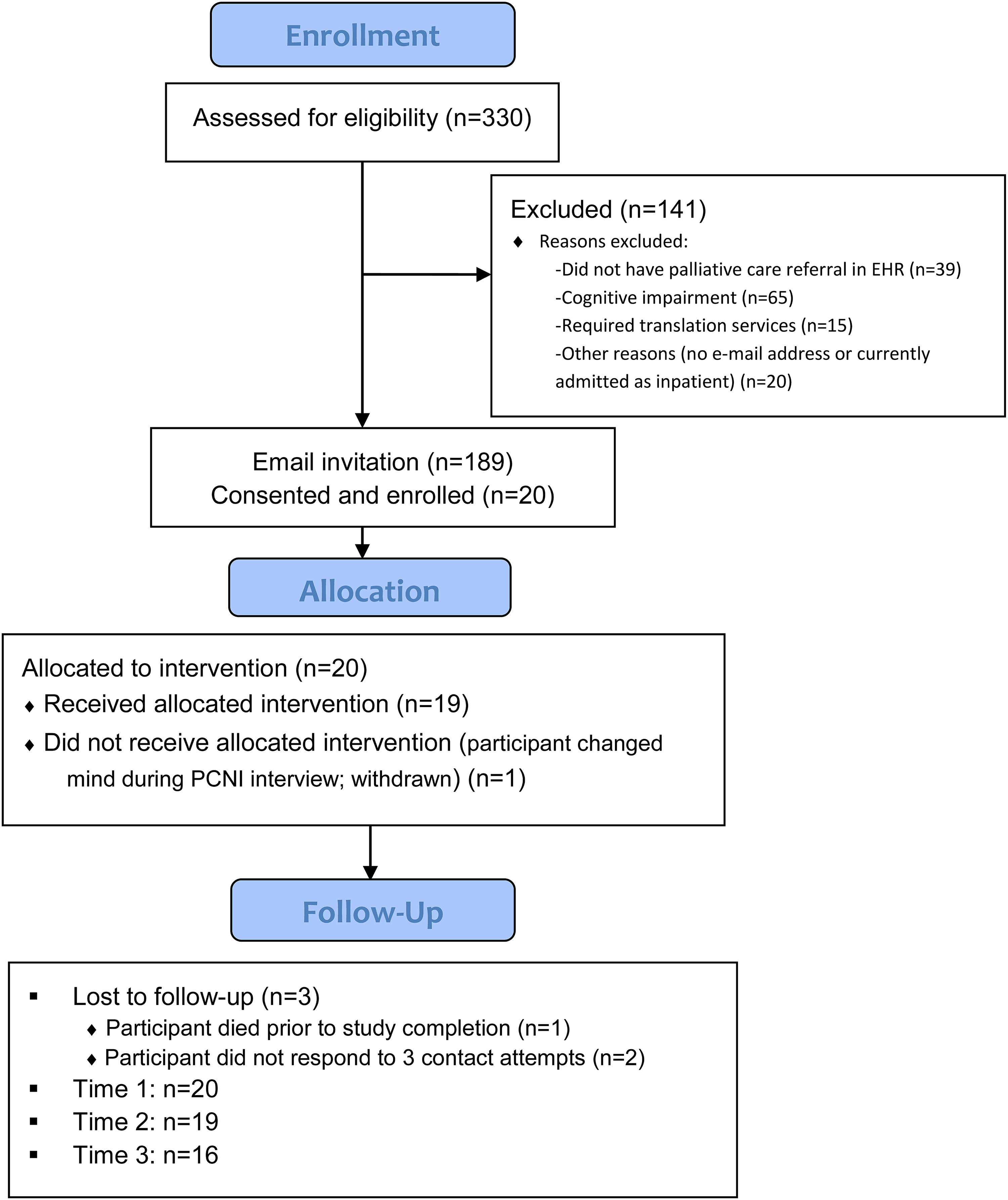
CONSORT diagram.
Overall, collection of patient-reported outcomes over time was feasible. Initially, 23 participants completed the electronic consent process for enrollment in the study. Three participants were lost to follow-up post enrollment, leaving 20 participants who completed T1 baseline measures. However, only 19 participants completed the narrative interview and T2 outcome measures, where one participant after completing the baseline measures chose to not complete the narrative interview and did not continue in the study. Therefore, follow-up data were obtained by n = 19 at T2 (94.7% retention) and n = 16 at T3 (84.2%). This demonstrates the feasibility of obtaining longitudinal patient-reported outcome data in the outpatient palliative care setting. However, there was variability in the amount of time elapsed between follow-ups, where T2 data were obtained a mean of 16.33 days after T1 data collection (SD = 6.50, range from 7 to 29 days) and T3 data were obtained a mean of 30.38 days after T1 data collection (SD = 9.14, range from 13 to 49 days).
Sample size, mean values (SDs), and ranges for patient-reported outcome measures at all waves are presented in Table 4. There were varied improvements across time on the subscales of the PROMIS-29 patient-reported outcomes, where lower values reflect an improvement in the outcomes (eg, lower anxiety, lower depression). For example, there was a decrease in depression values from T1 to T2 and a decrease in pain interference from T1 to T3, suggesting an improvement in depression and pain interference over the 3 time points, suggesting directions for future research. Although the study was not powered to detect significant changes over time, these results suggest directions for future research.
Patient-Reported Outcomes.

Abbreviation: SD, standard deviation.
Discussion
Findings from this pre-post single-arm feasibility study provided insights into areas to improve the integration of the PCNI into clinical flow. Evaluating the barriers and facilitators to implementation for the adapted virtual delivery of the PCNI is essential for effective translation to practice. For Aim 1, the findings provide a deeper understanding of the barriers/facilitators PCNI, not just from the research team but also from the patient participants’ perspectives. For Aim 2, the study was feasible for conducting the virtual narrative interview and collecting pre-post intervention patient-reported outcomes. However, the feasibility of electronic e-mail recruitment was not feasible. We only had a 10.6% response rate to 189 e-mails sent to potential patient participants. In our prior studies with in-person recruitment, our success in recruitment has been much higher.9,10 Therefore, future studies should consider the use of in-person recruitment more feasible than e-mail recruitment. Using implementation as a framework, future research should consider differences in settings where PCNI are collected. In our studies of the PCNI, there was a noted difference in the facilitators and barriers in this outpatient versus our other studies of the PCNI which have been conducted in the inpatient setting. The concept of “Time” was a key point of discussion. The average total patient time to collect the narrative was 61.7 (range 37.5-97.5) minutes, with an average of 5 (range of 1-23) days to complete the member-check of editing their narrative before uploading it into their EHR. Due to the nature of this study, in which all study activities were completed remotely and relied on digital communication, the research team discovered early on that it was more challenging to achieve consistent time frames than in a traditional face-to-face research study. The time to conduct the narrative interview and other study activities for this virtual/outpatient setting was longer than our prior inpatient setting studies of the PCNI. Additionally,9,10 patient participants in this setting were more engaged in the member-check process and the member-check processes took longer to complete (mean length = 5 days) than in previous studies of PCNI in the inpatient setting. One participant did speak to lack of “time” which was a barrier. This person wanted more time to complete the narrative interview processes.
Therefore, the research assistant aimed to build flexibility in the schedule from the time of enrollment. For example, the aim was to complete the narrative interview at least 2 weeks prior to the participant's first palliative care appointment. This allowed for 24 to 48 h to write the narrative and return it to the participant followed by several days to complete the member-check process, upload the narrative to the EHR, and notify the palliative care clinician at least 2 days prior to the new patient visit in the palliative care clinic. This generous timeline also allowed for rescheduling interviews, which was a somewhat common occurrence since the participants were at home and had competing life/work/family obligations. The research assistant also communicated the timelines and the reasons for them to the participants to foster mutual understanding and investment in the process.
For the PCNI, the characteristics of patient participants should also be considered. The type and acuity of a person's illness and the functional limitation imposed by a person's illness must be considered. For example, the speech and motor difficulties secondary to neurological conditions for some of these patient participants did create additional challenges. One of the outpatient clinics in which recruitment took place was neuropalliative care, thus there were some study participants who had challenges with speech or motor skills due to their neurological conditions. The key to success was offering options to accommodate ability and comfort at various steps in the study process. For example, one participant had severe ataxia that impaired speech. In this case, all phone calls and video conferences were scheduled such that the participant's sister could be present to assist the research assistant in understanding the participant's words when needed. Another participant experienced stuttering, and part of the consent process was discussing how to accommodate this. The participant had concerns about being understood and how much time the interview would take; the research assistant told him that there were no time limits on the interview and no concerns about the participant having to repeat for clarity, if he was comfortable with it. This challenge was best met with transparency, discussing options up front during initial conversations about study participation, and continuing to offer flexibility as needs arose throughout the study process. It was also key to exhibit patience and sensitivity in all interactions, to foster an experience that felt comfortable and accepting to the participants.
Limitations
For the PCNI, the type of setting and participant demographics should also be considered. The outpatient setting is distinctly different compared to our acute care setting studies, where time and space for conducting PCNI data collection is more limited.9,10 For this study in the outpatient setting, participating in the PCNI and the resulting narrative seemed to have more of a legacy aspect. Whereas in the acute care setting, patient participants have less physical/mental ability and time to spend with the cocreation processes.9,10 Additionally, the acute care setting can create disempowerment for the hospitalized person, as they are trying to survive the acute phase of their illness during their hospitalization and understandably may invest less energy into the narrative process.9,10 Another limitation was the lack of a diverse demographic representation. In this study, the sample was mostly female (74%), white (95%), Non-Hispanic/Latino (74%), and almost half had some graduate-level education (47%). We did not collect demographics of those individuals who did not respond to recruitment e-mails. Future studies should consider evaluating the demographics of those who did not respond and consider the feasibility in a more diverse sample.
Digital literacy with this all virtually delivered PCNI also had significant differences that our in-person PCNI studies. Among these study participants, there was a range of ease and comfort with the technology required to complete the all-virtual study. To ensure accessibility, the research assistant provided step-by-step (“click-by-click”) instructions for completing the electronic consent and electronic surveys. The research assistant also offered to have an additional phone call to guide the participant through the steps of the electronic consent if needed. Additionally, the member-check process was initiated by sending the participant a Word document of their narrative and offering them the option to incorporate changes themselves or meet with the research assistant via phone or videoconference so they could dictate the desired changes. Some participants (n = 4) stated they did not feel adept using Word and opted for the latter option. The key to success was to offer options at each step to accommodate varying degrees of digital literacy and comfort. In addition to digital literacy, all of the persons enrolled in our study had access to digital technology of email, Internet, and Zoom. Issues related to digital literacy and equitable access to digital technology must not be forgotten. Larger studies must incorporate into their designs dedicated ways to not exclude marginalized populations that may not use digital technology or have access to the technology.21,22
Conclusions
An increased understanding of psychological, social, and spiritual needs from the perspective of the person with a serious illness will help clinicians develop tailored person-centered interventions based on that person's values, beliefs, and preferences. The use and integration of narrative interventions into the EHR is a way to deploy dedicated tools to shift dehumanized healthcare delivery. A delivery of care that treats people as experts in their own life narratives by incorporating their beliefs, values, and preferences into their plan of care. This study continues to (1) build on the foundations of our prior PCNI research and (2) contribute important information for the refinement of the PCNI in a new setting with a new mode of delivery.
Close collaboration with key stakeholders and end users such as patients, families, and clinicians allows for the early identification of barriers and facilitators that can jeopardize the success of the integration of PCNI. This work provides insight into implementation of a complex intervention into complex health systems. The knowledge gained from this study provides the necessary information to guide future PCNI implementation, inform the best practices to scale up PCNI, and sustain implementation of PCNI across different health care settings.
Supplemental Material
sj-docx-1-jpx-10.1177_23743735231202729 - Supplemental material for Use of a Person-Centered Narrative Intervention in an Outpatient Palliative Care Setting: A Feasibility Study
Supplemental material, sj-docx-1-jpx-10.1177_23743735231202729 for Use of a Person-Centered Narrative Intervention in an Outpatient Palliative Care Setting: A Feasibility Study by Heather Coats, Nadia Shive, C Robert Bennett, Bonnie Adrian, Andrew D Boyd, Ardith Z Doorenbos and Sarah J Schmiege in Journal of Patient Experience
Footnotes
Acknowledgments
The authors wish to acknowledge the patient and nurse participants who were part of this study, the nursing managers on the units where study was conducted.
Authors’ Note
Ethical Conduct of Research: This study was approved on March 12, 2021, by the Colorado Multiple Institutional Review Board: COMIRB Protocol # 21-2887.
Author Contributions
All authors made substantial contributions to the design of the work and participated in revising this document critically; provided final approval of the version to be published; and agree to be accountable for the work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Research reported in this article was supported by the Cambia Health Foundation (PI Coats), K24 AT011995(PI Doorenbos), and NIH/NCATS Colorado CTSA Grant Number ULI TR002535 (REDCap). The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health or the Cambia Health Foundation.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
