Abstract
The primary aim of this study is to characterize long-term quality of life (QOL) in patients with esophageal and gastroesophageal junction (EGEJ) cancers who underwent curative intent treatment. EGEJ survivors were recruited to participate in a one-time cross-sectional survey study using validated questionnaires assessing QOL. Chart review was conducted for patient demographics and clinical characteristics. Spearman correlation coefficients, Wilcoxon signed-rank test, and Fisher's exact test were used to assess relationships between patient characteristics and long-term outcomes. QOL was relatively high in this sample, as evidenced by high median scores on the functional scales and low median scores in the symptom domains of the European Organization for Research and Treatment of Cancer (EORTC) Quality of Life Questionnaire (QLQ)-C30, with an overall median global health score of 75.0 (range 66.7-83.3). Patients using opiates at the time of survey reported lower role functioning (P = .004), social functioning (P = .052), and overall global health (P = .041). Younger patients had significantly higher rates of reflux (P = .019), odynophagia (P = .045), choking (P = .005), and cough (P = .007). Patients using opiates or of younger age had lower QOL and higher symptoms in this cohort of long-term EGEJ survivors.
Keywords
Introduction
As improved therapies are developed to treat esophageal and gastroesophageal junction (EGEJ) cancers, it is anticipated that the population of EGEJ cancer survivors will also increase. While the total incidence of esophageal cancer has remained stable in the United States, the rates of esophageal adenocarcinoma are steadily increasing at a rate higher than any other malignancy subtype, largely due to increased prevalence of nongenetic risk factors such as obesity and reflux.1,2 Significant improvements in treatment modalities have improved 5-year survival rates from around 4% in 1975 to around 20% in 2017, although it remains a highly morbid disease with high mortality. 1
A multitude of treatment options exist for early-stage EGEJ cancers. Many patients undergo curative therapy for localized EGEJ cancer using the CROSS regimen, which involves neoadjuvant chemoradiation therapy (nCRT) with weekly carboplatin and paclitaxel followed by esophagectomy. 3 This regimen, along with the incorporation of immunotherapy in the curative approach based on CheckMate577, has been shown to reduce risk of recurrence, improve progression-free survival and overall survival. 4 EGEJ survivors face a variety of health issues after the completion of treatment, however, limited data exist on the quality of life (QOL) after curative intent therapy. Some data show that short-term QOL differences exist between those undergoing curative intent multimodality treatment versus those undergoing surgery alone; however, these differences diminish 1-year postsurgery. 5 Beyond the available long-term data regarding the impact of curative intent multimodality EGEJ therapy, there is also a lack of consensus on the management of symptom burden and optimal surveillance recommendations for evaluating treatment effects.
To address this empirical gap, we aimed to evaluate long-term QOL in EGEJ patients who underwent treatment with curative intent by assessing survivors’ perceptions of symptom burden and health behaviors. We also assessed the relationship between these variables with both disease characteristics and demographic data.
Methods
This study was a one-time cross-sectional survey mailed to EGEJ cancer survivors who had surgery between 1990 and 2014. Eligible participants for this study were identified using tumor registries maintained at our NCI-designated comprehensive cancer center in a large urban city in the northeast United States. Patients were recruited via a mailed survey and in-person during clinic visits for surveillance care between July 11, 2016 and May 31, 2017. Adult patients (>18 years old) with histologically confirmed diagnosis of stages I-III adenocarcinoma or squamous cell carcinoma of EGEJ were included in this study. Other inclusion criteria were: (1) surgical resection (esophagectomy) with curative intent at least 12 months prior to study initiation; (2) no evidence of metastatic, recurrent, or persistent disease; and (3) English proficiency to complete the surveys. Patients who underwent endoscopic mucosal resection only for the treatment of esophageal cancer or gastrectomy for distal EGEJ cancer were excluded. Patients with medical or psychiatric disorders precluding them from informed consent were also excluded. Lastly, those with a secondary primary malignancy requiring treatment within 5 years of treatment for EGEJ with the exception of localized basal cell or squamous cell carcinoma of the skin treated definitively with local resection only, were ineligible.
This protocol was designed with the Dillman method to ensure the highest completion rate possible. 6 Patients were mailed an introduction letter explaining the purpose of the study, 2 copies of the informed consent document, the survey along with instructions, a stamped self-addressed return envelope, and an opt-out form if the participant did not wish to participate. The estimated time to complete the survey was between 30 and 40 minutes. The study was approved by the Institutional Review Board and all patients provided written informed consent. Off-study criteria included an expressed wish to no longer participate in the study, incomplete informed consent document or survey, return of the opt-out form, or failure to return the survey after 4 contacts. All patient data were de-identified and stored in accordance with the health insurance portability and accountability act (HIPAA).
We used multiple surveys to assess patients’ QOL, symptom burden, physical activity level, and appetite and nutrition-related challenges. QOL and symptoms were measured using the European Organization for Research and Treatment of Cancer (EORTC) Quality of Life Questionnaire (QLQ)-C30 and QLQ-OG25, which are well-validated and reliable (Cronbach α coefficient 0.67-0.87). 7 The QLQ-C30 consists of 30 questions addressing 5 functional domains (physical, emotional, cognitive, social, and role functioning), 2 symptom domains (fatigue and pain) and 1 global health status. 8 All measures have scores 0 to 100, with a higher score in functional domains indicating higher function, a higher score in the symptom domains indicating higher symptom burden, and a higher score in global health indicating higher QOL. The QLQ-OG25 consists of 25 questions addressing common symptoms, including dysphagia, eating restrictions, reflux, odynophagia, pain, and anxiety. Similarly, all measures have scores 0 to 100, with higher scores indicating higher symptom burden. 7
Physical activity was assessed using the Godin Leisure-Time Exercise Questionnaire (GLTEQ), a simple and brief questionnaire that can reliably measure leisure time exercise behavior and has been validated for use in patients with cancer.9,10 Two such studies reported a Pearson correlation coefficient between scores on the survey and pedometer step counts, one study of patients with breast cancer demonstrated a Pearson correlation coefficient of 0.53 and another study in leukemia survivors with Pearson correlation coefficient of 0.57.11,12 Nutrition self-efficacy was measured using the Nutrition Self-Efficacy Scale (NSEQ), 13 which assesses individuals’ abilities to manage their nutritional intake. The possible range of scores is 0 to 48, with higher scores indicating better appetite. Symptom burden as it relates to appetite was assessed by the Cancer Appetite and Symptom Questionnaire (CASQ), a 12-item scale that has been validated and found to have good internal reliability in patients with upper gastrointestinal cancers undergoing radiation therapy, with an intraclass correlation coefficient of 0.80 (confidence interval [CI] 0.68-0.92). 14 Scores range from 0 to 20 with higher scores indicating a better ability to maintain a healthy diet.
Disease characteristics, treatment history, prescribed medications, and medical history were obtained from medical chart review. Weight and body mass index (BMI) trends were also documented from the medical record.
The primary outcome of QOL as measured by the EORTC scales and the continuous secondary outcomes of symptom burden and health behaviors were summarized by medians, ranges, and interquartile ranges (IQR). For physical activity, as measured by the GLTEQ, the percentage of participants who were sedentary (total survey score <14), moderately active (total survey score 14-23), and active (total score ≥ 24) were reported and its 95% CI was summarized. Chi-square test, Wilcoxon test, and Spearman’s rank correlation were used to evaluate bivariate associations between patient characteristics and outcomes to identify potential risk factors.
Results
Of the 140 eligible patients who were mailed survey packets, 48 patients (34%) returned a survey packet including survey and informed consent document. However, 7 survey packets were incomplete, and therefore not included in the subsequent analyses (Figure 1). Patient characteristics are detailed in full in Table 1 (n = 41). Most patients (78%) were male and diagnosed with adenocarcinoma (83%). The primary tumor locations were in the lower esophagus (61%), gastroesophageal junction (GEJ) (27%), and thoracic region (12%). The population was 95% white (n = 39) with a median (IQR, range) age of 67 (60-75, 50-81) years. Seventy-six percent (n = 31) of patients were married and 59% (n = 24) completed an education higher than high school. Median time since diagnosis was 7 years (range 2-25). Twenty-two percent had pathological stage I disease (n = 9), 34% stage II (n = 14), and 39% stage III (n = 16). Seventy-one percent (n = 29) received neoadjuvant therapy and 59% (n = 24) had a complete pathological response. Of note, all patients were treated prior to the change in standard of care incorporating adjuvant nivolumab, and therefore no patients in this cohort received adjuvant immunotherapy at the time of curative treatment or survey.

CONSORT diagram for participant enrollment and completion.
Characteristics of Long-Term Survivors (N = 41) and Descriptive Summary of QOL Scores by Questionnaire.

*No clinical stage available for 2 patients.
**Three in the neoadjuvant CRT also received adjuvant chemotherapy.
Missing Data Count: QLQ30 Cognitive Functioning = 1, QLQ30 Social Functioning = 1, QLQ30 Global Health Score = 1, CASQ = 2, NSEQ = 5.
Abbreviations: CASQ, Cancer Appetite and Symptom Questionnaire; CRT, chemoradiation therapy; GEJ, gastroesophageal junction; GLTEQ, Godin Leisure-Time Exercise Questionnaire; NSEQ, Nutrition Self-Efficacy Scale; QLQ, Quality of Life Questionnaire; QOL, quality of life.
In this population of patients who had undergone esophagectomy at least 12 months prior to survey response, overall, QOL in this sample was relatively high, as evidenced by high median scores (MSs) on the functional scales and low MSs in the symptom domains of EORTC with an overall median (IQR) global health score of 75.0 (range 66.7-83.3). There were negative correlations between emotional functioning and symptom burden, including dysphagia (r = −0.34, P = .028), reflux (r = −0.47, P = .002), odynophagia (r = −0.37, P = .017), pain (r = −0.41, P = .008), anxiety (r = −0.54, P = .0003), and cough (r = −0.36, P = .021) in this cohort (Table 2). Similar correlations were seen between social functioning and dysphagia (r = −0.495, P = .001), restriction (r = −0.549, P = .0002), reflux (r = −0.371, P = .019), odynophagia (r = −0.508, P = .001), pain (r = −0.536, P = .0004), and anxiety (r = −0.566, P = .0001). Negative correlations between physical functioning and dysphagia (r = −0.413, P = .007), restriction (r = −0.396, P = .010), and odynophagia (r = −0.483, P = .001) were also seen (Table 2).
Correlation Between EORTC-QLQ-C30 Domains and QLQ-OG25 Symptoms.
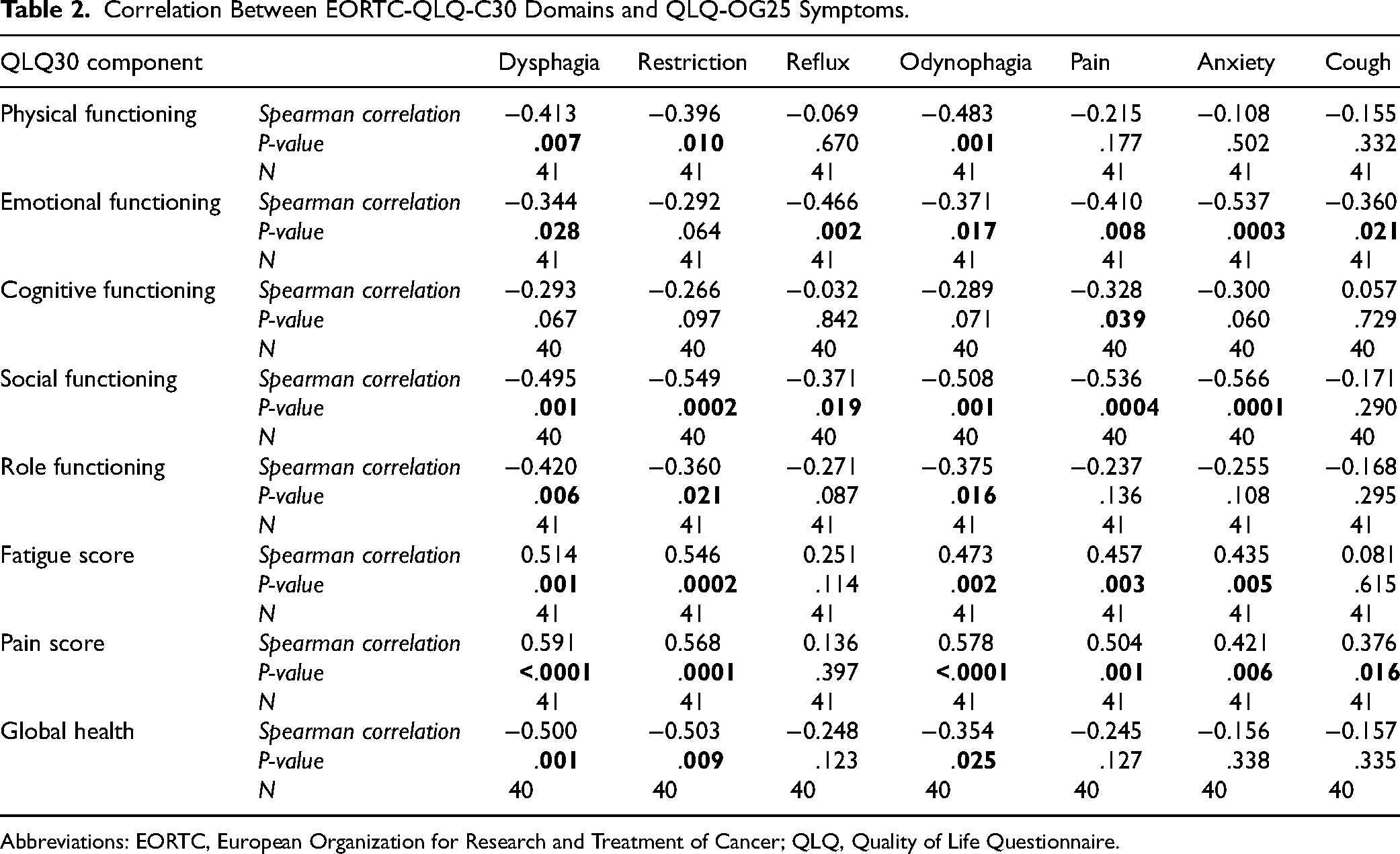
Abbreviations: EORTC, European Organization for Research and Treatment of Cancer; QLQ, Quality of Life Questionnaire.
Analyses also reviewed that QOL differed across patient subgroups. Compared to patients age ≥ 65 years (n = 26), younger patients age < 65 years (n = 15) had significantly higher rates of reflux (MS 33.3 vs 16.7; P = .019), odynophagia (MS 33.3 vs 0.0; P = .045), choking (MS 33.3 vs 0.0; P = .005), and cough (MS 33.3 vs 33.3; P = .007) as well as lower emotional functioning (MS 66.7 vs 95.8; P = .007; data not shown). Lower levels of education (HS or lower, n = 17), when compared with their counterparts (n = 24), were associated with higher symptom burden including higher dysphagia (MS 11.1 vs 0.0; P = .048), reflux (MS 33.3 vs 16.7; P = .039), pain (MS 33.3 vs 16.7; P = .005), anxiety (MS 50.0 vs 16.7; P = .001), and lower social functioning (MS 66.7 vs 100.0; P = .025). Patients using opiates at the time of survey (n = 8) reported lower role functioning (MS 66.7 vs 100.0; P = .004), higher rates of fatigue (MS 44.4 vs 22.2; P = .029), higher pain (MS 41.7 vs 0.0; P = .022), lower social functioning (MS 58.3 vs100.0; P = .052), and lower overall global health (MS 62.5 vs 83.3; P = .041) than those patients not using opiates (n = 32) (Figure 2).

Comparison of QLQ30 median scores in patients using opiates at the time of surgery (N = 8) versus patients not using opiates at time of surgery (N = 32). Abbreviation: QLQ, Quality of Life Questionnaire.
MS on the GLTEQ survey was 18.0 (range 0.0-91.0; IQR 6.0-30.0) (Table 1), with 46% of survivors in the sedentary category with GTLEQ scores <14. The CASQ questionnaire, yielded a MS of 33.0 (range 16.0-44.0; IQR 28.0-37.0) (Table 1). The NSEQ showed a MS of 10.5 (range 5.0-20.0; IQR 69.0-15.0) (Table 1). Demographic characteristics including age, marital status, gender, education, or income were not associated with GLTEQ, CASQ, or NSEQ scores (data not shown).
Median (IQR) BMI at diagnosis was 28.7 (23.9-34.3) and 23.1 (21.0-27.3) at the most recent clinic visit. Obesity was present in 37% of patients at the time of cancer diagnosis, but the obesity rate was only 7% at the most recent visit. Sedentary lifestyle was associated with having an overweight BMI (MS 26.3 for GLTEQ <14 vs 22.1 for GLTEQ ≥ 14; P = .04). There was a significant positive correlation between recent BMI and NSEQ score (n = 10, rs = 0.68; P = .03). Rates of obesity (BMI ≥ 30) decreased over time, while rates of low/normal BMI (<25) increased over time. Six-month postoperative BMI was significantly correlated with physical activity level (P = .04). There were no statistically significant correlations between the CASQ score and BMI nor the NSEQ score.
Discussion
In this cohort of long-term EGEJ survivors, overall QOL is relatively high. However, younger patients, those with a lower level of education, or currently receiving opiate therapy tended to have higher symptom burden and lower QOL. While the majority of esophageal cancer diagnoses are in older populations, our study suggests that patients under age 65 years in particular could benefit from regular screening of QOL measures. In our study, those with lower levels of education have higher symptom burden. This is likely multifactorial, and could be secondary to less access to resources that may alleviate symptom burden when compared to those with higher education and presumably better access to healthcare and other resources. While providers should counsel and educate all patients, our findings suggest younger patients and those with lower levels of education could benefit the most from multidisciplinary interventions, regular screening and interventions aimed at improving QOL measures.
Pain control is a major issue affecting QOL in EGEJ cancer survivors. 15 Esophagectomy has shifted to minimally invasive techniques worldwide, allowing earlier enteral nutrition and ambulation. Even with less invasive techniques, esophagectomy remains a major operation requiring pain control in many patients. The Enhanced Recovery After Surgery (ERAS) protocol for esophagectomy was created in 2019 to address pain in this population. ERAS favors multimodal nonopioid analgesia to decrease the negative side effects of opioids, which often prevent early ambulation and postoperative eating shown to improve outcomes. 16 Our cohort of 8 patients requiring opiates in the postoperative setting reported lower QOL and higher symptom burden. Prolonged opiate use in this cohort of patients is likely due to residual treatment-related pain and not secondary to active disease progression, as active metastatic of progressed disease at the time of survey was exclusionary. Additionally, the time of survey was prior to wide-spread implantation of ERAS, and may explain the role of opioids in this patient population. Ongoing efforts to minimize long-term opioid use through ERAS and other interventions can maximize long-term postoperative QOL, and should be implemented early in the treatment course.
Our study shows that poor nutrition and physical activity patterns exist in many EGEJ survivors, with almost one-half of EGEJ survivors qualifying as sedentary. Many patients were overweight or obese at diagnosis and subsequently had lower BMI with curative therapy, with weight changes primarily occurring between the time of initial cancer diagnosis and 6 months after surgery. These weight changes may be multifactorial in nature, including changes in eating habits necessitated by esophagectomy, but EGEJ survivors in our study population have a high burden of appetite-related symptoms that persist at a median follow up of 7 years following completion of therapy. Survivors with a higher nutrition self-efficacy (ie, those most likely to consume healthy foods) have higher current BMIs. Those who are physically active are more likely to have a normal BMI, and this combination may be beneficial. Lower levels of education were associated with higher symptom burden scores including dysphagia, reflux, pain, anxiety, and lower social functioning.
Our results are consistent with existing literature. Several studies have reported that QOL is reduced the first year after surgery and poor QOL is highly associated with swallowing dysfunction, reflux, and pain.5,17 In one study, QOL did not improve from 1 year after surgery up to 23 years after treatment in long-term survivors who underwent esophagectomy. 18 Another long-term study found that survivors 15 years postoperatively continued to have reduced QOL in 10 of 25 domains, with most symptom burden related to digestive tract issues. 19 However, not all studies confirm long-lasting QOL impairment, with a study from Japan demonstrating an initial negative impact of esophagectomy on QOL, but with transient effect. While QOL decreased significantly after starting chemoradiotherapy, there was a statistically significant improvement at 4 to 5 months compared to those seen at baseline, with continued improvement over time until at least 13 to 18 months after definite CRT. Follow up beyond 2 years was not reported in this study. 20
With regard to physical activity and body weight, our data is consistent with the existing literature. In a study comparing esophageal cancer survivors following curative treatment with controls, survivors showed significantly lower amounts of physical activity, as well as decreased physical and role functioning domains. 21 Esophageal survivors with overweight BMIs at diagnosis tend to experience the most weight loss and have an almost 5-fold increased rate of malnutrition compared to those with normal BMIs on presentation. 22 To our knowledge, there have not yet been studies using the NSEQ and CASQ in patients with EGEJ cancer. However, studies using similar assessment tools have shown that lack of appetite is common, with one study finding severe anorexia present in 52% of patients within 1 year of curative intent treatment. 23
Strengths of this study include the long-term nature of our results, with median time from diagnosis of 7 years, which show that symptoms and altered health behaviors persist over time for survivors. Future studies should evaluate strategies to routinely screen for poor lifestyle habits and reduced QOL in EGEJ patients starting from the time of diagnosis through the treatment continuum into survivorship. In a recent nationwide survey in Japan of institutions treating those with esophageal cancer, only 5% of patients who underwent definitive chemoradiotherapy were screened for health-related QOL measures at year 1 after treatment. 24 Our study supports the potential utility of these validated tools well after treatment given the high incidence of long-term symptom burden and its effects on various functions. Studies should also evaluate targeted interventions aimed at improving activity levels and nutritional status, ideally starting at the time of diagnosis. Indeed, studies of physical activity in patients with postoperative EGEJ cancer have demonstrated a mortality benefit compared to survivors with lower levels of activity. 25 The ongoing randomized control PERFECT trial is currently investigating the potential benefits of physical activity and will also help provide a clearer picture of physical activity's role in extending life in patients with EGEJ cancer. 26 Research into optimal modalities of physical activity, the impact of weight change, and ways to impact appetite and eating is warranted in this growing survivor population.
Limitations
There are several limitations to this study. First, the survey completion rate led to a small sample size. Additionally, the participants in this study were largely white and male; while this demographic profile is consistent with esophageal trends in the United States, it makes the results less generalizable to populations of different gender and race. This cohort also did not include patients who received adjuvant immunotherapy, as patients were treated prior to the change in standard of care.
Conclusion
Our results add to the current literature that EGEJ cancer survivors can have significant symptom burden and suggest that support should be provided on a longer-term basis for this population. As survivors continue to benefit from curative treatment regimens often comprising of neoadjuvant chemoradiotherapy, surgery, and now adjuvant immunotherapy, more research must be done to evaluate and address the lasting symptom burden to optimize QOL for patients. Additional research is needed to understand the specific mechanisms contributing to differences between the most vulnerable populations, and help design interventions to target EGEJ survivors that are at highest risks of long-term impairments.
Footnotes
Author Contributions
Study conception and design performed by CD and JYS. Material preparation, data collection, and analysis were performed by RJ, CR, JYS, EG, EM, DY, and XL. The first draft of the manuscript was written by EM. All the authors commented on previous versions of the manuscript and read and approved the final manuscript.
Declaration of Conflict of Interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This cross-sectional study was approved by the Temple University Human Research Protection Program.
Funding
The author(s) disclosed receipt of the following financial support for the research and/or authorship of this article: This work was supported by the Fox Chase Cancer Center Supported Grant P30 CA006927 and the Fox Chase Cancer Center GI Cancer Research Fund.
Statement of Human and Animal Rights
All procedures in this study were conducted in accordance with Temple University Human Research Protection Program's IRB# 13-828 approved protocols.
Statement of Informed Consent
Written informed consent was obtained for patients’ anonymized information to be published in this article.
