Abstract
This study examined Morbidity and Mortality (M&M) review practices and perspectives of physicians and parents regarding parent participation in M&M review. Surveys were distributed to parents of children with a prior hospitalization for congenital heart disease (CHD) and physicians caring for pediatric CHD patients. Response distributions and Fisher’s exact tests were performed to compare parent and physician responses. Qualitative survey data were thematically analyzed. Ninety-two parent and 36 physician surveys were analyzed. Physicians reported parent input or participation was rarely sought in M&M review. Parents with direct experience of adverse events or death of their child reported providers discussed events with them in a timely manner and answered their questions; however, nearly half wished their healthcare team had done something differently during the disclosure. There was no statistical difference between groups regarding transparency (P = .37, .79); however, there was a significant difference in perspectives regarding parental involvement in the M&M review (P < .001). Common themes important to parents which emerged from the qualitative analysis were being adequately informed, feeling their perspectives were acknowledged and respected, having attentive and empathetic providers, and receiving consistent messaging. Although rarely included in current practice, parent participation in M&M could offer unique insight and increase accountability to proposed change elucidated by M&M review.
Introduction
When provided the option, patients and families overwhelmingly prefer the opportunity to participate in a range of activities from which they have been customarily excluded including participation in daily rounds, acting as primary caregivers in the neonatal intensive care unit, accompanying their critically ill child during interhospital transport, and being present during invasive procedures and resuscitations (1–7). As a consequence of increased presence at these events, parents report reduced stress (6). Furthermore, when adverse events occur, families often desire not only full disclosure but to be active participants in improving the health care system to ensure that such events are avoided in the future (8–10).
The Morbidity and Mortality (M&M) review conference is one mechanism that healthcare organizations use to engage in system-wide learning and improvement. The M&M conference was created with the goal of reviewing medical practices, evaluating adverse outcomes, enhancing medical education, and fostering quality assurance (11,12). While not completely universal, M&M review is an established proceeding in most US hospitals and has become a requirement of the Accreditation Council for Graduate Medical Education (ACGME) as part of resident education (13–15). Given the ACGME mandate, there has been widespread adoption of the M&M conference across US hospitals (16). Given its prevalence, the M&M review has the potential to significantly influence patient care and quality improvement (17–20).
Despite its prevalence, the M&M conference and its findings are not standardized, with notable differences in M&M structure and processes across institutions (14,15,21–23). This manifests in significant variations including how cases for review are chosen, amount of time dedicated to case presentation versus case discussion, and number and style of moderators (14). In their study of surgical programs in the United States and Canada, Anderson et al. found that less than half (44%) of departments had written guidelines specifying which complications should be included in the M&M review (15). Furthermore, when asked “When a case is closed, what do you do with the information,” 20% of departments reported not retaining any records nor using the information for further analysis (15). The general expectation is that M&M should serve both an educational and quality improvement role, however, the process often falls short in its potential to impact system-wide quality improvement (14,21–24). Additionally, there is still uncertainty to the best structure for the M&M process to manifest meaningful change (24).
Several studies have examined the M&M conference and provided suggestions for restructuring and improving the M&M process. These suggestions include (1) standardizing case selection and presentation (23,25,26), (2) learning not only from errors but also near misses and successes (24,26), (3) fostering discussion by appointing a skilled facilitator to moderate discussion, allocating sufficient time to allow innovative thinking, and ensuring a safe environment to express opinions (23,25,26), (4) attaining and valuing input from multiple sources which consider individual, team, and system-level factors (21,26), (5) reviewing and assessing prior quality improvement initiatives derived from previous M&M processes (23), and (5) deriving action plans and assigning follow-up reflective of learnings identified during the M&M conference (21,25,26).
Expanding on the idea of obtaining input from multiple sources, several institutions have recognized that expanding the M&M review to a broader, interdisciplinary audience has allowed for better identification of systems-based issues (18,19,27). However, as noted by the Lucian Leape Institute, traditional M&M meetings often fall short of their potential, in part because they fail to include the patient’s point of view (28). While some programs may share their M&M review findings with families afterward, there are no published reports of hospitals routinely including patients or their families in the review process (18).
This study sought to investigate current practices pertaining to the inclusion of parents in the M&M review process and assess the perspectives of physicians and parents regarding the broad topic of transparency in healthcare, as well as the prospect of parent participation in the M&M review process.
Methods
This study was designed using mixed methods participatory research design. We partnered with a family advisory team in the design of the data collection tool and in defining a strategy for participant recruitment. Two surveys were developed—one specific to physicians directly involved in the care of pediatric congenital heart disease (CHD) patients and one specific to parents of pediatric patients with a history of hospitalization for CHD. Surveys were comprised of a combination of Likert scale and open-ended questions and were disseminated electronically between February 2017 and May 2017 using secure web-based Research Electronic Data Capture (REDCap) tools hosted at Cincinnati Children’s Hospital Medical Center. Full copies of the surveys are included in online Supplemental material.
Participants received a detailed overview of the survey topics prior to beginning and had the option to withdraw at any time. Surveys were launched, and responses were collected for 2 months. Parents were eligible for inclusion if they were at least 18 years of age and the parent of a child with a history of hospitalization due to CHD. Physicians were eligible for inclusion if they were directly involved in providing care to pediatric CHD patients. Eligibility was assessed via initial screening questions on the survey. The study protocol was submitted to the institutional IRB (2016-6718) and deemed exempt in accordance with 45 CFR 46.101 (b).
The physician survey was distributed via the Pediatric Cardiac Intensive Care Society (PCICS) contact list. PCICS is an international society focused on furthering “excellence in clinical care and research in the field of pediatric cardiac critical care.” At the time of the survey, PCICS had about 630 physician members. The parent survey link was distributed via social media websites for Sisters by Heart, a support network for parents of children with hypoplastic left heart syndrome, and Hayden’s Heart a, nonprofit created to raise awareness of CHD and help families affected by CHD. Both groups posted links on their publicly accessible website/Facebook page, with a total of approximately 24,000 followers at the time of recruitment. To limit multiple responses from the same user, survey submission was limited to one per unique Internet IP address.
Physicians were queried about the existing mechanisms for M&M review at their institutions and the manner in which patients and families were included in such review processes. Parents whose child had experienced a morbidity or death were asked about related communication and follow-up. Both physicians and parents were asked about their perspectives regarding information sharing in the context of the hospitalization and accompanying complications, errors, and/or mortality, including the potential for inclusion of families in the M&M review process. Physician and parents rated their level of agreement with a total of 17 statements about transparency in the context of adverse events (7 statements) and possible parent roles in the M&M review process (10 statements). The terms “complication” and “medical error” were explicitly defined on the parent survey, and a description of the M&M conference was provided (online Supplemental material). “Transparency” was defined in both surveys as “the free and uninhibited flow of information.”
Distributions of responses were examined for any missing data or possible transcription or data entry errors. Mean and range were used to describe the distribution of parents’ age. Counts and proportions were used to describe responses on binary or categorical measures. Fisher’s exact tests were performed to compare the distributions of responses between the two groups (parents and physicians). A P-value <.05 was considered to be statistically significant. Responses to open-ended survey questions were analyzed thematically and were used to supplement our understanding and interpretation of the Likert scale responses.
Results
Ninety-two of the 144 parents and 36 of the 60 physician surveys were validated by the screening questions, complete, and included in the final analysis. Table 1 provides demographic data for the final study cohort (parent and physician).
Parent and Physician Respondent Demographics.

Current M&M Review Practices
Most physician respondents (94%) reported that their organization had a formal M&M review with cases selected for inclusion because they were mortality cases (74%) or were identified by department/unit leadership for review (65%). Cases were generally reviewed within a month of the event or mortality (62%). Participants in the formal M&M review most often included attending physicians (97%), cardiothoracic surgeons (71%), nurse managers (74%), and fellows (82%). Physicians indicated that the review rarely focused on individual mistakes/errors (6%), but rather on root causes at the system level (73%). A majority of physicians (72%) also noted that their organization had an informal review process, with most informal reviews taking place within 72 h of an adverse event (81%).
Whether formal or informal, physicians reported that parents were rarely included in the review process. Six percent of physician respondents with formal M&M processes indicated that parents/patients were either “always” or “frequently” invited to participate in the formal reviews, and 4% of physician respondents with informal review processes indicated that parents or patients were “always” or “frequently” invited to participate in the informal process. Figure 1 summarizes physician-reported survey data pertaining to M&M review.

Physician responses about current Morbidity and Mortality (M&M) review.
Less than half of physician respondents (47%) indicated that their organization had a disclosure policy in place to address discussing adverse events, unexpected outcomes, or mortalities with families and 17% reported that their organizations offered formal training to help providers with such discussions.
Parent Experiences
Sixty-one percent of parent respondents reported that their child experienced at least one complication or medical error during their hospitalization, and another 22% either suspected or were unsure whether their child experienced a complication or error. Of those who were certain their child had experienced an error/complication, 82% reported the healthcare team discussed the error/complication with them. Respondents most often reported that the discussions included the cardiology attending (76%), followed by surgeon (52%), and registered nurse (48%). Discussions usually occurred immediately or within hours after the event (91%), and most parents (80%) felt that their questions were answered in a timely fashion. Despite being informed of the event and having their questions addressed in a timely manner, 44% of survey respondents whose child had experienced an error or complication wished that the team had done something differently when addressing the complication or error. One parent shared, “An apology for the error would have been very much appreciated, and a clearer explanation of what happen. The gap in medication happened overnight when we weren’t there…would have liked to have known exactly what happened and when.” Another parent noted, “Although she went into cardiac arrest in my arms, it was unclear to me what was going on as I am not medically trained. An explanation of the connection between the aspirating of her food to the cardiac event that occurred would have been extremely helpful.”
Thirty-six percent of parent respondents reported experiencing the death of their child during a hospitalization. Similar to the morbidity discussions, conversations following mortality most often involved the cardiology attending (88%), surgeon (52%), and registered nurse (67%), with the addition of social worker involved with more than half (58%) of bereaved respondents. The majority (76%) of bereaved respondents reported that their questions were answered in a timely fashion. However, as with the morbidity cohort, nearly half (45%) wished the healthcare team had done something differently when discussing the death with them. For example, one parent responded, “I wish that someone had discussed with us the option of an autopsy. In the midst of our grief I quickly said no, without asking for an explanation of the advantages of getting one, and realized after my son's death that perhaps that would have yielded helpful information.” Another parent offered, “Give guided questions instead of asking “is there anything I can do for you?”. Most families are in shock and are not in a frame of mind to think clinically about their child.”
When prompted to describe what had gone well or what they wish had been different, parents provided insight into aspects of the communication and interactions with the healthcare teams they appreciated, as well as experiences that they found problematic. For example, one parent shared: “The attending immediately asked if I wanted to donate my son's heart & lungs, which made us feel value in his life that they would learn more from his unique set of defects, but they never followed up to ever tell us that they learned anything.” Another parent noted “They could have been more considerate, understanding, apologetic on such a traumatic day. The staff made unnecessary comments and it was frustrating not to have real answers.”
The common themes that emerged from the qualitative analysis indicated that parents were deeply affected by whether they felt adequately informed versus uninformed, the perceived attentiveness and empathy of providers, consistency in messaging received, and whether they believed that their perspectives were heard and respected.
Parents rarely (15%) reported being offered an opportunity to provide their providers or hospital with feedback regarding their experience related to M&M. Less than half of respondents (45%) reported that a member of the healthcare team or hospital reached out to answer unresolved questions after the fact. One parent explained, “We basically left the hospital that night after our daughter had died with no idea what to do next or how to proceed. We never spoke with a social worker and didn't know until later that some hospitals have teams and plans to deal with the death of a child. We're grateful that the nurse spoke to us about getting a hand mold made and a lock of hair taken as we never would have thought to ask about those things. There should be procedures that all staff are trained for in the event a child dies so they can provide parents with resources and support.” In line with this feeling, another parent shared, “In the moments after our son died, we weren't in the mindset to ask the questions that needed to be asked. We just wanted to be with our baby. It would have been nice to come back a few weeks later and sit down with the medical team to discuss the day our son died.”
Transparency
The majority of physicians and parents agreed that healthcare providers should disclose both suspected and confirmed errors that result in prolonged treatment, discomfort, disability, or death of a child, and that healthcare providers should answer with honesty any questions that parents may have about their child’s clinical care or outcomes. Qualitative responses to open-ended prompts further validated this alignment.
However, physicians and parents varied in their responses regarding parent participation in the M&M review process. Parent respondents were generally in favor of being notified of the plan for their child’s case to be reviewed through M&M as well as being involved in the actual M&M review process, while physicians expressed more uncertainty or disagreement with parent notification or involvement. Figure 2 shows the breakdown of parent and physician responses regarding the parent role in the M&M review.
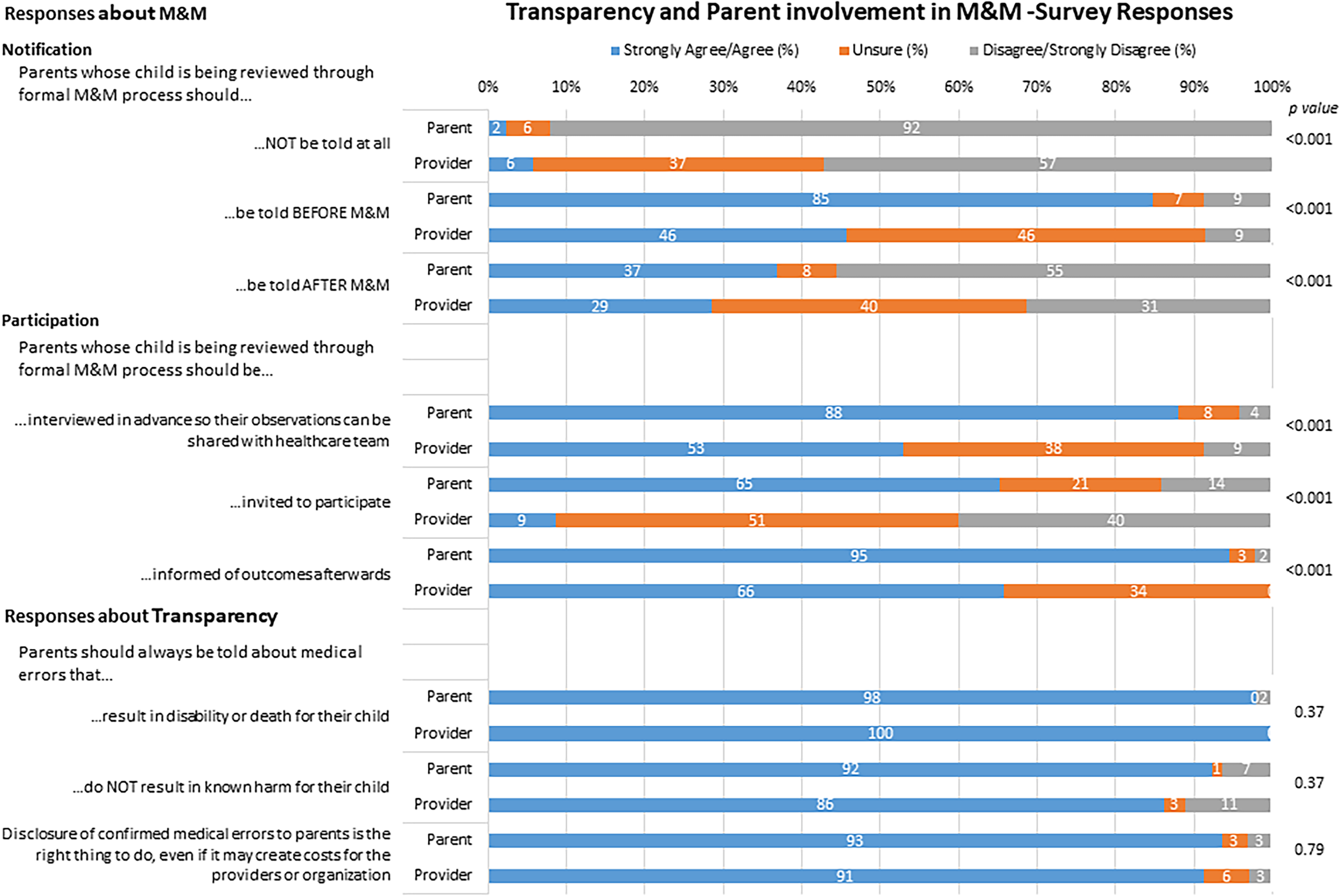
Parent and physician responses regarding parent involvement in Morbidity and Mortality (M&M) review.
Survey respondents elaborated on their ratings through the use of several open-ended questions and prompts. One parent explained, “I like the idea of sharing the outcomes of the morbidity review with the family…it would help them to feel that the healthcare providers learned from the situation.” Yet another parent considered the need for follow-up in their own circumstance: “I wish they had offered a meeting and way to discuss what went wrong—not that they would admit any wrongdoing. I know they are doing the best they can with the technology they have to use, but they should take parental input seriously as well.”
Physicians were concerned that parent participation in the M&M process could be detrimental and hamper the process. Qualitative comments provided by physicians provided some details of the hesitancy seen in the Likert scale responses: “I do worry that the presence of families will inhibit the team from full disclosure about their actions, and free flow of information. It will also likely heighten defensive behaviors and is less likely to reveal true system errors.” Others highlighted M&M as quality improvement, and that presence of family members would limit discussion of other cases that may be pertinent to an overall system failure. Concerns were also expressed regarding variability in families and that some could benefit from the process while it would be detrimental for other families.
Discussion
On the surface, there was a shared perspective regarding the importance of transparency and open disclosure in the context of adverse events. However, this study also highlights important differences in how physician respondents versus parent respondents defined transparency in the context of M&M review processes.
Organizations rarely include parents in the M&M review process; however, our study suggests a parental desire to be involved. This is in line with what Bakhbakhi and colleagues found in their study which examined bereaved parents’ perspectives on the perinatal mortality review in South West England (29). Their study found that the majority of participants were unaware their child was discussed at the perinatal mortality review, yet all of them voiced a desire to know when the review process was taking place and to be advised of the pertinent findings (29).
In our study, physicians were reluctant to have parent participation in M&M given concerns of obstructing the M&M process and that the process may be more detrimental than helpful for some families. However, physicians also reported that M&M doesn’t always serve the purpose it was meant to, resulting in changes to processes and practices in less than half of the cases. This reflects multiple other studies which recognize that M&M often falls short of affecting lasting and concrete change (14,21–24). However, many parents desire an understanding of what happened during an adverse event and the assurance that improvements will arise from it (29). Hence, both physicians and parents desire lasting impact and effective quality improvements to arise from the M&M review. Parent involvement in M&M could contribute to important changes to the M&M process to bring about meaningful impact.
Given this, we propose a stepwise approach to work toward integrating parent input into the M&M process, with the goal of improving the process and its objective of effecting positive change within the healthcare system. Integral to this approach is modifying the structure of M&M as needed to ensure that both patient confidentiality remains intact and team learning is not inhibited. Initial potential changes may include interviewing parents to obtain their perspective prior to the conference, incorporating this feedback into the M&M review, and ultimately having a formalized process to discuss the findings of M&M and the resultant changes with parents after the fact.
Some organizations may find that there are mechanisms by which interested families may be engaged as full participants in M&M review and conference, even if that may require modifications to the way in which the process is currently operationalized, thus allowing for patient confidentiality and protecting an organization’s ability to engage in QI. Families also recognize that there needs to be flexibility for parental involvement, accommodating the degree to which each family feels comfortable with participating, as well as the preferred timing of their participation. Some families may want to participate soon after an event while details remain fresh in their minds while others may prefer involvement further down the line, when they may feel more emotionally ready to participate (29).
Including families in the M&M review process may pose untoward effects that must be considered. Similar to open disclosure, concern exists around the impact on individual and organizational reputation and patient relationships (30,31). The legal ramifications of including families in the commonly protected M&M forum are also unclear (most states have a nondiscovery clause, but current apology law provides incomplete protection to providers) (31,32). Inclusion of families may also disrupt the collaborative effort, as participants try to distance themselves from association with “wrongdoings,” thus hampering, rather than contributing to, discovery and learning from the proceeding. Despite these challenges, the M&M review may offer a platform to support a family-centered approach to identifying opportunities for system improvement and foster co-creation of knowledge by health care providers and families. Incorporating parents in the M&M review could increase the sense of responsibility to execute and provide feedback regarding changes arising from the review (33).
While physicians and parents agree that errors should be disclosed openly and honestly, parent respondents wished something had been done differently nearly half of the time. Most hospitals do not provide guidance or formal training for the disclosure process, which may contribute to the high rate of parent dissatisfaction. Further educational investment in disclosure best practices may result in improved adverse event disclosure satisfaction. Educating physicians about what families desire from the disclosure process (comprehensive, transparent, and sensitively conveyed information, an apology as well as assurance that the root cause for the event will be addressed) and equipping them with practical skills to use during the disclosure process are crucial. Interventions such as workshops, professional patient scenarios, and interactive videos may be useful tools to address educational needs.
While navigating the process of restructuring M&M to make room for parent involvement will require some vulnerability and innovation, it has the potential to positively affect the process for physicians and parents, alike. Providing answers to parents can help alleviate parent self-blame and ensure accountability for system change (29). Additionally, parents identified wanting to incorporate positive feedback into the review process as well, highlighting excellence in care received, a concept not dissimilar from the suggestion of incorporating successes into the M&M process and humanizing the process (16,25,26,29,33,34).
Limitations
Our study has several limitations from the sampling bias in the use of social media and listserv for recruitment. First, most parent respondents were highly educated, white mothers whose views on the healthcare system may not be reflective of the entire population. Given respondents were self-selected, they may represent individuals with significant positive or negative experiences who desire to provide feedback to the medical team. Thus they may be more likely to voice interest in participating in activities like M&M. Additionally, respondents were predominantly parents of children with single ventricle physiology, influenced by posting through Sisters by Heart, which is an organization focused on children with hypoplastic left heart. Their views may not be generalizable to children with other types of heart disease. Finally, parents were asked to report their wishes in retrospect introducing potential recall bias as their responses may not represent their feelings immediately following the event. Similarly, the physician sample may not be representative of the broader group, and thus our ability to extrapolate our findings is limited.
Conclusion
This study highlights an important opportunity for meaningful family engagement. A segment of the parent population desires to be more informed members of their child’s team, and contributors to system improvement. That this may differ from physician perspectives underscores the need for additional research to understand how broadly held these perspectives may be.
Patient expectations have changed from passively accepting decisions of the healthcare team to co-creation. This study expands on this evolution and demonstrates that families desire not only better disclosure of adverse events, but involvement in the M&M review process. Parents and physicians agree on the principles of transparency and recognize that parents want to be involved in the M&M review. There is now an opportunity for sequential tests of change to meet the needs of both physicians and parents alike.
Supplemental Material
sj-docx-1-jpx-10.1177_23743735221102674 - Supplemental material for Parent Participation in Morbidity and Mortality Review: Parent and Physician Perspectives
Supplemental material, sj-docx-1-jpx-10.1177_23743735221102674 for Parent Participation in Morbidity and Mortality Review: Parent and Physician Perspectives by Sarah R de Loizaga, Katherine Clarke-Myers, Philip R Khoury and Samuel P Hanke in Journal of Patient Experience
Footnotes
Acknowledgments
The authors would like to thank the Heart Institute Research Core at CCHMC for project support. Special thanks to Sisters by Heart and Hayden’s Heart, to our family advisory team, and to all survey respondents.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This study was submitted to the institutional IRB (2016-6718) and deemed exempt in accordance with 45 CFR 46.101 (b).
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Statement of Human and Animal Rights
All procedures in this study were conducted in accordance with the Cincinnati Children’s Hospital Medical Center Institutional Review Board.
Statement of Informed Consent
Written informed consent was obtained from participants for their participation in this study.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
