Abstract
Nearly all new product development teams at pharmaceutical companies will routinely conduct patient advisory boards. These board meetings will help collect and document the experience of patients and caregivers for medical product development and regulatory decision-making. Recently, in June 2020, The US Food and Drug Administration (FDA) published a final guidance on methodological patient-focused drug development (PFDD) to address, in a stepwise manner, how stakeholders (patients, researchers, medical product developers, and others) can successfully use these patient forums. In the process of developing this guidance, the FDA acknowledged that leading its own PFDD meetings, especially when limited to organized disease advocacy groups, cannot address the gaps in information on the patient perspective. So, it has expressed support for advancing the science and utilization of patient input other means. Because traditional methods of conducting patient advisory boards often do not achieve the full potential of patient centricity, the authors of this article share an approach to consider when selecting patient advisors, in order to gain the most actionable input to a product development team.
Keywords
Introduction
Patient advisory boards have a well-established place in the commercialization process (1, 2, 3, 4, 5). There is a trend of conducting these advisory boards earlier in drug development across all clinical trial phases. They help explore unmet needs, generate hypotheses, create research plans, and design trials. In these groups, patients can advocate for new treatments and advise on clinical study design (6).
Although these initiatives are positive contributors to patient centricity (7), they may be limited to “getting feedback,” or worse, just “checking the box” of an expected tactic (8).
We advocate for a systematic approach to listening and understanding that informs the type of data a study will capture, for example, quality of life metrics, physical functioning, social participation or productivity (absenteeism and presenteeism), or other aspects of patient experiences (6).
Collectively, the authors have planned, executed, and facilitated dozens of patient advisory boards and related group initiatives. Through our experience, we share lessons learned and a practical 5-step model to identify, recruit, and involve patients in drug development.
Description
In the following case study, we applied a systematic, multiplatform, stepwise process (see Figure 1) for a company developing a new therapeutic to treat a rare autoimmune-related disease that led to many actionable outputs, such as clarity of patient language, and a better understanding of the role of patient influencers in this disease. The process is akin to the funnel approach used to identify, profile, and recruit candidates for clinical trials.
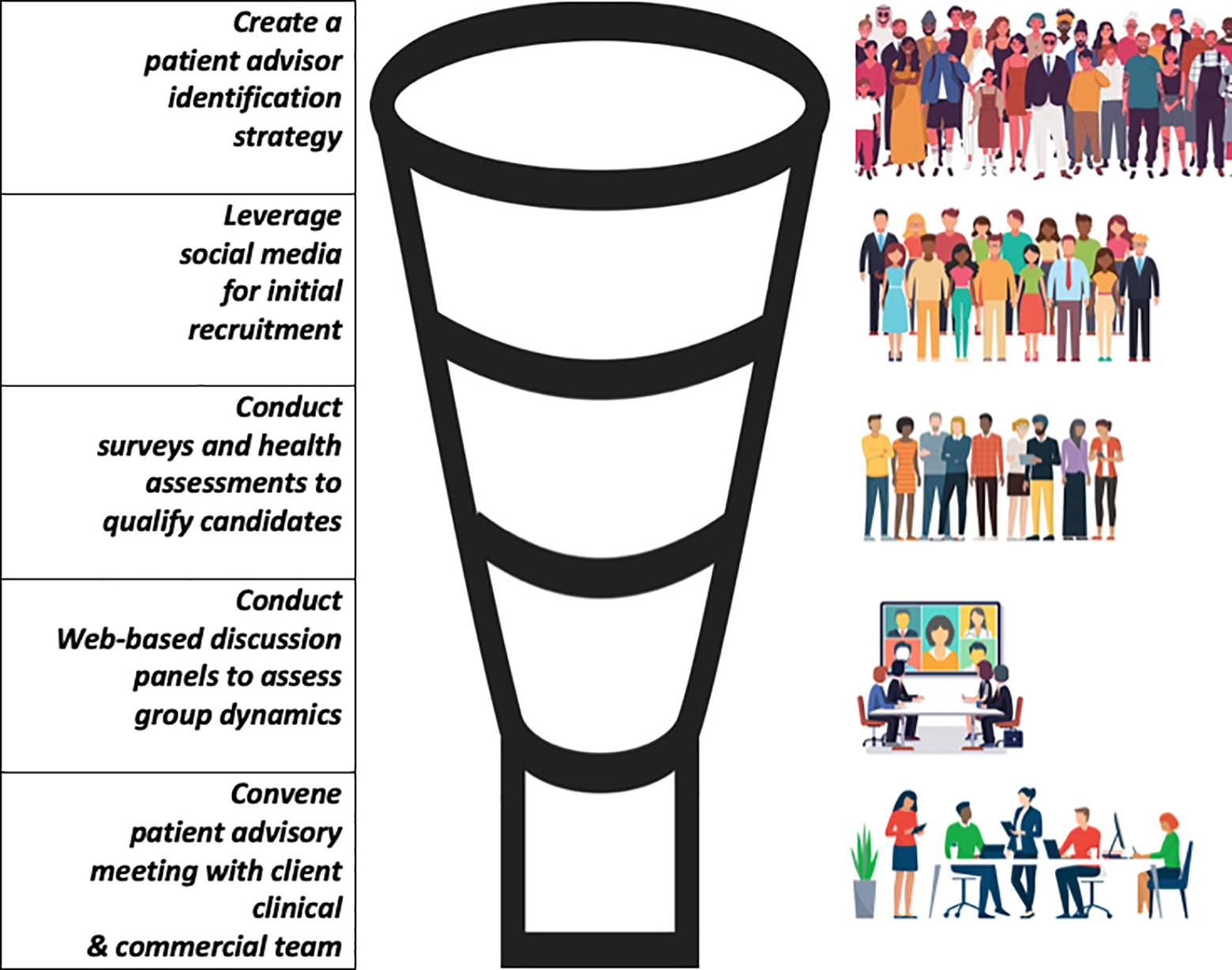
Five-step model to finding and engaging patient advisors.
Step 1: Create a patient advisor identification strategy—begin with the end in mind
What does the client want to learn from engaging patients? What will be produced as a result? The answers to these questions will direct the patient profiles most likely to deliver the required outcome.
In our rare disease case, the development team’s goal was to better understand the overall patient journey, in order to identify care touchpoints at which the new product might make a difference to patients. We also wanted input on a target product profile (TPP) (9).
Examples of other types of goals could include guidance on protocol design, consent forms, patient instructions, and experience with existing products. Each of these may require a different patient profile for optimal results.
Step 2: Leverage social media to get real-world patients for initial recruitment
Identifying the right patient advisors becomes more complicated in a rare disease category because the patient pool is so much smaller. To complicate things further, in our rare disease category, the vast majority of potential patients were still undiagnosed. For this reason, the traditional strategy of targeting an advocacy organization for diagnosed patients was not possible. We first learned this when working with a grassroots patient community leader who was building a database of like-minded patients seeking educational resources—and finding what a limited number of patients would be accessed this way.
Therefore, we cast a wide net across message boards, Facebook groups, and other social media with personal stories and a clear call to action. Multiple campaign platforms were tested for patient feedback to optimize the message and drive qualified responses. For example, we learned that the word “development” was more motivating than “research” or “studies” because it connoted progress. We also formalized a process to ask each qualified patient to provide another referral of someone they might know. As a result of this outreach approach, approximately 100 patients met the criteria to be considered.
Step 3: Conduct online surveys and health assessment questionnaires to qualify candidates
To further evaluate the patients’ qualifications, the 100 potential advisors participated in a series of online surveys related to their current experiences and the emotional impact of their condition. It was useful to reference quality of life instruments, which are available for many disease conditions (10 –13) and patient activation measures, such as PAM-13 (Patient Activation Measures-13) and other validated tools (14).
For example, our survey asked if the patient had experienced the following in the past week: difficulty enjoying meals, missed work, skipped social or family event, and changed usual behavior to work around symptoms. It also asked the patient which word best described his or her feeling: for example, embarrassed, anxious, frustrated, cautious, and nervous.
To assess their personal health engagement, the survey asked patients to rate agreement with statements such as: “I am responsible for taking care of my health,” “I tell my doctor my concerns even if he or she does not ask,” and “I can maintain lifestyle changes even during times of stress.” On the basis of these responses, we narrowed the field to approximately 50 potential advisor candidates.
Step 4: Conduct Web-based discussion panels to assess group dynamics
In this step, we assessed how individuals might interact together. A high level of interactivity and the ability to communicate personal insights in a group setting is critically important to the success of an in-person patient advisory board meeting.
This step provided us an opportunity to hear patient advisor candidates define the disease in their own terms, to follow the varied diagnostic pathways, and to understand the wide range of treatment approaches.
In our rare disease example, we wanted patient advisors who had varied experiences and who also could relate to others at different stages of their rare disease journey. Using webcams with the moderated group allowed for a live platform with participants from across the country. Topics ranged from attitudes about doctor visits to the social, emotional, and health impact of their condition. At the conclusion of this step, we were able to compile the final invitation list of 10 individuals.
Step 5: Convene patient advisory meeting with client clinical and commercial team
We facilitated a one-and-a-half-day workshop with Patient Journey as the centerpiece, using highly interactive mapping exercises. The emotional language of the patient was key for our client to hear the outcomes that the end customer really wants, in contrast to the clinical language of trial efficacy measures and Food and Drug Administration definitions—ie, the usual lexicon of the clinical development team.
Other industry colleagues have published similar examples illustrating the need to balance medical outcome with the medical journey and individual aspirations. One case study reads: “a father with diabetes and heart disease wants to follow his doctor’s orders to reach his desired medical outcome to be in better health, but his journey to reach that outcome is difficult to manage. He is a bus driver and the prescribed medications make him drowsy. This man’s aspiration is to make sure he can work to provide a better life for his children. It is only by prioritizing this aspiration and altering the journey by finding a treatment that will allow him to keep working that the patient can achieve the desired medical outcome” (15).
Results
Taking this different approach yielded meaningful benefits. Specifically, we gained potentially significant input for changes in protocol design, suggestions for word changes for the TPP and labeling, and key patient experiences that can be shared with health care providers, trialists, guideline developers, and others. From a patient’s perspective, we conducted a post-meeting survey in which participants expressed gaining new knowledge of their disease, a renewed attitude of empowerment, and a sense of actions they could take to improvement their own well-being, as well as contribute to new drug development.
In our work with other companies and contract research organizations, we also have used patient advisors to seek perspectives on clinical trial kit designs, a digital diary app, study outreach communications, and packaging.
Furthermore, one important side benefit emerges within the company when cross-functional teams are involved: shared ownership roles that help create continuity from research and development to launch, including appropriate outcomes and communications.
Lessons Learned
Investing early in patient advisors is a sound strategic move that increases a company’s chances of successfully developing a product with the patient/customer in mind. In addition, it helps the sponsor team embrace advocacy as its members work together toward patient-focused goals.
As health care systems continue to evolve and establish patients as the primary stakeholder in their health care decision-making, the pharmaceutical industry will need to be innovative to demonstrate the value of their products relative to the outcomes experienced by patients. Pharmaceutical companies should recognize the value of involving patients across the entire product life cycle and work to transform present perceptions and practices throughout their organizations (16).
Conclusion
The practical 5-step model summarized in the article can improve the way drug development teams identify, recruit, and engage patients as advisors and partners in the new drug development process.
When you reorient the way patients are identified and engaged, the common everyday tactic of a patient advisory board takes on a new potential. And true patient-centricity emerges.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
